Submitted:
18 April 2026
Posted:
20 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Subjects
| Characteristics | (Mean ± SD) |
| Age (years) | 19.6 ± 1.5 |
| Height (cm) | 175.6 ± 3.5 |
| Weight (kg) | 78.3 ± 13.8 |
| Note: SD = Standard Deviation | |
2.2. Experimental Design
2.2.1. Overview
2.2.2. One Repetition Maximum Back Squat Assessment
2.2.3. Main Experiment - Back Squat Protocol
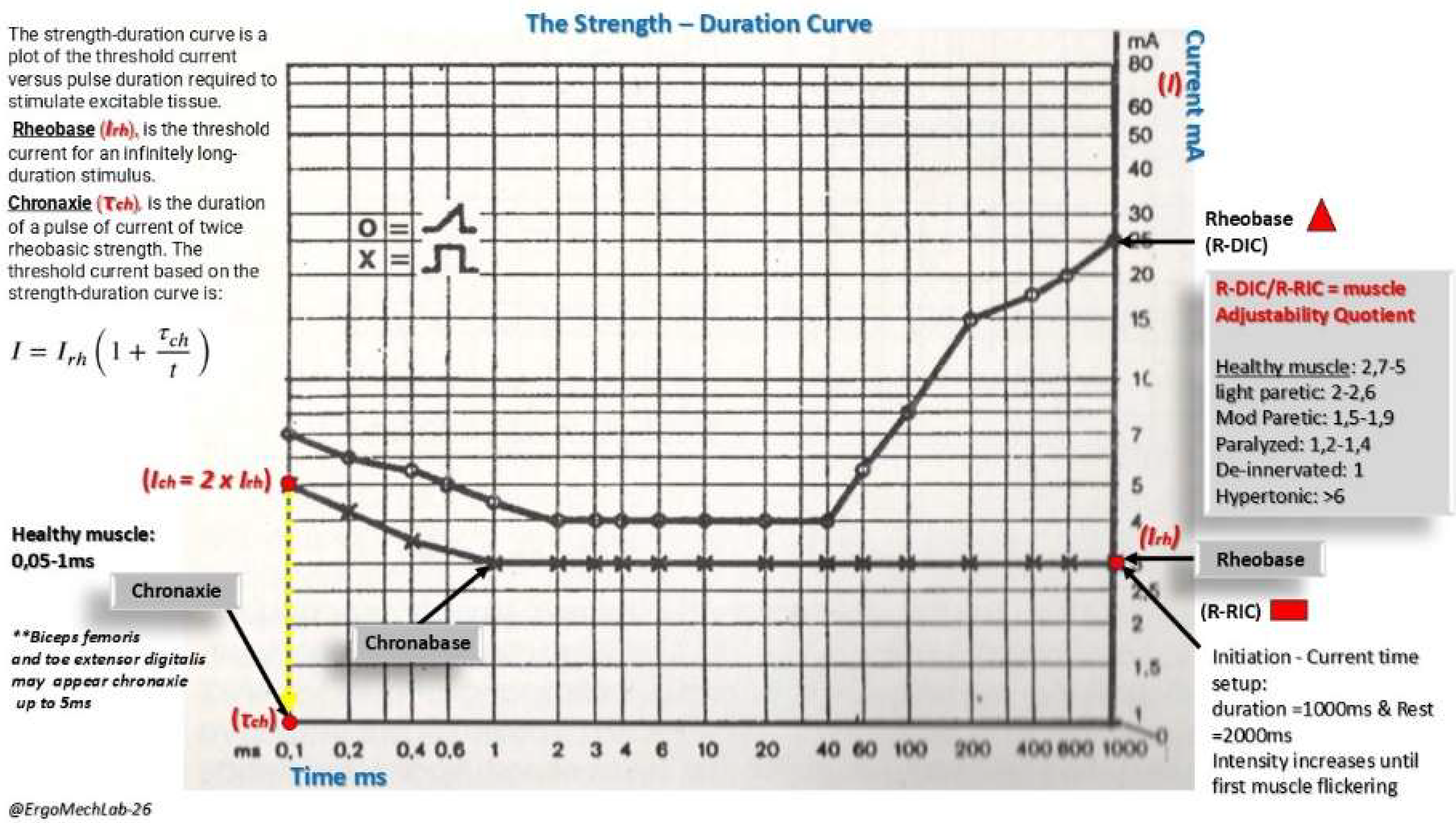
2.2.4. Collection of Rheobase Parameters and Chronaxie
2.3. Formulas for Threshold Charge and Muscle Adjustability Quotient
2.4. Statistical Analysis
3. Results
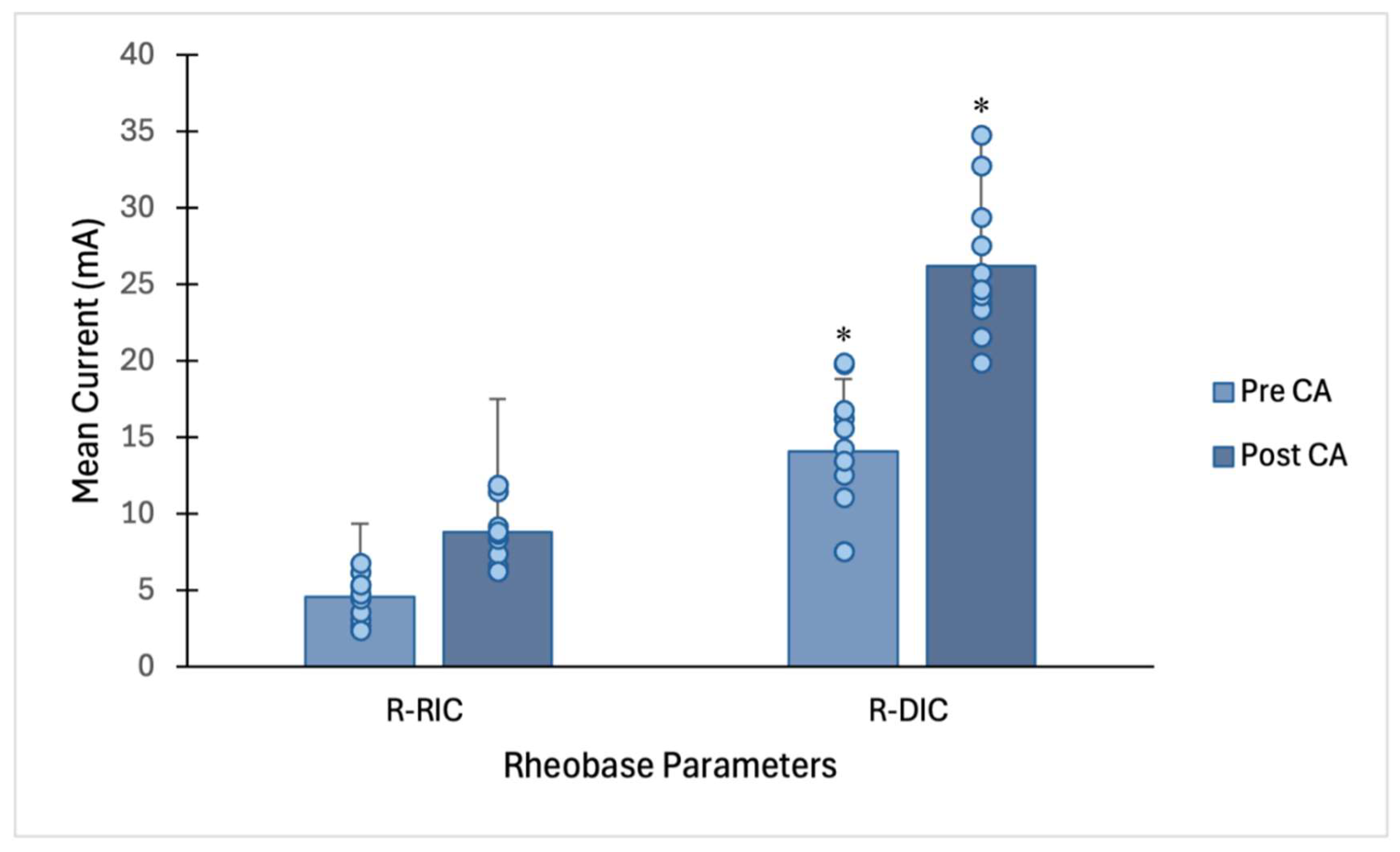
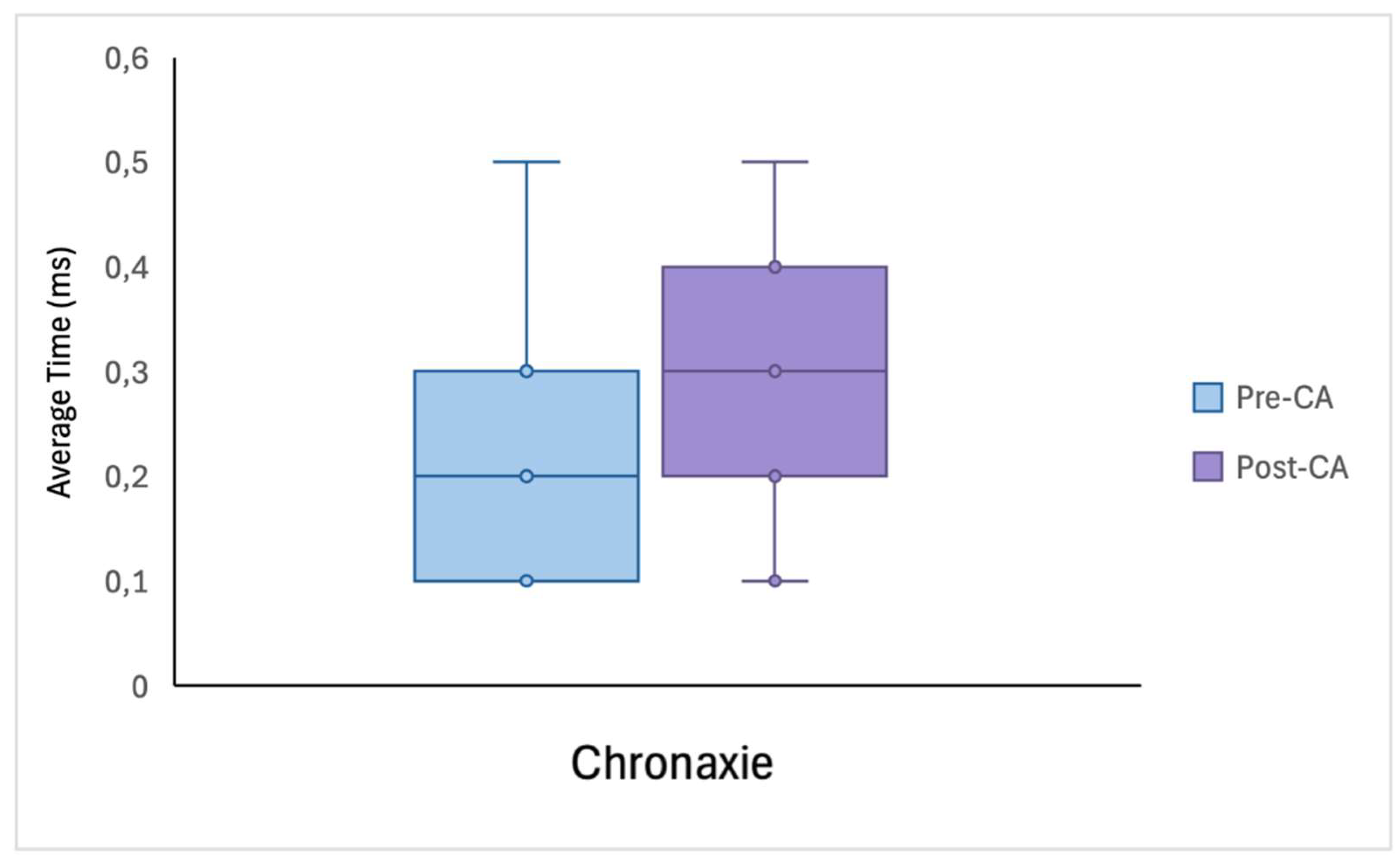
3.1. Rheobase Parameters and Chronaxie
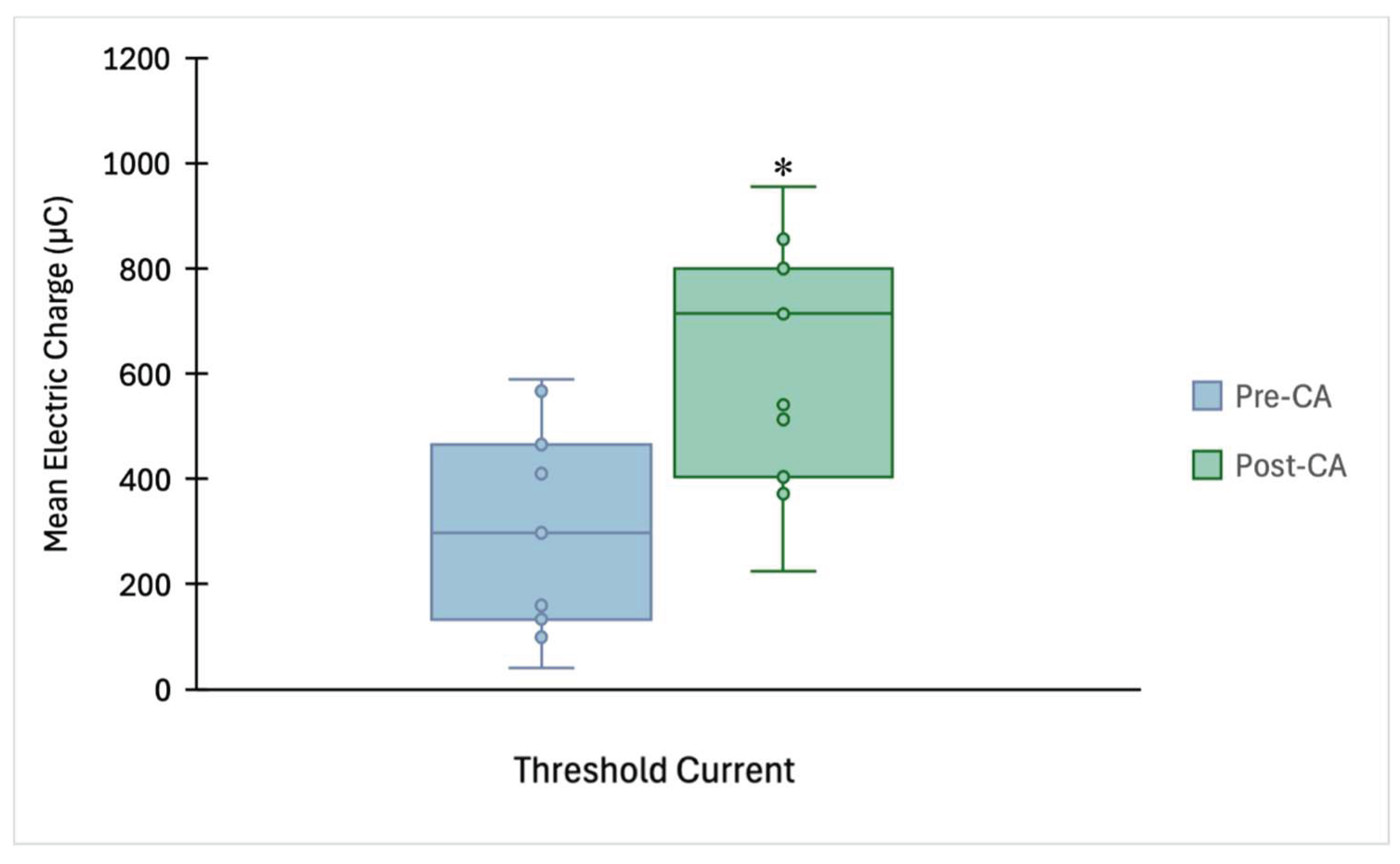
3.2. Threshold Charge and Muscle Adjustability Quotient
4. Discussion
4.1. Practical Applications
4.2. Limitations and Future Recommendations
5. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| [K+]0 | Extracellular Potassium Concentrations |
| [K+]i | Intracellular Potassium Concentrations |
| 1 RM | One-Repetition Maximum |
| BS | Back Squat |
| CA | Conditioning Activity |
| Ca+ | Calcium Ion |
| CNS | Central Nervous System |
| EEG | Electroencephalogram |
| M-wave | Compound Muscle Action Potential |
| MAQ | Muscle Adjustability Quotient |
| MRLC | Myosin Regulatory Light Chain |
| MVC | Maximum Voluntary Contraction |
| NMJ | Neuromuscular Junction |
| PAP | Post-Activation Potentiation |
| PAPE | Post-Activation Performance Enhancement |
| PTT | Peak Twitch Torque |
| Q | Threshold Charge |
| R-DIC | Triangular Rheobase |
| R-RIC | Rectangular Rheobase |
| Reps | Repetitions |
| S-D | Strength-Duration |
| SD | Standard Deviations |
| SDC | Strength-Duration Curve |
| SR | Sarcoplasmic Reticulum |
| TMS | Transcranial Magnetic Stimulation |
| VGSCs | Voltage-Gated Sodium Channels |
| VM | Vastus Medialis |
| μC | Microcoulomb |
References
- Plant, D.R.; Lynch, G.S. Excitation-contraction coupling and sarcoplasmic reticulum function in lechanically skinned fibres frog fast skeletal muscles of aged mice. The Journal of Physiology 2002, 543, 169–76. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Ochoa, E.O.; Schneider, M.F. Voltage sensing mechanism in skeletal muscle excitation-contraction coupling: coming of age or midlife crisis? Skeletal Muscle 2018, 8, 22. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Dudko, O.K. A theory of synaptic transmission. eLife 2021, 10, e73585. [Google Scholar] [CrossRef]
- Stull, J.T.; Kamm, K.E.; Vandenboom, R. Myosin light chain kinase and the role of myosin light chain phosphorylation in skeletal muscle. Arch Biochem Biophys. 2011, 510, 120–8. [Google Scholar] [CrossRef]
- Pitake, S.; Ochs, R.S. Membrane depolarization increases ryanodine sensitivity to Ca2+ release to the cytosol in L6 skeletal muscle cells: Implications for excitation–contraction coupling. Exp Biol Med (Maywood) 2016, 241, 854–62. [Google Scholar] [CrossRef]
- Allen, D.G.; Lamb, G.D.; Westerblad, H. Skeletal muscle fatigue: cellular mechanisms. Physiol Rev. 2008, 88, 287–332. [Google Scholar] [CrossRef]
- Cairns, S.P.; Taberner, A.J.; Loiselle, D.S. Changes of surface and t-tubular membrane excitability during fatigue with repeated tetani in isolated mouse fast- and slow-twitch muscle. Journal of Applied Physiology 2009, 106, 101–12. [Google Scholar] [CrossRef]
- Fauler, M.; Jurkat-Rott, K.; Lehmann-Horn, F. Membrane excitability and excitation–contraction uncoupling in muscle fatigue. Neuromuscular Disorders 2012, 22, 162–7. [Google Scholar] [CrossRef]
- Padilla, C.J.; Harrigan, M.E.; Harris, H.; Schwab, J.M.; Rutkove, S.B.; Rich, M.M.; et al. Correction to: Profiling age-related muscle weakness and wasting: neuromuscular junction transmission as a driver of age-related physical decline. GeroScience 2021, 43, 2835–6. [Google Scholar] [CrossRef] [PubMed]
- Miao, Y.; Xie, L.; Song, J.; Cai, X.; Yang, J.; Ma, X.; et al. Unraveling the causes of sarcopenia: Roles of neuromuscular junction impairment and mitochondrial dysfunction. Physiological Reports 2024, 12, e15917. [Google Scholar] [CrossRef]
- Kwai, N.C.G.; Arnold, R.; Wickremaarachchi, C.; Lin, C.S.Y.; Poynten, A.M.; Kiernan, M.C.; et al. Effects of Axonal Ion Channel Dysfunction on Quality of Life in Type 2 Diabetes. Diabetes Care 2013, 36, 1272–7. [Google Scholar] [CrossRef]
- Silva, P.E.; Maldaner, V.; Vieira, L.; De Carvalho, K.L.; Gomes, H.; Melo, P.; et al. Neuromuscular electrophysiological disorders and muscle atrophy in mechanically-ventilated traumatic brain injury patients: New insights from a prospective observational study. Journal of Critical Care 2018, 44, 87–94. [Google Scholar] [CrossRef]
- Wróbel, B.; Dolibog, P.; Penkala, A.; Kierszniok, K.; Król, P. The Determination of Normative Values for the Median Nerve Using Classic Electrodiagnostic Methods. Med Rehabil. 2022, 25, 9–14. [Google Scholar] [CrossRef]
- Lee, W.D.; Kim, J.H.; Lee, J.U.; Kim, M.Y.; Lee, L.K.; Yang, S.M.; et al. Differences in Rheobase and Chronaxie between the Paretic and Non-Paretic Sides of Hemiplegic Stroke Patients: a Pilot Study. J Phys Ther Sci. 2013, 25, 717–9. [Google Scholar] [CrossRef] [PubMed]
- Bostock, H. The strength-duration relationship for excitation of myelinated nerve: computed dependence on membrane parameters. The Journal of Physiology 1983, 341, 59–74. [Google Scholar] [CrossRef]
- Smit, J.E.; Hanekom, T.; Hanekom, J.J. Predicting action potential characteristics of human auditory nerve fibres through modification of the Hodgkin–Huxley equations. South African Journal of Science 2008, 104, 284–292. [Google Scholar]
- Arnold, D.; Thielker, J.; Klingner, C.M.; Puls, W.C.; Misikire, W.; Guntinas-Lichius, O.; et al. Selective Surface Electrostimulation of the Denervated Zygomaticus Muscle. Diagnostics 2021, 11, 188. [Google Scholar] [CrossRef] [PubMed]
- De-la-Cruz-Torres, B.; Abuín-Porras, V.; Navarro-Flores, E.; Calvo-Lobo, C.; Romero-Morales, C. Ultrasound-Guided Percutaneous Neuromodulation in Patients with Chronic Lateral Epicondylalgia: A Pilot Randomized Clinical Trial. IJERPH 2021, 18, 4877. [Google Scholar] [CrossRef]
- Geddes, L.A. Accuracy limitations of chronaxie values. IEEE Trans Biomed Eng. 2004, 51, 176–81. [Google Scholar] [CrossRef] [PubMed]
- Salian, S.; Tulsankar, G. Strength-duration curve: A measure for assessing pain in trapezius spasm. Indian J Pain. 2017, 31, 41. [Google Scholar] [CrossRef]
- Abalkhail, T.M.; MacDonald, D.B.; AlThubaiti, I.; AlOtaibi, F.A.; Stigsby, B.; Mokeem, A.A.; et al. Intraoperative direct cortical stimulation motor evoked potentials: Stimulus parameter recommendations based on rheobase and chronaxie. Clinical Neurophysiology 2017, 128, 2300–8. [Google Scholar] [CrossRef]
- Krarup, C.; Moldovan, M. Nerve conduction and excitability studies in peripheral nerve disorders. Current Opinion in Neurology 2009, 22, 460–6. [Google Scholar] [CrossRef]
- Fortune, E.; Lowery, M.M. Effect of membrane properties on skeletal muscle fiber excitability: a sensitivity analysis. Med Biol Eng Comput. 2012, 50, 617–29. [Google Scholar] [CrossRef] [PubMed]
- Boërio, D.; Bostock, H.; Spescha, R.; Z’Graggen, W.J. Potassium and the Excitability Properties of Normal Human Motor Axons In Vivo. PLoS ONE 2014, 9, e98262. [Google Scholar] [CrossRef]
- Kiernan, M.C.; Walters, R.J.L.; Andersen, K.V.; Taube, D.; Murray, N.M.F.; Bostock, H. Nerve excitability changes in chronic renal failure indicate membrane depolarization due to hyperkalaemia. Brain 2002, 125, 1366–78. [Google Scholar] [CrossRef] [PubMed]
- Weiss, G. Sur la possibilite de rendre comparables entre eux les appareils servant a l’excitation electrique. Arch Ital Biol. 1901, 35, 413. [Google Scholar]
- Mogyoros, I.; Kiernan, M.C.; Burke, D. Strength-duration properties of human peripheral nerve. Brain 1996, 119, 439–47. [Google Scholar] [CrossRef] [PubMed]
- Shellock, F.G.; Prentice, W.E. Warming-Up and Stretching for Improved Physical Performance and Prevention of Sports-Related Injuries: Sports Medicine. 1985, 2, 267–78. [Google Scholar] [CrossRef]
- Lee, J.; Park, D.; Lee, J.Y.; Park, J. Effect of Warm-Up Exercise on Functional Regulation of Motor Unit Activation during Isometric Torque Production. Journal of Human Kinetics 2024, 92, 29–41. [Google Scholar] [CrossRef]
- McGowan, C.J.; Pyne, D.B.; Thompson, K.G.; Rattray, B. Warm-Up Strategies for Sport and Exercise: Mechanisms and Applications. Sports Med. 2015, 45, 1523–46. [Google Scholar] [CrossRef]
- Blazevich, A.J.; Babault, N. Post-activation Potentiation Versus Post-activation Performance Enhancement in Humans: Historical Perspective, Underlying Mechanisms, and Current Issues. Front Physiol. 2019, 10, 1359. [Google Scholar] [CrossRef]
- Miyamoto, N.; Kanehisa, H.; Fukunaga, T.; Kawakami, Y. Effect of postactivation potentiation on the maximal voluntary isokinetic concentric torque in humans. J Strength Cond Res. 2011, 25, 186–92. [Google Scholar] [CrossRef]
- Cuenca-Fernández, F.; Smith, I.C.; Jordan, M.J.; MacIntosh, B.R.; López-Contreras, G.; Arellano, R.; et al. Nonlocalized postactivation performance enhancement (PAPE) effects in trained athletes: a pilot study. Appl Physiol Nutr Metab. 2017, 42, 1122–5. [Google Scholar] [CrossRef]
- Hodgson, M.J.; Docherty, D.; Zehr, E.P. Postactivation potentiation of force is independent of h-reflex excitability. Int J Sports Physiol Perform. 2008, 3, 219–31. [Google Scholar] [CrossRef]
- Folland, J.P.; Wakamatsu, T.; Fimland, M.S. The influence of maximal isometric activity on twitch and H-reflex potentiation, and quadriceps femoris performance. Eur J Appl Physiol. 2008, 104, 739–48. [Google Scholar] [CrossRef]
- Hamada, T.; Sale, D.G.; MacDougall, J.D.; Tarnopolsky, M.A. Postactivation potentiation, fiber type, and twitch contraction time in human knee extensor muscles. Journal of Applied Physiology 2000, 88, 2131–7. [Google Scholar] [CrossRef]
- Hicks, A.; Fenton, J.; Garner, S.; McComas, A.J. M wave potentiation during and after muscle activity. J Appl Physiol (1985) 1989, 66, 2606–10. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Falces, J.; Place, N. Sarcolemmal Excitability, M-Wave Changes, and Conduction Velocity During a Sustained Low-Force Contraction. Front Physiol. 2021, 12, 732624. [Google Scholar] [CrossRef] [PubMed]
- Gallardo, P.; Giakas, G.; Sakkas, G.K.; Tsaklis, P.V. Are Surface Electromyography Parameters Indicative of Post-Activation Potentiation/Post-Activation Performance Enhancement, in Terms of Twitch Potentiation and Voluntary Performance? A Systematic Review. JFMK 2024, 9, 106. [Google Scholar] [CrossRef] [PubMed]
- Seitz, L.B.; Trajano, G.S.; Dal Maso, F.; Haff, G.G.; Blazevich, A.J. Postactivation potentiation during voluntary contractions after continued knee extensor task-specific practice. Appl Physiol Nutr Metab. 2015, 40, 230–7. [Google Scholar] [CrossRef]
- Johnson, M.; Baudin, P.; Ley, A.L.; Collins, D.F. A Warm-Up Routine That Incorporates a Plyometric Protocol Potentiates the Force-Generating Capacity of the Quadriceps Muscles. J Strength Cond Res. 2019, 33, 380–9. [Google Scholar] [CrossRef]
- Boullosa, D. Post-activation performance enhancement strategies in sport: a brief review for practitioners. Hum Mov. 2021, 22, 101–9. [Google Scholar] [CrossRef]
- Esformes, J.I.; Bampouras, T.M. Effect of Back Squat Depth on Lower-Body Postactivation Potentiation. Journal of Strength and Conditioning Research 2013, 27, 2997–3000. [Google Scholar] [CrossRef]
- Mina, M.A.; Blazevich, A.J.; Tsatalas, T.; Giakas, G.; Seitz, L.B.; Kay, A.D. Variable, but not free-weight, resistance back squat exercise potentiates jump performance following a comprehensive task-specific warm-up. Scand J Med Sci Sports 2019, 29, 380–92. [Google Scholar] [CrossRef] [PubMed]
- Conrado De Freitas, M.; Rossi, F.E.; Colognesi, L.A.; De Oliveira, J.V.N.S.; Zanchi, N.E.; Lira, F.S.; et al. Postactivation Potentiation Improves Acute Resistance Exercise Performance and Muscular Force in Trained Men. Journal of Strength & Conditioning Research 2021, 35, 1357–63. [Google Scholar] [CrossRef]
- Fuentes Del Toro, S.; Wei, Y.; Olmeda, E.; Ren, L.; Guowu, W.; Díaz, V. Validation of a Low-Cost Electromyography (EMG) System via a Commercial and Accurate EMG Device: Pilot Study. Sensors 2019, 19, 5214. [Google Scholar] [CrossRef]
- Nguyen, A.T.; Aris, I.M.; Snyder, B.D.; Harris, M.B.; Kang, J.D.; Murray, M.; et al. Musculoskeletal health: an ecological study a ssessing disease burden and research funding. Lancet Reg Health Am. 2024, 29, 100661. [Google Scholar] [CrossRef]
- Thomas, K.; Brownstein, C.G.; Dent, J.; Parker, P.; Goodall, S.; Howatson, G. Neuromuscular Fatigue and Recovery after Heavy Resistance, Jump, and Sprint Training. Medicine & Science in Sports & Exercise 2018, 50, 2526–35. [Google Scholar] [CrossRef]
- Tsoukos, A.; Brown, L.E.; Veligekas, P.; Terzis, G.; Bogdanis, G.C. Postactivation Potentiation of Bench Press Throw Performance Using Velocity-Based Conditioning Protocols with Low and Moderate Loads. Journal of Human Kinetics 2019, 68, 81–98. [Google Scholar] [CrossRef] [PubMed]
- Eken, Ö; Mainer-Pardos, E.; Yagin, F.H.; Eken, I.; Prieto-González, P.; Nobari, H. Motoric performance variation from morning to evening: 80% intensity post-activation potentiation protocol impacts performance and its diurnal amplitude in basketball players. Front Psychol. 2022, 13, 1066026. [Google Scholar] [CrossRef]
- Botter, A.; Oprandi, G.; Lanfranco, F.; Allasia, S.; Maffiuletti, N.A.; Minetto, M.A. Atlas of the muscle motor points for the lower limb: implications for electrical stimulation procedures and electrode positioning. Eur J Appl Physiol. 2011, 111, 2461–71. [Google Scholar] [CrossRef]
- Babb, T.L.; Soper, H.V.; Lieb, J.P.; Brown, W.J.; Ottino, C.A.; Crandall, P.H. Electrophysiological studies of long-term electrical stimulation of the cerebellum in monkeys. Journal of Neurosurgery 1977, 47, 353–65. [Google Scholar] [CrossRef]
- Kloth, L.C. Electrical Stimulation Technologies for Wound Healing. Adv Wound Care (New Rochelle) 2014, 3, 81–90. [Google Scholar] [CrossRef]
- Rodríguez-Fernández, ÁL.; Rebollo-Roldán, J.; Jiménez-Rejano, J.J.; Güeita-Rodríguez, J. Strength-Duration Curves of the Common Fibular Nerve Show Hypoexcitability in People With Functional Ankle Instability. PM&R 2016, 8, 536–44. [Google Scholar] [CrossRef]
- Zagatto, A.M.; Dutra, Y.M.; Claus, G.; Malta E de, S.; de Poli, R.A.B.; Brisola, G.M.P.; et al. Drop jumps improve repeated sprint ability performance in professional basketball players. Biol Sport. 2022, 39, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Baudry, S.; Duchateau, J. Postactivation potentiation in a human muscle: effect on the rate of torque development of tetanic and voluntary isometric contractions. J Appl Physiol (1985) 2007, 102, 1394–401. [Google Scholar] [CrossRef] [PubMed]
- Bigland-Ritchie, B.; Jones, D.A.; Hosking, G.P.; Edwards, R.H. Central and peripheral fatigue in sustained maximum voluntary contractions of human quadriceps muscle. Clin Sci Mol Med. 1978, 54, 609–14. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Falces, J.; Place, N. Sarcolemmal membrane excitability during repeated intermittent maximal voluntary contractions. Exp Physiol. 2019, 104, 136–48. [Google Scholar] [CrossRef]
- Renaud, J.M.; Ørtenblad, N.; McKenna, M.J.; Overgaard, K. Exercise and fatigue: integrating the role of K+, Na+ and Cl− in the regulation of sarcolemmal excitability of skeletal muscle. Eur J Appl Physiol. 2023, 123, 2345–78. [Google Scholar] [CrossRef]
- Juel, C. Muscle action potential propagation velocity changes during activity. Muscle and Nerve 1988, 11, 714–9. [Google Scholar] [CrossRef]
- Cairns, S.P.; Flatman, J.A.; Clausen, T. Relation between extracellular [K+], membrane potential and contraction in rat soleus muscle: modulation by the Na+-K+ pump. Pflugers Arch. 1995, 430, 909–15. [Google Scholar] [CrossRef]
- Cairns, S.P.; Leader, J.P.; Higgins, A.; Renaud, J.M. The peak force-resting membrane potential relationships of mouse fast- and slow-twitch muscle. American Journal of Physiology-Cell Physiology 2022, 322, C1151–65. [Google Scholar] [CrossRef]
- Sale, D.G. Postactivation potentiation: role in human performance. Exerc Sport Sci Rev. 2002, 30, 138–43. [Google Scholar] [CrossRef]
- Robbins, D.W. Postactivation Potentiation and Its Practical Applicability: A Brief Review. J Strength Cond Res. 2005, 19, 453. [Google Scholar] [CrossRef]
- Tillin, N.A.; Bishop, D. Factors Modulating Post-Activation Potentiation and its Effect on Performance of Subsequent Explosive Activities: Sports Medicine. 2009, 39, 147–66. [Google Scholar] [CrossRef] [PubMed]
- Rocchi, L.; Brown, K.; Di Santo, A.; Smith, H.; Peterchev, A.V.; Rothwell, J.C.; et al. Distinct impacts of sodium channel blockers on the strength–duration properties of human motor cortex neurons. Epilepsia 2025, 66, 4223–34. [Google Scholar] [CrossRef] [PubMed]
- Hodgkin, A.L.; Huxley, A.F. A quantitative description of membrane current and its application to conduction and excitation in nerve. The Journal of Physiology 1952, 117, 500–44. [Google Scholar] [CrossRef]
- Catterall, W.A. Voltage-gated sodium channels at 60: structure, function and pathophysiology. The Journal of Physiology 2012, 590, 2577–89. [Google Scholar] [CrossRef]
- Xie, Y.F.; Yang, J.; Ratté, S.; Prescott, S.A. Similar excitability through different sodium channels and implications for the analgesic efficacy of selective drugs. eLife 2024, 12, RP90960. [Google Scholar] [CrossRef] [PubMed]
- Goldfarb, M. Voltage-gated sodium channel-associated proteins and alternative mechanisms of inactivation and block. Cell Mol Life Sci. 2012, 69, 1067–76. [Google Scholar] [CrossRef]
- Vandael, D.H.F.; Ottaviani, M.M.; Legros, C.; Lefort, C.; Guérineau, N.C.; Allio, A.; et al. Reduced availability of voltage-gated sodium channels by depolarization or blockade by tetrodotoxin boosts burst firing and catecholamine release in mouse chromaffin cells. The Journal of Physiology 2015, 593, 905–27. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J.; Clement, E.M.; Sumsky, S.L.; Xiang, Y.; Park, I.H.; Santaniello, S.; et al. The critical role of persistent sodium current in hippocampal gamma oscillations. Neuropharmacology 2020, 162, 107787. [Google Scholar] [CrossRef]
- Hennings, K.; Arendt-Nielsen, L.; Andersen, O.K. Breakdown of accommodation in nerve: a possible role for persistent sodium current. Theor Biol Med Model. 2005, 2, 16. [Google Scholar] [CrossRef]
- Mogyoros, I.; Lin, C.S.; Kuwabara, S.; Cappelen-Smith, C.; Burke, D. Strength-duration properties and their voltage dependence as measures of a threshold conductance at the node of Ranvier of single motor axons. Muscle Nerve 2000, 23, 1719–26. [Google Scholar] [CrossRef]
- Stern, S.; Agudelo-Toro, A.; Rotem, A.; Moses, E.; Neef, A. Chronaxie Measurements in Patterned Neuronal Cultures from Rat Hippocampus. PLoS ONE 2015, 10, e0132577. [Google Scholar] [CrossRef]
- Rattay, F.; Paredes, L.P.; Leao, R.N. Strength-duration relationship for intra- versus extracellular stimulation with microelectrodes. Neuroscience 2012, 214, 1–13. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, B.L.; Pfeiffer, E.R.; Sundnes, J.; Wall, S.T.; McCulloch, A.D. Increased cell membrane capacitance is the dominant mechanism of stretch-dependent conduction slowing in the rabbit heart: a computational study. Cell Mol Bioeng. 2015, 8, 237–46. [Google Scholar] [CrossRef]
- Dougherty, P.M.; Chen, J. Relationship of membrane properties, spike burst responses, laminar location, and functional class of dorsal horn neurons recorded in vitro. Journal of Neurophysiology 2016, 116, 1137–51. [Google Scholar] [CrossRef] [PubMed]
- Boinagrov, D.; Loudin, J.; Palanker, D. Strength-duration relationship for extracellular neural stimulation: numerical and analytical models. J Neurophysiol. 2010, 104, 2236–48. [Google Scholar] [CrossRef]
- Rozand, V.; Grosprêtre, S.; Stapley, P.J.; Lepers, R. Assessment of Neuromuscular Function Using Percutaneous Electrical Nerve Stimulation. J Vis Exp. 2015, 103, 52974. [Google Scholar] [CrossRef]
- Brümmer, V.; Schneider, S.; Strüder, H.K.; Askew, C.D. Primary motor cortex activity is elevated with incremental exercise intensity. Neuroscience 2011, 181, 150–62. [Google Scholar] [CrossRef]
- Collins, B.W.; Gale, L.H.; Buckle, N.C.M.; Button, D.C. Corticospinal excitability to the biceps brachii and its relationship to postactivation potentiation of the elbow flexors. Physiol Rep. 2017, 5, e13265. [Google Scholar] [CrossRef]
| Specific Warm Up Conditioning Protocol – For Back Squats | ||
| Intensity (% of 1RM) | Reps | Rest/set |
| <50 (20 kg) | 10 | 2 |
| 50 | 6 | 2 |
| 60-65 | 5 | 3 |
| 70-75 | 5 | 3 |
| 80 (Top Set) | 5 | |
| Parameter | Trial | Cohen’s d | p-value | |
| Pre (n = 11) | Post (n = 11) | |||
| R-RIC (mA) | 4.56 ± 1.45 | 8.83 ± 2.15 | 2.8 | <0.001 |
| R-DIC (mA) | 14.01 ± 4.2 | 26.18 ± 4.6 | 2.1 | <0.001 |
| τch (ms) | 0.20 ± 0.13 | 0.27 ± 0.13 | 0.66 | 0.054 |
| Parameter | Trial | Cohen’s d | p-value | |
| Pre (n = 11) | Post (n = 11) | |||
| Q (μC) | 293.14 ± 192.78 | 627.36 ± 231.13 | 1.4 | <0.001 |
| MAQ | 3.12 ± 0.34 | 3.05 ± 0.54 | 0.16 | 0.61 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




