Submitted:
21 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
3. Materials and Methods
3.1. Typical Reaction Procedure
3.2. Characterization
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, Z.; Ganewatta, M. S.; Tang, C. Sustainable polymers from biomass: bridging chemistry with materials and processing. Prog. Polym. Sci. 2020, 101, 101197. [Google Scholar] [CrossRef]
- Rosenboom, J. G.; Langer, R.; Traverso, G. Bioplastics for a circular economy. Nat. Rev. Mater. 2022, 7, 117–137. [Google Scholar] [CrossRef]
- Mülhaupt, R. Green polymer chemistry and bio-based plastics: Dreams and reality. Macromol. Chem. Phys. 2013, 214, 159–174. [Google Scholar] [CrossRef]
- Zhu, Y.; Romain, C.; Williams, C. K. Sustainable polymers from renewable resources. Nature 2016, 540, 354–362. [Google Scholar] [CrossRef]
- Kristufek, S.L.; Wacker, K.T.; Tsao, Y.-Y.T.; Su, L.; Wooley, K.L. Monomer Design Strategies to Create Natural Product-Based Polymer Materials. Nat. Prod. Rep. 2017, 34, 433–459. [Google Scholar] [CrossRef]
- Ricci, G.; Pampaloni, G.; Sommazzi, A.; Masi, F. Dienes Polymerization: Where We Are and What Lies Ahead. Macromolecules 2021, 54, 5879–5914. [Google Scholar] [CrossRef]
- Zhang, G.; Guo, B.; Zhang, L. Significant Trends in Rubber Research Driven by Environment, Resource, and Sustainability. Macromolecules 2025, 58, 5883–5903. [Google Scholar] [CrossRef]
- Hanssens, J.; Meneses, D.; Saya, J.M.; Orru, R.V.A. Terpenes and Terpenoids: How can we use them? Eur. J. Org. Chem. 2025, 28, e202401151. [Google Scholar] [CrossRef]
- Loughmari, S.; Hafid, A.; Bouazza, A.; El Bouadili, A.; Zinck, P.; Visseaux, M. Highly stereoselective coordination polymerization of β-myrcene from a lanthanide-based catalyst: access to biosourced elastomers. J. Polym. Sci. Polym. Chem. 2012, 50, 2898–2905. [Google Scholar] [CrossRef]
- Anastasiou, D. E. Critical review and perspective on the production of synthetic and natural poly-β-myrcene. Polym. Bull. 2025, 82, 2717–2750. [Google Scholar] [CrossRef]
- Lamparelli, D.H.; Kleybolte, M. M.; Winnacker, M.; Capacchione, C. Sustainable Myrcene-Based Elastomers via a Convenient Anionic Polymerization. Polymers 2021, 13, 838. [Google Scholar] [CrossRef]
- Lamparelli, D. H.; Paradiso, V.; Monica, F. D.; Proto, A.; Guerra, S.; Giannini, L.; Capacchione, C. Toward more sustainable elastomers: stereoselective copolymerization of linear terpenes with butadiene. Macromolecules 2020, 53, 1665–1678. [Google Scholar] [CrossRef]
- Lamparelli, D. H.; Winnacker, M.; Capacchione, C. Stereoregular polymerization of acyclic terpenes. ChemPlusChem 2022, 87, e202100366. [Google Scholar] [CrossRef] [PubMed]
- Grieco, S.; Di Girolamo, R.; Ritacco, R.; Falivene, L.; Leone, G. Comonomer Discrimination in Copolymerization of β-Myrcene: Ethylene Inhibition, Spectators, and Soft Elastomers with Isoprene. ACS Polym. Au 2025, 5, 645–655. [Google Scholar] [CrossRef]
- Nomura, K.; Jantawan, K. Modified half-titanocenes as polymerization catalysts: Basic concept, displayed promising characteristics and some mechanistic insights. Catalysts 2026, 16, 221. [Google Scholar] [CrossRef]
- Kitphaitun, S.; Chaimongkolkunasin, S.; Manit, I.; Makino, R.; Kadota, J.; Hirano, H.; Nomura, K. Ethylene/Myrcene Copolymers as New Bio-Based Elastomers Prepared by Coordination Polymerization Using Titanium Catalysts. Macromolecules 2021, 54, 10049–10058. [Google Scholar] [CrossRef]
- Niknam, F.; Buonerba, A.; Lamparelli, D. H.; Capacchione, C. An old tool to obtain new polymers from renewable resources: [OSSO]-type titanium-catalysed ethylene and myrcene copolymers. Faraday Discuss. 2026, 262, 152–168. [Google Scholar] [CrossRef]
- Ren, X.; Guo, F.; Fu, H.; Song, Y.; Li, Y.; Hou, Z. Scandium-catalyzed copolymerization of myrcene with ethylene and propylene: convenient syntheses of versatile functionalized polyolefins. Polym. Chem. 2018, 9, 1223–1233. [Google Scholar] [CrossRef]
- Wang, W.; Qu, S.; Li, X.; Chen, J.; Guo, Z.; Sun, W.-H. Transition metal complex catalysts promoting copolymers of cycloolefin with propylene/higher olefins. Coord. Chem. Rev. 2023, 494, 215351. [Google Scholar] [CrossRef]
- Nomura, K. Half-titanocenes containing anionic ancillary donor ligands as promising new catalysts for precise olefin polymerisation. Dalton Trans. 2009, 38, 8811–8823. [Google Scholar] [CrossRef] [PubMed]
- Losio, S.; Boggioni, L.; Vignali, A.; Bertini, F.; Nishiyama, A.; Nomura, K.; Tritto, I. Poly(propene-co-norbornene)s with high molar masses, tunable norbornene contents and properties, in high yield by ketimide-modified half-titanocene catalysts. Polym. Chem. 2025, 16, 3709–3719. [Google Scholar] [CrossRef]
- Losio, S.; Bertini, F.; Vignali, A.; Fujioka, T.; Nomura, K.; Tritto, I. Amorphous elastomeric ultra-high molar mass polypropylene in high yield by half-titanocene catalysts. Polymers 2024, 16, 512. [Google Scholar] [CrossRef]
- Resconi, L.; Jones, R.L.; Rheingold, A.L.; Yap, G.P.A. High-Molecular-Weight Atactic Polypropylene from Metallocene Catalysts. 1. Me2Si(9-Flu)2ZrX2 (X = Cl, Me). Organometallics 1996, 15, 998–1005. [Google Scholar] [CrossRef]
- Carvill, A.; Zetta, L.; Zannoni, G.; Sacchi, M. C. ansa-Zirconocene-Catalyzed Solution Polymerization of Propene: Influence of Polymerization Conditions on the Unsaturated Chain-End Groups. Macromolecules 1998, 31, 3783–3789. Available online: https://pubs.acs.org/doi/10.1021/ma9800156. [CrossRef]
- Yamamoto, Y. Transition-Metal-Catalyzed Cycloisomerizations of α,ω-Dienes. Chem. Rev. 2012, 112, 4736–4769. [Google Scholar] [CrossRef]
- Galimberti, M.; Albizzati, E.; Abis, L.; Bacchilega, G. 13C NMR analysis of α-olefins copolymers with 1,3-butadiene obtained with zirconocenes/methylalumoxane catalysts. Makromol. Chem. Macromol. Symp. 1991, 192, 2591–2601. [Google Scholar] [CrossRef]
- Pragliola, S.; Costabile, C.; Di Bartolomeo, F.; Longo, P. Copolymerization of Propene and Buta-1,3-diene in the Presence of Highly Hindered C2-Symmetric Zirconocene-Based Catalyst. Macromol. Rapid Comm. 2004, 25, 995–999. [Google Scholar] [CrossRef]
- Sun, B.; Wu, C.; Cui, D. Coordination Terpolymerization of Ethylene, Butadiene, and Styrene Using Thioanisole-Modified Rare-Earth Catalysts. Macromolecules 2025, 58, 12528–12538. [Google Scholar] [CrossRef]
- Guo, L.; Makino, R.; Shimoyama, D.; Kadota, J.; Hirano, H.; Nomura, K. Synthesis of Ethylene/Isoprene Copolymers Containing Cyclopentane/Cyclohexane Units as Unique Elastomers by Half-Titanocene Catalysts. Macromolecules 2023, 56, 899–914. [Google Scholar] [CrossRef]
- Ricci, G.; Boccia, A.C.; Palucci, B.; Sommazzi, A.; Masi, F.; Scoti, M.; De Stefano, F.; De Rosa, C. Synthesis of stereoregular polymyrcenes using neodymium-, iron- and copper-based catalysts. Polym. Chem. 2024, 15, 1367–1376. [Google Scholar] [CrossRef]
- Zhang, J.; Aydogan, C.; Patias, G.; Smith, T.; Al-Shok, L.; Liu, H.; Eissa, A. M.; Haddleton, D. M. Polymerization of Myrcene in Both Conventional and Renewable Solvents: Postpolymerization Modification via Regioselective Photoinduced Thiol–Ene Chemistry for Use as Carbon Renewable Dispersant. ACS Sustainable Chem. Eng. 2022, 10, 9654–9664. Available online: https://pubs.acs.org/doi/10.1021/acssuschemeng.2c03755. [CrossRef] [PubMed]
- Nomura, K.; Fujita, K.; Fujiki, M. Olefin polymerization by (cyclopentadienyl)(ketimide)-titanium(IV) complexes of the type. J. Mol. Catal. A Chem. 2004, 220, 133–144. [Google Scholar] [CrossRef]
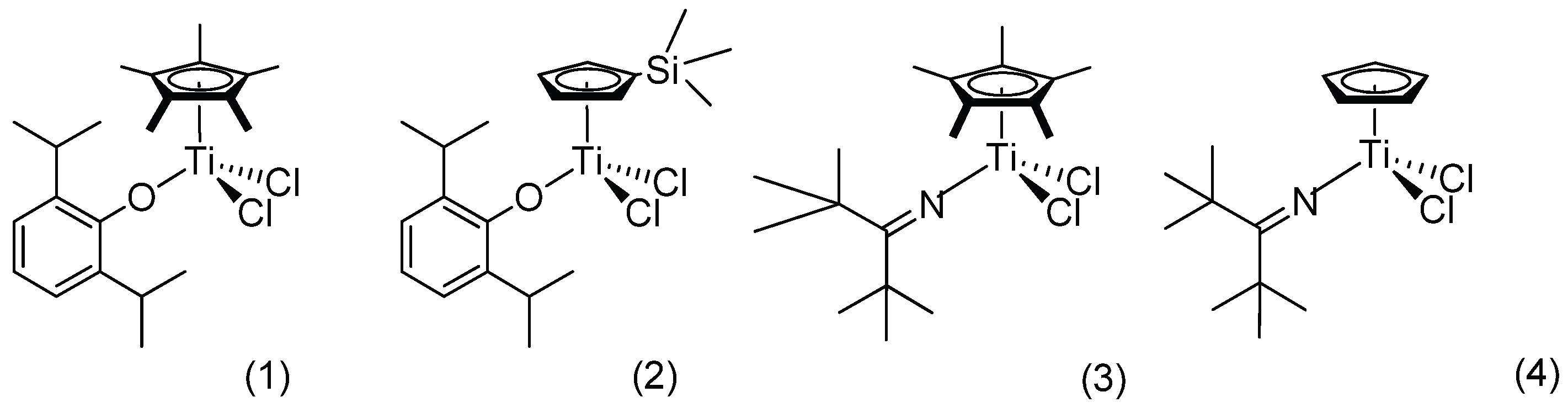
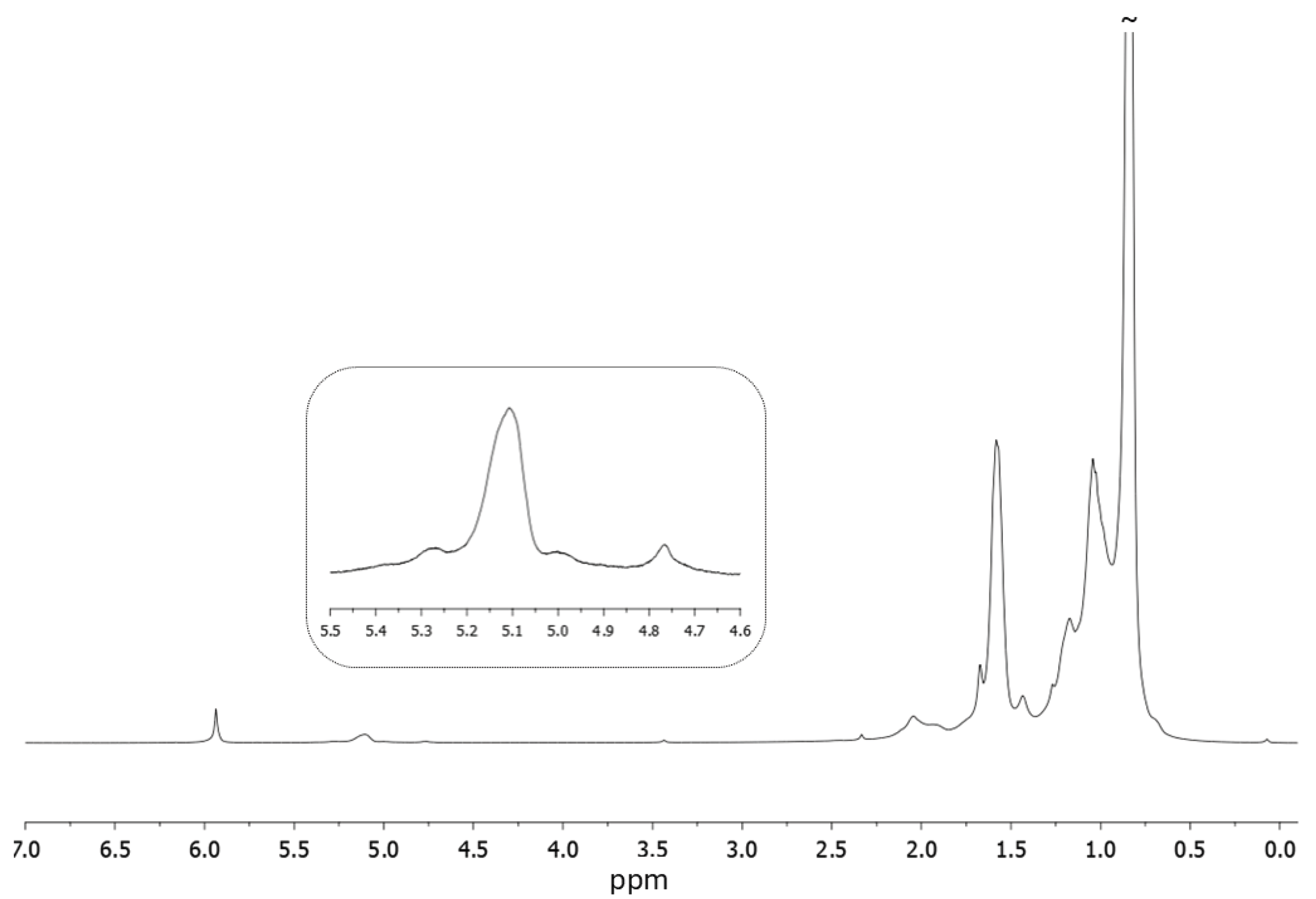
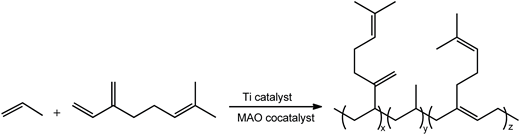 | |||||||||||
| entry | catalyst | P/MYb | T (°C) |
yield (g) |
activity kg (mol-Ti·h)-1 |
Mw c (kg mol-1) |
Ðc |
MY %d (mol) |
1,4 (%) | 3,4 (%) |
Tge (°C) |
| 1 | 1 | -- | 25 | 5.93 | 593 | 830 | 1.9 | -- | -- | -- | 2 |
| 2 | 1/0.50 | 25 | 0.83 | 20.7 | 188 | 2.9 | 0.51 | 57 | 43 | –10 | |
| 3 | -- | 40 | 8.48 | 424.0 | 309 | 1.9 | -- | -- | -- | –2 | |
| 4 | 1/0.50 | 40 | 1.98 | 79.3 | 249 | 1.9 | 0.83 | 62 | 38 | –7 | |
| 5 | -- | 60 | 1.47 | 36.9 | 225 | 7.9 | -- | -- | -- | –1 | |
| 6 | 1/0.50 | 60 | 0.92 | 92.4 | 87.5 | 2.2 | 0.50 | 55 | 45 | –10 | |
| 7 | 1/0.75 | 60 | 0.81 | 81.1 | 96.9 | 2.4 | 0.84 | 58 | 42 | –8 | |
| 8 | 1/1.0 | 60 | 1.02 | 102.2 | 91.4 | 1.9 | 1.11 | 62 | 38 | –11 | |
| 9 | 1/1.5 | 60 | 0.18 | 17.6 | 58.2 | 2.0 | 3.10 | 60 | 40 | –17 | |
| 10 | 2 | -- | 40 | 0.695 | 34.8 | 130 | 4.0 | -- | -- | -- | –11 |
| 11 | 1/0.50 | 40 | Traces | -- -- |
|||||||
| 12 | -- | 60 | Traces | ||||||||
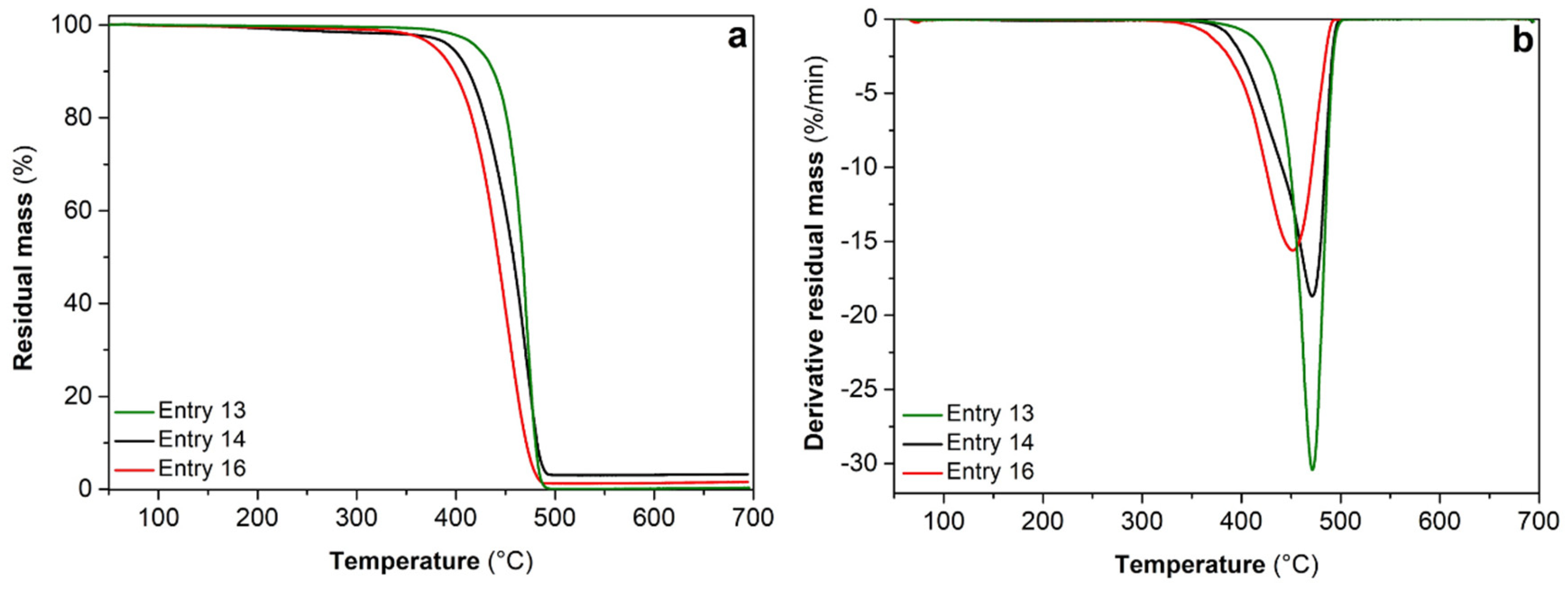
| 13f | 3 | -- | 40 | 11.20 | 4480 | 832 | 2.6 | -- | -- | -- | 2 |
| 14 | 1/0.50 | 40 | 1.17 | 117.3 | 343 | 2.5 | 0.33 | 67 | 33 | 0 | |
| 15 | 1/0.75 | 40 | 3.15 | 105.0 | 280 | 1.5 | 0.98 | 88 | 12 | –4 | |
| 16 | 1/1.5 | 40 | 0.43 | 21.6 | 144 | 1.6 | 2.52 | 83 | 17 | –9 | |
| 17f | -- | 60 | 12.93 | 5173 | 650 | 1.7 | -- | -- | -- | 0 | |
| 18 | 1/0.50 | 60 | 2.57 | 385.3 | 293 | 2.1 | 0.65 | 64 | 36 | –7 | |
| 19 | 1/0.75 | 60 | 2.90 | 96.7 | 287 | 1.5 | 1.98 | 78 | 22 | –13 | |
| 20 | 1/1.0 | 60 | 1.40 | 140.0 | 183 | 4.7 | 2.06 | 81 | 19 | –11 | |
| 21f | 4 | -- | 40 | 6.08 | 2433 | 1404 | 1.7 | -- | -- | -- | 1 |
| 22 | 1/0.50 | 40 | Traces | -- | |||||||
| 23f | -- | 60 | 3.85 | 1543 | 663 | 7.3 | -- | -- | -- | –2 | |
| 24 | 1/0.50 | 60 | Traces | -- | |||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




