Submitted:
20 April 2026
Posted:
21 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
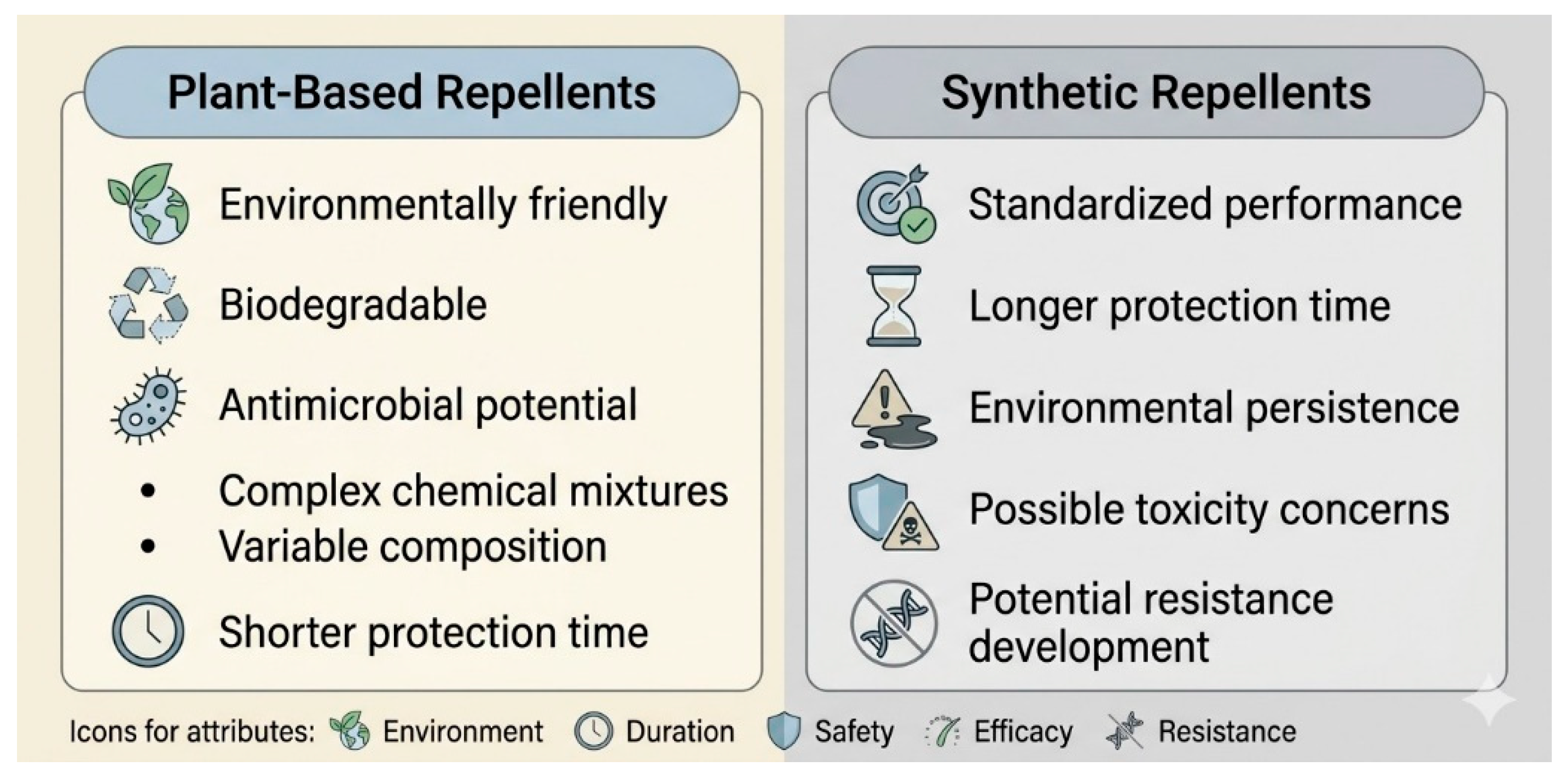
3.1. Synthetic Repellents
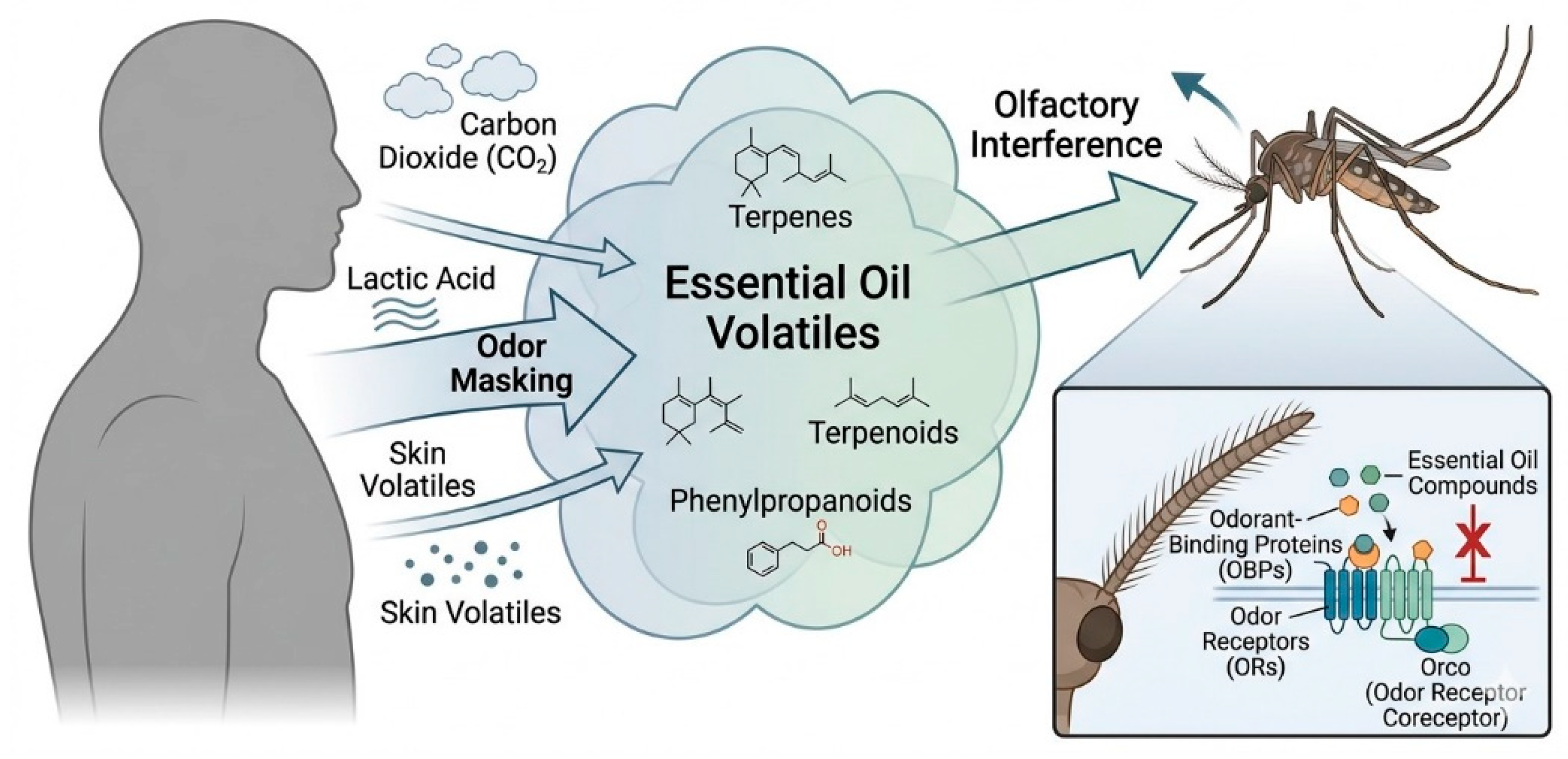
3.2. Essential Oils as Natural Repellents
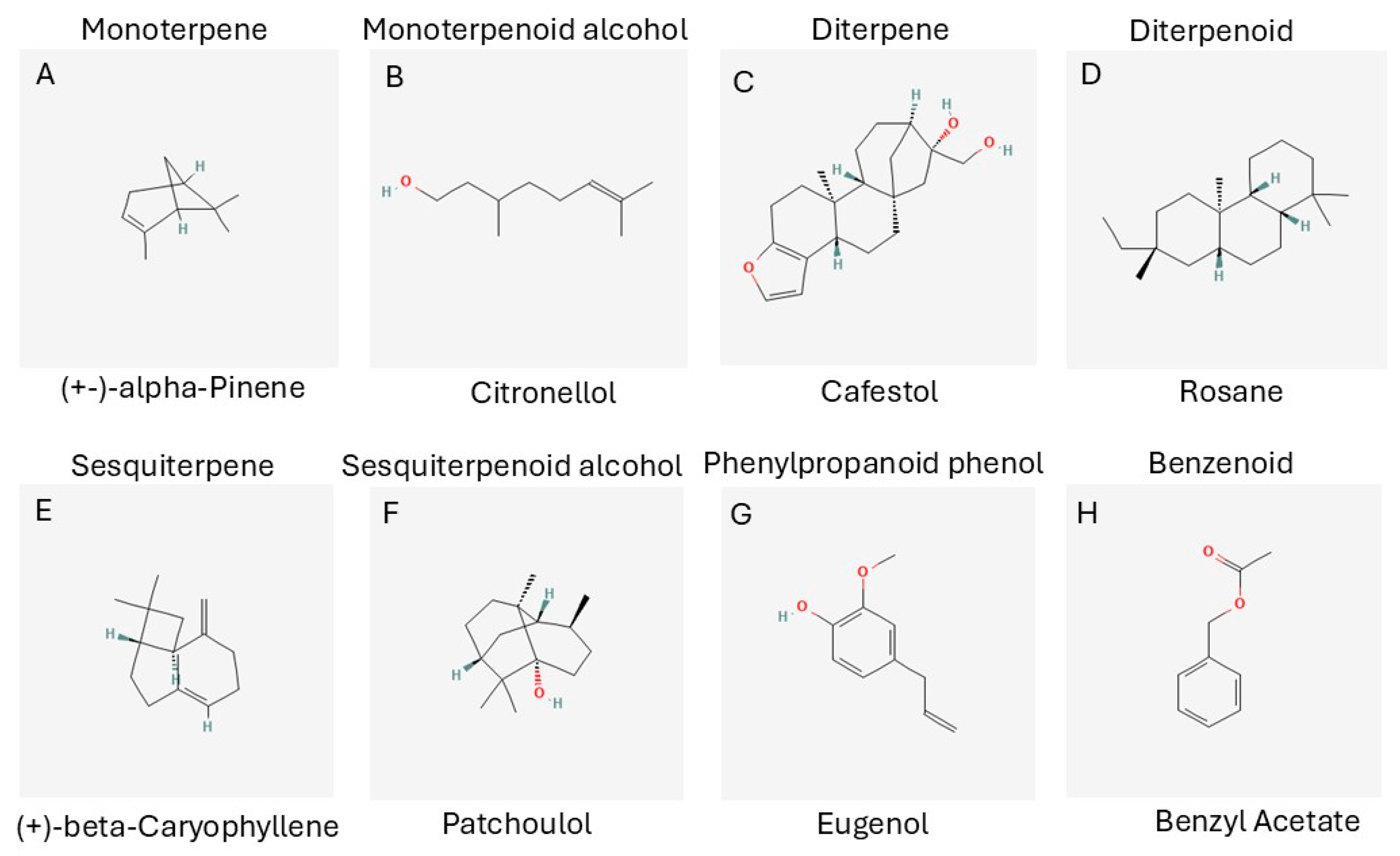
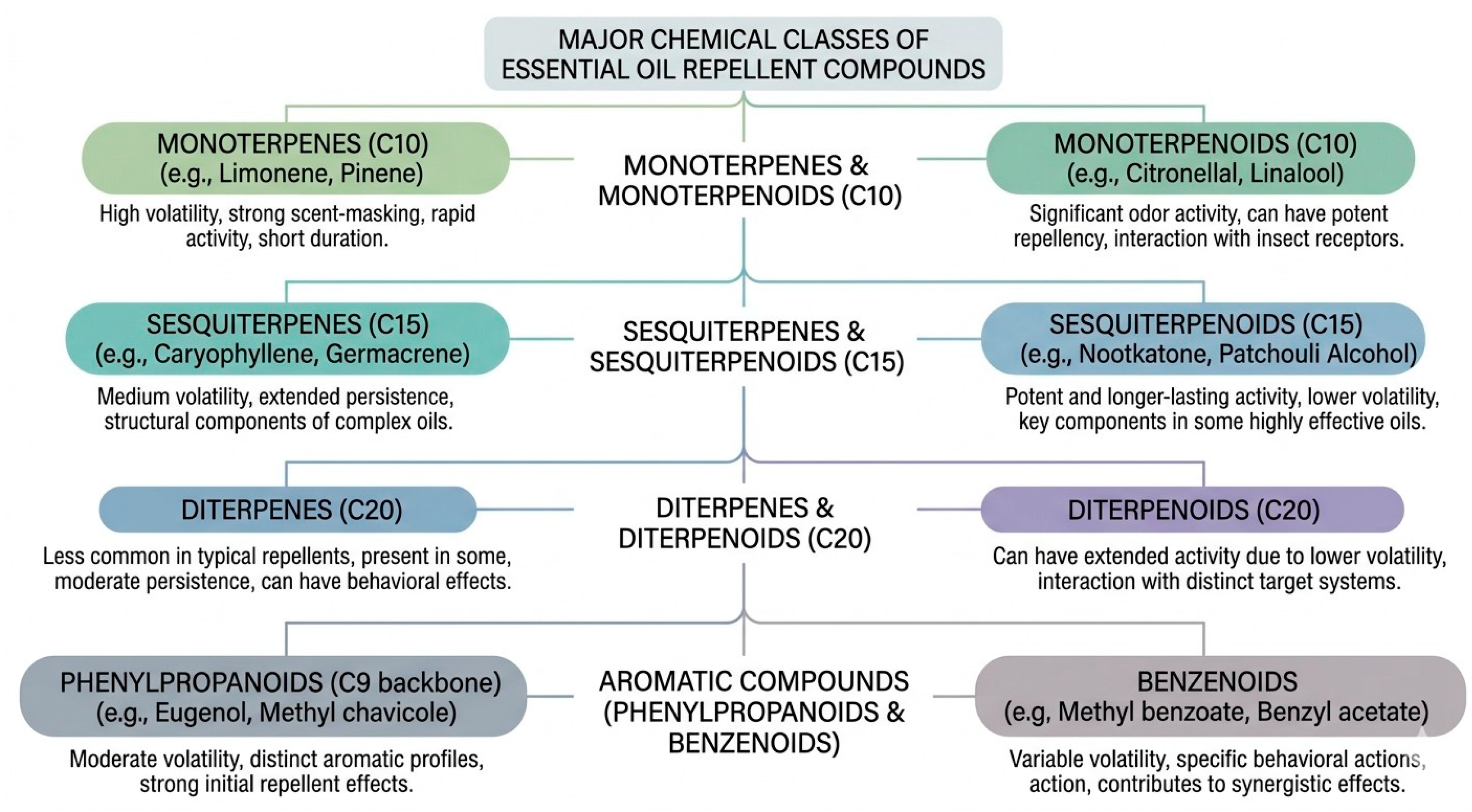
3.3. Chemical Classes of Essential Oils
3.3.1. Terpenes and Terpenoids
3.3.2. Monoterpenes and Monoterpenoids
3.3.3. Diterpenes and Diterpenoids
3.3.4. Sesquiterpenes and Sesquiterpenoids
3.3.5. Phenylpropanoids
3.3.6. Benzenoids
3.4. Chemical Composition and Dominant Constituents of Essential Oils as Repellents
3.5. Essential Oils as Insect Repellents
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| EPA WHO DEET EPA WHO NHANES DDT OBPs Orco ORx PMD TRPA1 SAR |
Environmental Protection Agency World Heath Organization N,N-diethyl-meta-toluamide Environmental Protection Agency World Health Organization National Health and Nutrition Examination Survey Dichlorodiphenyltrichloroethane Odorant-binding proteins Odorant Receptor Co-receptor Variable ligand-binding odorant receptor subunits Para-menthane-3,8-diol Transient Receptor Potential Ankyrin 1 Structure Activity Relationship |
References
- Dahiya, N.; Yadav, M.; Kataria, D.; Janjoter, S.; Sehrawat, N. Mosquito-based transmission-blocking vaccine candidates for malaria: Progress, challenges, and innovations. Molecular Biology Reports 2025, 52, 908. [Google Scholar] [CrossRef]
- Tang, J.; Amin, M. A.; Campian, J. L. Past, present, and future of viral vector vaccine platforms: A comprehensive review. Vaccines 2025, 13(5), 524. [Google Scholar] [CrossRef]
- Diaz, J.H. Chemical and plant-based insect repellents: Efficacy, safety, and toxicity. Wilderness Environ Med. 2016, 27(1), 153–63. [Google Scholar] [CrossRef] [PubMed]
- Fradin, M. S.; Day, J. F. Comparative efficacy of insect repellents against mosquito bites. The New England Journal of Medicine 2002, 347(1), 13–18. [Google Scholar] [CrossRef]
- Yadav, D. K.; Rathee, S.; Sharma, V.; Patil, U. K. A comprehensive review on insect repellent agents: Medicinal plants and synthetic compounds. In Anti-Inflammatory & Anti-Allergy Agents in Medicinal Chemistry; 2024. [Google Scholar] [CrossRef]
- Hagstrum, D. W.; Phillips, T. W.; Cuperus, G. (Eds.) Stored product protection (S156); Kansas State University, 2012; Available online: https://bookstore.ksre.ksu.edu/pubs/stored-product.
- Ghali, H.; Albers, S. E. An updated review on the safety of N,N-diethyl-meta toluamide insect repellent use in children and the efficacy of natural alternatives. Pediatric Dermatology 2024, 41(3), 403–409. [Google Scholar] [CrossRef]
- Lee, M. Y. Essential oils as repellents against arthropods. In BioMed Research International; 2018; pp. 1–9. [Google Scholar] [CrossRef]
- Hazarika, H.; Krishnatreyya, H. Technological advancements in mosquito repellents: Challenges and opportunities in plant-based repellents. Acta Parasitologica 2025, 70(3), 117. [Google Scholar] [CrossRef]
- Tavares, M.; da Silva, M. R. M.; de Oliveira de Siqueira, L. B.; Rodrigues, R. A. S.; Bodjolle d’Almeida, L.; Dos Santos, E. P.; Ricci-Júnior, E. Trends in insect repellent formulations: A review. International Journal of Pharmaceutics 2018, 539(1–2), 190–209. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Chen, Y.; Gao, M.; Li, W.; Zhao, Y.; Wang, Y. An updated review on essential oils from Lauraceae plants: Chemical composition and genetic characteristics of biosynthesis. International Journal of Molecular Sciences 2025, 26(12), 5690. [Google Scholar] [CrossRef]
- Shields, V. D. C.; Hildebrand, J. G. Recent advances in insect olfaction, specifically regarding the morphology and sensory physiology of antennal sensilla of the female sphinx moth Manduca sexta. Microscopy Research and Technique 2001, 55(5), 307–329. [Google Scholar] [CrossRef] [PubMed]
- Heinbockel, T.K.; Alzyoud, R.O.; Raheel, S.; Shields, V.D.C. Selected Essential Oils Act as Repellents Against the House Cricket, Acheta domesticus. Insects 2026, 17(1), 106. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sritabutra D, Soonwera M. Repellent activity of herbal essential oils against Aedes aegypti (Linn.) and Culex quinquefasciatus (Say.). Asian Pac J Trop Dis. 2013 Aug;3(4):271–6. U.S. Environmental Protection Agency. (n.d.). DEET (N,N-Diethyl-meta-toluamide). Retrieved November 6, 2025, from https://www.epa.gov/insect-repellents/deet. [CrossRef] [PubMed Central]
- Sathantriphop, S.; Kongmee, M.; Bangs, M. J. The effects of plant essential oils on escape response and mortality rate of Aedes aegypti and Anopheles minimus. Journal of Vector Ecology 2015, 40(2), 318–326. [Google Scholar] [CrossRef] [PubMed]
- U.S. Environmental Protection Agency. DEET—Registration review: Human health and environmental risk conclusions [Docket EPA-HQ-OPP-2012-0162-0012]. 2014. Available online: https://www.regulations.gov/document/EPA-HQ-OPP-2012-0162-0012.
- Almeida, A. R.; Oliveira, N. D.; Pinheiro, F. A. S. D.; Morais, W. A.; Ferreira, L. S. Challenges encountered by natural repellents: Since obtaining until the final product. Pesticide Biochemistry and Physiology 2023, 195, 105538. [Google Scholar] [CrossRef]
- Calafat, A. M.; Baker, S. E.; Wong, L. Y.; Bishop, A. M.; Morales-A., P.; Valentin-Blasini, L. Novel exposure biomarkers of N,N-diethyl-m-toluamide (DEET): Data from the 2007 2010 National Health and Nutrition Examination Survey. Environment International 2016, 92–93, 398–404. [Google Scholar] [CrossRef]
- Wickerham, E. L.; Lozoff, B.; Shao, J.; Kaciroti, N.; Xia, Y.; Meeker, J. D. Reduced birth weight in relation to pesticide mixtures detected in cord blood of full-term infants. Environment International 2012, 47, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Fang, W.; Krauss, M.; Brack, W.; Wang, Z.; Li, F.; Zhang, X. Screening hundreds of emerging organic pollutants in surface water from the Yangtze River Delta: Occurrence, distribution, ecological risk. Environmental Pollution 2018, 241, 484–493. [Google Scholar] [CrossRef] [PubMed]
- Osimitz, T.G.; Murphy, J.V. Neurological effects associated with use of the insect repellent N,N-diethyl-m-toluamide (DEET). J Toxicol Clin Toxicol. 1997, 35(5), 435–41. [Google Scholar] [CrossRef] [PubMed]
- Lipscomb, J. W.; Kramer, J. E.; Leikin, J. B. Seizure following brief exposure to the insect repellent N,N-diethyl-m-toluamide. Annals of Emergency Medicine 1992, 21(3), 315–317. [Google Scholar] [CrossRef]
- Shin, N.; Lascarez-Lagunas, L.I.; Henderson, A.L.; Martínez-García, M.; Karthikraj, R.; Barrera, V.; Sui, S.H.; Kannan, K.; Colaiácovo, M.P. Altered gene expression linked to germline dysfunction following exposure of Caenorhabditis elegans to DEET. iScience 2024, 27(1), 108699. [Google Scholar] [CrossRef]
- Yan, S.; Wang, J.; Xu, J.; Jiang, W.; Xiong, M.; Cao, Z.; Wang, Y.; Wang, Z.; Zhang, T.; Wang, Z.; Sun, C.; Hou, S.; Wei, W. Exposure to N,N-diethyl-m-toluamide and cardiovascular diseases in adults. Frontiers in Public Health 2022, 10, 922005. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, C.; Sun, Y.; Tang, S.; Lei, Y.; Zhang, W.; Cheng, B.; Zhao, Y.; Luo, Q. Toxicity assessment of N,N-diethyl-meta-toluamide (DEET) in zebrafish embryos. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology 2025, 297, 110293. [Google Scholar] [CrossRef]
- Cui, Q.; Zhu, X.; Guan, G.; Hui, R.; Zhu, L.; Wang, J. Association of N,N-diethyl-m toluamide (DEET) with obesity among adult participants: Results from NHANES 2007–2016. Chemosphere 2022, 307, 135669. [Google Scholar] [CrossRef]
- Wei, C.; He, J.; Wei, Z.; Huang, Y.; Xiong, M.; Deng, C.; Chen, Z.; Li, W.; Zhang, X. Association between N,N-diethyl-m-toluamide exposure and the odds of kidney stones in U.S. adults: A population-based study. Frontiers in Public Health 2023, 11, 1248674. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Zhu, X.; Xing, Y.; Guan, G.; Zhang, Y.; Hui, R.; Cui, Q.; Liu, Z.; Zhu, L. Association of N,N-diethyl-m-toluamide (DEET) with hyperuricemia among adult participants. Chemosphere 2023, 338, 139320. [Google Scholar] [CrossRef]
- Liu, L.; Qin, W.; Nie, L.; Wang, X.; Dong, X. Correlation between environmental DEET exposure and the mortality rate of cancer survivors: A large-sample cross-sectional investigation. BMC Cancer 2024, 24, 1410. [Google Scholar] [CrossRef] [PubMed]
- Li, B.A.; Li, B.M.; Bao, Z.; Li, Q.; Xing, M.; Li, B. Dichlorodiphenyltrichloroethane for malaria and agricultural uses and its impacts on human health. Bull Environ Contam Toxicol 2023, 111(4), 45. [Google Scholar] [CrossRef] [PubMed]
- Turek, C.; Stintzing, F.C. Stability of essential oils: A review. Comprehensive Reviews in Food Science and Food Safety 2013, 12, 40–53. Available online: https://www.sciencedirect.com/science/article/pii/S0031942202001747. [CrossRef]
- Wu, W.; Yang, Y.; Feng, Y.; Ren, X.; Li, Y.; Li, W.; Huang, J.; Kong, L.; Chen, X.; Lin, Z. Study of the repellent activity of 60 essential oils and their main constituents against Aedes albopictus, and nano-formulation development. Insects 2022, 13(12), 1077. [Google Scholar] [CrossRef] [PubMed]
- Nerio, L. S.; Olivero-Verbel, J.; Stashenko, E. Repellent activity of essential oils: A review. Bioresource Technology 2010, 101(1), 372–378. [Google Scholar] [CrossRef]
- Bruce, T. J.; Pickett, J. A. Perception of plant volatile blends by herbivorous insects: Finding the right mix. Phytochemistry 2011, 72(13), 1605–1611. [Google Scholar] [CrossRef]
- Kamaraj, C.; Satish Kumar, R. C.; Al-Ghanim, K. A.; Nicoletti, M.; Sathiyamoorthy, V.; Sarvesh, S.; Ragavendran, C.; Govindarajan, M. Novel essential oils blend as a repellent and toxic agent against disease-transmitting mosquitoes. Toxics 2023, 11(6), 517. [Google Scholar] [CrossRef]
- Uniyal, A.; Tikar, S. N.; Mendki, M. J.; Singh, R.; Shukla, S. V.; Agrawal, O. P.; Veer, V.; Sukumaran, D. Behavioral response of Aedes aegypti mosquito towards essential oils using olfactometer. Journal of Arthropod-Borne Diseases 2016, 10(3), 370–380. [Google Scholar]
- Ray, A. Reception of odors and repellents in mosquitoes. Current Opinion in Neurobiology 2015, 34, 158–164. [Google Scholar] [CrossRef]
- Webster, B.; Lacey, E. S.; Cardé, R. T. Waiting with bated breath: Opportunistic orientation to human odor in the malaria mosquito, Anopheles gambiae, is modulated by minute changes in carbon dioxide concentration. Journal of Chemical Ecology 2015, 41(1), 59–66. [Google Scholar] [CrossRef] [PubMed]
- Paluch, G.; Bartholomay, L.; Coats, J. Mosquito repellents: A review of chemical structure diversity and olfaction. Pest Management Science 2010, 66(9), 925–935. [Google Scholar] [CrossRef]
- Meier, C. J.; Nguyen, M. N.; Potter, C. J. Making scents of mosquito repellents. Trends in Parasitology 2025, 41(4), 280–289. [Google Scholar] [CrossRef] [PubMed]
- Dhifi, W.; Bellili, S.; Jazi, S.; Bahloul, N.; Mnif, W. Essential oils' chemical characterization and investigation of some biological activities: A critical review. Medicines (Basel) 2016, 3(4), 25. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Abbas, M. G.; Binyameen, M.; Azeem, M.; Majeed, S.; Sarwar, Z. M.; Nazir, A.; Sharif, M. M. I.; Parveen, A.; Mozūratis, R. Chemical analysis, repellent, larvicidal, and oviposition deterrent activities of plant essential oils against Aedes aegypti, Anopheles gambiae, and Culex quinquefasciatus. Frontiers in Insect Science 2025, 5, 1582669. [Google Scholar] [CrossRef]
- Bohbot, J.D.; Dickens, J.C. Insect repellents: Modulators of mosquito odorant receptor activity. PLoS ONE 2010, 5(8), e12138. [Google Scholar] [CrossRef] [PubMed]
- Tsitoura, P.; Koussis, K.; Iatrou, K. Inhibition of Anopheles gambiae odorant receptor function by mosquito repellents. J Biol Chem. 2015, 290(12), 7961–72. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Farina, P.; Conti, B. Liabilities of essential oils as insect repellents. Current Opinion in Environmental Science & Health 2024, 40, 100564. Available online: https://www.sciencedirect.com/science/article/pii/S2468584424000345. [CrossRef]
- Konopka, J. K.; Task, D.; Afify, A.; Raji, J.; Deibel, K.; Maguire, S.; Lawrence, R.; Potter, C. J. Olfaction in Anopheles mosquitoes. Chemical Senses 2021, 46, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Masyita, A.; Mustika Sari, R.; Dwi Astuti, A.; Yasir, B.; Rahma Rumata, N.; Emran, T.B.; Nainu, F.; Simal-Gandara, J. Terpenes and terpenoids as main bioactive compounds of essential oils, their roles in human health and potential application as natural food preservatives. Food Chem X 2022, 13, 100217. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Katz, T. M.; Miller, J. H.; Hebert, A. A. Insect repellents: Historical perspectives and new developments. Journal of the American Academy of Dermatology 2008, 58(5), 865–871. [Google Scholar] [CrossRef] [PubMed]
- Corzo-Gómez, J. C.; Espinosa-Juárez, J. V.; Ovando-Zambrano, J. C.; Briones-Aranda, A.; Cruz Salomón, A.; Esquinca-Avilés, H. A. A review of botanical extracts with repellent and insecticidal activity and their suitability for managing mosquito-borne disease risk in Mexico. Pathogens 2024, 13(9), 737. [Google Scholar] [CrossRef]
- Degenhardt, J.; Gershenzon, J.; Baldwin, I.T.; Kessler, A. Attracting friends to feast on foes: engineering terpene emission to make crop plants more attractive to herbivore enemies. In Current Opinion in Biotechnology; ISSN 0958-1669, 2003; Volume 14, 2, pp. 169–176. Available online: https://www.sciencedirect.com/science/article/pii/S0958166903000259. [CrossRef]
- Gershenzon, J.; Dudareva, N. The function of terpene natural products in the natural world. Nat Chem Biol 2007, 3, 408–414. [Google Scholar] [CrossRef]
- Sadgrove, N.J.; Padilla-González, G.F.; Phumthum, M. Fundamental Chemistry of Essential Oils and Volatile Organic Compounds, Methods of Analysis and Authentication. Plants (Basel) 2022, 11(6), 789. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sharmeen, J.B.; Mahomoodally, F.M.; Zengin, G.; Maggi, F. Essential Oils as Natural Sources of Fragrance Compounds for Cosmetics and Cosmeceuticals. Molecules 2021, 26(3), 666. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ulland, S.; Borg-Karlson, E. A.-K.; Mustaparta, H. Discrimination between enantiomers of linalool by olfactory receptor neurons in the cabbage moth Mamestra brassicae (L.). Chem Senses Epub (2006) Feb 22. 2006, 31(4), 325–34. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, M.; Pereira, D.M. The Chemical Space of Terpenes: Insights from Data Science and AI. Pharmaceuticals (Basel) 2023, 16(2), 202. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Handbook of Essential Oils: Science, Technology, and Applications, 3rd ed.; Başer, K. H. C., Buchbauer, G., Eds.; CRC Press, 2020; ISBN 9780815370963; eBook ISBN 9781351246460. [Google Scholar]
- Benomari, F.Z.; Sarazin, M.; Chaib, D.; Pichette, A.; Boumghar, H.; Boumghar, Y.; Djabou, N. Chemical Variability and Chemotype Concept of Essential Oils from Algerian Wild Plants. Molecules 2023, 28(11), 4439. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Khan, M.H.; Dar, N.A.; Alie, B.A.; Dar, S.A.; Lone, A.A.; Mir, G.H.; Fayaz, U.; Ali, S.; Tyagi, A.; El-Sheikh, M.A.; Alansi, S. Unraveling the Variability of Essential Oil Composition in Different Accessions of Bunium persicum Collected from Different Temperate Micro-Climates. Molecules 2023, 28, 2404. [Google Scholar] [CrossRef]
- Sadgrove, N.; Jones, G. A Contemporary Introduction to Essential Oils: Chemistry, Bioactivity and Prospects for Australian Agriculture. Agriculture 2015, 5, 48–102. [Google Scholar] [CrossRef]
- Zielińska-Błajet, M.; Feder-Kubis, J. Monoterpenes and their derivatives- Recent development in biological and medical applications. Int J Mol Sci 2020, 21(19), 7078. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bedini, S.; Flamini, G.; Ascrizzi, R.; Venturi, F.; Ferroni, G.; Bader, A.; Giraldi, J.; Conti, B. Essential oils sensory quality and their bioactivity against the mosquito Aedes albopictus. Sci Rep 2018, 8, 17857. [Google Scholar] [CrossRef] [PubMed]
- Maia, M.F.; Moore, S.J. Plant-based insect repellents: a review of their efficacy, development and testing. Malar J 2011, 10 Suppl 1(Suppl 1), S11. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- İşcan, G. Antibacterial and anticandidal activities of common essential oil constituents. Rec. Nat. Prod. 11(4), 374–388. Available online: https://acgpubs.org/doc/2018080514581047-RNP-EO_1701-003.pdf.
- Müller, G.C.; Junnila, A.; Butler, J.; Kravchenko, V.D.; Revay, E.E.; Weiss, R.W.; Schlein, Y. Efficacy of the botanical repellents geraniol, linalool, and citronella against mosquitoes. J Vector Ecol. 2009, 34(1), 2–8. [Google Scholar] [CrossRef] [PubMed]
- Carroll, S.P.; Loye, J. PMD, a registered botanical mosquito repellent with deet-like efficacy. J Am Mosq Control Assoc. 2006, 22(3), 507–14. [Google Scholar] [CrossRef] [PubMed]
- Melo, N.; Capek, M.; Arenas, O.M.; Afify, A.; Yilmaz, A.; Potter, C.J.; Laminette, P.J.; Para, A.; Gallio, M.; Stensmyr, M.C. The irritant receptor TRPA1 mediates the mosquito repellent effect of catnip. Curr Biol. 2021, 31(9), 1988–1994.e5. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Birkett, M.A.; Hassanali, A.; Hoglund, S.; Pettersson, J.; Pickett, J.A. Repellent activity of catmint, Nepeta cataria, and iridoid nepetalactone isomers against Afro-tropical mosquitoes, ixodid ticks and red poultry mites. Phytochemistry 2011, 72(1), 109–14. [Google Scholar] [CrossRef] [PubMed]
- Reichert, W.; Ejercito, J.; Guda, T.; Dong, X.; Wu, Q.; Ray, A.; Simon, J. Repellency assessment of Nepeta cataria essential oils and isolated nepetalactones on Aedes aegypti. Sci Rep 2019, 9, 1524. [Google Scholar] [CrossRef] [PubMed]
- Batume, C.; Mulongo, I.M.; Ludlow, R.; Ssebaale, J.; Randerso, P.; Pickett, J.A.; Mukisa, I.M.; Scofield, S. Evaluating repellence properties of catnip essential oil against the mosquito species Aedes aegypti using a Y-tube olfactometer. Sci Rep 2024, 14, 2269. [Google Scholar] [CrossRef]
- Fichan; Larroche, C.; Gros, J. B. Water solubility, vapor pressure, and activity coefficients of terpenes and terpenoids. Journal of Chemical & Engineering Data 1998, 44(1), 56–62. [Google Scholar] [CrossRef]
- Pub Chem. National Library of Medicine NIH. Available online: https://pubchem.ncbi.nlm.nih.gov/ (accessed on 2/23/26).
- Keeling, C.; Bohlmann, J. Diterpene resin acids in conifers. Phytochemistry 2006, 67(22), Pages 2415–2423. Available online: https://www.sciencedirect.com/science/article/pii/S0031942206005449. [CrossRef]
- Sosa, M.E.; Tonn, C.E.; Giordano, O.S. Insect antifeedant activity of clerodane diterpenoids. J Nat Prod. 1994, 57(9), 1262–5. [Google Scholar] [CrossRef] [PubMed]
- Li, B. A.; Li, B. M.; Bao, Z.; Li, Q.; Xing, M.; Li, B. Dichlorodiphenyltrichloroethane for malaria and agricultural uses and its impacts on human health. Bulletin of Environmental Contamination and Toxicology 2023, 111(4), 45. [Google Scholar] [CrossRef]
- Gebbinck, E.A.K.; Jansen, B.J.M. Jansen; de Groot, A. Insect antifeedant activity of clerodane diterpenes and related model compounds. Phytochemistry 61(7), 2002, 737–770. [CrossRef]
- Li, R.; Morris-Natschke, S.L.; Lee, K.H. Clerodane diterpenes: sources, structures, and biological activities. Nat Prod Rep. 2016, 33(10), 1166–226. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Scheffrahn, R.H.; Hsu, R.C.; Su, N.Y.; Huffman, J.B.; Midland, S.L.; Sims, J.J. Allelochemical resistance of bald cypress, Taxodium distichum, heartwood to the subterranean termite, Coptotermes formosanus. J Chem Ecol. 1988, 14(3), 765–76. [Google Scholar] [CrossRef] [PubMed]
- Kusumoto, N.; Ashitani, T.; Hayasaka, Y.; Murayama, T.; Ogiyama, K.; Takahashi, K. Antitermitic activities of abietane-type diterpenes from Taxodium distichum cones. J Chem Ecol. 2009, 35(6), 635–42. [Google Scholar] [CrossRef] [PubMed]
- Nieto, G.; Ros, G.; Castillo, J. Antioxidant and Antimicrobial Properties of Rosemary (Rosmarinus officinalis, L.): A Review. Medicines (Basel) 2018, 5(3), 98. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- McAndrew, B.A. Sesquiterpenoids: The lost dimension of perfumery. Perfumer & Flavorist 1992, 17, 1–12. Available online: https://img.perfumerflavorist.com/files/base/allured/all/document/2016/03/pf.9229.pdf.
- Barreto, I.C.; de Almeida, A.S.; Sena Filho, J.G. Taxonomic Insights and Its Type Cyclization Correlation of Volatile Sesquiterpenes in Vitex Species and Potential Source Insecticidal Compounds: A Review. Molecules 2021, 26(21), 6405. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Christensson, J.B.; Forsström, P.; Wennberg, A.-M.; Karlberg, A.-T.; Matura, M. Air oxidation increases skin irritation from fragrance terpenes. Contact Dermatitis Digital Object Identifier (DOI). 2009. [Google Scholar] [CrossRef] [PubMed]
- Dittmar, D.; Schuttelaar, M.L.A. Contact sensitization to hydroperoxides of limonene and linalool: Results of consecutive patch testing and clinical relevance. Contact Dermatitis 2019, 80(2), 101–109. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Fichan, I.; Larroche, C.; Gros, J. B. Water solubility, vapor pressure, and activity coefficients of terpenes and terpenoids. J. Chem. Eng. Data 1999, 44, 56–62. [Google Scholar] [CrossRef]
- Clarkson, T.C.; Janich, A.J.; Sanchez-Vargas, I.; Markle, E.D.; Gray, M.; Foster, J.R.; Black, W.C., IV; Foy, B.D.; Olson, K.E. Nootkatone Is an Effective Repellent against Aedes aegypti and Aedes albopictus. Insects 2021, 12, 386. [Google Scholar] [CrossRef]
- Gokulakrishnan, J.; Kuppusamy, E.; Shanmugam, D.; Appavu, A.; Kaliyamoorthi, K. Pupicidal and repellent activities of Pogostemon cablin essential oil chemical compounds against medically important human vector mosquitoes. Asian Pac J Trop Dis. 2013, 3(1), 26–31. [Google Scholar] [CrossRef] [PubMed Central]
- da Silva, R.C.; Milet-Pinheiro, P.; Bezerra da Silva, P.C.; da Silva, A.G.; da Silva, M.V.; Navarro, D.M.; da Silva, N.H. (E)-caryophyllene and α-humulene: Aedes aegypti oviposition deterrents elucidated by gas chromatography-electrophysiological assay of Commiphora leptophloeos leaf oil. PLoS One 2015, 10(12), e0144586. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Liakakou, A.; Angelis, A.; Papachristos, D.P.; Fokialakis, N.; Michaelakis, A.; Skaltsounis, L.A. Isolation of volatile compounds with repellent properties against Aedes albopictus (Diptera: Culicidae) Using CPC Technology. Molecules 2021, 26(11), 3072. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kunert, G.; Reinhold, C.; Gershenzon, J. Constitutive emission of the aphid alarm pheromone, (E)-β-farnesene, from plants does not serve as a direct defense against aphids. BMC Ecol 2010, 10, 23. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rasmann, S.; Köllner, T.G.; Degenhardt, J.; Hiltpold, I.; Toepfer, S.; Kuhlmann, U.; Gershenzon, J.; Turlings, T.C. Recruitment of entomopathogenic nematodes by insect-damaged maize roots. Nature 2005, 434(7034), 732–7. [Google Scholar] [CrossRef] [PubMed]
- Noel, J.P.; Austin, M.B.; Bomati, E.K. Structure-function relationships in plant phenylpropanoid biosynthesis. Curr Opin Plant Biol. 2005, 8(3), 249–53. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jankowska, M.; Rogalska, J.; Wyszkowska, J.; Stankiewicz, M. Molecular Targets for Components of Essential Oils in the Insect Nervous System-A Review. Molecules 2017, 23(1), 34. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- de Souza, R.F.; Amaro, T.R.; Palacio-Cortés, A.M.; da Silva, M.A.N.; Dionisio, J.F.; Pezenti, L.F.; Lopes, T.B.F.; Mantovani, M.S.; Zequi, J.A.C.; da Rosa, R. Comparative transcriptional analysis between susceptible and resistant populations of Aedes (Stegomyia) aegypti (Linnaeus, 1762) after malathion exposure. Mol Genet Genomics 2024, 299(1), 92. [Google Scholar] [CrossRef] [PubMed]
- Fabbro, S.D.; Nazzi, F. From Chemistry to Behavior. Molecular Structure and Bioactivity of Repellents against Ixodes ricinus Ticks. PLoS ONE 2013, 8(6), e67832. [Google Scholar] [CrossRef] [PubMed]
- Barrozo, M.M.; Santos, E.F.; Chagas, H.D.F.; Carvalho, R.A.; Silva, I.S.; de Souza Oliveira, A.; Faria, L.C.F.; Teixeira, A.L.C.; Zeringota, V.; Luz, H.R.; et al. Repellent Activity of the Botanical Compounds Thymol, Carvacrol, Nootkatone, and Eugenol Against Amblyomma sculptum Nymphs. Pathogens 2025, 14, 926. [Google Scholar] [CrossRef]
- Kuang, C.; Cao, J.; Zhou, Y.; Zhang, H.; Wang, Y.; Zhou, J. HL-TRP channel is required for various repellents for the parthenogenetic Haemaphysalis longicornis. Parasit Vectors 2025, 18(1), 139. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Badji, C.A.; Dorland, J.; Kheloul, L.; Bréard, D.; Richomme, P.; Kellouche, A.; Azevedo de Souza, C.R.; Bezerra, A.L.; Anton, S. Behavioral and Antennal Responses of Tribolium confusum to Varronia globosa Essential Oil and Its Main Constituents: Perspective for Their Use as Repellent. Molecules 2021, 26(15), 4393. [Google Scholar] [CrossRef] [PubMed]
- Muhlemann, J.K.; Klempien, A.; Dudareva, N. Floral volatiles: from biosynthesis to function. Plant Cell Environ 2014, 37(8), 1936–49. [Google Scholar] [CrossRef] [PubMed]
- Lv, M.; Zhang, L.; Wang, Y.; Ma, L.; Yang, Y.; Zhou, X.; Wang, L.; Yu, X.; Li, S. Floral volatile benzenoids/phenylpropanoids: biosynthetic pathway, regulation and ecological value. Hortic Res 2024, 11(10), uhae220. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Groux, R.; Hilfiker, O.; Gouhier-Darimont, C.; Peñaflor, M.F.; Erb, M.; Reymond, P. Role of methyl salicylate on oviposition deterrence in Arabidopsis thaliana. J Chem Ecol. 2014, 40(7), 754–9. [Google Scholar] [CrossRef] [PubMed]
- Gale, C.C.; Ferguson, B.; Rodriguez-Saona, C.; Shields, V.D.C.; Zhang, A. Evaluation of a push-pull strategy for spotted-wing Drosophila management in highbush blueberry. Insects 2024, 15(1), 47. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Stevens, G.R.; Clark, L. Bird repellents: development of avian-specific tear gases for resolution of human–wildlife conflicts. In International Biodeterioration & Biodegradation; ISSN 0964-8305, 1998; Volume 42, Issues 2–3, pp. Pages 153–160. Available online: https://www.sciencedirect.com/science/article/pii/S0964830598000560. [CrossRef]
- Hadani, A.; Ziv, M.; Rechav, Y. A laboratory study of tick repellents. 1977, 22(1), 53–59. [Google Scholar] [CrossRef]
- Hayden, M.L.; Rose, G.; Diduch, K.B.; Domson, P.; Chapman, M.D.; Heymann, P.W.; Platts-Mills, T.A. Benzyl benzoate moist powder: investigation of acaricidal [correction of acarical] activity in cultures and reduction of dust mite allergens in carpets. J Allergy Clin Immunol. 1992, 89(2), 536–45. [Google Scholar] [CrossRef] [PubMed]
- Siddique, A.; Naeem, J.; Ang, K.L.; Abid, S.; Xu, Z.; Khawar, M.T.; Saleemi, S.; Abdullah, M.; Adeel. Cinnamon and Eucalyptus Extracts: A Promising Natural Approach for Durable Mosquito-Repellent Fabrics with Multifunctionality. ACS Omega 2024, 9(40), 41468–41479. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lopez, A.D.; Whyms, S.; Luker, H.A.; Galvan, C.J.; Holguin, F.O.; Hansen, I.A. Repellency of essential oils and plant-derived compounds against Aedes aegypti mosquitoes. Insects 2025, 16(1), 51. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Barra, A. Factors affecting chemical variability of essential oils: a review of recent developments. Nat Prod Commun 2009, 4(8), 1147–54. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Liu, H.; Liu, M.; Liu, Z. Selective hydrogenation of citronellal to citronellol over polymer-stabilized noble metal colloids. Reactive and Functional Polymers 2000, 44. 21–29. [Google Scholar] [CrossRef]
- Fitri, N.; Riza, R.; Akbari, M.K.; Khonitah, N.; Fahmi, R.L.; Fatimah, I. Identification of citronella oil fractions as efficient bio-additive for diesel engine fuel. Designs 2022, 6, 15. [Google Scholar] [CrossRef]
- Müller, G.C.; Junnila, A.; Butler, J.; Kravchenko, V.D.; Revay, E.E.; Weiss, R.W.; Schlein, Y. Efficacy of the botanical repellents geraniol, linalool, and citronella against mosquitoes. J Vector Ecol. 2009, 34(1), 2–8. [Google Scholar] [CrossRef] [PubMed]
- Krause, S.T.; Liao, P.; Crocoll, C.; Boachon, B.; Förster, C.; Leidecker, F.; Wiese, N.; Zhao, D.; Wood, J.C.; Buell, C.R.; Gershenzon, J.; Dudareva, N.; Degenhardt, J. The biosynthesis of thymol, carvacrol, and thymohydroquinone in Lamiaceae proceeds via cytochrome P450s and a short-chain dehydrogenase. Proc Natl Acad Sci U S A 2021, 118(52), e2110092118. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hudaib, M.; Speroni, E.; Di Pietra, A.M.; Cavrini, V. GC/MS evaluation of thyme (Thymus vulgaris L.) oil composition and variations during the vegetative cycle. Journal of Pharmaceutical and Biomedical Analysis 2002, 29(4), Pages 691–700. Available online: https://www.sciencedirect.com/science/article/pii/S073170850200119X. [CrossRef]
- Zinno, P.; Guantario, B.; Lombardi, G.; Ranaldi, G.; Finamore, A.; Allegra, S.; Mammano, M.M.; Fascella, G.; Raffo, A.; Roselli, M. Chemical composition and biological activities of essential oils from Origanum vulgare genotypes belonging to the carvacrol and thymol chemotypes. Plants 2023, 12, 1344. [Google Scholar] [CrossRef]
- Pluhár, Z.; Kun, R.; Cservenka, J.; Neumayer, É; Tavaszi-Sárosi, S.; Radácsi, P.; Gosztola, B. Variations in essential oil composition and chemotype patterns of wild thyme (Thymus) species in the natural habitats of Hungary. Horticulturae 2024, 10, 150. [Google Scholar] [CrossRef]
- Alfikri, F.N.; Pujiarti, R.; Wibisono, M.G.; Hardiyanto, E.B. Yield, quality, and antioxidant activity of clove (Syzygium aromaticum L.) bud oil at the different phenological stages in young and mature trees. Scientifica (Cairo) 2020, 2020, 9701701. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Behbahani, B.A.; Falah, F.; Arab, F.A.; Vasiee, M.; Tabatabaee, F. Chemical composition and antioxidant, antimicrobial, and antiproliferative activities of Cinnamomum zeylanicum bark essential oil. Evid Based Complement Alternat Med. 2020 Apr 29;2020:5190603. [CrossRef] [PubMed] [PubMed Central]
- Lee M.Y. Essential oils as repellents against arthropods. BioMed Res Int. 2018;6860271.
- Lee, J.; Choi, D.B.; Liu, F.; Grieco, J.P.; Achee, N.L. Effect of the topical repellent para-Menthane-3,8-diol on blood feeding behavior and fecundity of the dengue virus vector Aedes aegypti. Insects 2018, 9(2), 60. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Solomon, B.; Sahle, F.F.; Gebre-Mariam, T.; Asres, K.; Neubert, R.H.H. Microencapsulation of citronella oil for mosquito-repellent application: Formulation and in vitro permeation studies. European Journal of Pharmaceutics and Biopharmaceutics 2012, 80(1), 61–66. Available online: https://www.sciencedirect.com/science/article/pii/S0939641111002402. [CrossRef]
- Higuchi, C.T.; Sales, C.C.; Andréo-Filho, N.; Martins, T.S.; Ferraz, H.O.; Santos, Y.R.; Lopes, P.S.; Grice, J.E.; Benson, H.A.E.; Leite-Silva, V.R. Development of a Nanotechnology Matrix-Based Citronella Oil Insect Repellent to Obtain a Prolonged Effect and Evaluation of the Safety and Efficacy. Life (Basel) 2023, 13(1), 141. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sousa, D. L.; Xavier, E. O.; da Cruz, R. C. D.; de Souza, I. A.; de Oliveira, R. A.; da Silva, D. C.; Gualberto, S. A.; de Freitas, J. S. Chemical composition and repellent potential of essential oil from Croton tetradenius (Euphorbiaceae) leaves against Aedes aegypti (Diptera: Culicidae). Biocatalysis and Agricultural Biotechnology 2023, 47, 102549. [Google Scholar] [CrossRef]
- Mukarram, M.; Choudhary, S.; Khan, M.A.; Poltronieri, P.; Khan, M.M.A.; Ali, J.; Kurjak, D.; Shahid, M. Lemongrass Essential Oil Components with Antimicrobial and Anticancer Activities. Antioxidants 2022, 11, 20. [Google Scholar] [CrossRef]
- Dangol, S.; Poudel, D.K.; Ojha, P.K.; Maharjan, S.; Poudel, A.; Satyal, R.; Rokaya, A.; Timsina, S.; Dosoky, N.S.; Satyal, P.; Setzer, W.N. Essential Oil Composition Analysis of Cymbopogon Species from Eastern Nepal by GC-MS and Chiral GC-MS, and Antimicrobial Activity of Some Major Compounds. Molecules 2023, 28, 543. [Google Scholar] [CrossRef] [PubMed]
- Reichert, W.; Ejercito, J.; Guda, T.; Dong, X.; Wu, Q.; Ray, A.; Simon, J.E. Repellency Assessment of Nepeta cataria Essential Oils and Isolated Nepetalactones on Aedes aegypti. Sci Rep 2019, 9, 1524. [Google Scholar] [CrossRef]
- Birkett, M.A.; Hassanali, A.; Hoglund, S.; Pettersson, J.; Pickett, J.A. Repellent activity of catmint, Nepeta cataria, and iridoid nepetalactone isomers against Afro-tropical mosquitoes, ixodid ticks and red poultry mites. Phytochemistry 2011, (72), 1:109. Available online: https://www.sciencedirect.com/science/article/pii/S0031942210003729. [CrossRef]
- Haro-González, J.N.; Castillo-Herrera, G.A.; Martínez-Velázquez, M.; Espinosa-Andrews, H. Clove Essential Oil (Syzygium aromaticum L. Myrtaceae): Extraction, Chemical Composition, Food Applications, and Essential Bioactivity for Human Health. Molecules 2021, 26(21), 6387. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Silva, M.V.; de Lima, A.D.C.A.; Silva, M.G.; Caetano, V.F.; de Andrade, M.F.; da Silva, R.G.C.; de Moraes Filho, L.E.P.T.; de Lima Silva, I.D.; Vinhas, G.M. Clove essential oil and eugenol: A review of their significance and uses. Food Bioscience Volume 62(2024), 105112. Available online: https://www.sciencedirect.com/science/article/pii/S2212429224015426. [CrossRef]
- Mitra, S.; Rodriguez, S.D.; Vulcan, J.; Cordova, J.; Chung, H.-N.; Moore, E.; Kandel, Y.; Hansen, I.A. Efficacy of Active Ingredients From the EPA 25(B) List in Reducing Attraction of Aedes aegypti to Humans. Journal of Medical Entomology 2020, 57(2), 477–484. [Google Scholar] [CrossRef]
- Li, Y.-Q.; Kong, D.-X.; Wu, H. Analysis and evaluation of essential oil components of cinnamon barks using GC–MS and FTIR spectroscopy. Industrial Crops and Products 41 2013, 269–278. Available online: https://www.sciencedirect.com/science/article/pii/S0926669012002622. [CrossRef]
- Perdones, A.; Vargas, M.; Atarés, L.; Chiralt, A. Physical, antioxidant and antimicrobial properties of chitosan–cinnamon leaf oil films as affected by oleic acid. Food Hydrocolloids,Volume;ISSN 0268-005X Volume 36, Pages 256–26. Available online: https://www.sciencedirect.com/science/article/pii/S0268005X13003184. [CrossRef]
- Leyva-López, N.; Gutiérrez-Grijalva, E.P.; Vazquez-Olivo, G.; Heredia, J.B. Essential Oils of Oregano: Biological Activity beyond Their Antimicrobial Properties. Molecules 2017, 22(6), 989. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tabari, M.A.; Youssefi, M.R.; Maggi, F.; Benelli, G. Toxic and repellent activity of selected monoterpenoids (thymol, carvacrol and linalool) against the castor bean tick, Ixodes ricinus (Acari: Ixodidae). Veterinary Parasitology 2017, Volume 245, Pages 86–91. Available online: https://www.sciencedirect.com/science/article/pii/S0304401717303618. [CrossRef]
- Borugă, O.; Jianu, C.; Mişcă, C.; Goleţ, I.; Gruia, A.T.; Horhat, F.G. Thymus vulgaris essential oil: chemical composition and antimicrobial activity. J Med Life 2014, 7 Spec(No. 3(Spec Iss 3)), 56–60. [Google Scholar] [PubMed] [PubMed Central]
- Satyal, P.; Murray, B.L.; McFeeters, R.L.; Setzer, W.N. Essential Oil Characterization of Thymus vulgaris from Various Geographical Locations. Foods 2016, 5(4), 70. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Najar, B.; Pistelli, L.; Ferri, B.; Angelini, L.G.; Tavarini, S. Crop Yield and Essential Oil Composition of Two Thymus vulgaris Chemotypes along Three Years of Organic Cultivation in a Hilly Area of Central Italy. Molecules 2021, 26, 5109. [Google Scholar] [CrossRef]
- Zinno, P.; Guantario, B.; Lombardi, G.; Ranaldi, G.; Finamore, A.; Allegra, S.; Mammano, M.M.; Fascella, G.; Raffo, A.; Roselli, M. Chemical Composition and Biological Activities of Essential Oils from Origanum vulgare Genotypes Belonging to the Carvacrol and Thymol Chemotypes. Plants (Basel) 2023, 12(6), 1344. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Walasek-Janusz, M.; Grzegorczyk, A.; Malm, A.; Nurzyńska-Wierdak, R.; Zalewski, D. Chemical Composition, and Antioxidant and Antimicrobial Activity of Oregano Essential Oil. Molecules 2024, 29, 435. [Google Scholar] [CrossRef] [PubMed]
- De Martino, L.; De Feo, V.; Formisano, C.; Mignola, E.; Senatore, F. Chemical Composition and Antimicrobial Activity of the Essential Oils from Three Chemotypes of Origanum vulgare L. ssp. hirtum (Link) Ietswaart Growing Wild in Campania (Southern Italy). Molecules 2009, 14, 2735–2746. [Google Scholar] [CrossRef] [PubMed]
- Hudz, N.; Kobylinska, L.; Pokajewicz, K.; Horčinová Sedláčková, V.; Fedin, R.; Voloshyn, M.; Myskiv, I.; Brindza, J.; Wieczorek, P.P.; Lipok, J. Mentha piperita: Essential Oil and Extracts, Their Biological Activities, and Perspectives on the Development of New Medicinal and Cosmetic Products. Molecules 2023, 28(21), 7444. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Batish, D.R.; Singh, H.P.; Kohli, R.K.; Kaur, S. Eucalyptus essential oil as a natural pesticide. Forest Ecology and Management 2008, Volume 256(Issue 12), Pages 2166–2174. Available online: https://www.sciencedirect.com/science/article/pii/S0378112708006166. [CrossRef]
- Barbosa, L.C.; Filomeno, C.A.; Teixeira, R.R. Chemical variability and biological activities of Eucalyptus spp. essential oils. Molecules 2016, 21(12), 1671. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sheikh, Z.; Amani, A.; Basseri, H.R.; Kazemi, S.H.M.; Sedaghat, M.M.; Azam, K.; Azizi, M.; Amirmohammadi, F. Repellent efficacy of Eucalyptus globulus and Syzygium aromaticum: Essential oils against malaria vector, Anopheles stephensi (Diptera: Culicidae). Iran J Public Health 2021, 50(8), 1668–1677. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Čmiková, N.; Galovičová, L.; Schwarzová, M.; Vukic, M.D.; Vukovic, N.L.; Kowalczewski, P.Ł; Bakay, L.; Kluz, M.I.; Puchalski, C.; Kačániová, M. Chemical Composition and Biological Activities of Eucalyptus globulus Essential Oil. Plants 2023, 12, 1076. [Google Scholar] [CrossRef] [PubMed]
- Faria, J.M.S.; Pereira, G.; Figueiredo, A.C.; Barbosa, P. In vivo and in vitro grown lemon-scented gum as a source of nematicidal essential oil compounds. Plants (Basel) 2025, 14(13), 1892. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gillij, Y.G.; Gleiser, R.M.; Zygadlo, J.A. Mosquito repellent activity of essential oils of aromatic plants growing in Argentina. Bioresour Technol. 2008, 99(7), 2507–15. [Google Scholar] [CrossRef] [PubMed]
- Luker, H. A.; Salas, K. R.; Esmaeili, D.; Holguin, F. O.; Bendzus-Mendoza, H.; Hansen, I. A. Repellent efficacy of 20 essential oils on Aedes aegypti mosquitoes and Ixodes scapularis ticks in contact-repellency assays. Scientific Reports 2023, 13, 1705. [Google Scholar] [CrossRef]
- Oyarce, G.A.; Loyola, P.; Iubini-Aravena, M.; Romero, Á; Rodríguez-Maciel, J.C.; Becerra, J.; Silva-Aguayo, G. Adulticidal and repellent activity of essential oils from three cultivated aromatic plants against Musca domestica L. Insects 2025, 16(5), 542. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Nenaah, G.E.; Alasmari, S.; Almadiy, A. A.; Albogami, B.Z.; Shawer, D.A.; Fadl, A.E. Bio-efficacy of Salvia officinalis essential oil, nanoemulsion and monoterpene components as eco-friendly green insecticides for controlling the granary weevil, Industrial Crops and Products. Part A, 2023, 117298, ISSN 0926-6690 Volume 204. Available online: https://www.sciencedirect.com/science/article/pii/S0926669023010634. [CrossRef]
- Craft, J.D.; Satyal, P.; Setzer, W.N. The chemotaxonomy of common sage (Salvia officinalis) based on the volatile constituents. Medicines (Basel) 2017, 4(3), 47. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mot, M.-D.; Gavrilaș, S.; Lupitu, A.I.; Moisa, C.; Chambre, D.; Tit, D.M.; Bogdan, M.A.; Bodescu, A.-M.; Copolovici, L.; Copolovici, D.M.; Bungau, S.G. Salvia officinalis L. Essential oil: Characterization, antioxidant properties, and the effects of aromatherapy in adult patients. Antioxidants 2022, 11, 808. [Google Scholar] [CrossRef]
- Jaenson, T.G.T.; Garboui, S.; Pålsson, K. Repellency of oils of lemon eucalyptus, geranium, and lavender and the mosquito repellent MyggA natural to Ixodes ricinus (Acari: Ixodidae) in the laboratory and field. Journal of Medical Entomology 2006, 43(4), 731–736. [Google Scholar] [CrossRef] [PubMed]
- Tabanca, N.; Wang, M.; Avonto, C.; Chittiboyina, A. G.; Parcher, J. F.; Carroll, J. F.; Kramer, M.; Khan, I. A. Bioactivity-guided investigation of geranium essential oils as natural tick repellents. Journal of Agricultural and Food Chemistry 2013, 61, 4101–4107. [Google Scholar] [CrossRef]
- Rana, V. S.; Juyal, J. P.; Blázquez, M. A. Chemical constituents of the essential oil of Pelargonium graveolens leaves. International Journal of Aromatherapy 2002, 12, 216–218. [Google Scholar] [CrossRef]
- Sharopov, F. S.; Zhang, H.; Setzer, W. N. Composition of geranium (Pelargonium graveolens) essential oil from Tajikistan. American Journal of Essential Oils and Natural Products 2014, 2(2), 13–16. [Google Scholar]
- Yohana, R.; Chisulumi, P.S.; Kidima, W.; Tahghighi, A.; Maleki-Ravasan, N.; Kweka, E.J. Anti-mosquito properties of Pelargonium roseum (Geraniaceae) and Juniperus virginiana (Cupressaceae) essential oils against dominant malaria vectors in Africa. Malar J 2022, 21(1), 219. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Shellie, R.; Mondello, L.; Marriott, P.; Dugo, G. Characterisation of lavender essential oils by using gas chromatography–mass spectrometry with correlation of linear retention indices and comparison with comprehensive two-dimensional gas chromatography. In Journal of Chromatography A; ISSN 0021-9673, 2002; Volume 970, Issues 1–2, pp. 225–234. Available online: https://www.sciencedirect.com/science/article/pii/S0021967302006532. [CrossRef]
- Nedeltcheva-Antonova, D.; Gechovska, K.; Bozhanov, S.; Antonov, L. Exploring the chemical composition of Bulgarian lavender absolute (Lavandula Angustifolia Mill.) by GC/MS and GC-FID. Plants (Basel) 2022, 11(22), 3150. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Pokajewicz, K.; Białoń, M.; Svydenko, L.; Fedin, R.; Hudz, N. Chemical composition of the essential oil of the new cultivars of Lavandula angustifolia Mill. Bred in Ukraine. Molecules 2021, 26(18), 5681. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wińska, K.; Mączka, W.; Łyczko, J.; Grabarczyk, M.; Czubaszek, A.; Szumny, A. Essential oils as antimicrobial agents—Myth or real alternative? Molecules 2019, 24(11), 2130. [Google Scholar] [CrossRef] [PubMed]
- Hammer, K. A.; Carson, C. F.; Riley, T. V. Antimicrobial activity of essential oils and other plant extracts. Journal of Applied Microbiology 1999, 86(6), 985–990. [Google Scholar] [CrossRef] [PubMed]
- Chouhan, S.; Sharma, K.; Guleria, S. Antimicrobial activity of some essential oils— Present status and future perspectives. Medicines 2017, 4(3), 58. [Google Scholar] [CrossRef]
- Donelli, D.; Antonelli, M.; Bellinazzi, C.; Gensini, G. F.; Firenzuoli, F. Effects of lavender on anxiety: A systematic review and meta-analysis. Phytomedicine 2019, 65, 153099. [Google Scholar] [CrossRef]
- Lee, M. S.; Choi, J.; Posadzki, P.; Ernst, E. Aromatherapy for health care: An overview of systematic reviews. Maturitas 2012, 71(3), 257–260. [Google Scholar] [CrossRef] [PubMed]
- Tisserand, R.; Young, R. Essential oil safety: A guide for health care professionals, 2nd ed.; Churchill Livingstone, 2014. [Google Scholar]
- Göbel, H.; Schmidt, G.; Soyka, D. Effect of peppermint and eucalyptus oil preparations on neurophysiological and experimental algesimetric headache parameters. Cephalalgia 1994, 14(3), 228–234. [Google Scholar] [CrossRef] [PubMed]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils: A review. Food and Chemical Toxicology 2008, 46(2), 446–475. [Google Scholar] [CrossRef] [PubMed]
- Mills, S.; Bone, K. Principles and practice of phytotherapy: Modern herbal medicine, 2nd ed.; Churchill Livingstone, 2013. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



