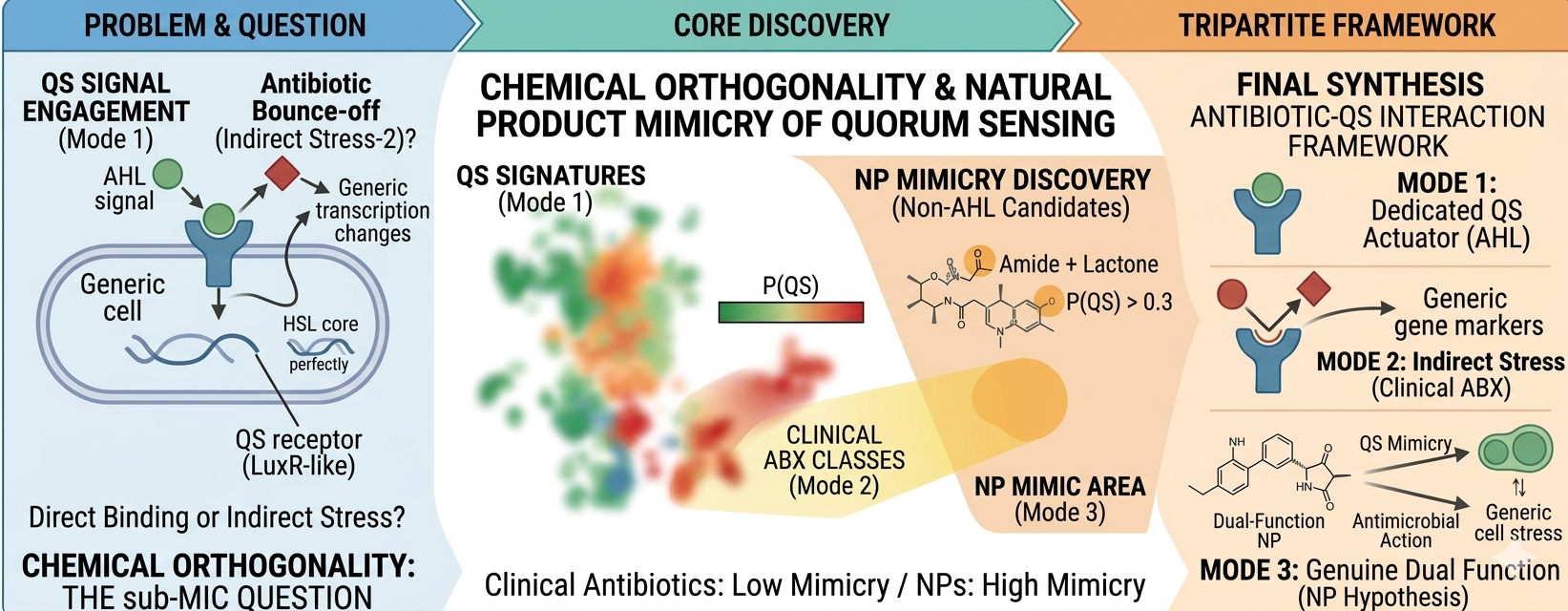
Sub-inhibitory (sub-MIC) antibiotics modulate bacterial quorum-sensing (QS) networks, but whether this modulation involves direct receptor engagement or indirect stress-mediated mechanisms remains unresolved. To address this, we trained a Random Forest classifier (ECFP4; scaffold-split AUC = 0.958; Y-randomization separation = 0.482) on 3,324 ChEMBL-curated compounds to predict engagement with LuxR-family N-acyl-homoserine lactone (AHL) receptors across six bacterial species. Clinical antibiotics (n = 54) scored near zero (mean P(QS) = 0.014), including those with documented sub-MIC QS effects (p = 0.36 vs. undocumented), suggesting that sub-MIC modulation operates via transcriptional reprogramming rather than direct binding. Large-scale screening of 36,132 antibacterial compounds (Broad Institute) confirmed a negative correlation between P(QS) and antibacterial activity (Spearman ρ = −0.086; p < 10−60), robust across species-stratified and MW-stratified analyses. Screening of 731,587 natural products (NPs) from the COCONUT database identified 41 non-AHL candidates with P(QS) > 0.3 within the applicability domain, including the confirmed QS inhibitor Honaucin A and the marine antibiotics Korormicins. Independent pharmacophore analysis against a 10-AHL reference panel confirmed greater similarity of NP candidates to AHLs relative to antibiotics (Gobbi-Tanimoto: 0.108 vs. 0.037; Mann-Whitney p = 0.006). The results demonstrate quantitative chemical orthogonality between antibacterials and QS modulators and identify NPs as priority hypotheses for experimental validation of dual function — QS modulation at sub-MIC and antibacterial activity at elevated concentrations.