Submitted:
17 April 2026
Posted:
21 April 2026
You are already at the latest version
Abstract
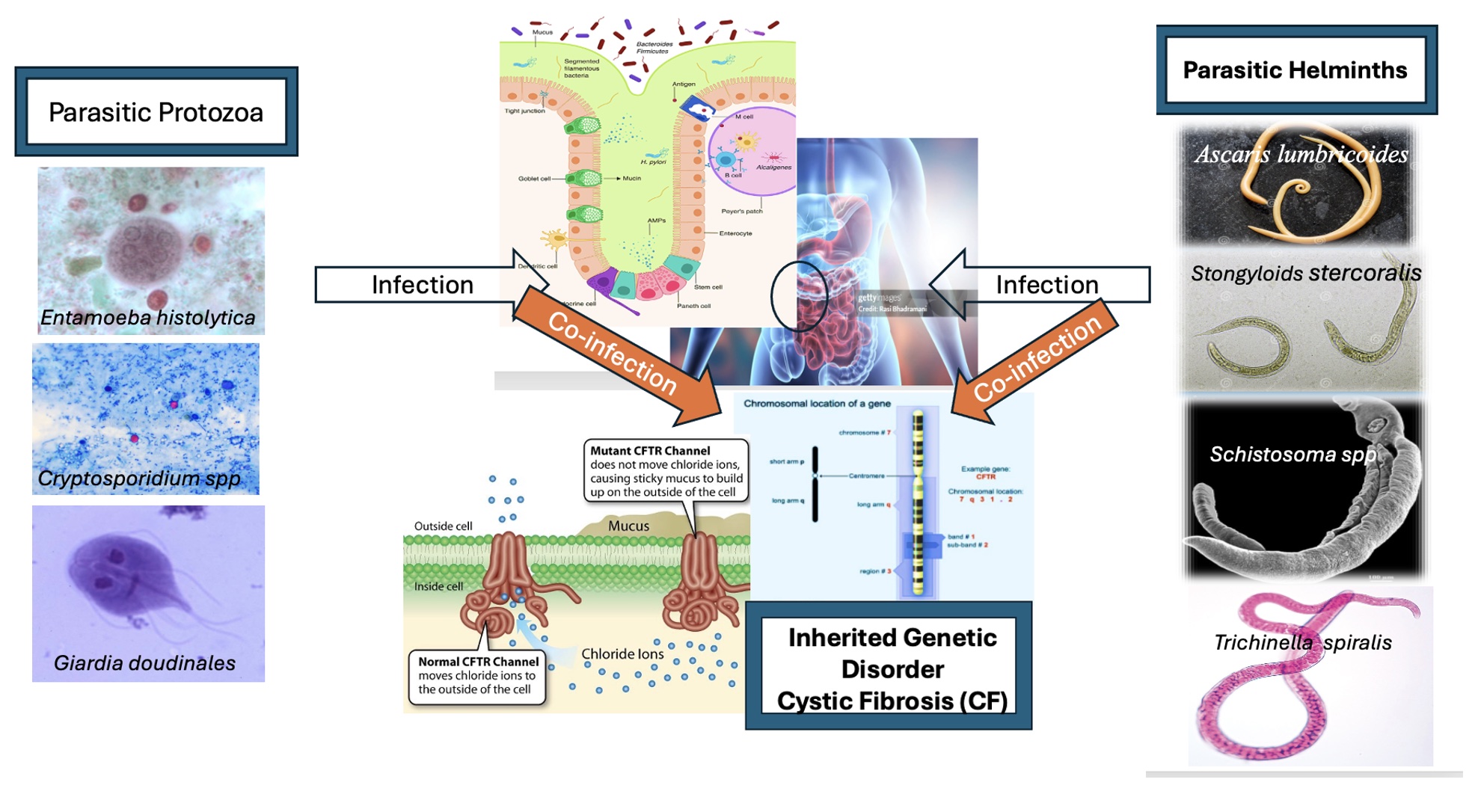
Keywords:
1. Introduction
2. Pathophysiological Basis of Cystic Fibrosis and Mucosal Immunity
3. Pathobiological Basis of Major Human Intestinal Protozoa and Mucosal Immunity
4. Is There a Correlation Between CF and Intestinal parasites?
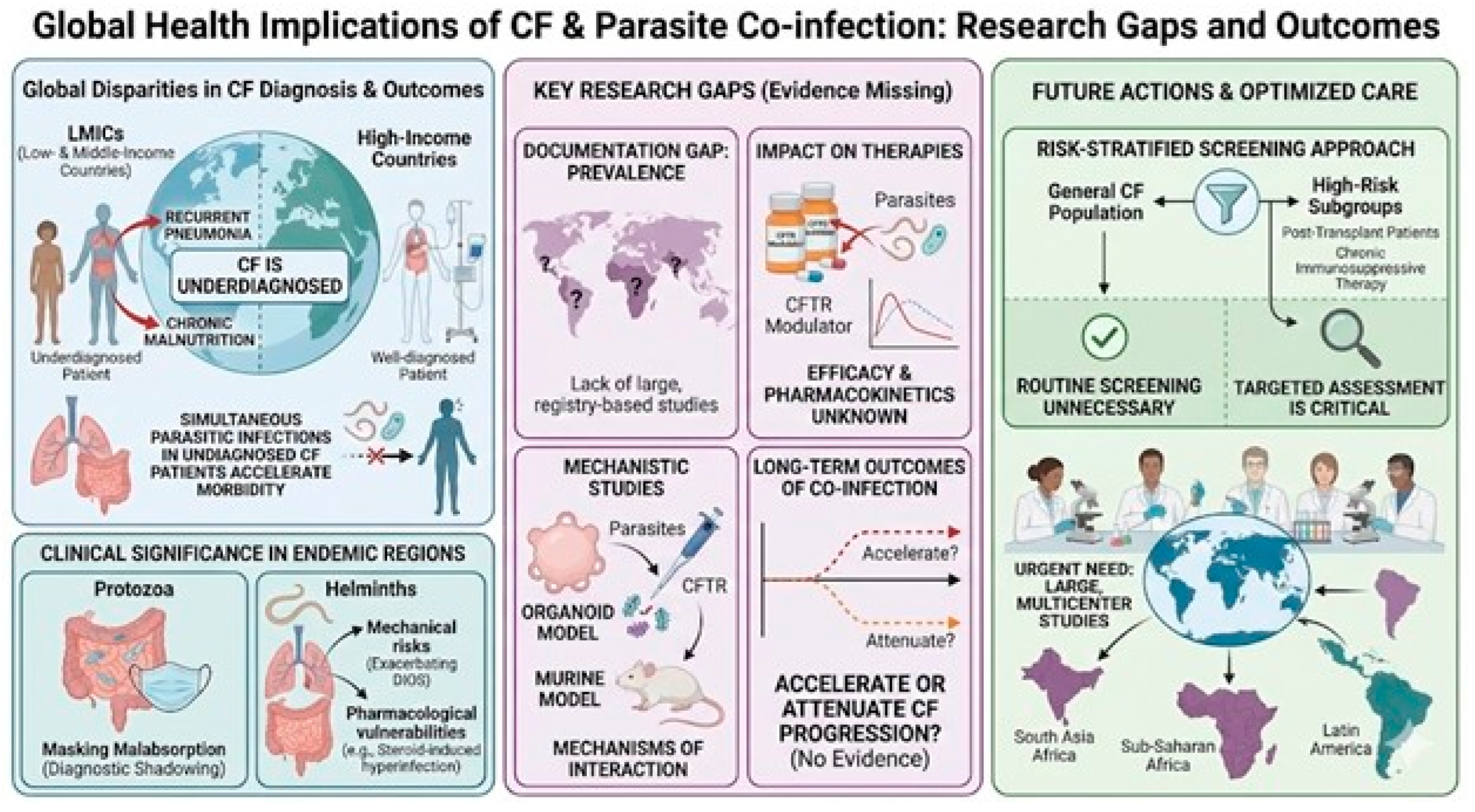
5. Research Gaps
6. Conclusion
Funding
Conflicts of Interest
References
- CFF. Annual report. 2023. Available online: https://www.cff.org/about-us/2023-annual-report.
- Abubakar Bobbo, K.; Ahmad, U.; Chau, D.M.; Nordin, N.; Abdullah, S. A comprehensive review of cystic fibrosis in Africa and Asia. Saudi J Biol Sci. 2023, 30(7), 103685. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- El Makhzen, N.; Daimi, H.; Bouguenouch, L.; Abriel, H. The burden of cystic fibrosis in North Africa. Front Genet. 2024, 14, 1295008. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Riordan, J.R.; Rommens, J.M.; Kerem, B.; Alon, N.; Rozmahel, R.; Grzelczak, Z.; Zielenski, J.; Lok, S.; Plavsic, N.; Chou, J.L.; et al. Identification of the cystic fibrosis gene: cloning and characterization of complementary DNA. Science Erratum in: Science 1989 Sep 29;245(4925):1437. PMID: 2475911. 1989, 245(4922), 1066–73. [Google Scholar] [CrossRef]
- Rowe, S.M.; Miller, S.; Sorscher, E.J. Cystic fibrosis. N Engl J Med 2005, 352(19), 1992–2001. [Google Scholar] [CrossRef] [PubMed]
- Cutting, G.R. Cystic fibrosis genetics: from molecular understanding to clinical application. Nat Rev Genet. 2015, 16(1), 45–56. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- De Lisle, R.C.; Borowitz, D. The cystic fibrosis intestine. Cold Spring Harb Perspect Med. 2013, 3(9), a009753. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yibcharoenporn, C.; Kongkaew, T.; Worakajit, N.; Khumjiang, R.; Saetang, P.; Satitsri, S.; Rukachaisirikul, V.; Muanprasat, C. Inhibition of CFTR-mediated intestinal chloride secretion by nornidulin: Cellular mechanisms and anti-secretory efficacy in human intestinal epithelial cells and human colonoids. PLoS One. 2024, 19(12), e0314723. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Esposito, S.; De Palma, F.D.E.; Cernera, G.; Zarrilli, F.; Scialò, F.; Maiuri, M.C.; Amato, F.; Castaldo, G.; Villella, V.R. Mouse intestine as a useful model for CFTR electrophysiology function analysis. Methods Cell Biol. 2025, 197, 25–40. [Google Scholar] [CrossRef] [PubMed]
- Curley, C.E.; Mobbs, C.L.; Mroz, M.S.; McLean, M.H.; Keely, S.J. The secondary bile acid, lithocholic acid, inhibits cystic fibrosis transmembrane conductance regulator expression and activity in colonic epithelial cells. Am J Physiol Gastrointest Liver Physiol. 2026, 330(2), G110–G122. [Google Scholar] [CrossRef]
- Ballout, J.; Waeber, N.B.; Diener, M. Ionic dependency, sensitivity to transport inhibitors and signal transduction of propionate-induced anion secretion in rat caecum. Eur J Pharmacol. 2026, 30;1013, 178511. [Google Scholar] [CrossRef] [PubMed]
- Françoise, A.; Héry-Arnaud, G. The Microbiome in Cystic Fibrosis Pulmonary Disease. Genes (Basel) 2020, 11(5), 536. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Marsland, B.J.; Gollwitzer, E.S. Host–microorganism interactions in lung diseases. Nat. Rev Immunol. 2014, 14, 827–835. [Google Scholar] [CrossRef]
- Anand, S.; Mande, S.S. Diet, microbiota and gut-lung connection. Front. Microbiol. 2018, 9, 2147. [Google Scholar] [CrossRef]
- Zhang, D.; Li, S.; Wang, N.; Tan, H.-Y.; Zhang, Z.; Feng, Y. The cross-talk between gut microbiota and lungs in common lung diseases. Front. Microbiol. 2020, 11, 301. [Google Scholar] [CrossRef]
- Bazett, M.; Bergeron, M.-E.; Haston, C.K. Streptomycin treatment alters the intestinal microbiome, pulmonary T cell profile and airway hyperresponsiveness in a cystic fibrosis mouse model. Sci. Rep. 2016, 6, 19189. [Google Scholar] [CrossRef]
- Nielsen, S.; Needham, B.; Leach, S.T.; Day, A.S.; Ja e, A.; Thomas, T.; Ooi, C.Y. Disrupted progression of the intestinal microbiota with age in children with cystic fibrosis. Sci. Rep. 2016, 6, 24857. [Google Scholar] [CrossRef]
- Stanton, B.A.; Coutermarsh, B.; Barnaby, R.; Hogan, D. Pseudomonas aeruginosa Reduces VX-809 Stimulated F508del-CFTR Chloride Secretion by Airway Epithelial Cells. PLOS ONE 2015, 10(5), e0127742. [Google Scholar] [CrossRef]
- World Health Organisation (WHO). Global report on neglected tropical diseases, 2025; ISBN 978-92-4-011404-3 (electronic version) ISBN 978-92-4-011405-0 (print version).
- Álvarez Fernández, A.; Traversi, L.; Polverino, E. The ageing of Cystic Fibrosis patients with new modulators: current gaps and challenges. Expert Review of Respiratory Medicine 2023, 17(12), 1091–1094. [Google Scholar] [CrossRef] [PubMed]
- Kley, A.C.; White, A.C., Jr. Parasitic Infections in Pulmonary and ICU Patients: Presentation, Diagnosis, and Treatment. Chest 2025, 167(3), 686–693. [Google Scholar] [CrossRef] [PubMed]
- Felipe Montiel, A.; Parisi, G.F.; Papale, M.; Pecora, G.; Presti, S.; Tosto, M.; Mulé, E.; Ornato, V.; Aloisio, D.; Leonardi, S. Evolving Cystic Fibrosis Care: Lung Immunology and Emerging Health Challenges in the Era of CFTR Modulators. Biomolecules 2025, 15(10), 1460. [Google Scholar] [CrossRef]
- Slae, M.; Wilschanski, M. Cystic fibrosis and the gut. Frontline Gastroenterol 2020, 12(7), 622–628. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wang, H.; Antinozzi, P.A.; Hagenfeldt, K.A.; Maechler, P.; Wollheim, C.B. Molecular targets of a human HNF1α mutation responsible for pancreatic β-cell dysfunction. The EMBO Journal 2000, 19(16), 4257–64. [Google Scholar] [CrossRef]
- Scull CE, Luo M, Jennings S, Taylor CM, & Wang G. Cftr deletion in mouse epithelial and immune cells differentially influences the intestinal microbiota. Communications Biology 2022, 5(1), 1130. [CrossRef]
- Delpire, E. Advances in the development of novel compounds targeting cation-chloride cotransporter physiology. American Journal of Physiology-Cell Physiology 2021, 320(3), C324–C340. [Google Scholar] [CrossRef]
- Alberini G, Alexis Paz S, Corradi B, Abrams CF, Benfenati F, & Maragliano L. Molecular dynamics simulations of ion permeation in human voltage-gated sodium channels. Journal of Chemical Theory and Computation 2023, 19(10), 2953–2972. [CrossRef]
- Castagna A, Mango G, Martinelli N, Marzano L, Moruzzi S, Friso S, & Pizzolo F. Sodium chloride cotransporter in hypertension. Biomedicines 2024, 12(11), 2580. [CrossRef]
- Aksel-Uylar, A.A.; Topcuoglu, C.; Saglam, M.; Vardar-Yagli, N. Effects of inspiratory muscle training on physical fitness in cystic fibrosis: a systematic review and meta-analysis. BMC Sports Sci Med Rehabil. 2025, 17(1), 384. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Aziz, W.; Sultana, H.; Kumar, V.; Tyagi, A. The Relationship Between NETosis and Biofilm Formation in Chronic Infections. Biomolecules 2025, 15(12), 1692. [Google Scholar] [CrossRef]
- Bateman, G.; Guo-Parke, H.; Harvey, C.; Rodgers, A.; Krasnodembskaya, A.; Linden, D.; et al. Bronchial epithelial cell-derived extracellular vesicles drive inflammasome activation and NTHi infection in COPD. Front Immunol. 2026, 2;16, 1713012. [Google Scholar] [CrossRef]
- Tiringer, K.; Treis, A.; Fucik, P.; Gona, M.; Gruber, S.; Renner, S.; et al. A Th17- and Th2-skewed cytokine profile in cystic fibrosis lungs represents a potential risk factor for Pseudomonas aeruginosa infection. Am J Respir Crit Care Med. 2013, 187(6), 621–9. [Google Scholar] [CrossRef] [PubMed]
- Scull, C.E.; Hu, Y.; Jennings, S.; Wang, G. Normalization of Cystic Fibrosis Immune System Reverses Intestinal Neutrophilic Inflammation and Significantly Improves the Survival of Cystic Fibrosis Mice. Cell Mol Gastroenterol Hepatol. 2025, 19(2), 101424. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Goswami, D.; Gabriel, S.E.; Cotton, C.U. Bicarbonate and CFTR: Implications for cystic fibrosis. Journal of Cystic Fibrosis 2017, 16 (Suppl 2), S45–S51. [Google Scholar] [CrossRef]
- Yamada, A.; Komaki, Y.; Komaki, F.; Micic, D.; Zullow, S.; Sakuraba, A. Risk of gastrointestinal cancers in patients with cystic fibrosis: a systematic review and meta-analysis. Lancet Oncol. 2018, 19(6), 758–767. [Google Scholar] [CrossRef]
- Lazzarotto, E.S.; Vasco, J.F.M.; Führ, F.; Riedi, C.A.; Filho, N.A.R. Systematic review on fecal calprotectin in cystic fibrosis. J Pediatr (Rio J). 2023, 99(1), 4–10. [Google Scholar] [CrossRef] [PubMed]
- McDonald, C.M.; Reid, E.K.; Pohl, J.F.; Yuzyuk, T.K.; Padula, L.M.; Vavrina, K.; Altman, K. Cystic fibrosis and fat malabsorption: Pathophysiology of the cystic fibrosis gastrointestinal tract and the impact of highly effective CFTR modulator therapy. Nutr Clin Pract. 2024, 39 Suppl 1, S57–S77. [Google Scholar] [CrossRef] [PubMed]
- Haack, A.; Aragão, G.G.; Novaes, M.R. Pathophysiology of cystic fibrosis and drugs used in associated digestive tract diseases. World J Gastroenterol. 2013, 19(46), 8552–61. [Google Scholar] [CrossRef]
- Hemphill, A.; Müller, N.; Müller, J. Comparative Pathobiology of the Intestinal Protozoan Parasites Giardia lamblia, Entamoeba histolytica, and Cryptosporidium parvum. Pathogens. 2019, 8(3), 116. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chabe, M.; Lokmer, A.; Segurel, L. Gut protozoa: Friends or foes of the human gut microbiota? Trends Parasitol. 2017, 33, 925–934. [Google Scholar] [CrossRef]
- Huang, D.B.; White, A.C. An updated review on Cryptosporidium and Giardia. Gastroenterol. Clin. North Am. 2006, 35, 291–314. [Google Scholar] [CrossRef]
- Nakada-Tsukui, Kumiko; Nozaki, Tomoyoshi. Immune Response of Amebiasis and Immune Evasion by Entamoeba histolytica. Frontiers in Immunology 2016, 7. [Google Scholar] [CrossRef]
- Gonzalez-Ruiz, A.; Wright, S.G. Disparate amoebae. Lancet 1998, 351, 1672–1673. [Google Scholar] [CrossRef]
- Van Voorhis, W. Protozoan infections. Sci. Am. Med. 2014. [Google Scholar] [CrossRef]
- Checkley, W.; White, A.C., Jr.; Jaganath, D.; Arrowood, M.J.; Chalmers, R.M.; Chen, X.M.; et al. A review of the global burden, novel diagnostics, therapeutics, and vaccine targets for Cryptosporidium. Lancet. Infect. Dis. 2015, 15, 85–94. [Google Scholar] [CrossRef]
- Koh, W.H.; Geurden, T.; Paget, T.; O’Handley, R.; Steuart, R.F.; Thompson, R.C.; Buret, A.G. Giardia duodenalis assemblage-specific induction of apoptosis and tight junction disruption in human intestinal epithelial cells: Effects of mixed infections. J. Parasitol. 2013, 99, 353–358. [Google Scholar] [CrossRef]
- Cotton, J.A.; Beatty, J.K.; Buret, A.G. Host parasite interactions and pathophysiology in Giardia infections. Int. J. Parasitol. 2011, 41, 925–933. [Google Scholar] [CrossRef] [PubMed]
- Scott, K.G.; Logan, M.R.; Klammer, G.M.; Teoh, D.A.; Buret, A.G. Jejunal brush border microvillous alterations in Giardia muris-infected mice: Role of T lymphocytes and interleukin-6. Infect. Immun. 2000, 68, 3412–3418. [Google Scholar] [CrossRef]
- Dunn, N.; Juergens, A.L. Giardiasis: Diagnosis, Pathogenesis and Treatment. StatPearls / International Journal of Health & Medical Research 2024, 3(6). [Google Scholar]
- Ribeiro, M.R.S.; Carvalho, C.F.; Gomes, M.A.; Cassali, G.D.; Oliveira, D.R. Fructooligosaccharides (FOS) intake modulates oxidative stress and restores gut morphology in Giardia lamblia-infected gerbils. An Acad Bras Cienc. 2025, 97(4), e20250346. [Google Scholar] [CrossRef] [PubMed]
- Fink, M.Y.; Singer, S.M. The Intersection of Immune Responses, Microbiota, and Pathogenesis in Giardiasis. Trends Parasitol. 2017, 33, 901–913. [Google Scholar] [CrossRef] [PubMed]
- Allain, T.; Fekete, E.; Buret, A.G. Giardia cysteine proteases: The teeth behind the smile. Trends Parasitol. 2019, 35, 636–648. [Google Scholar] [CrossRef]
- Cotton, J.A.; Beatty, J.K.; Buret, A.G. Host-parasite interactions and pathophysiology in Giardia infections. Int. J. Parasitol. 2011, 41, 925–933. [Google Scholar] [CrossRef]
- Schneider, C.; O’Leary, C.E.; von Moltke, J.; Liang, H.E.; Ang, Q.Y.; Turnbaugh, P.J.; et al. A metabolite-triggered tuft cell-ILC2 circuit drives small intestinal remodeling. Cell 2018, 174, 271–284. [Google Scholar] [CrossRef]
- Kamda, J.D.; Nash, T.E.; Singer, S.M. Giardia duodenalis: Dendritic cell defects in IL-6-deficient mice contribute to susceptibility to intestinal infection. Exp. Parasitol. 2012, 130, 288–291. [Google Scholar] [CrossRef]
- Saghaug, C.S.; Sornes, S.; Peirasmaki, D.; Svard, S.; Langeland, N.; Hanevik, K. Human memory CD4+ T cell immune responses against Giardia lamblia. Clin. Vaccine Immunol. 2015, 23, 11–18. [Google Scholar] [CrossRef]
- Stäger, S.; Gottstein, B.; Sager, H.; Jungi, T.W.; Müller, N. Influence of antibodies in mother’s milk on antigenic variation of Giardia lamblia in the murine mother-o spring model of infection. Infect. Immun. 1998, 66, 1287–1292. [Google Scholar] [CrossRef]
- Lopez-Romero, G.; Quintero, J.; Astiazaran-Garcia, H.; Velazquez, C. Host defenses against Giardia lamblia. Parasite Immunol. 2015, 37, 394–406. [Google Scholar] [CrossRef] [PubMed]
- Bénéré, E.; Van Assche, T.; Cos, P.; Maes, L. Intrinsic susceptibility of Giardia duodenalis assemblage subtypes A(I), A(II), B and E(III) for nitric oxide under axenic culture conditions. Parasitol. Res. 2012, 110, 1315–1319. [Google Scholar] [CrossRef] [PubMed]
- Holthaus, D.; Kraft, M.R.; Krug, S.M.; Wolf, S.; Müller, A.; Delgado Betancourt, E.; et al. Dissection of Barrier Dysfunction in Organoid-Derived Human Intestinal Epithelia Induced by Giardia duodenalis. Gastroenterology. 2022, 162(3), 844–858. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, V.P.; Nash, T.E.; Hubbard, V.S. Severe giardiasis in a patient with cystic fibrosis. J Pediatr Gastroenterol Nutr. 1985, 4(2), 320–2. [Google Scholar] [CrossRef] [PubMed]
- Roberts, D.M.; Craft, J.C.; Mather, F.J.; Davis, S.H.; Wright, J.A., Jr. Prevalence of giardiasis in patients with cystic fibrosis. The Journal of Paediatrics 1988, 112(4), 555–559. [Google Scholar] [CrossRef]
- Baxter, P.S.; Dickson, J.A.; Variend, S.; Taylor, C.J. Intestinal disease in cystic fibrosis. Arch Dis Child. 1988, 63(12), 1496–7. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lopez, J.; Wright, A.J.; Hammer, R.A.; Atilla, E. Chronic Pancreatitis is Associated with a High Prevalence of Giardiasis. Canadian Journal of Gastroenterology 1992, 6, 73–76. [Google Scholar] [CrossRef]
- Hernell, O.; Ward, H.; Bläckberg, L.; Pereira, M.E. Killing of Giardia lamblia by human milk lipases: an effect mediated by lipolysis of milk lipids. J Infect Dis. 1986, 153(4), 715–20. [Google Scholar] [CrossRef] [PubMed]
- Klimczak, S.; Packi, K.; Rudek, A.; Wenclewska, S.; Kurowski, M.; Kurczabińska, D.; Śliwińska, A. The Influence of the Protozoan Giardia lamblia on the Modulation of the Immune System and Alterations in Host Glucose and Lipid Metabolism. Int J Mol Sci. 2024, 25(16), 8627. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ahmadpour, E.; Safarpour, H.; Xiao, L.; Zarean, M.; Hatam-Nahavandi, K.; Barac, A.; et al. Cryptosporidiosis in HIV-positive patients and related risk factors: A systematic review and meta-analysis. Parasite 2020, 27, 27. [Google Scholar] [CrossRef]
- Piazzesi, A.; Pane, S.; Russo, A.; Del Chierico, F.; Francalanci, P.; Cotugno, N. Case Report: The impact of severe cryptosporidiosis on the gut microbiota of a pediatric patient with CD40L immunodeficiency. Front Cell Infect Microbiol. 2023, 13, 1281440. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Dupuy, F.; Valot, S.; Dalle, F.; Sterin, A.; L’Ollivier, C. Disseminated Cryptosporidium infection in an infant with CD40L deficiency. IDCases 2021, 24, e01115. [Google Scholar] [CrossRef]
- Fayer, R.; Ungar, B.L. Cryptosporidium spp. and cryptosporidiosis. Microbiol Rev. 1986, 50(4), 458–83. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chen, X.M.; Keithly, J.S.; Paya, C.V.; LaRusso, N.F. Cryptosporidiosis. N Engl J Med. 2002, 346(22), 1723–1731. [Google Scholar] [CrossRef]
- Bouzid, M.; Hunter, P.R.; Chalmers, R.M.; Tyler, K.M. Cryptosporidium pathogenicity and virulence. Clin Microbiol Rev. 2013, 26(1), 115–134. [Google Scholar] [CrossRef] [PubMed]
- Sponseller, J.K.; Griffiths, J.K.; Tzipori, S. The evolution of respiratory Cryptosporidiosis: evidence for transmission by inhalation. Clin Microbiol Rev. 2014, 27(3), 575–86. [Google Scholar] [CrossRef]
- Chaudhary, H.; Cochrane, A.B.; Tanna, S.; Aryal, S.; Kattih, Z.; Wilkinson, J.D.; et al. A Case Series of Lung Transplant Recipients Who Developed Life-Threatening Diarrhoea from Cryptosporidium Contaminated Water Supply. Archives of Clinical and Medical Case Reports 2026, 10, 06–10. [Google Scholar] [CrossRef]
- Perkins, M.E.; Riojas, Y.A.; Wu, T.W.; Le Blancq, S.M. CpABC, a Cryptosporidium parvum ATP-binding cassette protein at the host-parasite boundary in intracellular stages. Proc Natl Acad Sci U S A 1999, 96(10), 5734–9. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Turkeltaub, J.A.; McCarty, TR3rd; Hotez, P.J. The intestinal protozoa: Emerging impact on global health and development. Curr. Opin. Gastroenterol. 2015, 31, 38–44. [Google Scholar] [CrossRef]
- Iyer, L.R.; Verma, A.K.; Paul, J.; Bhattacharya, A. Phagocytosis of Gut Bacteria by Entamoeba histolytica. Front. Cell. Infect. Microbiol. 2019, 9, 34. [Google Scholar] [CrossRef]
- Guillen, N. The interaction between Entamoeba histolytica and enterobacteria sheds light on an ancient antibacterial response. Cell. Microbiol. 2019, 21, e13039. [Google Scholar] [CrossRef] [PubMed]
- Leon-Coria, A.; Kumar, M.; Chadee, K. The delicate balance between Entamoeba histolytica, mucus and microbiota. Gut Microbes 2020, 11, 118–125. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



