Submitted:
20 April 2026
Posted:
21 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
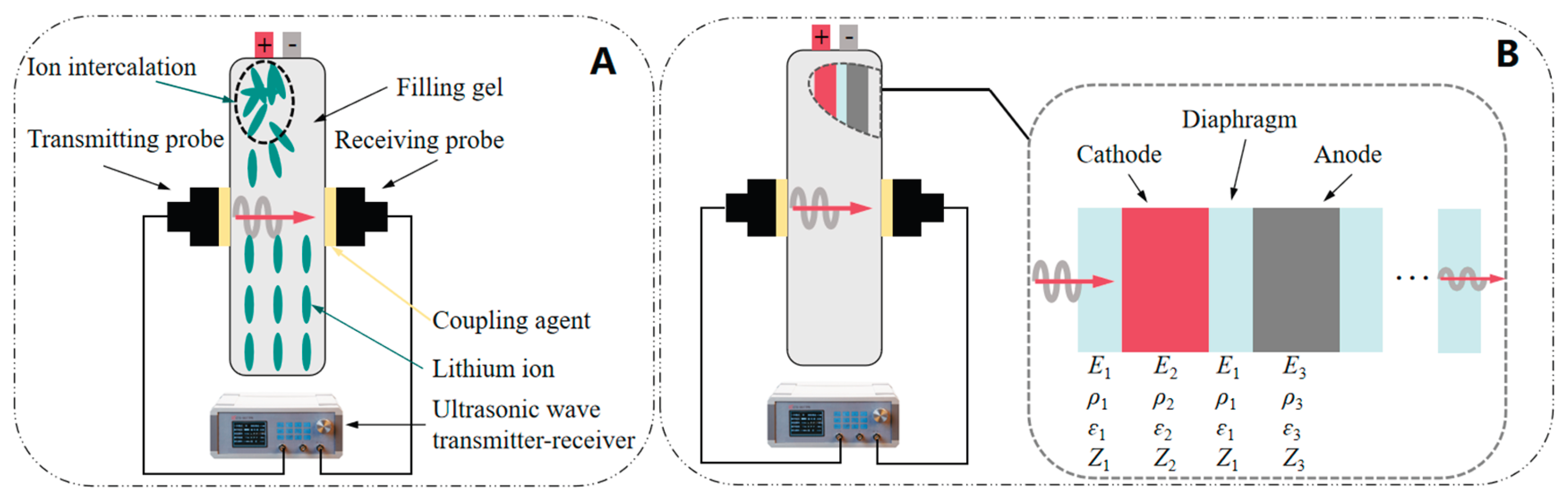
2.1. Theoretical Analysis on Ultrasonic Characterization of SOC
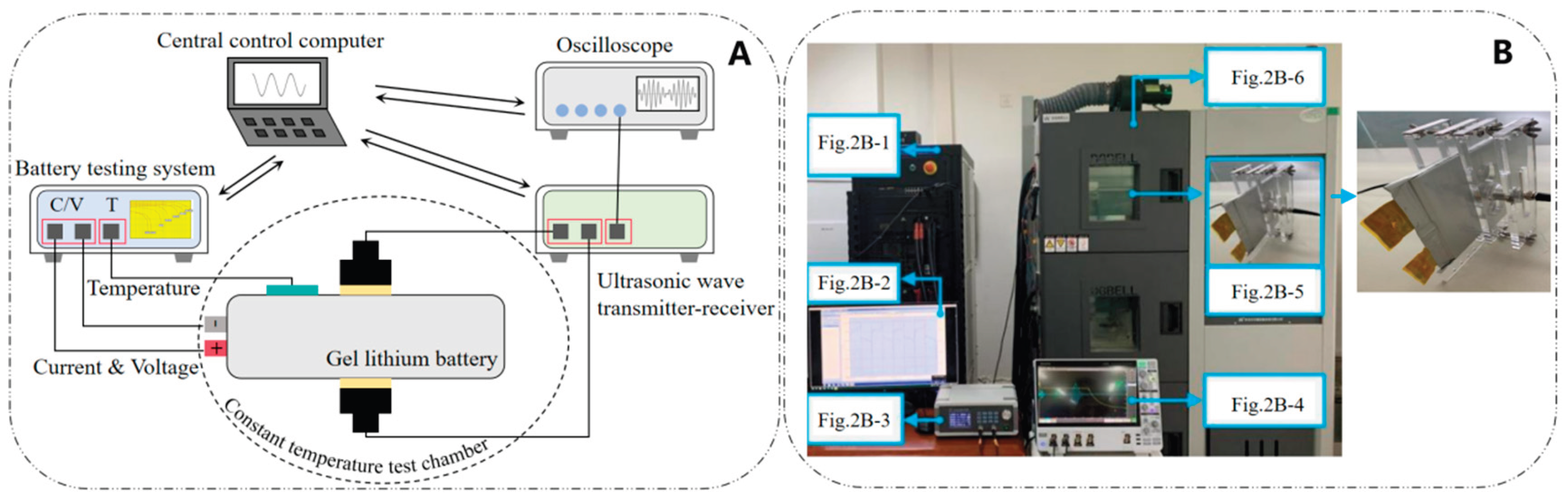
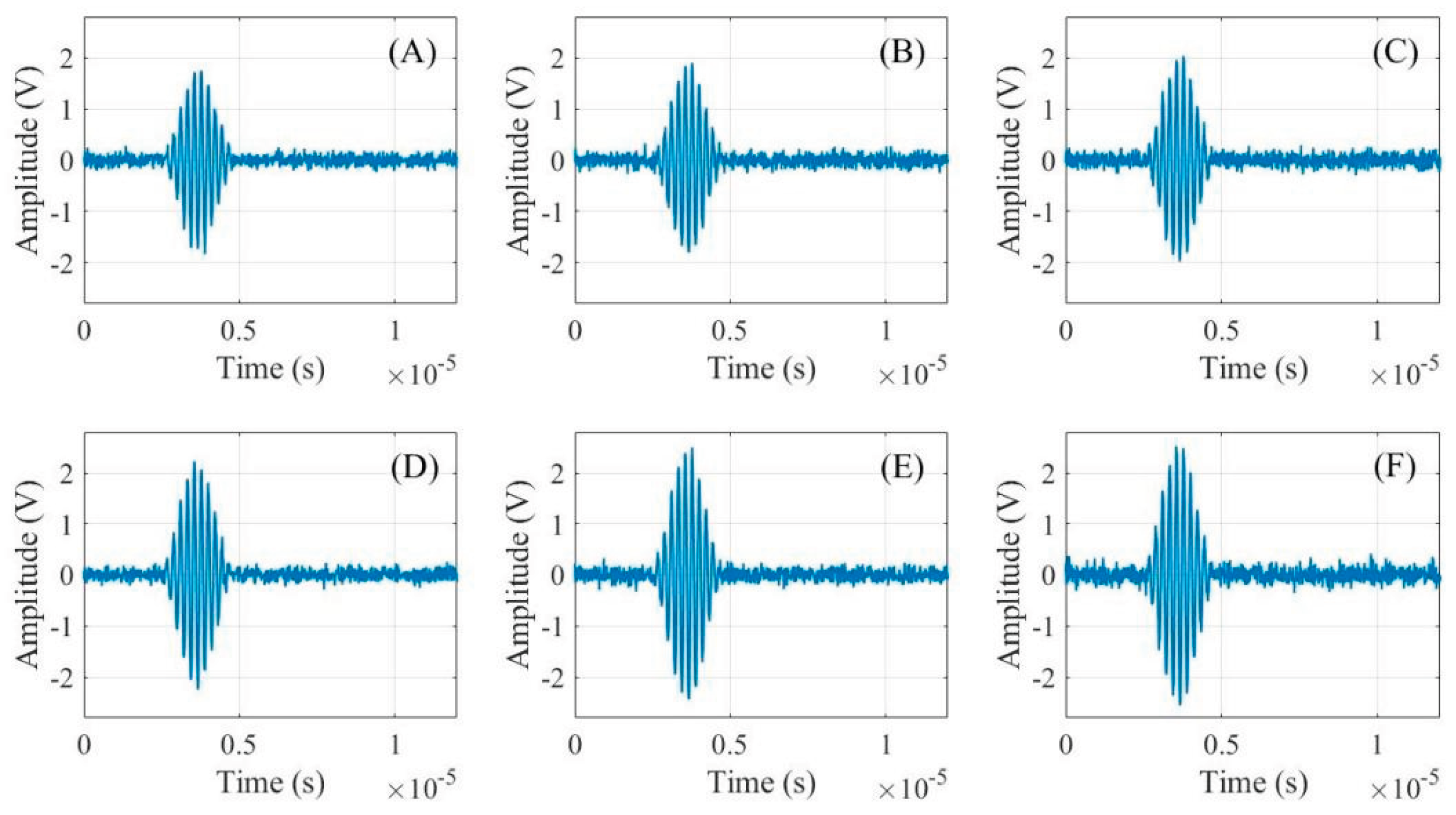
2.2. Analysis of Ultrasonic Time-Domain Signals for Gel Battery SOC
2.3. Analysis of Ultrasonic Frequency-Domain Signals for Gel Battery SOC
2.4. Analysis of Ultrasonic Signal’s Time-Frequency Transformation Features
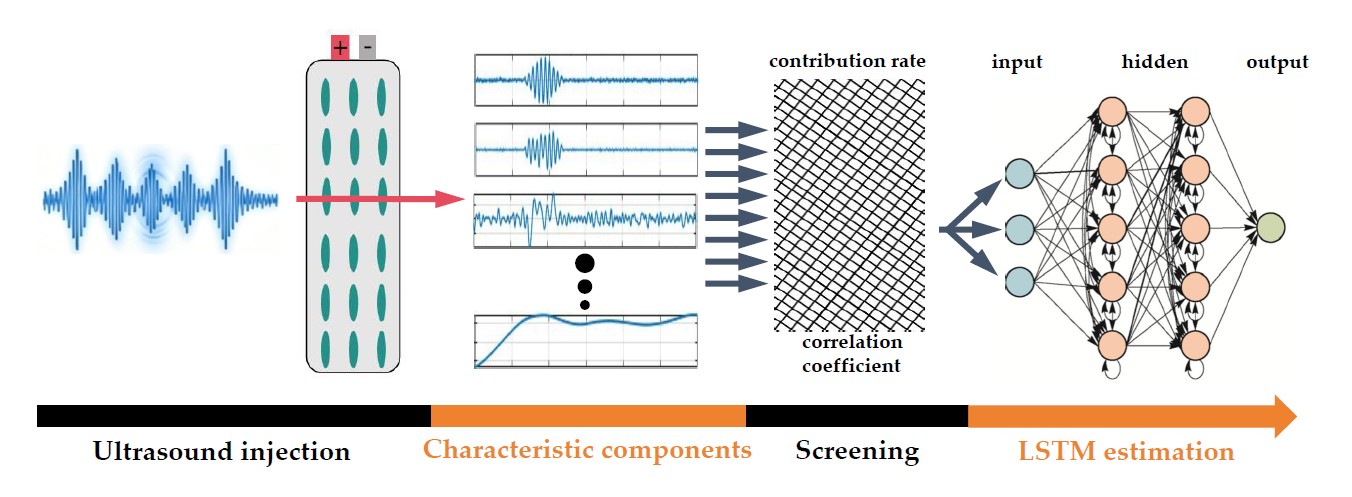
2.5. Gel Battery SOC Eestimation Based on Ultrasonic Time-Frequency Features
2.5. Analysis of the Feature Choice on SOC Estimation Accuracy
3. Conclusions
- (1)
- Multi-dimensional ultrasonic time-frequency domain features are extracted, including time-domain features, frequency-domain features and time-frequency transform features. The analysis demonstrates that these ultrasonic features can effectively characterize the electrochemical processes inside the gel battery, and eight characteristic indicators (SA, TOF, Fm, k1, k2, mf1, mf2, mf3) exhibit a strong correlation with the battery SOC.
- (2)
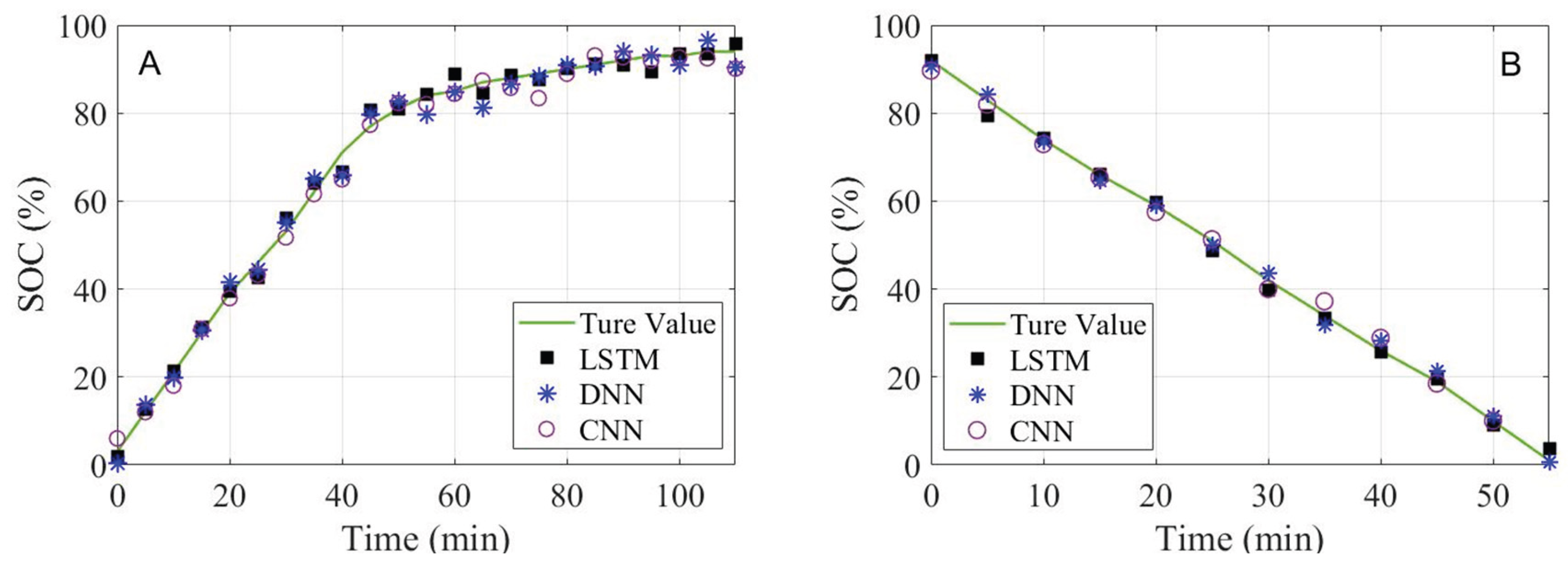
- An SOC estimation model for gel lithium battery is established based on LSTM, which fuses multi-dimensional ultrasonic time-frequency domain features. The results demonstrate that for the battery with the same aging degree, during the charging process, the SOC estimation RMSE is within 0.90% and the MAE within 0.65%; during the discharging process, the RMSE is within 0.42% and the MAE within 0.36%. The estimation results indicate that the accuracy of this method is excellent.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yao, J.; Kowal, J. Towards a smarter battery management system: A critical review on deep learning-based state of charge estimation of lithium-ion batteries. Energy and AI 2025, 21, 100585. [Google Scholar] [CrossRef]
- Hannan, M. A.; Hoque, M. M.; Mohamed, A.; Ayob, A. Review of energy storage systems for electric vehicle applications: Issues and challenges. Renewable and Sustainable Energy Reviews 2017, 69, 771–789. [Google Scholar] [CrossRef]
- Wei, Z.; Leng, F.; He, Z.; Zhang, W.; Li, K. Online state of charge and state of health estimation for a lithium-ion battery based on a data–model fusion method. Energies 2018, 11, 1810. [Google Scholar] [CrossRef]
- Zou, C.; Klintberg, A.; Wei, Z.; Fridholm, B.; Wik, T.; Egardt, B. Power capability prediction for lithium-ion batteries using economic nonlinear model predictive control. J. Power Sources. 2018, 396, 580–589. [Google Scholar] [CrossRef]
- Sultana, I.; Chen, Y.; Huang, S.; Rahman, M. M. Recycled value-added circular energy materials for new battery application: Recycling strategies, challenges, and sustainability-a comprehensive review. Journal of Environmental Chemical Engineering 2022, 10, 108728. [Google Scholar] [CrossRef]
- Peng, S.; Zhu, J.; Wu, T.; Tang, A.; Kan, J.; Pecht, M. SOH early prediction of lithium-ion batteries based on voltage interval selection and features fusion. Energy 2024, 308, 132993. [Google Scholar] [CrossRef]
- Sepasi, S.; Ghorbani, R.; Liaw, B. Y. A novel on-board state-of-charge estimation method for aged Li-ion batteries based on model adaptive extended Kalman filter. Journal of Power Sources 2014, 245, 337–344. [Google Scholar] [CrossRef]
- Suzhen, L.; Luhang, Y.; Chuang, Z. State of charge estimation of LiFeO4 batteries based on time domain features of ultrasonic waves and random forest. Transactions of China Electrotechnical Society 2022, 37, 5872–5885. [Google Scholar]
- Schneider, D. Silicon anodes will give lithiumion batteries a boost. IEEE Spectrum 2018, 56, 48–49. [Google Scholar] [CrossRef]
- Omariba, Z. B.; Zhang, L.; Sun, D. Review on health management system for lithium-ion batteries of electric vehicles. Electronics 2018, 7, 72. [Google Scholar] [CrossRef]
- Zhang, M.; Fan, X. Review on the state of charge estimation methods for electric vehicle battery. World Electric Vehicle Journal. 2020, 11, 23. [Google Scholar] [CrossRef]
- Hsieh, A. G.; Bhadra, S.; Hertzberg, B. J.; Gjeltema, P. J.; Goy, A.; Fleischer, J. W.; Steingart, D. A. Electrochemical-acoustic time of flight: in operando correlation of physical dynamics with battery charge and health. Energy & environmental science 2015, 8, 1569–1577. [Google Scholar]
- Chang, J. J.; Zeng, X. F.; Wan, T. L. Real-time measurement of lithium-ion batteries’ state-of-charge based on air-coupled ultrasound. Aip Advances 2019, 9. [Google Scholar] [CrossRef]
- Ladpli, P.; Nardari, R.; Kopsaftopoulos, F.; Chang, F. K. Multifunctional energy storage composite structures with embedded lithium-ion batteries. Journal of Power Sources 2019, 414, 517–529. [Google Scholar] [CrossRef]
- Chang, W.; Mohr, R.; Kim, A.; Raj, A.; Davies, G.; Denner, K.; Steingart, D. Measuring effective stiffness of Li-ion batteries via acoustic signal processing. Journal of Materials Chemistry A 2020, 8, 16624–16635. [Google Scholar] [CrossRef]
- Danko, M.; Adamec, J.; Taraba, M.; Drgona, P. Overview of batteries State of Charge estimation methods. Transportation Research Procedia 2019, 40, 186–192. [Google Scholar] [CrossRef]
- Liu, S.; Ren, J.; Yuan, L.; Xu, Z.; Zhang, C. Multi-Source Data Feature Extraction Method for State of Charge Estimation of LiFePO4 Battery. Transactions of China Electrotechnical Society 2025, 40, 3349–3361. [Google Scholar]
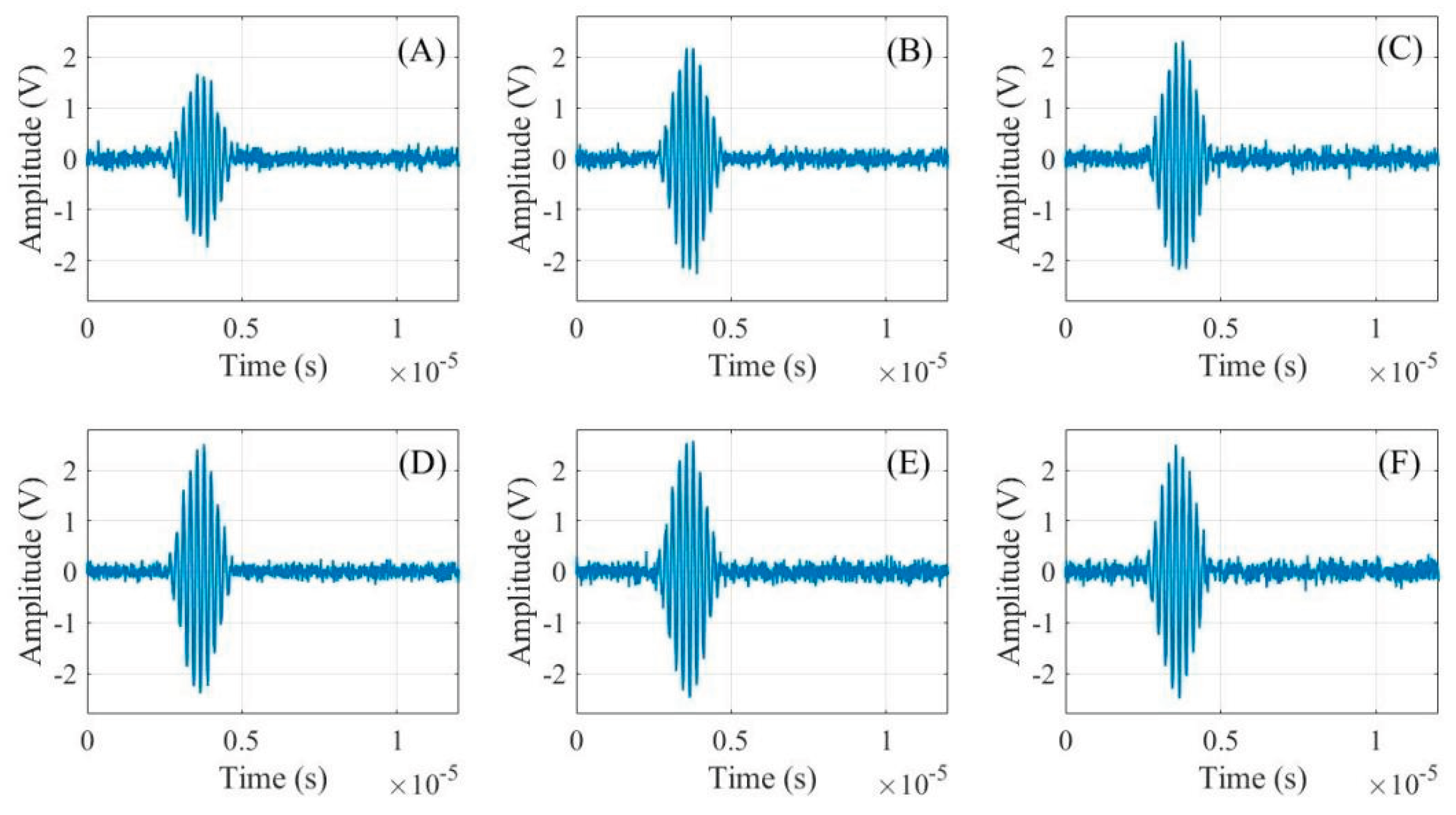
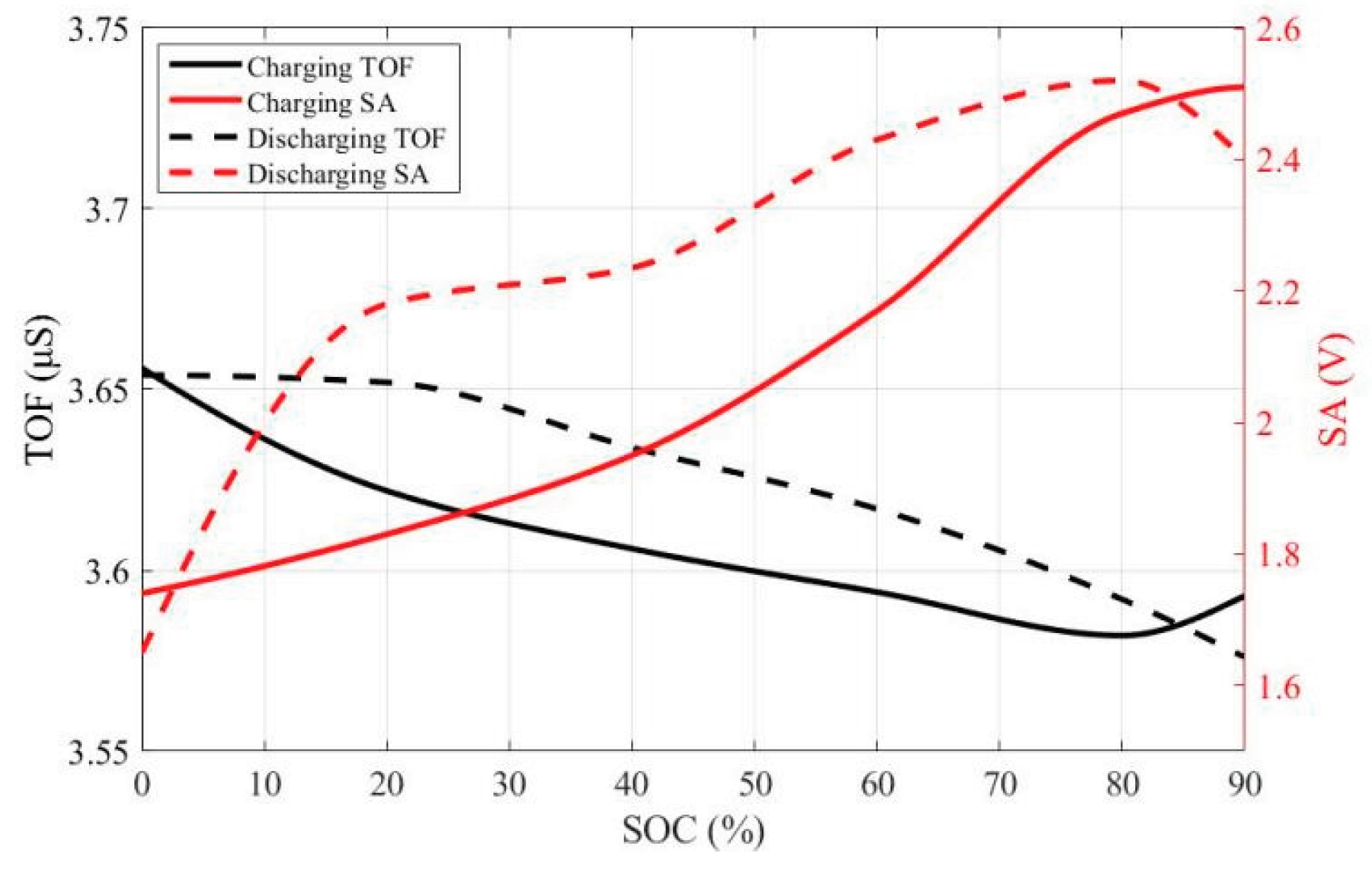
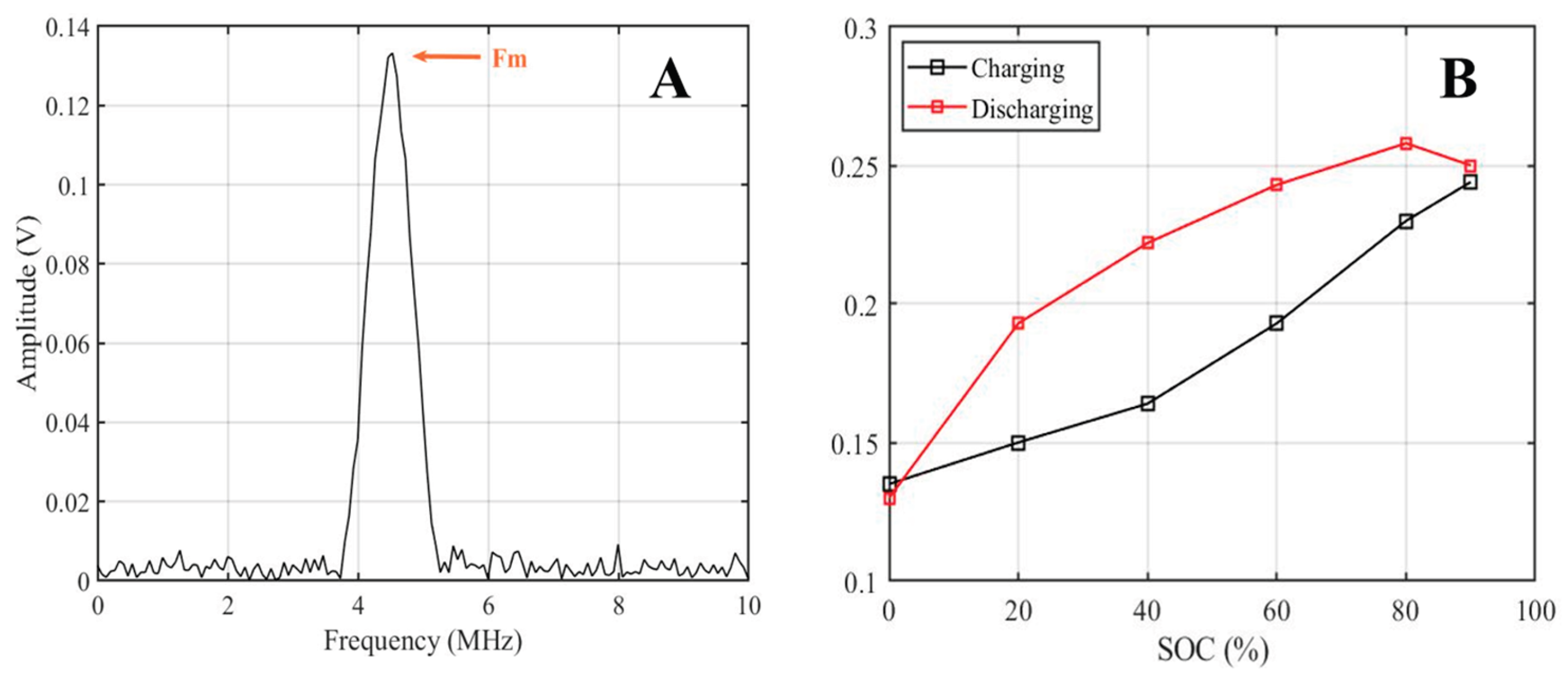
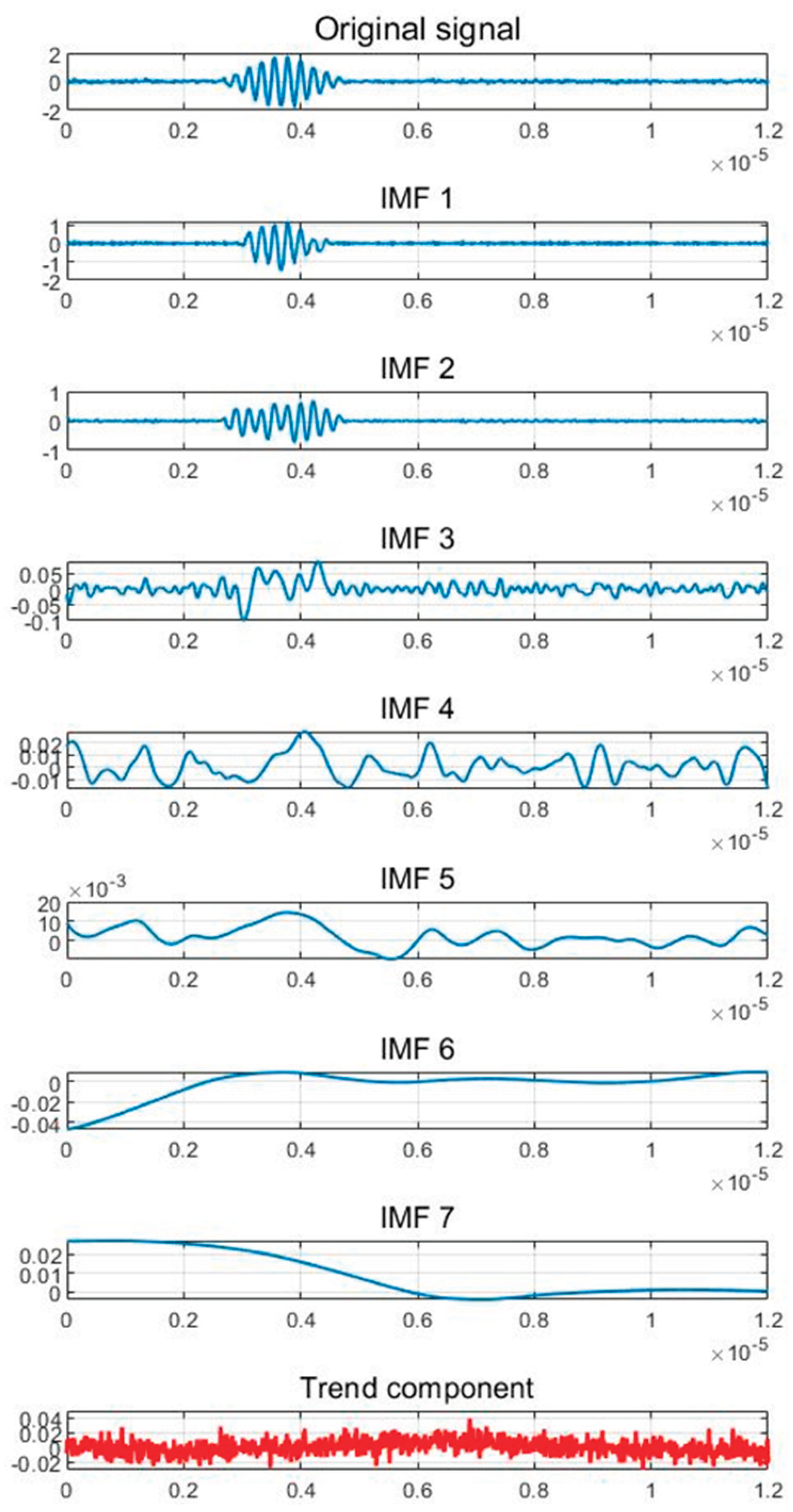
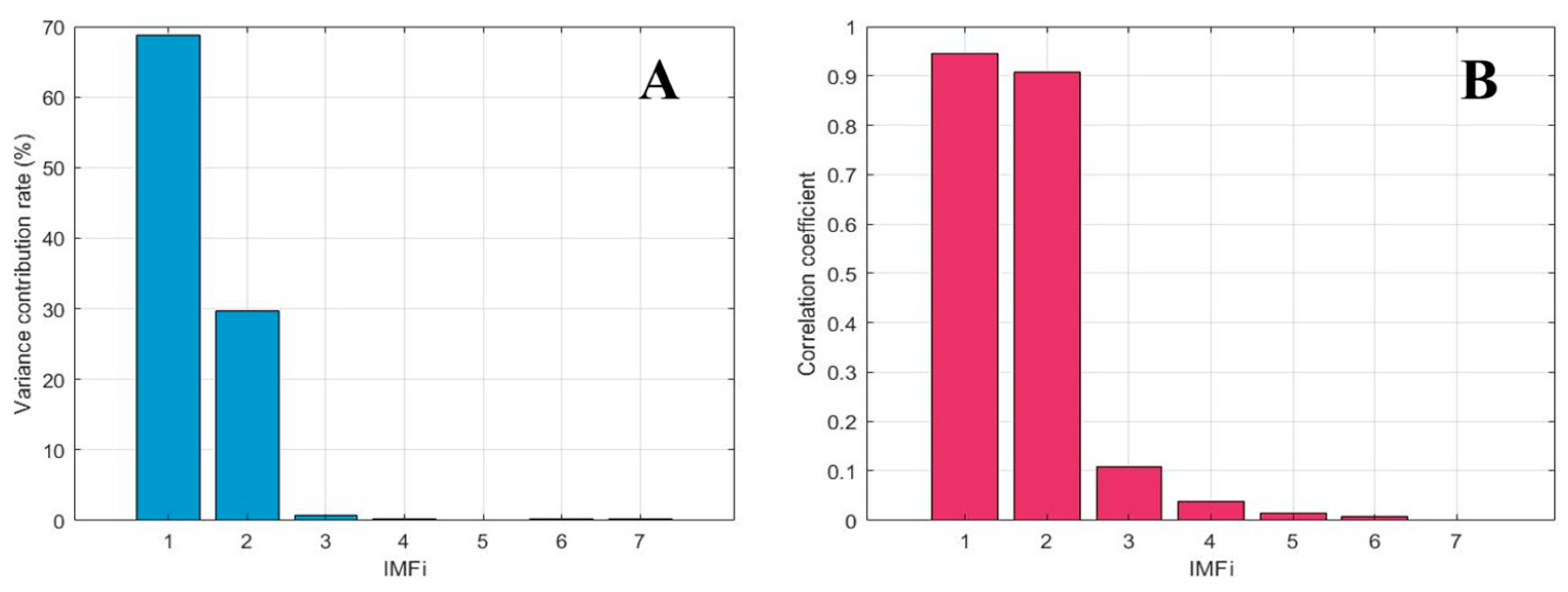
| Parameter | IMF1 | IMF2 | IMF3 |
| s | 0.6322 | 0.4258 | 0.3361 |
| k | 0.9012 | 0.8344 | 0.4321 |
| cf | 0.7125 | 0.6588 | 0.4003 |
| mf | 0.9255 | 0.9012 | 0.8143 |
| sf | 0.6827 | 0.4876 | 0.3256 |
| if | 0.2101 | 0.1571 | 0.0145 |
| Evaluation Indicator | State | LSTM | DNN | CNN |
| RMSE (%) | charging | 0.8901 | 1.1043 | 1.3008 |
| discharging | 0.4153 | 0.9933 | 1.2019 | |
| MAE (%) | charging | 0.6419 | 1.0347 | 0.9473 |
| discharging | 0.3541 | 1.1021 | 0.9215 | |
| R2 | charging | 0.9904 | 0.9254 | 0.9016 |
| discharging | 0.9913 | 0.9417 | 0.9570 |
| Evaluation Indicator | State | 3 features | 5 features |
| RMSE (%) | charging | 1.4020 | 1.2017 |
| discharging | 1.2017 | 1.1340 | |
| MAE (%) | charging | 1.0834 | 0.9301 |
| discharging | 0.9893 | 0.8936 | |
| R2 | charging | 0.9214 | 0.9471 |
| discharging | 0.9331 | 0.9601 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




