Submitted:
19 April 2026
Posted:
21 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
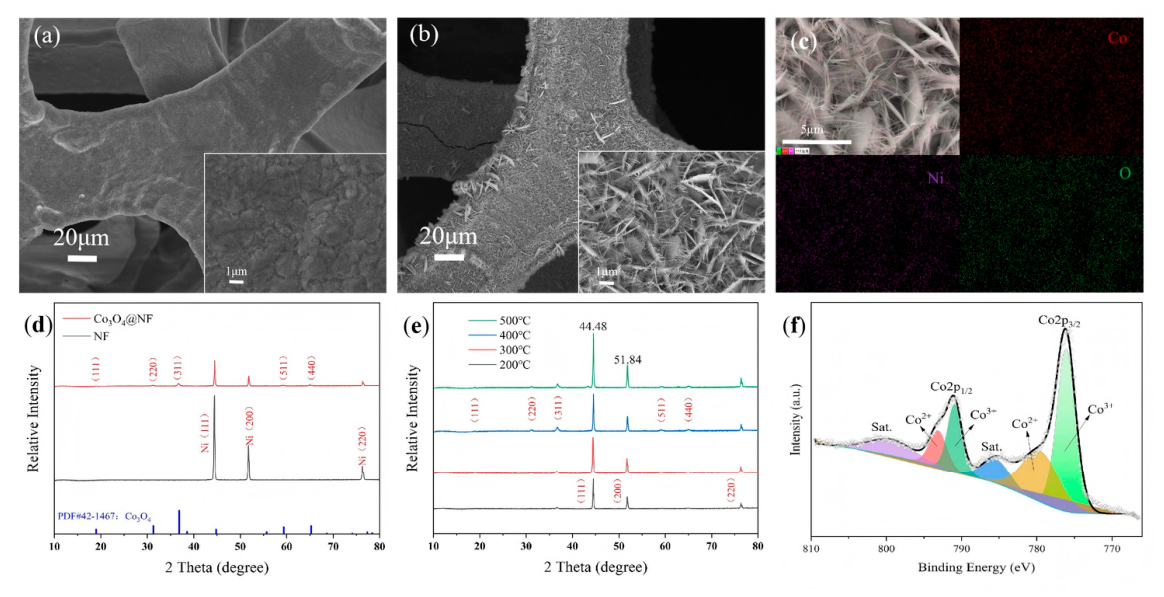
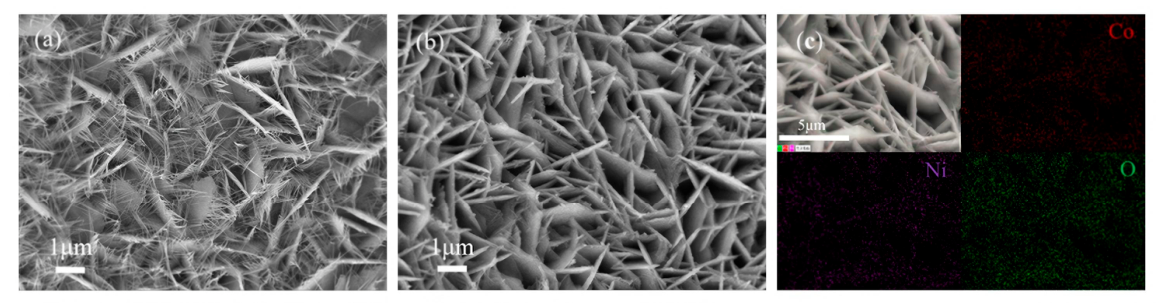
2.1. Structural Characterization of the Co3O4@NF Electrode
2.2. Electrocatalytic Performance and Parameter Optimization
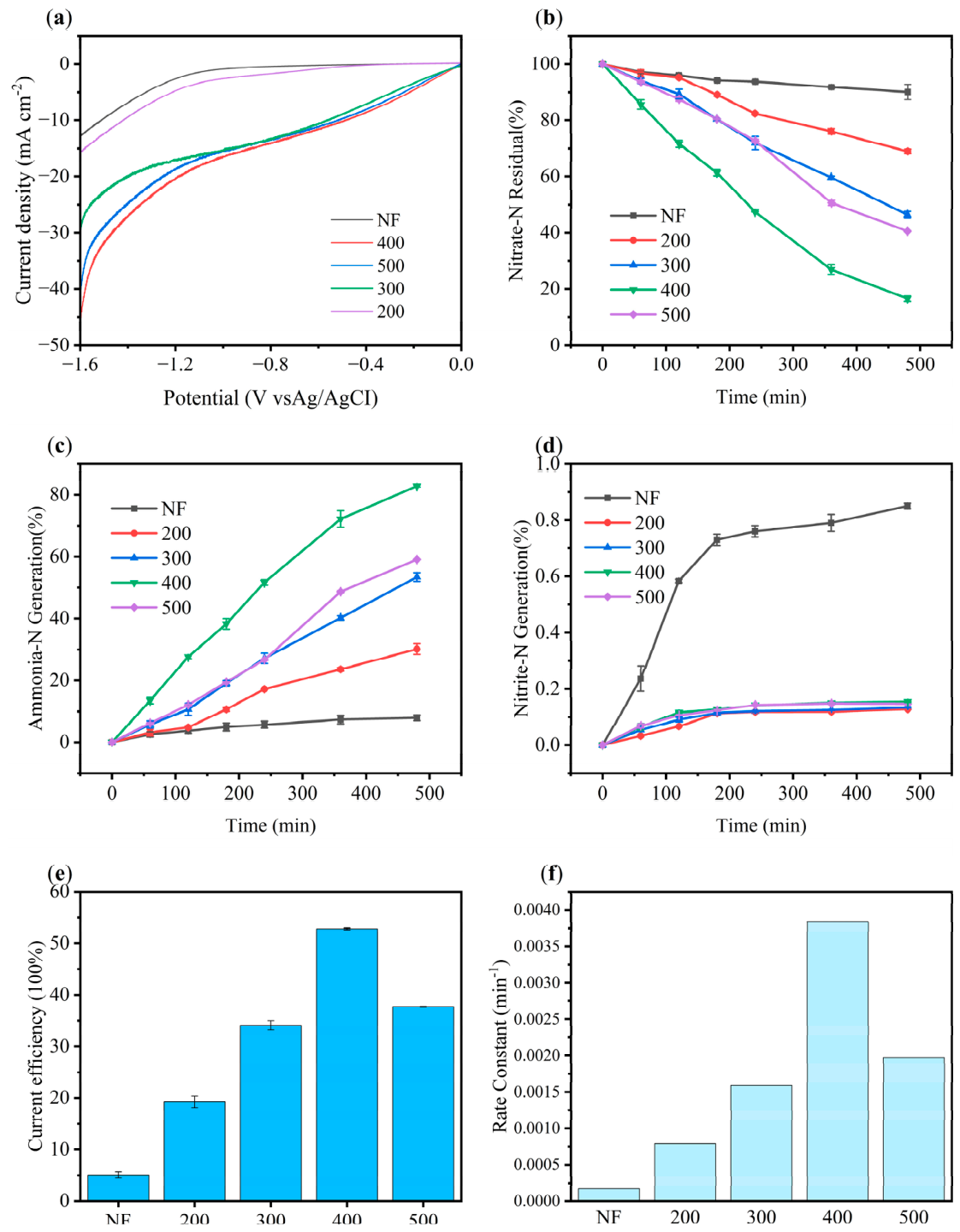
2.2.1. Optimization of Electrode Calcination Temperature
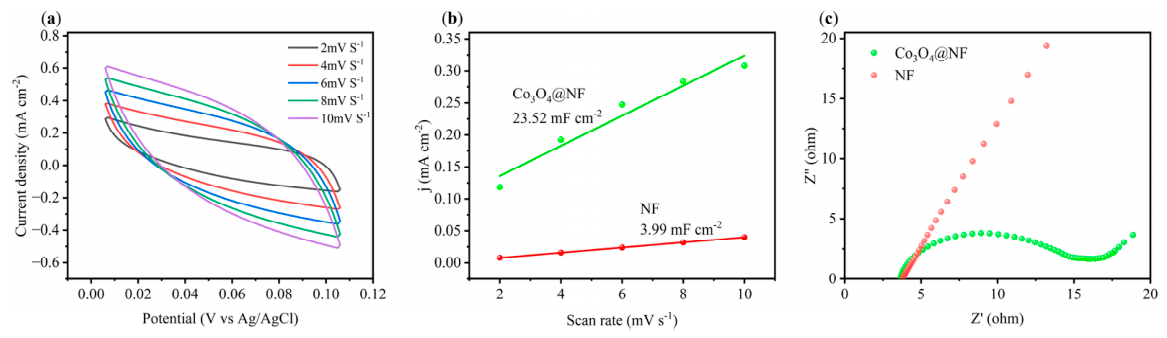
2.2.2. Electrochemical Testing
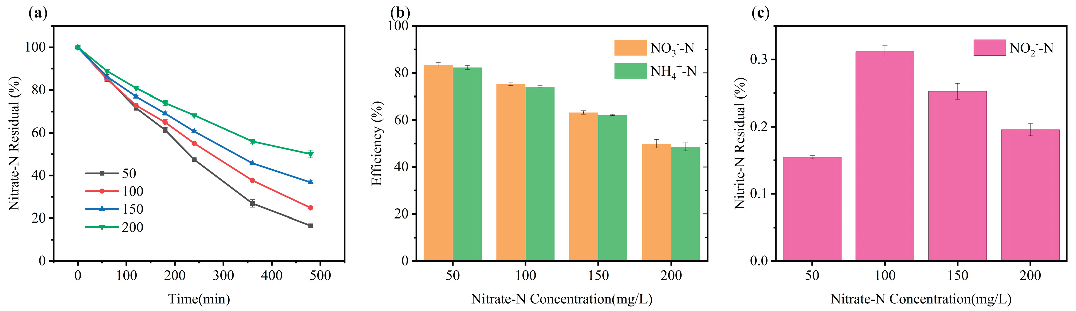
2.2.3. Effect of Initial Nitrate Concentration
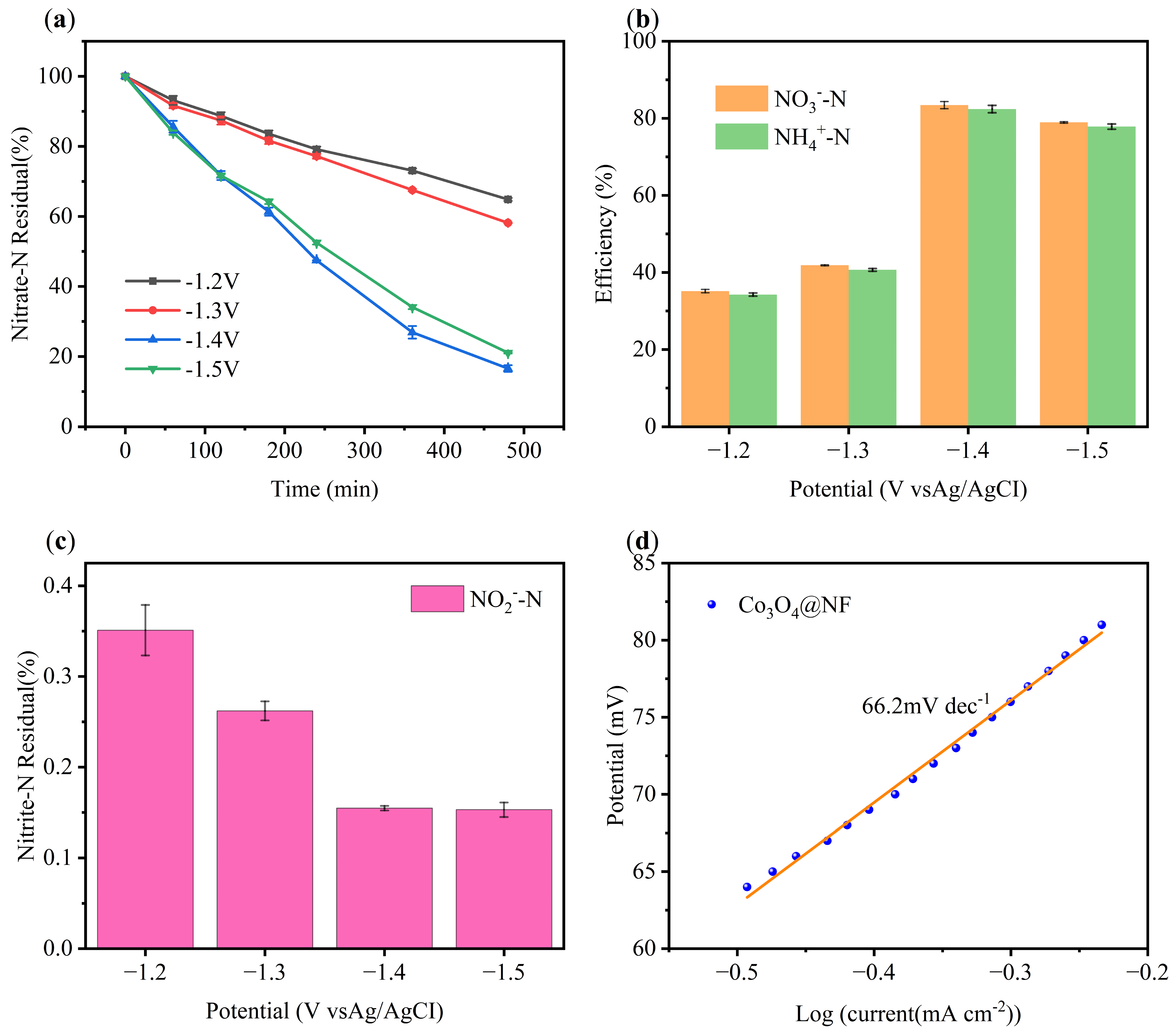
2.2.4. Effect of Initial Voltage Intensity
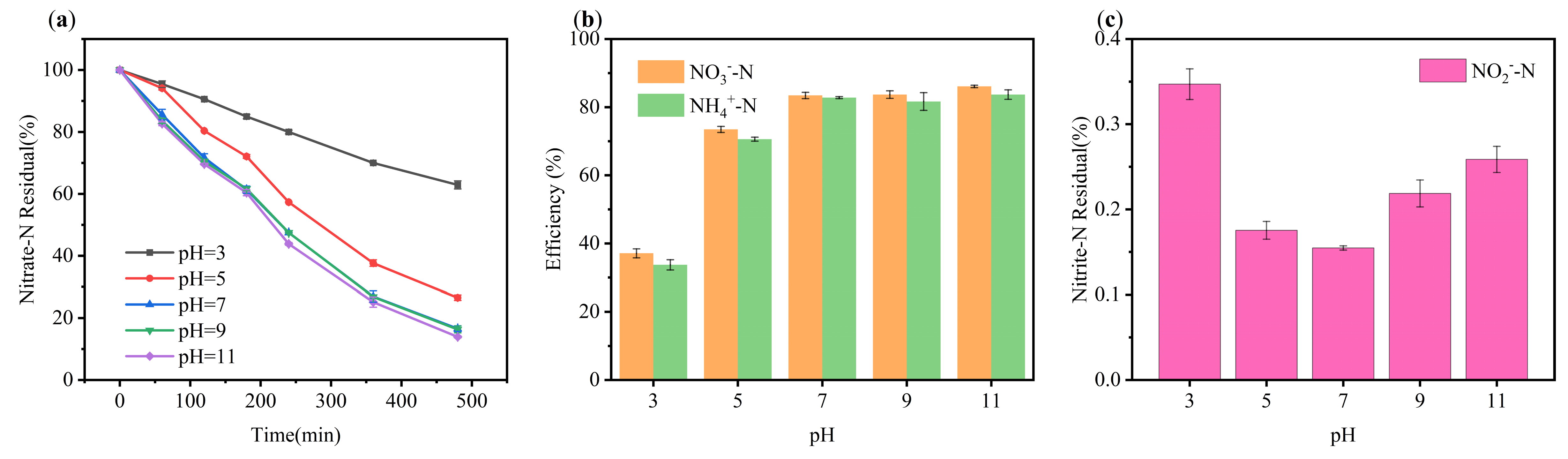
2.2.5. Effect of Initial pH
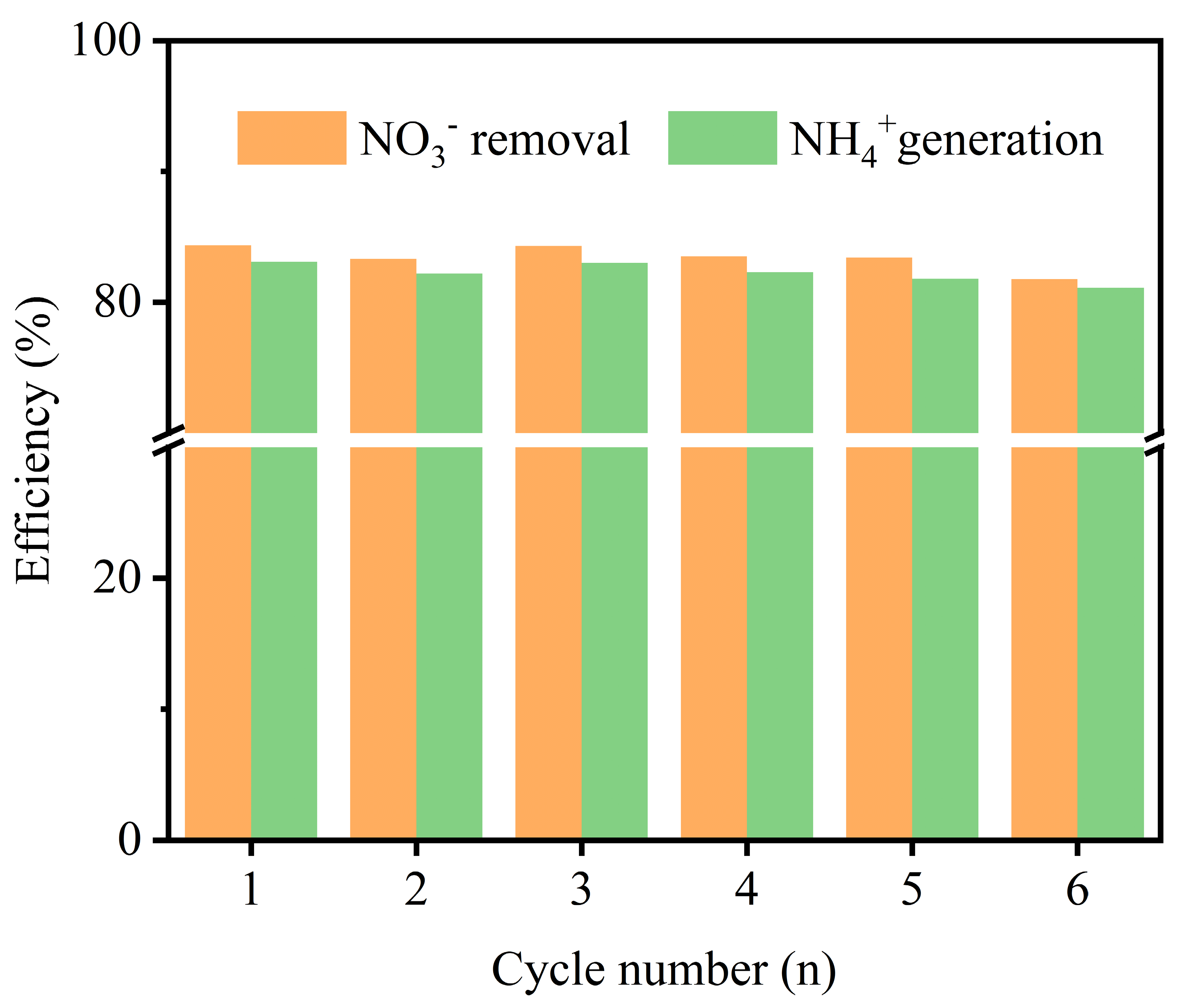
2.3. Stability and Structural Integrity
2.4. Electrocatalytic Nitrate Reduction Mechanism
2.5. Performance in Real Water Matrices
3. Discussion
4. Materials and Methods
4.1. Reagents and Materials
4.2. Fabrication of the Binder-Free Co3O4@NF Cathode
4.3. Electrochemical Nitrate Removal Experiments
4.4. Characterization Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| NF | Nickel Foam |
| LSV | Linear Sweep Voltammetry |
| CV | Cyclic Voltammetry |
| ECSA | Electrochemical Active Surface Area |
| Cdl | Double-layer Capacitance |
| EIS | Electrochemical Impedance Spectroscopy |
| HER | Hydrogen Evolution Reaction |
| TBA | tert-butanol |
| FE-SEM | Field-Emission Scanning Electron Microscopy |
| EDS | Energy-Dispersive X-ray Spectrometer |
| XRD | X-ray Diffraction |
| XPS | X-ray Photoelectron Spectroscopy |
| UV-Vis | Ultraviolet-visible Spectrophotometry |
| RDS | rate-determining step |
| DFT | Density Functional Theory |
References
- Moloantoa, K.M.; Khetsha, Z.P.; van Heerden, E.; Castillo, J.C.; Cason, E.D. Nitrate Water Contamination from Industrial Activities and Complete Denitrification as a Remediation Option. Water 2022, 14. [Google Scholar] [CrossRef]
- Su, H.; Kang, W.; Li, Y.; Li, Z. Fluoride and nitrate contamination of groundwater in the Loess Plateau, China: Sources and related human health risks. Environmental Pollution 2021, 286. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, X.; Jia, S.; Mao, B. Multi-methods to investigate spatiotemporal variations of nitrogen-nitrate and its risks to human health in China's largest fresh water lake (Poyang Lake). Science of The Total Environment 2023, 863. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Li, Z.; He, G.; Liu, G.; Ji, M.; Liu, W. Effect of combined cadmium and doxycycline pollution on the nitrogen cycle in constructed ditch wetlands. Environmental Chemistry Letters 2024, 22, 989–995. [Google Scholar] [CrossRef]
- Xing, Y.-F.; Zhu, X.-Y.; Dong, H.-B.; Huang, J.-H.; Duan, Y.-F.; Zhang, J.-S. Microplastics and Nitrite Stress Affect Physiological and Metabolic Functions of the Hepatopancreas in Marine Shrimp. Journal of Xenobiotics 2026, 16. [Google Scholar] [CrossRef]
- Bae, B.-U.; Jung, Y.-H.; Han, W.-W.; Shin, H.-S. Improved brine recycling during nitrate removal using ion exchange. J Water Research 2002, 36, 3330–3340. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Perumal, P.; Sithole, T.; Luukkonen, T. Recovery of ammonium and nitrate from wastewater using adsorption-based techniques: A review. Journal of Cleaner Production 2025, 519. [Google Scholar] [CrossRef]
- Zhang, J.; Wu, J.; Chao, J.; Shi, N.; Li, H.; Hu, Q.; Yang, X.J. Simultaneous removal of nitrate, copper and hexavalent chromium from water by aluminum-iron alloy particles. Journal of Contaminant Hydrology 2019, 227. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, Y.; Shi, Z.; Chen, Y. Linking Genome-Centric Metagenomics to Kinetic Analysis Reveals the Regulation Mechanism of Hydroxylamine in Nitrite Accumulation of Biological Denitrification. Environmental Science & Technology 2022, 56, 10317–10328. [Google Scholar] [CrossRef]
- Yan, L.; Miao, J.; Li, M.; Zhang, Y.; Si, W.; Liu, X.; Qiao, J.; Yang, Z.; Wang, P. Removal of nitrate by aerobic denitrification granular sludge under strongly alkaline and low carbon to nitrogen ratio conditions: Performance and mechanism. Bioresource Technology 2026, 449. [Google Scholar] [CrossRef]
- Li, X.; Wang, B.; Ma, Y.; Jiang, T.; Peng, Y. Enhanced mesophilic fermentation of waste activated sludge by integration with in-situ nitrate reduction. Bioresource Technology 2023, 368. [Google Scholar] [CrossRef]
- Chen, J.; Zhang, C.; Liu, Y.; Tian, J.; Guo, J. In-Situ Improvement of the Sediment Microenvironment by Nitrate in Tailwater of Wastewater Treatment Plants Combined with Aerobic Denitrifying Bacteria under Low-DO Regulation. Water 2024, 16. [Google Scholar] [CrossRef]
- Chang, Y.S.; Ooi, B.S. Multiple nutrient recovery from aquaculture wastewater through vacuum membrane distillation. Chemical Engineering Journal 2026, 527. [Google Scholar] [CrossRef]
- Liu, B.; Lu, D.; Zhang, T.; Shen, Y.; Qiu, Z.; Mao, X.; Wu, R. Sustainable approach for landfill leachate treatment through dielectric barrier discharge/ferrate (DBD/Fe(VI)) enhanced nanofiltration. Journal of Membrane Science 2025, 714. [Google Scholar] [CrossRef]
- Lu, D.; Mao, X.; Wu, R.; Liu, B. Dielectric Barrier Discharge (DBD) enhanced Fenton process for landfill leachate nanofiltration: Organic matter removal and membrane fouling alleviation. Water Research 2024, 266. [Google Scholar] [CrossRef] [PubMed]
- Tugaoen, H.O.N.; Garcia-Segura, S.; Hristovski, K.; Westerhoff, P. Challenges in photocatalytic reduction of nitrate as a water treatment technology. Science of The Total Environment 2017, 599-600, 1524–1551. [Google Scholar] [CrossRef]
- Liu, B.; Sun, X.; Sun, W.; Zhang, Y.; Wang, L.; Bai, J.; Xu, G. La-doped carbon nanofibers loaded with Cu-Co nanocubes enable highly efficient series electrochemical catalysis for nitrate reduction in ammonia synthesis. Journal of Alloys and Compounds 2026, 1059. [Google Scholar] [CrossRef]
- Hu, L.; Teng, M.; Zhao, Y.; Zhang, W.; Hu, H.; Cheng, X.; Yuan, W.; Yuan, J.; Zhang, C.; Li, F.; et al. Recent progress of two-dimensional materials for efficient electrochemical nitrate reduction. Journal of Environmental Chemical Engineering 2026, 14. [Google Scholar] [CrossRef]
- Martínez, J.; Ortiz, A.; Ortiz, I. State-of-the-art and perspectives of the catalytic and electrocatalytic reduction of aqueous nitrates. Applied Catalysis B: Environmental 2017, 207, 42–59. [Google Scholar] [CrossRef]
- Meng, S.; Ling, Y.; Yang, M.; Zhao, X.; Osman, A.I.; Al-Muhtaseb, A.a.H.; Rooney, D.W.; Yap, P.-S. Recent research progress of electrocatalytic reduction technology for nitrate wastewater: A review. Journal of Environmental Chemical Engineering 2023, 11. [Google Scholar] [CrossRef]
- Yan, C.; Lee, K.-L.; Troutman, J.P.; Brady, C.E.; Humphrey, S.M.; Cwiertny, D.M.; Mubeen, S.; Werth, C.J. Tailored copper-based cathode design advances economic viability of electrocatalytic nitrate treatment with ammonia recovery in a scalable flow reactor. Applied Catalysis B: Environment and Energy 2024, 357. [Google Scholar] [CrossRef]
- Guo, J.; Zhang, L.H.; Guo, Y.; Shi, C.; Han, A.; Wang, D.; Yu, F. Oxygen-Incorporation-Engineered Interfacial Water Modulation on Single-Atom Cu Sites for Enhanced Dilute Nitrate Electroreduction. Angewandte Chemie International Edition 2025, 65. [Google Scholar] [CrossRef]
- Meng, Z.; Priyadarsini, A.; Shi, K.; Ren, Z.; Subedi, D.; Israel, D.; Kaden, W.E.; Feng, X.; Kattel, S. pH-Dependent Electroreduction of Nitrate on Fe Single-Atom Catalyst. ChemSusChem 2025, 18. [Google Scholar] [CrossRef]
- Wu, C.; Liang, G.; Xia, S.; Zhou, J.; Lichtfouse, E.; Cai, X.; Xu, S.; Liu, H. In situ-generated palladium nanoparticles promoted co-reduction of bromate and nitrate in hydrogenotrophic biofilms. Water Research 2025, 286. [Google Scholar] [CrossRef] [PubMed]
- García-Fernández, M.J.; Pastor-Blas, M.M.; Epron, F.; Sepúlveda-Escribano, A. Proposed mechanisms for the removal of nitrate from water by platinum catalysts supported on polyaniline and polypyrrole. Applied Catalysis B: Environmental 2018, 225, 162–171. [Google Scholar] [CrossRef]
- Zhou, F.; Lv, B.; Zhang, Y.; Wu, Y.; Wang, Y.; Luo, W. Carbon-constrained CoRu bimetallic catalysts enabling efficient electrocatalytic reduction of nitrate to ammonia. Journal of Power Sources 2025, 657. [Google Scholar] [CrossRef]
- Qu, Y.; Li, X.; Xia, Y.; Lan, H.; Ding, L.; Zhong, J.; Chang, X. Enhanced nitrate reduction to ammonia using Cu-Ni catalyst: Synergistic mechanisms and reaction pathways. Journal of Environmental Sciences 2026, 159, 23–32. [Google Scholar] [CrossRef]
- Zhao, C.; Wang, T.-H.; Chen, Z.W.; Wen, Z.; Jiang, Q. Multi-site activation in metal-doped CuPd alloy catalysts enhances nitrate electroreduction to ammonia. Materials Today Sustainability 2025, 30. [Google Scholar] [CrossRef]
- Peng, C.; Wu, R.; Yang, Y.; Li, C.; Lin, Y.; Chen, S.; Kuai, Z.; Li, L. Hydrothermal formation of controllable hexagonal holes and Er2O3/Er2O3-RGO particles on silicon wafers toward superhydrophobic surfaces. Journal of Colloid and Interface Science 2020, 580, 768–775. [Google Scholar] [CrossRef]
- Cerrón-Calle, G.A.; Maya, A.; Leon, D.; Roldan, M.; Fajardo, A.S.; Sánchez-Sánchez, C.M.; Garcia-Segura, S. Unlocking sustainable nitrate reduction: Earth-abundant bimetallic electrodes under galvanostatic evaluation. Electrochimica Acta 2024, 489. [Google Scholar] [CrossRef]
- Sahoo, S.; Wickramathilaka, K.Y.; Njeri, E.; Silva, D.; Suib, S.L. A review on transition metal oxides in catalysis. Frontiers in Chemistry 2024, 12. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, A.; Swarnalal, A.; Das, D.; Krishnan, M.; Saji, V.S.; Shibli, S.M.A. A review on transition metal oxides based photocatalysts for degradation of synthetic organic pollutants. Journal of Environmental Sciences 2024, 139, 389–417. [Google Scholar] [CrossRef]
- Gao, N.; Guo, M.; Tong, H.; Hou, G.; Ba, J.; Zhang, X.; Zhang, R.; Zhang, H.; Fu, X.; Cancellara, L.; et al. Atomically Dispersed Cobalt on Ionic Carbon Nitrides for Selective and Efficient Nitrate Electroreduction to Ammonia. Angewandte Chemie International Edition 2026. [Google Scholar] [CrossRef]
- Farid, S.; Rashid, A. One-step dealloying-derived cobalt nanoelectrodes for highly efficient electroreduction of carbon dioxide. Fuel 2026, 411. [Google Scholar] [CrossRef]
- Dharmaraj, K.; Hanna, R.; Ruske, F.; Douglas-Henry, D.; Lauermann, I.; Prathapani, S.; Reyes-Figueroa, P.; Rodriguez-Ayllon, Y.; Lu, Y.; Nicolosi, V.; et al. Ammonia tolerant alkaline oxygen reduction reaction on bimetallic cobalt spinels. Chemical Engineering Journal 2025, 522. [Google Scholar] [CrossRef]
- Lin, Y.-P.; Chen, W.-H.; Fan, S.-C.; Cheng, K.-W. Evaluation of cell performance for Zn-CO2 batteries using bifunctional honeycomb-like ZnS/CuxS and S-doped NiO/Co3O4 electrocatalysts on Ni foams as gas cathodes. Journal of Energy Storage 2025, 139. [Google Scholar] [CrossRef]
- He, L.; Zeng, T.; Yao, F.; Zhong, Y.; Tan, C.; Pi, Z.; Hou, K.; Chen, S.; Li, X.; Yang, Q. Electrocatalytic reduction of nitrate by carbon encapsulated Cu-Fe electroactive nanocatalysts on Ni foam. Journal of Colloid and Interface Science 2023, 634, 440–449. [Google Scholar] [CrossRef]
- Li, X.; Zhao, X.; Lv, J.; Jia, X.; Zhou, S.; Huang, Y.; Chang, F.; Zhang, H.; Hu, G. Self-supported porous copper oxide nanosheet arrays for efficient and selective electrochemical conversion of nitrate ions to nitrogen gas. Journal of Materials Science & Technology 2023, 137, 104–111. [Google Scholar] [CrossRef]
- Jonoush, Z.A.; Rezaee, A.; Ghaffarinejad, A. Electrocatalytic nitrate reduction using Fe0/Fe3O4 nanoparticles immobilized on nickel foam: Selectivity and energy consumption studies. Journal of Cleaner Production 2020, 242. [Google Scholar] [CrossRef]
- Yan, K.-L.; Shang, X.; Gao, W.-K.; Dong, B.; Li, X.; Chi, J.-Q.; Liu, Y.-R.; Chai, Y.-M.; Liu, C.-G. Ternary MnO 2 /NiCo 2 O 4 /NF with hierarchical structure and synergistic interaction as efficient electrocatalysts for oxygen evolution reaction. Journal of Alloys and Compounds 2017, 719, 314–321. [Google Scholar] [CrossRef]
- Wu, H.S.; Tao, M.; Che, X.; Sun, Z.; Chen, K.; Wang, R.T. Visualization of gas–liquid two-phase flow in open-cell metal foams. Chemical Industry and Engineering Progress. 2021, 40, 4152–4164. [Google Scholar] [CrossRef]
- Pierożyński, B.; Kuczyński, M.; Mikołajczyk, T. Simple Nickel Foam Modification Procedures for Enhanced Ni Foam Supercapacitor Applications. Crystals 2024, 14. [Google Scholar] [CrossRef]
- Station, H.E.P.M. Water Quality Determination of Nitrate Nitrogen Phenol Disulfonic Acid Spectrophotometry. 1987; GB/T 7480-1987, p. 8.
- Station, H.E.M.C. Water Quality Determination of Nitrite Nitrogen Spectrophotometry. 1987; GB/T 7493-1987, p. 8.
- Station, S.E.M.C. Water Quality. Determination of Ammonia Nitrogen. Nessler's Reagent Spectrophotometry 2009, HJ 535-2009, 9P.;A4. [Google Scholar]
- Su, L.; Li, K.; Zhang, H.; Fan, M.; Ying, D.; Sun, T.; Wang, Y.; Jia, J. Electrochemical nitrate reduction by using a novel Co3O4/Ti cathode. Water Res 2017, 120, 1–11. [Google Scholar] [CrossRef]
- Duan, W.; Li, G.; Lei, Z.; Zhu, T.; Xue, Y.; Wei, C.; Feng, C. Highly active and durable carbon electrocatalyst for nitrate reduction reaction. Water Res 2019, 161, 126–135. [Google Scholar] [CrossRef]
- Zhu, T.; Chen, Q.; Liao, P.; Duan, W.; Liang, S.; Yan, Z.; Feng, C. Single-Atom Cu Catalysts for Enhanced Electrocatalytic Nitrate Reduction with Significant Alleviation of Nitrite Production. Small 2020, 16, e2004526. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Xu, Y.; Shi, W.; Wang, W.; Zhang, R.; Bao, X.; Zhang, B.; Li, L.; Cui, F. Electrochemical-catalytic reduction of nitrate over Pd–Cu/γAl2O3 catalyst in cathode chamber: Enhanced removal efficiency and N2 selectivity. Chemical Engineering Journal 2016, 290, 201–208. [Google Scholar] [CrossRef]
- Li, M.; Feng, C.; Zhang, Z.; Lei, X.; Chen, R.; Yang, Y.; Sugiura, N. Simultaneous reduction of nitrate and oxidation of by-products using electrochemical method. J Hazard Mater 2009, 171, 724–730. [Google Scholar] [CrossRef]
- Chen, M.; Bi, J.; Huang, X.; Wang, T.; Wang, Z.; Hao, H. Bi2O3 nanosheets arrays in-situ decorated on carbon cloth for efficient electrochemical reduction of nitrate. Chemosphere 2021, 278, 130386. [Google Scholar] [CrossRef] [PubMed]
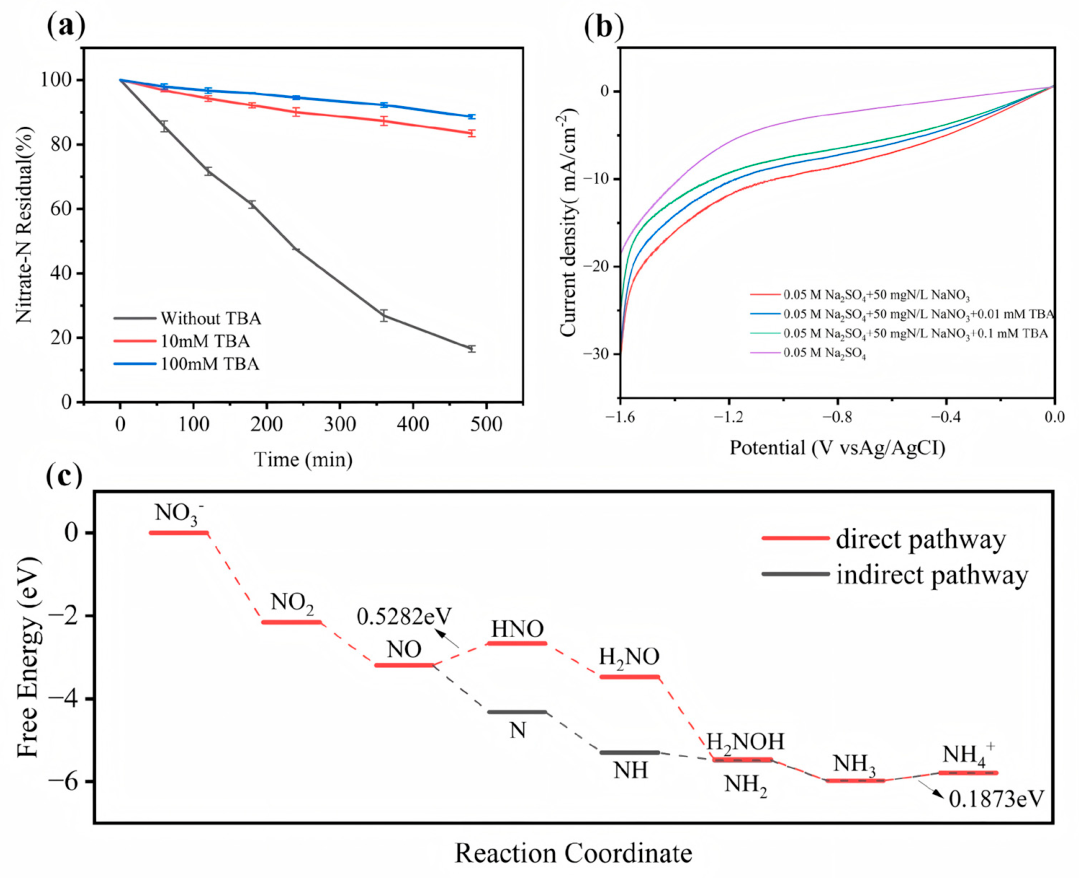
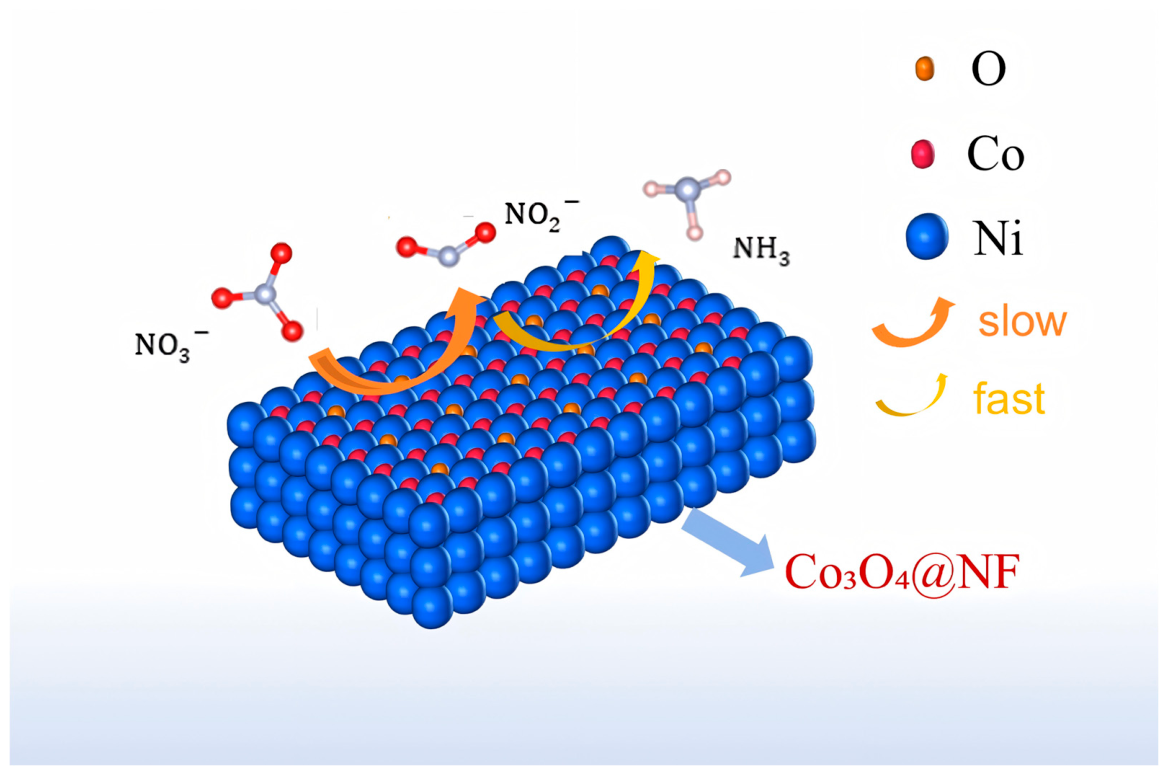
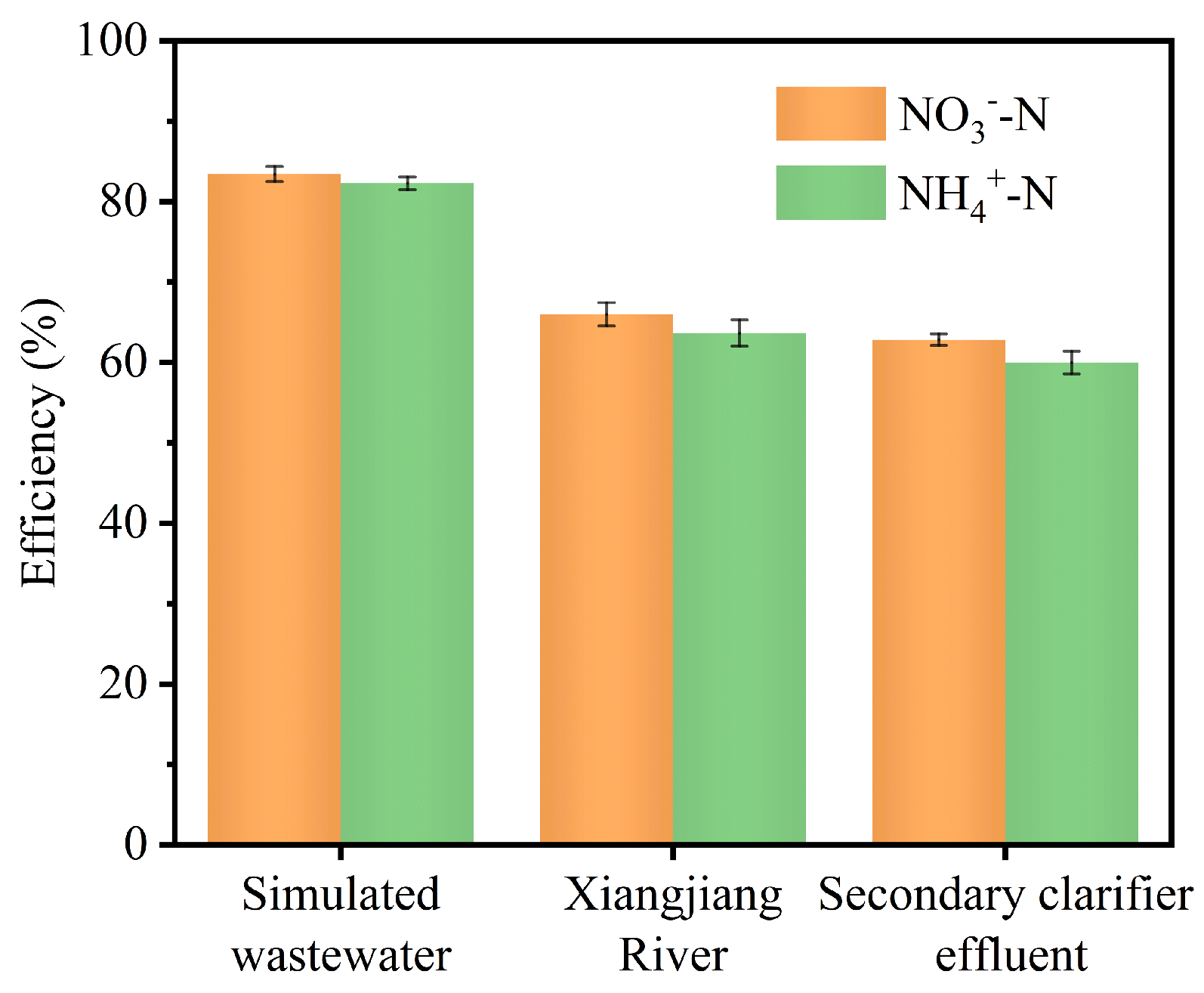
| Samples | NH₃ yield rate (mg‧h-1‧cm-2) | |||
|---|---|---|---|---|
| 200°C | 300°C | 400°C | 500°C | |
| Co₃O₄@NF | 0.229 | 0.405 | 0.628 | 0.448 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




