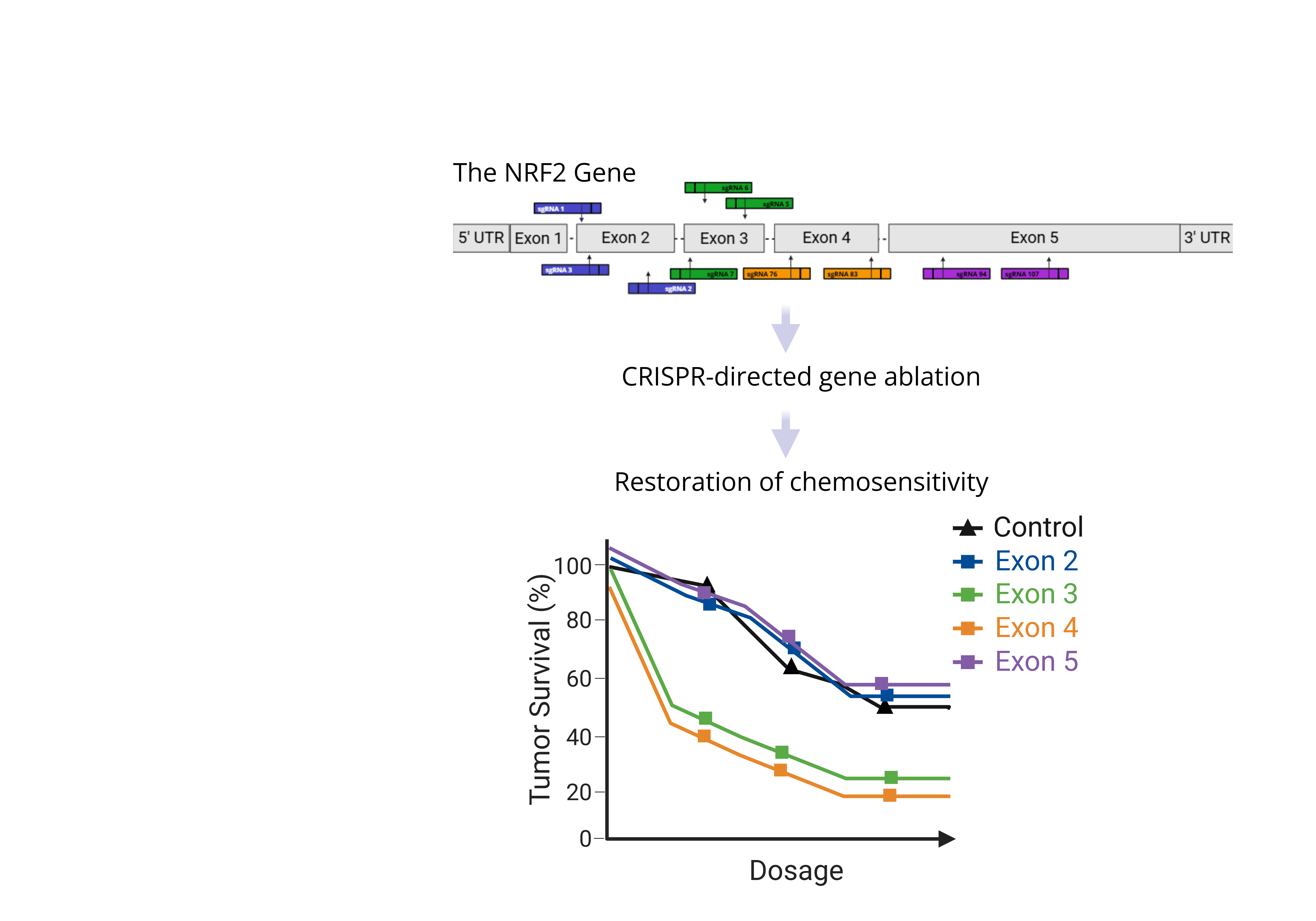
Pancreatic ductal adenocarcinoma (PDAC) presents unique treatment challenges often due to the development of anti-cancer drug resistance. Previously, we demonstrated that CRISPR-directed gene ablation disabled the master regulator gene NRF2, a tran-scription factor known to control drug resistance in squamous cell carcinoma tumor cells restored chemosensitivity. In this short study, we evaluated a broad range of CRISPR/Cas9 molecules for their capacity to elicit similar response in PDAC cells. Synthetic single guide RNAs (sgRNAs) were designed to target multiple functional domains encoded by NRF2. These molecules were delivered to cells via nucleofection with outcomes analyzed by genotypic, phenotypic and functional assays. We observed targeting efficiencies ranging from 25% to 100% with a high level of random insertions and deletions (indels). sgRNAs targeting exons 2, 3 and 4 demonstrated produced a high degree of genotypic, phenotypic and functional outcomes. Targeted disruption of exons 3 and 4 reveals significant loss of cell viability while overcoming dug resistance through the restoration of sensitivity to Gemcitabine (>1.75 uM). Our study identifies do-main-specific sites within NRF2 that, when disabled, restore sensitivity to Gemcitabine potentiating a more in-depth analyses of this novel augmentative therapeutic approach.