Submitted:
20 April 2026
Posted:
21 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
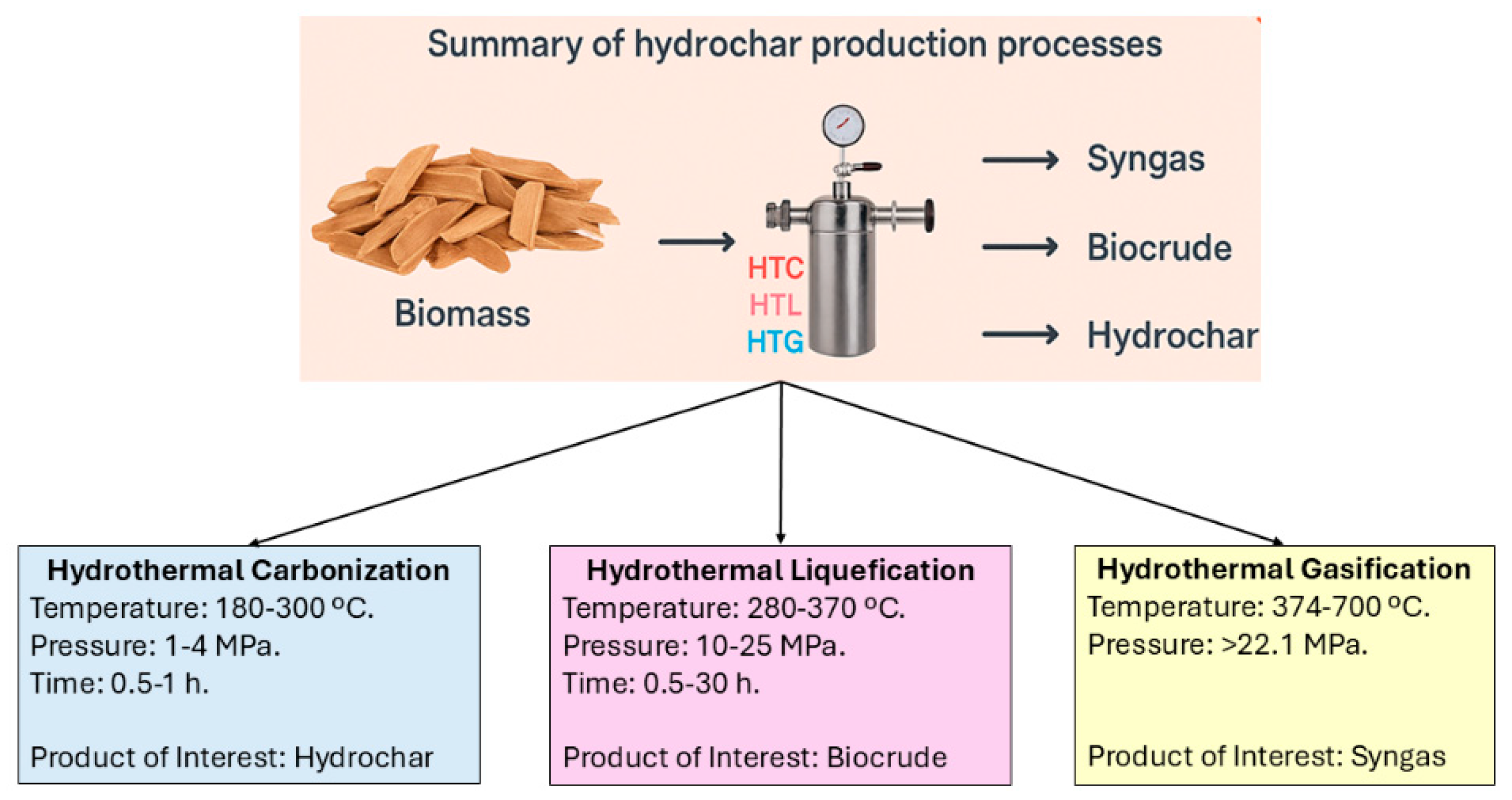
2. Thermal Conversion of Bio-Based Residues
3. Hydrothermal- Liquification, Biocrude Upgradation Techniques and Factors Affecting Biocrude Upgradation
3.1. Hydro-Processing Methods of HTL-Biocrude
3.2. Hydrodeoxygenation
4. Applications for Hydrochar Produced from Hydrothermal Liquification
4.1. Bio-Coal for Energy
4.2. Soil Enrichment
4.3. Catalysts from Hydrochar
5. Application of Artificial Intelligence and Machine Learning in Biomass Valorization
6. Future Research Opportunities
- Design-by-Specification: Setting explicit electronic targets for hydrochar catalysts (like, minimum conductivity, carrier mobility, interfacial electron-transfer rate constants) and use physics-informed models together with operando measurements to backtrack the catalyst synthesis parameters, for achieving the set targets.
- b. Metal-Support Electronic Coupling for Hydro-denitrogenation: Engineering bimetallic catalyst systems on electronically tailored hydrochars to promote C-N bond scission in refractory N- species. Further, use operando FTIR and transient methods to resolve rate-limiting steps and inhibition, extending the catalysts lifetime.
- c. Interface Engineering in Biphasic Media: Using hydrochar to stabilize Pickering emulsions responsible for co-localizing H₂ activation, acid sites, and electron transfer at water-oil boundaries. Furthermore, application of microfluidics or interfacial spectroscopy could help quantify the coupled mass and charge-transport, suppressing coking or polymerization.
- d. Data & AI Integration for Scale-Up: Combining explainable AI with physics-informed neural networks (PNN), and uncertainty quantification can guide experimental selection, while ensuring model reliability beyond lab scale.
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HTL | Hydrothermal Liquification |
| HTC | Hydrothermal Carbonization |
| HDO | Hydrodeoxygenation |
| HDS | Hydro-desulfurization |
| HDN | Hydro-denitrogenation |
| AI | Artificial Intelligence |
| ML | Machine Learning |
| TRL | Technology Readiness Level |
References
- Arodudu, O.; Holmatov, B.; Voinov, A. Ecological Impacts and Limits of Biomass Use: A Critical Review. Clean Technol. Environ. Policy 2020, 22, 1591–1611. [Google Scholar] [CrossRef]
- Igwebuike, C.M.; Awad, S.; Andrès, Y. Renewable Energy Potential: Second-Generation Biomass as Feedstock for Bioethanol Production. Molecules 2024, 29, 1619. [Google Scholar] [CrossRef]
- Chavan, S.; Yadav, B.; Atmakuri, A.; Tyagi, R.D.; Wong, J.W.C.; Drogui, P. Bioconversion of Organic Wastes into Value-Added Products: A Review. Bioresour. Technol. 2022, 344, 126398. [Google Scholar] [CrossRef]
- Okolie, J.A.; Nanda, S.; Dalai, A.K.; Kozinski, J.A. Chemistry and Specialty Industrial Applications of Lignocellulosic Biomass. Waste and Biomass Valorization 2021, 12, 2145–2169. [Google Scholar] [CrossRef]
- Popp, J.; Kovács, S.; Oláh, J.; Divéki, Z.; Balázs, E. Bioeconomy: Biomass and Biomass-Based Energy Supply and Demand. N. Biotechnol. 2021, 60, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Diop, C.A.B.; Lo, M.; Snoussi, Y.; Gam-Derouich, S.; El Garah, M.; Jouini, M.; Gningue-Sall, D.; Chehimi, M.M. Functional Hydrochar/Biochar through Thermochemical Conversion of Millet Bran from Senegal: Physicochemical, Morphological and Electrochemical Properties. Emergent Mater. 2025, 8, 2663–2678. [Google Scholar] [CrossRef]
- Huber, G.W.; Iborra, S.; Corma, A. Synthesis of Transportation Fuels from Biomass: Chemistry, Catalysts, and Engineering. Chem. Rev. 2006, 106, 4044–4098. [Google Scholar] [CrossRef] [PubMed]
- McKendry, P. Energy Production from Biomass (Part 2): Conversion Technologies. Bioresour. Technol. 2002, 83, 47–54. [Google Scholar] [CrossRef]
- Sarker, M.I.; Liu, L.; Yadav, M.P.; Yosief, H.O.; Hussain, S.A. Conversion of Renewable Biomass into Bioproducts; 2021; pp. 1–5. [Google Scholar]
- Vuppaladadiyam, A.K.; Jena, M.K.; Hakeem, I.G.; Patel, S.; Veluswamy, G.; Thulasiraman, A.V.; Surapaneni, A.; Shah, K. A Critical Review of Biochar versus Hydrochar and Their Application for H2S Removal from Biogas. Rev. Environ. Sci. Bio/Technology 2024, 23, 699–737. [Google Scholar] [CrossRef]
- Vuppaladadiyam, A.K.; Varsha Vuppaladadiyam, S.S.; Sikarwar, V.S.; Ahmad, E.; Pant, K.K.; S, M.; Pandey, A.; Bhattacharya, S.; Sarmah, A.; Leu, S.-Y. A Critical Review on Biomass Pyrolysis: Reaction Mechanisms, Process Modeling and Potential Challenges. J. Energy Inst. 2023, 108, 101236. [Google Scholar] [CrossRef]
- Nanda, S.; Pattnaik, F.; Borugadda, V.B.; Dalai, A.K.; Kozinski, J.A.; Naik, S. Catalytic and Noncatalytic Upgrading of Bio-Oil to Synthetic Fuels: An Introductory Review 2021, 1–28.
- Verdier, S.; Mante, O.D.; Hansen, A.B.; Poulsen, K.G.; Christensen, J.H.; Ammtizboll, N.; Gabrielsen, J.; Dayton, D.C. Pilot-Scale Hydrotreating of Catalytic Fast Pyrolysis Biocrudes: Process Performance and Product Analysis. Sustain. Energy Fuels 2021, 5, 4668–4679. [Google Scholar] [CrossRef]
- Zacher, A.H.; Olarte, M. V.; Santosa, D.M.; Elliott, D.C.; Jones, S.B. A Review and Perspective of Recent Bio-Oil Hydrotreating Research. Green Chem. 2014, 16, 491–515. [Google Scholar] [CrossRef]
- Tang, Z.; Lu, Q.; Zhang, Y.; Zhu, X.; Guo, Q. One Step Bio-Oil Upgrading through Hydrotreatment, Esterification, and Cracking. Ind. Eng. Chem. Res. 2009, 48, 6923–6929. [Google Scholar] [CrossRef]
- Pulungan, A.N.; Goei, R.; Kembaren, A.; Nurfajriani, N.; Sihombing, J.L.; Gea, S.; Wong, H.R.; Hasibuan, M.I.; Rahayu, R.; Tok, A.I.Y. Two Stages Upgrading of Bio-Oil through Esterification and Hydrodeoxygenation Reactions Using Fe2O3-CoO Supported Catalyst. Biomass Convers. Biorefinery 2024, 14, 20655–20664. [Google Scholar] [CrossRef]
- Brebu, M.; Ioniță, D.; Stoleru, E. Thermal Behavior and Conversion of Agriculture Biomass Residues by Torrefaction and Pyrolysis. Sci. Rep. 2025, 15, 11505. [Google Scholar] [CrossRef] [PubMed]
- Ramos, A. Integrating Waste Thermal Conversion and Lifecycle Analysis for Sustainable Energy Production: Reflecting upon Environmental and Economic Impacts. Sustain. Energy Technol. Assessments 2025, 78, 104342. [Google Scholar] [CrossRef]
- Kamran, M. Bioenergy; INC, 2021; ISBN 9780128235386. [Google Scholar]
- Biller, P.; Ross, A.B. 17 - Production of Biofuels via Hydrothermal Conversion; Elsevier Ltd, 2016; ISBN 9780081004555. [Google Scholar]
- Ramos, A. Integrating Waste Thermal Conversion and Lifecycle Analysis for Sustainable Energy Production: Reflecting upon Environmental and Economic Impacts. Sustain. Energy Technol. Assessments 2025, 78, 104342. [Google Scholar] [CrossRef]
- Makarichi, L.; Jutidamrongphan, W.; Techato, K. The Evolution of Waste-to-Energy Incineration: A Review. Renew. Sustain. Energy Rev. 2018, 91, 812–821. [Google Scholar] [CrossRef]
- Dong, J.; Tang, Y.; Nzihou, A.; Chi, Y.; Weiss-Hortala, E.; Ni, M. Life Cycle Assessment of Pyrolysis, Gasification and Incineration Waste-to-Energy Technologies: Theoretical Analysis and Case Study of Commercial Plants. Sci. Total Environ. 2018, 626, 744–753. [Google Scholar] [CrossRef]
- Khalil, M.; Berawi, M.A.; Heryanto, R.; Rizalie, A. Waste to Energy Technology: The Potential of Sustainable Biogas Production from Animal Waste in Indonesia. Renew. Sustain. Energy Rev. 2019, 105, 323–331. [Google Scholar] [CrossRef]
- Roy, P.; Dias, G. Prospects for Pyrolysis Technologies in the Bioenergy Sector: A Review. Renew. Sustain. Energy Rev. 2017, 77, 59–69. [Google Scholar] [CrossRef]
- Morris, M.; Waldheim, L. Energy Recovery from Solid Waste Fuels Using Advanced Gasification Technology. Waste Manag. 1998, 18, 557–564. [Google Scholar] [CrossRef]
- Alizad Oghyanous, F.; Eskicioglu, C. Hydrothermal Liquefaction vs. Fast/Flash Pyrolysis for Biomass-to-Biofuel Conversion: New Insights and Comparative Review of Liquid Biofuel Yield, Composition, and Properties. Green Chem. 2025, 27, 7009–7041. [Google Scholar] [CrossRef]
- de Caprariis, B.; De Filippis, P.; Petrullo, A.; Scarsella, M. Hydrothermal Liquefaction of Biomass: Influence of Temperature and Biomass Composition on the Bio-Oil Production. Fuel 2017, 208, 618–625. [Google Scholar] [CrossRef]
- Moreira-Mendoza, C.A.; Essounani-Mérida, S.; Molina-Ramírez, S.; Cortés-Reyes, M.; Herrera, C.; Larrubia, M.Á; Alemany, L.J. Biocrude Upgrading with Tandem Catalysts Hydrothermal Liquefaction of Biomass Feedstocks. Catal. Today 2026, 461, 115496. [Google Scholar] [CrossRef]
- Adams, P.; Bridgwater, T.; Lea-Langton, A.; Ross, A.; Watson, I. Biomass Conversion Technologies. In Greenhouse Gases Balances of Bioenergy Systems; Elsevier, 2018; pp. 107–139. [Google Scholar]
- Adams, P.; Bridgwater, T.; Lea-Langton, A.; Ross, A.; Watson, I. Biomass Conversion Technologies. In Greenhouse Gases Balances of Bioenergy Systems; Elsevier, 2018; pp. 107–139. [Google Scholar]
- Gai, C.; Zhang, Y.; Chen, W.-T.; Zhang, P.; Dong, Y. An Investigation of Reaction Pathways of Hydrothermal Liquefaction Using Chlorella Pyrenoidosa and Spirulina Platensis. Energy Convers. Manag. 2015, 96, 330–339. [Google Scholar] [CrossRef]
- Scarsella, M.; de Caprariis, B.; Damizia, M.; De Filippis, P. Heterogeneous Catalysts for Hydrothermal Liquefaction of Lignocellulosic Biomass: A Review. Biomass and Bioenergy 2020, 140, 105662. [Google Scholar] [CrossRef]
- Liu, W.-J.; Yu, H.-Q. Thermochemical Conversion of Lignocellulosic Biomass into Mass-Producible Fuels: Emerging Technology Progress and Environmental Sustainability Evaluation. ACS Environ. Au 2022, 2, 98–114. [Google Scholar] [CrossRef] [PubMed]
- Bao, R.; Wang, S.; Feng, J.; Duan, Y.; Liu, K.; Zhao, J.; Liu, H.; Yang, J. A Review of Hydrothermal Biomass Liquefaction: Operating Parameters, Reaction Mechanism, and Bio-Oil Yields and Compositions. Energy & Fuels 2024, 38, 8437–8459. [Google Scholar] [CrossRef]
- U.S. Energy Information Administration Biomass Explained Available online: https://www.eia.gov/energyexplained/biomass/#:~:text=Wood and wood processing waste,producing biogas (renewable natural gas).
- Ghadge, R.; Nagwani, N.; Saxena, N.; Dasgupta, S.; Sapre, A. Design and Scale-up Challenges in Hydrothermal Liquefaction Process for Biocrude Production and Its Upgradation. Energy Convers. Manag. X 2022, 14, 100223. [Google Scholar] [CrossRef]
- Biller, P.; Ross, A.B. Potential Yields and Properties of Oil from the Hydrothermal Liquefaction of Microalgae with Different Biochemical Content. Bioresour. Technol. 2011, 102, 215–225. [Google Scholar] [CrossRef]
- Li, H.; Liu, Z.; Zhang, Y.; Li, B.; Lu, H.; Duan, N.; Liu, M.; Zhu, Z.; Si, B. Conversion Efficiency and Oil Quality of Low-Lipid High-Protein and High-Lipid Low-Protein Microalgae via Hydrothermal Liquefaction. Bioresour. Technol. 2014, 154, 322–329. [Google Scholar] [CrossRef]
- Usman, M.; Cheng, S.; Boonyubol, S.; Cross, J.S. From Biomass to Biocrude: Innovations in Hydrothermal Liquefaction and Upgrading. Energy Convers. Manag. 2024, 302, 118093. [Google Scholar] [CrossRef]
- Rath, G.K.; Borugadda, V.B.; Dalai, A.K. Advancements in Co-Refining Biocrude with Conventional Crude Oil: A Comparative Review of Upgradation Techniques. In Next-Generation Biofuels; Elsevier, 2026; pp. 261–286. [Google Scholar]
- Guo, Y.; Xu, D.; Wang, S. Bio-Oil Production in Hydrothermal Liquefaction of Biomass and Upgrading of Biocrude. In Hydrothermal Processing of Biomass for Hydrogen and Bio-oil Production; Springer Nature Singapore: Singapore, 2025; pp. 89–150. [Google Scholar]
- Badoga, S.; Alvarez-Majmutov, A.; Rodriguez, J.K.; Chen, J. Upgrading of Hydrothermal Liquefaction Biocrude from Forest Residues Using Solvents and Mild Hydrotreating for Use as Co-Processing Feed in a Refinery. Energy & Fuels 2023, 37, 13104–13114. [Google Scholar] [CrossRef]
- Nath, S.; Boahene, P.; Dalai, A. Conversion of Co-Hydrothermal Liquefaction Biocrude into High-Quality Biofuels via Hydrodeoxygenation: Process Optimization and Analysis. J. Anal. Appl. Pyrolysis 2025, 192. [Google Scholar] [CrossRef]
- Marangon, B.B.; Castro, J. de S.; Ballotin, F.C.; Silva, L.S.; Assemany, P.; do Couto, E.A.; Silva, T.A.; Jesus Junior, M.M.; Ribeiro, V.J.; Ribeiro Júnior, J.I.; et al. One-Step Hydrothermal Liquefaction and Catalytic Upgrading of Wastewater-Grown Microalgae for Potential Sustainable Aviation Fuel Precursors. ACS Omega 2026, 11, 6073–6083. [Google Scholar] [CrossRef] [PubMed]
- Browning, B.; Batalha, N.; Costa Gomes, M.; Laurenti, D.; Lebaz, N.; Geantet, C.; Tayakout-Fayolle, M. Hydrotreatment of Sewage Sludge-Derived Hydrothermal Liquefaction Biocrude. Part I: Experimental. Energy & Fuels 2024, 38, 11793–11804. [Google Scholar] [CrossRef]
- Subramaniam, S.; Santosa, D.M.; Brady, C.; Swita, M.; Ramasamy, K.K.; Thorson, M.R. Extended Catalyst Lifetime Testing for HTL Biocrude Hydrotreating to Produce Fuel Blendstocks from Wet Wastes. ACS Sustain. Chem. Eng. 2021, 9, 12825–12832. [Google Scholar] [CrossRef]
- Yan, P.; Kennedy, E.M.; Rabiee, H.; Weng, Y.; Peng, H.; Ma, B.; Zhu, Z.; Stockenhuber, M. Recent Advances in Heterogeneous Catalysts for Biocrude Hydrodeoxygenation. Green Chem. 2025, 27, 3375–3397. [Google Scholar] [CrossRef]
- Wang, J.; Wang, R. Recent Advances in Simultaneous Desulfurization and Denitrogenation of Fuel Oil. Molecules 2026, 31, 279. [Google Scholar] [CrossRef] [PubMed]
- Chand, R.; Borugadda, V.B.; Dalai, A.K. Catalytic Hydrodeoxygenation of Bio-Crude and Heavy Gas Oil Blends Using Carbon-Supported Molybdenum Catalysts. Energy & Fuels 2025, 39, 10435–10451. [Google Scholar] [CrossRef]
- Morais, A.R.C.; da Costa Lopes, A.M.; Costa, P.; Fonseca, I.; Nogueira, I.N.; Oliveira, A.C.; Bogel-Lukasik, R. Cattle Fat Valorisation through Biofuel Production by Hydrogenation in Supercritical Carbon Dioxide. RSC Adv. 2014, 4, 32081. [Google Scholar] [CrossRef]
- Barroso-Martín, I.; Ballesteros-Plata, D.; Infantes-Molina, A.; Guerrero-Pérez, M.O.; Santamaría-González, J.; Rodríguez-Castellón, E. An Overview of Catalysts for the Hydrodeoxygenation Reaction of Model Compounds from Lignocellulosic Biomass. IET Renew. Power Gener. 2022, 16, 3009–3022. [Google Scholar] [CrossRef]
- Wei, H.; Wang, Z.; Li, H. Sustainable Biomass Hydrodeoxygenation in Biphasic Systems. Green Chem. 2022, 24, 1930–1950. [Google Scholar] [CrossRef]
- Vankelecom, I.F.J. Polymeric Membranes in Catalytic Reactors. Chem. Rev. 2002, 102, 3779–3810. [Google Scholar] [CrossRef]
- Qing, W.; Li, X.; Shao, S.; Shi, X.; Wang, J.; Feng, Y.; Zhang, W.; Zhang, W. Polymeric Catalytically Active Membranes for Reaction-Separation Coupling: A Review. J. Memb. Sci. 2019, 583, 118–138. [Google Scholar] [CrossRef]
- Mortensen, P.M.; Grunwaldt, J.-D.; Jensen, P.A.; Jensen, A.D. Screening of Catalysts for Hydrodeoxygenation of Phenol as a Model Compound for Bio-Oil. ACS Catal. 2013, 3, 1774–1785. [Google Scholar] [CrossRef]
- Yohe, S.L.; Choudhari, H.J.; Mehta, D.D.; Dietrich, P.J.; Detwiler, M.D.; Akatay, C.M.; Stach, E.A.; Miller, J.T.; Delgass, W.N.; Agrawal, R.; et al. High-Pressure Vapor-Phase Hydrodeoxygenation of Lignin-Derived Oxygenates to Hydrocarbons by a PtMo Bimetallic Catalyst: Product Selectivity, Reaction Pathway, and Structural Characterization. J. Catal. 2016, 344, 535–552. [Google Scholar] [CrossRef]
- Utikar, R.P.; Ranade, V. V. Intensifying Multiphase Reactions and Reactors: Strategies and Examples. ACS Sustain. Chem. Eng. 2017, 5, 3607–3622. [Google Scholar] [CrossRef]
- Wang, X.; Arai, M.; Wu, Q.; Zhang, C.; Zhao, F. Hydrodeoxygenation of Lignin-Derived Phenolics – a Review on the Active Sites of Supported Metal Catalysts. Green Chem. 2020, 22, 8140–8168. [Google Scholar] [CrossRef]
- Kim, H.; Yang, S.; Lim, Y.H.; Ha, J.-M.; Kim, D.H. Upgrading Bio-Oil Model Compound over Bifunctional Ru/HZSM-5 Catalysts in Biphasic System: Complete Hydrodeoxygenation of Vanillin. J. Hazard. Mater. 2022, 423, 126525. [Google Scholar] [CrossRef]
- Wang, Z.C.; Chen, D.; Shan, Y.Q.; Lin, L.X.; Duan, P.G. Catalytic Hydrotreatment of the High-Boiling-Point Fraction of Soybean Straw Biocrude in a Mixed Hydrogen Donor. Fuel 2022, 310, 122126. [Google Scholar] [CrossRef]
- Yang, C.; Wang, S.; Jiang, Z.; Li, J.; He, C.; Xu, T.; Xu, D. Catalytic Hydrotreatment Upgrading of Biocrude Oil Derived from Hydrothermal Liquefaction of Animal Carcass. Fuel 2022, 317, 123528. [Google Scholar] [CrossRef]
- Ebrahim, S.A.; Jankovic, M.; Jiang, X.; Kodra, O.; Toll, F.; Baranova, E.A.; Singh, D. Catalytic Hydrotreating of Food Waste-Derived Hydrothermal Liquefaction Bio-Crude: A Comparative Study of Pd/C, in-Situ Sulfided NiMo/Al2O3 and CoMo/Al2O3 Catalysts. Fuel 2026, 407, 137571. [Google Scholar] [CrossRef]
- Masoumi, S.; Dalai, A.K. NiMo Carbide Supported on Algal Derived Activated Carbon for Hydrodeoxygenation of Algal Biocrude Oil. Energy Convers. Manag. 2021, 231, 113834. [Google Scholar] [CrossRef]
- Jahromi, H.; Rahman, T.; Roy, P.; Adhikari, S. Hydrotreatment of Solvent-Extracted Biocrude from Hydrothermal Liquefaction of Municipal Sewage Sludge. Energy Convers. Manag. 2022, 263, 115719. [Google Scholar] [CrossRef]
- Achour, A.; Castello, D.; Haider, M.S.; Rosendahl, L.A. Lab-Scale Catalytic Hydrotreating of Hydrothermal Biocrude: Effects of Temperature and Space Velocity on Fuel Upgrading and Catalyst Performance. Chem. Eng. J. 2025, 520, 166233. [Google Scholar] [CrossRef]
- Badoga, S.; Alvarez-Majmutov, A.; Rodriguez, J.K.; Gieleciak, R.; Chen, J. Coprocessing Partially Hydrodeoxygenated Hydrothermal Liquefaction Biocrude from Forest Residue in the Vacuum Gas Oil Hydrocracking Process. Energy & Fuels 2023, 37, 13126–13136. [Google Scholar] [CrossRef]
- Dimitriadis, A.; Bezergianni, S. Towards Bio-Crude Refinery Integration: Hydrodeoxygenation and Co-Hydroprocessing with Light Cycle Oil. Energies 2024, 17, 6032. [Google Scholar] [CrossRef]
- Rautiainen, S.; Viertiö, T.; Vuorio, N.; Hyppönen, F.; Meca, L.; Kukula, P.; Lehtonen, J. Hydrodeoxygenation of Black Liquor HTL Oil Model Compounds in Supercritical Water. Reactions 2026, 7, 7. [Google Scholar] [CrossRef]
- Nath, S.; Boahene, P.; Dalai, A. Conversion of Co-Hydrothermal Liquefaction Biocrude into High-Quality Biofuels via Hydrodeoxygenation: Process Optimization and Analysis. J. Anal. Appl. Pyrolysis 2025, 192, 107275. [Google Scholar] [CrossRef]
- Kryeziu, A.; Slovák, V.; Parchaňská, A. Liquefaction of Cellulose for Production of Advanced Porous Carbon Materials. Polymers (Basel). 2022, 14, 1621. [Google Scholar] [CrossRef]
- Sarker, T.R.; Sarker, B.; Saha, B.; Khatun, M.L.; Dalai, A.K. Hydrothermal Liquefaction: Transforming Waste into Renewable Fuels and High-Value Bio-Chemicals. Renew. Sustain. Energy Rev. 2025, 222, 115974. [Google Scholar] [CrossRef]
- Summers, S.; Jing, Q.; Kawale, H.; Wang, Z.; Mirzaei, D.; Zhang, Y. Waste Biorefinery Concept for Production of Value-Added Products Through Hydrothermal Liquefaction Pathway: A Critical Review and Outlook. ACS ES&T Eng. 2025, 5, 2417–2449. [Google Scholar] [CrossRef]
- Feng, S.; Yuan, Z.; Leitch, M.; Shui, H.; Xu, C.C. Effects of Bark Extraction before Liquefaction and Liquid Oil Fractionation after Liquefaction on Bark-Based Phenol Formaldehyde Resoles. Ind. Crops Prod. 2016, 84, 330–336. [Google Scholar] [CrossRef]
- Feng, S.; Yuan, Z.; Leitch, M.; Xu, C.C. Adhesives Formulated from Bark Bio-Crude and Phenol Formaldehyde Resole. Ind. Crops Prod. 2015, 76, 258–268. [Google Scholar] [CrossRef]
- Ren, R.; Han, X.; Zhang, H.; Lin, H.; Zhao, J.; Zheng, Y.; Wang, H. High Yield Bio-Oil Production by Hydrothermal Liquefaction of a Hydrocarbon-Rich Microalgae and Biocrude Upgrading. Carbon Resour. Convers. 2018, 1, 153–159. [Google Scholar] [CrossRef]
- Ahmad, F.; Doddapaneni, T.R.K.C.; Toor, S.S.; Kikas, T. Reaction Mechanism and Kinetics of Hydrothermal Liquefaction at Sub- and Supercritical Conditions: A Review. Biomass 2025, 5, 9. [Google Scholar] [CrossRef]
- LeClerc, H.O.; Atwi, R.; Niles, S.F.; McKenna, A.M.; Timko, M.T.; West, R.H.; Teixeira, A.R. Elucidating the Role of Reactive Nitrogen Intermediates in Hetero-Cyclization during Hydrothermal Liquefaction of Food Waste. Green Chem. 2022, 24, 5125–5141. [Google Scholar] [CrossRef]
- Robertson, G.; Adiningtyas, K.V.; Ebrahim, S.A.; Scoles, L.; Baranova, E.A.; Singh, D. Understanding the Nature of Bio-Asphaltenes Produced during Hydrothermal Liquefaction. Renew. Energy 2021, 173, 128–140. [Google Scholar] [CrossRef]
- Hu, S.; Luo, X.; Li, Y. Polyols and Polyurethanes from the Liquefaction of Lignocellulosic Biomass. ChemSusChem 2014, 7, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Schmidt, A.; Valdez, P.; Snowden-Swan, L.; Edmundson, S. Hydrothermal Liquefaction and Upgrading of Wastewater-Grown Microalgae: 2021 State of Technology; Richland, WA (United States), 2022. [Google Scholar]
- Halleraker, H. V.; Barth, T. Quantitative NMR Analysis of the Aqueous Phase from Hydrothermal Liquefaction of Lignin. J. Anal. Appl. Pyrolysis 2020, 151, 104919. [Google Scholar] [CrossRef]
- Chen, P.H.; Venegas Jimenez, J.L.; Rowland, S.M.; Quinn, J.C.; Laurens, L.M.L. Nutrient Recycle from Algae Hydrothermal Liquefaction Aqueous Phase through a Novel Selective Remediation Approach. Algal Res. 2020, 46, 101776. [Google Scholar] [CrossRef]
- Shen, Y. A Review on Hydrothermal Carbonization of Biomass and Plastic Wastes to Energy Products. Biomass and Bioenergy 2020, 134, 105479. [Google Scholar] [CrossRef]
- Mumme, J.; Eckervogt, L.; Pielert, J.; Diakité, M.; Rupp, F.; Kern, J. Hydrothermal Carbonization of Anaerobically Digested Maize Silage. Bioresour. Technol. 2011, 102, 9255–9260. [Google Scholar] [CrossRef]
- Reza, M.T.; Andert, J.; Wirth, B.; Busch, D.; Pielert, J.; Lynam, J.G.; Mumme, J. Hydrothermal Carbonization of Biomass for Energy and Crop Production. Appl. Bioenergy 2014, 1. [Google Scholar] [CrossRef]
- Ighalo, J.O.; Akaeme, F.C.; Georgin, J.; de Oliveira, J.S.; Franco, D.S.P. Biomass Hydrochar: A Critical Review of Process Chemistry, Synthesis Methodology, and Applications. Sustainability 2025, 17, 1660. [Google Scholar] [CrossRef]
- Masoumi, S.; Borugadda, V.B.; Nanda, S.; Dalai, A.K. Hydrochar: A Review on Its Production Technologies and Applications. Catalysts 2021, 11, 939. [Google Scholar] [CrossRef]
- Liu, H.; Chen, Y.; Yang, H.; Gentili, F.G.; Söderlind, U.; Wang, X.; Zhang, W.; Chen, H. Hydrothermal Carbonization of Natural Microalgae Containing a High Ash Content. Fuel 2019, 249, 441–448. [Google Scholar] [CrossRef]
- Onyango, C.; Nyairo, W.; Shikuku, V. A Review on the Recent Advances in the Use of Hydrochar for Adsorption of Methylene Blue Dye from Aqueous Systems. Discov. Chem. 2026, 3, 20. [Google Scholar] [CrossRef]
- Chhabra, T.; Dwivedi, P.; Krishnan, V. Acid Functionalized Hydrochar as Heterogeneous Catalysts for Solventless Synthesis of Biofuel Precursors. Green Chem. 2022, 24, 898–910. [Google Scholar] [CrossRef]
- Demirbaş, A. Estimating of Structural Composition of Wood and Non-Wood Biomass Samples. Energy Sources 2005, 27, 761–767. [Google Scholar] [CrossRef]
- Mäkelä, M.; Benavente, V.; Fullana, A. Hydrothermal Carbonization of Lignocellulosic Biomass: Effect of Process Conditions on Hydrochar Properties. Appl. Energy 2015, 155, 576–584. [Google Scholar] [CrossRef]
- Liu, Z.; Quek, A.; Kent Hoekman, S.; Balasubramanian, R. Production of Solid Biochar Fuel from Waste Biomass by Hydrothermal Carbonization. Fuel 2013, 103, 943–949. [Google Scholar] [CrossRef]
- Baxter, L.L.; Miles, T.R.; Miles, T.R.; Jenkins, B.M.; Milne, T.; Dayton, D.; Bryers, R.W.; Oden, L.L. The Behavior of Inorganic Material in Biomass-Fired Power Boilers: Field and Laboratory Experiences. Fuel Process. Technol. 1998, 54, 47–78. [Google Scholar] [CrossRef]
- Afolabi, O.O.D.; Sohail, M. Comparative Evaluation of Conventional and Microwave Hydrothermal Carbonization of Human Biowaste for Value Recovery. Water Sci. Technol. 2017, 75, 2852–2863. [Google Scholar] [CrossRef]
- Zhuang, X.; Song, Y.; Zhan, H.; Yin, X.; Wu, C. Gasification Performance of Biowaste-Derived Hydrochar: The Properties of Products and the Conversion Processes. Fuel 2020, 260, 116320. [Google Scholar] [CrossRef]
- Feng, Y.; Yu, T.; Ma, K.; Xu, G.; Hu, Y.; Chen, D. Effect of Hydrothermal Temperature on the Steam Gasification Performance of Sewage Sludge: Syngas Quality and Tar Formation. Energy & Fuels 2018, 32, 6834–6838. [Google Scholar] [CrossRef]
- Álvarez-Murillo, A.; Ledesma, B.; Román, S.; Sabio, E.; Gañán, J. Biomass Pyrolysis toward Hydrocarbonization. Influence on Subsequent Steam Gasification Processes. J. Anal. Appl. Pyrolysis 2015, 113, 380–389. [Google Scholar] [CrossRef]
- Musa, U.; Castro-Díaz, M.; Uguna, C.N.; Snape, C.E. Effect of Process Variables on Producing Biocoals by Hydrothermal Carbonisation of Pine Kraft Lignin at Low Temperatures. Fuel 2022, 325, 124784. [Google Scholar] [CrossRef]
- Singh, A.; Gill, A.; Lim, D.L.K.; Kasmaruddin, A.; Miri, T.; Chakrabarty, A.; Chai, H.H.; Selvarajoo, A.; Massawe, F.; Abakr, Y.A.; et al. Feasibility of Bio-Coal Production from Hydrothermal Carbonization (HTC) Technology Using Food Waste in Malaysia. Sustainability 2022, 14, 4534. [Google Scholar] [CrossRef]
- Alper, K.; Auersvald, M.; Kejla, L.; Ercan, B.; Ucar, S.; Tekin, K.; Šimáček, P.; Karagoz, S. Sulfonic Acid-Catalyzed Biocoal Production from Lignocellulosic Biomass. Energy & Fuels 2024, 38, 8817–8828. [Google Scholar] [CrossRef]
- Smith, A.M.; Whittaker, C.; Shield, I.; Ross, A.B. The Potential for Production of High Quality Bio-Coal from Early Harvested Miscanthus by Hydrothermal Carbonisation. Fuel 2018, 220, 546–557. [Google Scholar] [CrossRef]
- Stępień, P.; Świechowski, K.; Hnat, M.; Kugler, S.; Stegenta-Dąbrowska, S.; Koziel, J.A.; Manczarski, P.; Białowiec, A. Waste to Carbon: Biocoal from Elephant Dung as New Cooking Fuel. Energies 2019, 12, 4344. [Google Scholar] [CrossRef]
- Islam, M.A.; Limon, M.S.H.; Romić, M.; Islam, M.A. Hydrochar-Based Soil Amendments for Agriculture: A Review of Recent Progress. Arab. J. Geosci. 2021, 14, 102. [Google Scholar] [CrossRef]
- Li, F.; Zimmerman, A.R.; Zheng, Y.; Yang, Y.; Huang, J.; Zhang, Y.; Hu, X.; Yu, Z.; Huang, J.; Gao, B. P-Enriched Hydrochar for Soil Remediation: Synthesis, Characterization, and Lead Stabilization. Sci. Total Environ. 2021, 783, 146983. [Google Scholar] [CrossRef]
- Sun, L.; Wang, J.J.; Wei, S.; Ye, P.; Deng, Y.; Meng, X.; Li, R.; Zhang, Z.; Su, X.; Xiao, R. Hydrochar as an Effective Amendment for Enhancing Soil Aggregation and Carbon Sequestration: Evidence from Comparative Microcosm Experiments. Biochar 2026, 8, 69. [Google Scholar] [CrossRef]
- Xu, S.; Chu, Q.; Lin, J.; Qin, F.; Li, D.; Liu, X.; Xu, X.; Yin, S.; Chen, C.; He, P.; et al. Hydrochar from Rice Straw as a Bio-Based Slow-Release Fertilizer: Tuning Temperature and Oxidation for Agronomic Performance. Ind. Crops Prod. 2026, 240, 122662. [Google Scholar] [CrossRef]
- Kravchenko, E.; Minkina, T.; Privizentseva, D.; Kazeev, K.; Chernikova, N.; Popov, V.; Cruz, T. Dela; Yuan, Z.; Baek, K. Effective Lead Immobilization in Contaminated Soil with Hydrochar Application. Water, Air, Soil Pollut. 2026, 237, 409. [Google Scholar] [CrossRef]
- Hu, C.; Wang, Y.; Cao, B.; Chen, L.; Qiu, X. Contrasting Cr(VI) and Cd(II) Immobilization in Contaminated Soils by Microalgae Derived Hydrochar 2026.
- Sun, K.; Han, L.; Yang, Y.; Xia, X.; Yang, Z.; Wu, F.; Li, F.; Feng, Y.; Xing, B. Application of Hydrochar Altered Soil Microbial Community Composition and the Molecular Structure of Native Soil Organic Carbon in a Paddy Soil. Environ. Sci. Technol. 2020, 54, 2715–2725. [Google Scholar] [CrossRef] [PubMed]
- Sudibyo, H.; Pangestu, R.A.; Athalia, A.T.; Salsabila, R.; Mahannada, A.; Suparmin, A. Catalyst- and Temperature-Driven Variations in Chemistry, Carbon Permanence, and Agronomic Performance of Hydrochar from Hydrothermal Processing of Biomass Waste. Environ. Res. 2026, 296, 124002. [Google Scholar] [CrossRef]
- Chen, X.; Galliane, T.F.J.; Zhao, C.; Li, M.; Huang, L. Phosphorus-Enriched Hydrochar Enhances Soybean Yield through Phosphorus Transformation and Microbial Community Modulation: A Novel Strategy for Reducing Phosphate Fertilizer Application in Calcareous Saline-Sodic Soil. Soil Tillage Res. 2026, 257, 106960. [Google Scholar] [CrossRef]
- Pantelopoulos, A.; Aronsson, H. Organic Waste and Their Respective Hydrochars: Characteristics, Carbon Stability and Nutrient Release Dynamics in Soil. J. Environ. Manage. 2026, 400, 128674. [Google Scholar] [CrossRef]
- Sun, K.; Han, L.; Yang, Y.; Xia, X.; Yang, Z.; Wu, F.; Li, F.; Feng, Y.; Xing, B. Application of Hydrochar Altered Soil Microbial Community Composition and the Molecular Structure of Native Soil Organic Carbon in a Paddy Soil. Environ. Sci. Technol. 2020, 54, 2715–2725. [Google Scholar] [CrossRef] [PubMed]
- Sudibyo, H.; Pangestu, R.A.; Athalia, A.T.; Salsabila, R.; Mahannada, A.; Suparmin, A. Catalyst- and Temperature-Driven Variations in Chemistry, Carbon Permanence, and Agronomic Performance of Hydrochar from Hydrothermal Processing of Biomass Waste. Environ. Res. 2026, 296, 124002. [Google Scholar] [CrossRef] [PubMed]
- Alfredo Quevedo-Amador, R.; Elizabeth Reynel-Avila, H.; Ileana Mendoza-Castillo, D.; Badawi, M.; Bonilla-Petriciolet, A. Functionalized Hydrochar-Based Catalysts for Biodiesel Production via Oil Transesterification: Optimum Preparation Conditions and Performance Assessment. Fuel 2022, 312, 122731. [Google Scholar] [CrossRef]
- Ghosh, N.; Halder, G. Transforming Discarded Cigarette Butts into Novel Hydrochar Catalyst towards Biodiesel Synthesis from Waste Cooking Oil: A Trash-to-Treasure Approach. Energy Convers. Manag. 2026, 352, 121138. [Google Scholar] [CrossRef]
- Eskikaya, O.; Isik, Z.; Arslantas, C.; Yabalak, E.; Balakrishnan, D.; Dizge, N.; Rao, K.S. Preparation of Hydrochar Bio-Based Catalyst for Fenton Process in Dye-Containing Wastewater Treatment. Environ. Res. 2023, 216, 114357. [Google Scholar] [CrossRef]
- Sá, H.; Michelin, M.; Tavares, T.; Sanroman, M.A.; Rosales, E.; Neves, I.C.; Silva, B. Immobilization of Laccase on Grape Seed-Derived Hydrochar and Biochar as Sustainable Biocatalysts for Efficient Pharmaceutical Degradation in Real Wastewater. J. Environ. Chem. Eng. 2025, 13, 118237. [Google Scholar] [CrossRef]
- Alfredo Quevedo-Amador, R.; Elizabeth Reynel-Avila, H.; Ileana Mendoza-Castillo, D.; Badawi, M.; Bonilla-Petriciolet, A. Functionalized Hydrochar-Based Catalysts for Biodiesel Production via Oil Transesterification: Optimum Preparation Conditions and Performance Assessment. Fuel 2022, 312, 122731. [Google Scholar] [CrossRef]
- Sá, H.; Michelin, M.; Tavares, T.; Sanroman, M.A.; Rosales, E.; Neves, I.C.; Silva, B. Immobilization of Laccase on Grape Seed-Derived Hydrochar and Biochar as Sustainable Biocatalysts for Efficient Pharmaceutical Degradation in Real Wastewater. J. Environ. Chem. Eng. 2025, 13, 118237. [Google Scholar] [CrossRef]
- Ghosh, N.; Halder, G. Transforming Discarded Cigarette Butts into Novel Hydrochar Catalyst towards Biodiesel Synthesis from Waste Cooking Oil: A Trash-to-Treasure Approach. Energy Convers. Manag. 2026, 352, 121138. [Google Scholar] [CrossRef]
- Yu, J.; Zhu, Z.; Zhang, H.; Chen, T.; Qiu, Y.; Xu, Z.; Yin, D. Efficient Removal of Several Estrogens in Water by Fe-Hydrochar Composite and Related Interactive Effect Mechanism of H2O2 and Iron with Persistent Free Radicals from Hydrochar of Pinewood. Sci. Total Environ. 2019, 658, 1013–1022. [Google Scholar] [CrossRef]
- Li, S.; Ma, Q.; Chen, L.; Yang, Z.; Aqeel Kamran, M.; Chen, B. Hydrochar-Mediated Photocatalyst Fe3O4/BiOBr@HC for Highly Efficient Carbamazepine Degradation under Visible LED Light Irradiation. Chem. Eng. J. 2022, 433, 134492. [Google Scholar] [CrossRef]
- Pereira, G.R.; Lopes, R.P.; Wang, W.; Guimarães, T.; Teixeira, R.R.; Astruc, D. Triazole-Functionalized Hydrochar-Stabilized Pd Nanocatalyst for Ullmann Coupling. Chemosphere 2022, 308, 136250. [Google Scholar] [CrossRef]
- Khan, L.A.; Liaquat, R.; Aman, M.; Kanan, M.; Saleem, M.; Khoja, A.H.; Bahadar, A.; Khan, W.U.H. Investigation of Novel Transition Metal Loaded Hydrochar Catalyst Synthesized from Waste Biomass (Rice Husk) and Its Application in Biodiesel Production Using Waste Cooking Oil (WCO). Sustainability 2024, 16, 7275. [Google Scholar] [CrossRef]
- Islam, I.U.; Hu, X.; Abdulghaffar, A.T.; Zhao, X.; Long, J.; Wang, X.; XU, Y.; Yabalak, E. PASP-Coated Bimetallic Mustard Husk Hydrochar for Dual Environmental Remediation and Green Energy: Pb(II) Removal, Enhanced Hydrogen Evolution, and DFT Insights Aligned with UN SDGs. Fuel 2026, 410, 137930. [Google Scholar] [CrossRef]
- Liu, R.-P.; Sha, R.; Cheng, A.-L.; Xue, Q.-S.; Gao, E.-Q. Enhanced Degradation of Organic Dyes with Hydrochar Supported Bimetallic NH2-MIL-101(Fe/Co) via Peroxymonosulfate Activation. Polyhedron 2026, 292, 118085. [Google Scholar] [CrossRef]
- Izghri, Z.; Rabichi, I.; Yaacoubi, F.E.; Beddach, Y.; Hanyny, J.; Ounas, A.; Sekkouri, C.; El Gaini, L.; Ennaciri, K.; Chahid, L.; et al. Activated FeCl3-Hydrochar/DWTS as a High-Performance Catalyst in the Photo-Fenton Process for Wastewater Treatment. Biomass Convers. Biorefinery 2026, 16, 54. [Google Scholar] [CrossRef]
- Naribi, Z.; Esserrar, S.; Salhi, A.; El Krati, M.; Tahiri, S. TiO2 Supported on Hydrochar Derived from Industrial Ice Cream Wastewater as a Photocatalytic Composite for Efficient Degradation of AR97 in Aqueous Media. Environ. Sci. Pollut. Res. 2026, 33, 3172–3188. [Google Scholar] [CrossRef] [PubMed]
- IEA ETP Clean Energy Technology Guide. 2026.
- Sajal Suhane; Rushali Rajaram Katkar; Smita Suhane; S. Sugumaran; Santosh Bhauso Takale; Surekha Dehu Khetree; Shyamsing Thakur; Shital Yashwant Waware; Anant Sidhappa Kurhade AI-Driven Optimization of Bio-Energy Systems: Models for Resource Assessment and Emission Reduction. Appl. Chem. Eng. 2025, 9. [CrossRef]
- Ezhumalai, M.; Govindasamy, M.; Dhairiyasamy, R.; Varshney, D.; Singh, S. Catalytic Performance and AI-Predicted Optimization of Hydrogen-Rich Syngas from Biomass-Derived Feedstocks. Int. J. Energy Water Resour. 2026, 10, 5. [Google Scholar] [CrossRef]
- Pallavi Vishnu Kharat; Beena Nawghare; N. Alangudi Balaji; Vishvas V. Kalunge; Charu P. Kumbhare; Tejasvini Rahul Katkar; Sagar Arjun Dalvi; Shital Yashwant Waware; Anant Sidhappa Kurhade Data- Driven Prediction of Biofuel Yield and Combustion Emissions Using AI Techniques Appl. Chem. Eng. 2025. [CrossRef]
- Owusu, W.A.; Marfo, S.A. Artificial Intelligence Application in Bioethanol Production. Int. J. Energy Res. 2023, 2023, 1–8. [Google Scholar] [CrossRef]
- Hosseini, M.; Amirfakhri, S.J.; Ghiaasiaan, R. Artificial Intelligence in Biofuels: Progress, Trends, and Directions. Chem. Biodivers. 2026, 23. [Google Scholar] [CrossRef]
- Deepankumar, S.; Senthil Kumar, K. L. Hydrothermal Liquefaction and Gasification of Industrial Waste Algae: Experimental and AI-Assisted Optimization for Biofuel and Hydrogen Production. Biomass Convers. Biorefinery 2026, 16, 125. [Google Scholar] [CrossRef]
- Alruqi, M.; Sharma, P.; Algburi, S.; Khan, M.A.; Alsubih, M.; Islam, S. Biomass Energy Transformation: Harnessing the Power of Explainable Ai to Unlock the Potential of Ultimate Analysis Data. Environ. Technol. Innov. 2024, 35, 103652. [Google Scholar] [CrossRef]
- Xiao, K.; Zhu, X. Machine Learning Approach for the Prediction of Biomass Waste Pyrolysis Kinetics from Preliminary Analysis. ACS Omega 2024, 9, 48125–48136. [Google Scholar] [CrossRef]
- Esfahanian, M.; Nikzad, M.; Najafpour, G.; Ghoreyshi, A. Modeling and Optimization of Ethanol Fermentation Using Saccharomyces Cerevisiae: Response Surface Methodology and Artificial Neural Network. Chem. Ind. Chem. Eng. Q. 2013, 19, 241–252. [Google Scholar] [CrossRef]
- Khandelwal, K.; Nanda, S.; Dalai, A.K. Machine Learning Modeling of Supercritical Water Gasification for Predictive Hydrogen Production from Waste Biomass. Biomass and Bioenergy 2025, 197, 107816. [Google Scholar] [CrossRef]
- khan, M.; Raza Naqvi, S.; Ullah, Z.; Ali Ammar Taqvi, S.; Nouman Aslam Khan, M.; Farooq, W.; Taqi Mehran, M.; Juchelková, D.; Štěpanec, L. Applications of Machine Learning in Thermochemical Conversion of Biomass-A Review. Fuel 2023, 332, 126055. [Google Scholar] [CrossRef]
- Wang, Z.; Peng, X.; Xia, A.; Shah, A.A.; Huang, Y.; Zhu, X.; Zhu, X.; Liao, Q. The Role of Machine Learning to Boost the Bioenergy and Biofuels Conversion. Bioresour. Technol. 2022, 343, 126099. [Google Scholar] [CrossRef]
- Mira, K.; Bugiotti, F.; Morosuk, T. Artificial Intelligence and Machine Learning in Energy Conversion and Management. Energies 2023, 16, 7773. [Google Scholar] [CrossRef]
- Sonali Shrikant Patil; P. Ramani; Snehal Mayur Banarase; Prafulla O. Bagde; Pushparaj Sunil Warke; N. Alangudi Balaji; Muralidhar Ingale; Shital Yashwant Waware; Anant Sidhappa Kurhade AI-Supported Forecasting of Biomass Availability under Changing Environmental and Resource Conditions. Appl. Chem. Eng. 2026. [CrossRef]
- Wang, R.; He, Z.; Chen, H.; Guo, S.; Zhang, S.; Wang, K.; Wang, M.; Ho, S.-H. Enhancing Biomass Conversion to Bioenergy with Machine Learning: Gains and Problems. Sci. Total Environ. 2024, 927, 172310. [Google Scholar] [CrossRef]
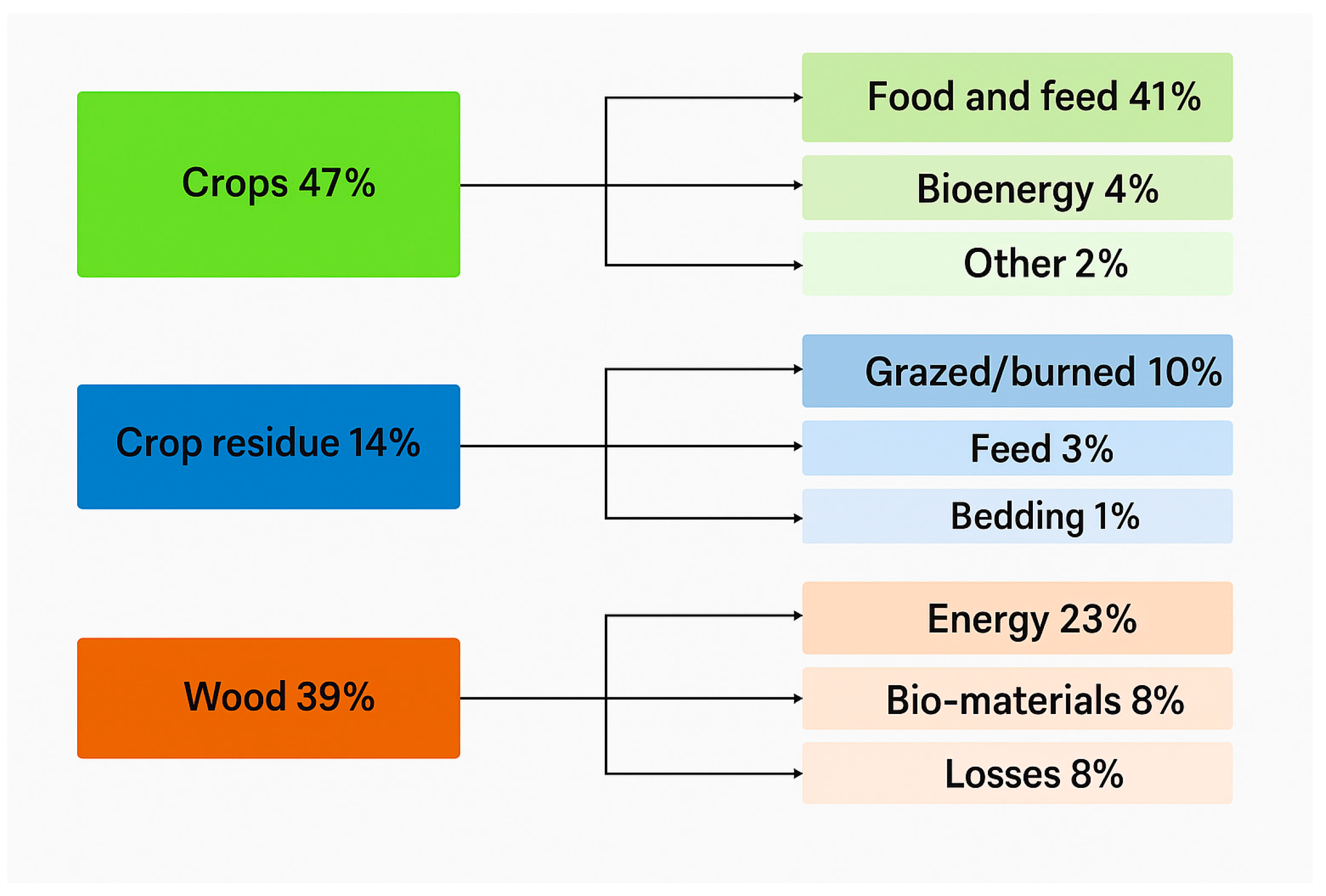
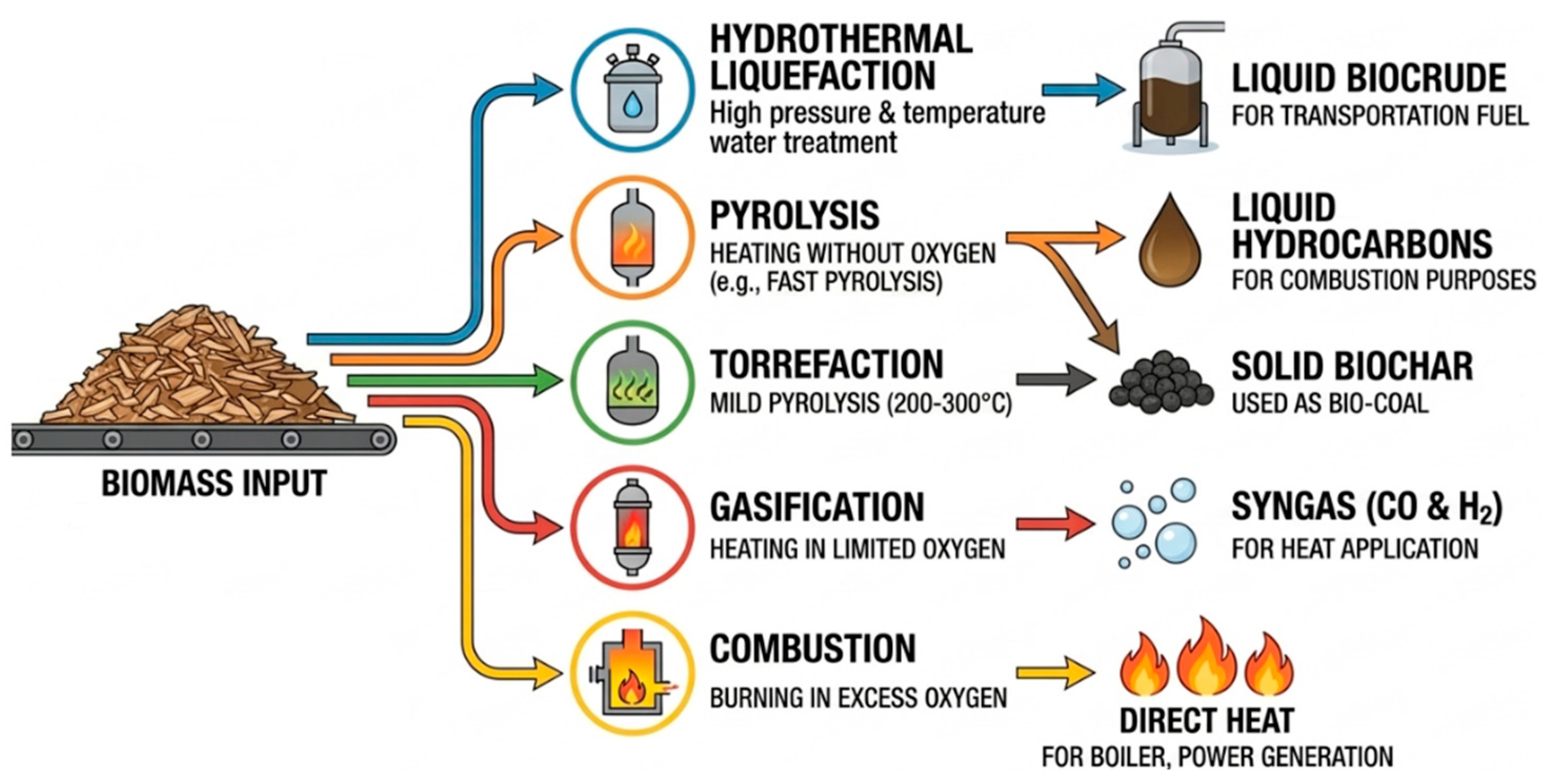
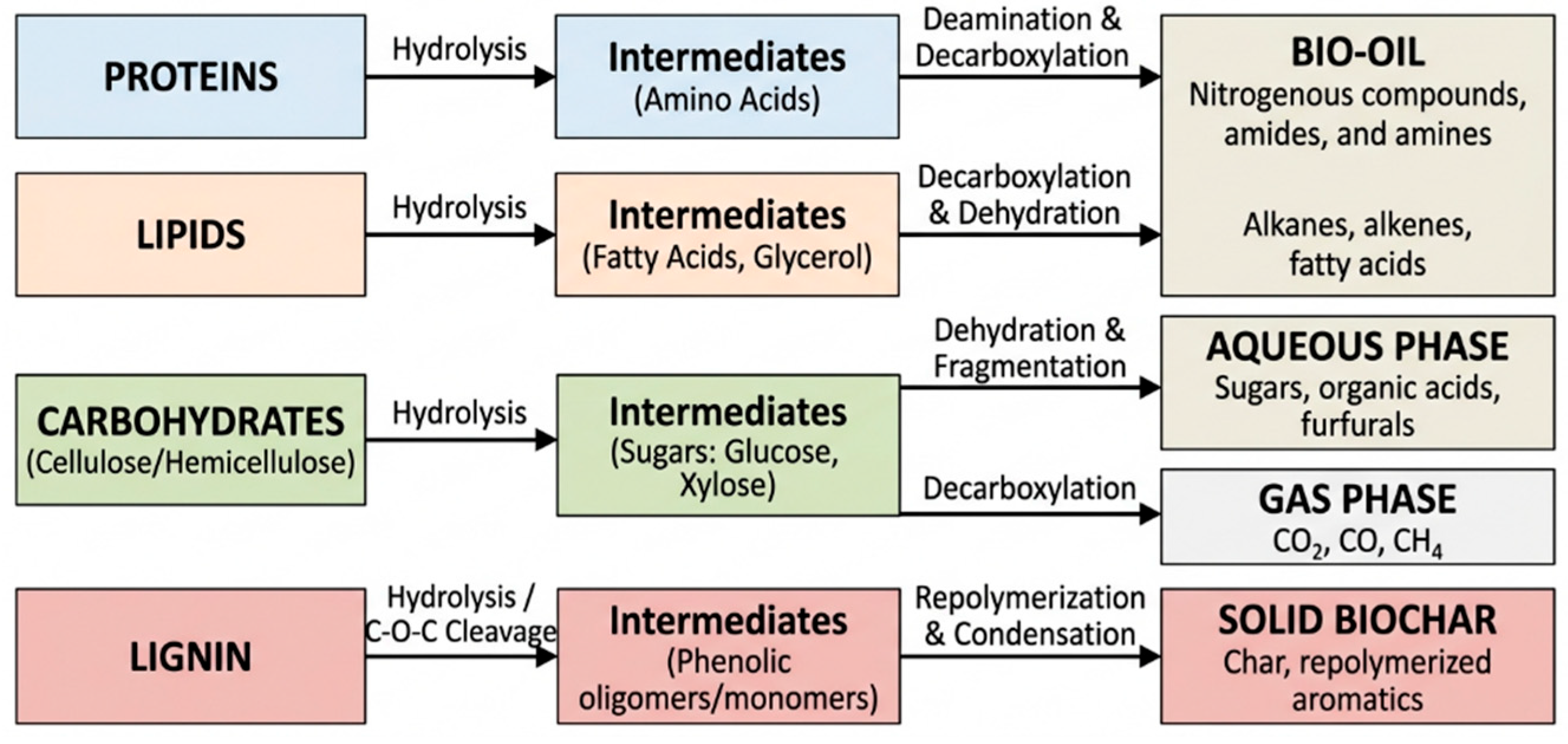
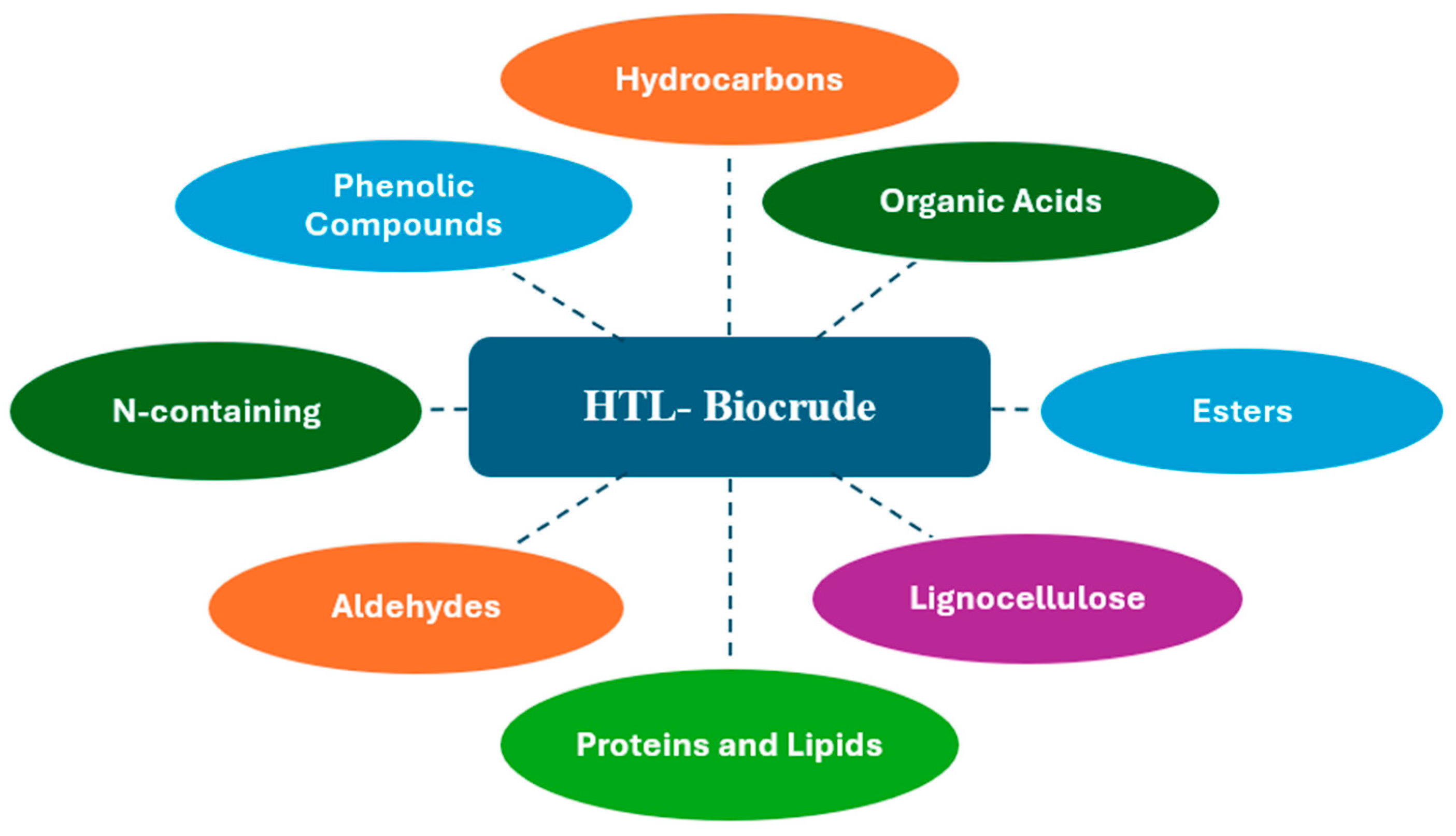
| Feedstock | Dominant Issues | Upgrading Strategy | Catalyst Type | Operating Conditions (T, P) | Products | References |
| Forest residues (50/50 spruce-pine wood) | High O2 (11 wt. %), phenolic compounds, higsh TAN (68 mgKOH g⁻¹) | Solvent de-asphalting (n-pentane, toluene, DCM, EtOAc); Mild hydrotreating | Sulfided NiMo/Al₂O₃ | 290-320 °C, 9 h, 1400 psi H₂ | Distillate-range hydrocarbons; O2 final 2.9 wt. % | [43] |
| Co-HTL of wheat straw + waste cooking oil | High oxygen (10 wt. %), high acidity. | Batch hydrodeoxygenation | Sulfided NiMo/γ Al₂O₃ | 350 °C, 8 h, 1500 psi H₂ | Diesel-range paraffins (C17–C19); O2 final 0.56 wt. %. | [44] |
| Wastewater-grown microalgae | High heteroatoms (O, N, S), aromatic-rich oil. | One-step HTL + in-situ catalytic upgrading | NiMo/Al₂O₃ | 320-370 °C, 30-120 min, 18-22 MPa | Aromatics + alkanes; SAF precursor; O2 final 5-10 wt% O2. | [45] |
| Sewage-sludge HTL biocrude | Extremely high N (6-7 wt.%), refractory carbazoles/indoles | Severe batch hydrotreatment | NiMoS/Al₂O3 | 350-390 °C, 0-5 h, 100 bar H₂ | Fuel-range liquids (73 wt% <350 °C); N2 residual of 1.4 wt%, high degree of O2 removal | [46] |
| Food waste and sewage-sludge | High N (4-5 wt.%), high metals, catalyst stability concerns | Continuous two-stage hydrotreating (guard+ main bed) | CoMo/Al₂O₃ (guard) + NiMo/Al₂O₃ (main) | 350-400 °C, 1500 psi, WHSV 2 h⁻¹ | Diesel-rich blend stock (70% diesel cut); 0.15-0.25 wt.% O | [47] |
| Feedstock | Upgrading strategy | Catalyst used (loading in wt.%) | Operating conditions | Observation | Ref. |
|---|---|---|---|---|---|
| High boiling fraction of Soyabean straw HTL biocrude | Catalytic hydrotreatment in H2-donor solvent (tetralin + decalin) | Pt/C (40 wt.%) |
T = 400 ºC, Pr. = 100 bar t = 4h |
98.6% S, 96.2% N, 87.1% O2 removed |
[61] |
| HTL biocrude of Animal carcass (streaky pork) | Catalytic hydrotreatment of biocrude in a water-free system | CoMo/γAl2O3 (20 wt.%) |
T = 400 ºC, Pr. = 150 bar t = 4h |
64.4% N, 84.6% O2 removed |
[62] |
| HTL biocrude of Food waste (mixed vegetables + ground meat) | Single-step catalytic HDO, HDN, and mild hydrocracking | Pd/C (20 wt.%) |
T = 350 ºC, Pr. = 30 bar t = 3h |
58% O2 removed, low H2 consumption | [63] |
| HTL biocrude derived from Microalgae (Nannochlopsis) | Catalytic hydrodeoxygenation (HDO) following non-catalytic HTL | NiMoC/AC (10 wt.%) |
T = 400 ºC, Pr. = 30 bar t = 2.75h |
94% O2 reduction, 72.5% oil yield, 43 MJ/kg HHV | [64] |
| HTL biocrude of municipal sweage sludge | Single-step HDO of solvent-extracted biocrudes (DCM, hexane, toluene, acetone) | Ni/SiO2- Al2O3 (5 wt.%) |
T = 350 ºC, Pr. = 70 bar t = 1h |
Hexane extract: 5 wt. % O2; Toluene extract 12 wt. % O2 | [65] |
| HTL biocrude of bio-pulp derived from food wastes. | 2-stage continuous catalytic HDO using trickle-bed reactors with guard-bed stabilization followed by deep hydrotreating | Mo/Al₂O₃ (guard bed- 60 g) + NiMo/Al₂O₃ (main catalyst 47 g) | Continuous process; 1st stage: 260 °C, 2nd stage: 400 °C; 10 MPa H₂, WHSV 0.2 h⁻¹ | Upgraded oil yield of 92 wt.%, 64% diesel production, 96% O2 removed. | [66] |
| Partially hydrotreated HTL biocrude from a 50/50 spruce-pine wood mixture | Partial HDO to render biocrude fully miscible in VGO, followed by 2-stage co-processing | Hydrotreating: 15 mL NiMo catalyst with 28 mL SiC; Hydrocracking: 10 mL Zeolite-based catalyst with 18.5 mL SiC | Hydrotreating: T = 330 ºC, Pr. = 100 bar WHSV = 0.5 h-1; Hydrocracking: T = 405 ºC, Pr. = 100 bar LHSV = 1.5 h-1 |
Partially HDO biocrude: 3.6 wt. % O2. co-processed blend: 0.16 wt. % O2 |
[67] |
| HTL biocrude derived from spent coffee grounds | Mild hydrotreatment (HDO stabilization) followed by refinery-relevant coprocessing | Sulfided NiMo/γAl₂O₃ (7.1 v/v blend with demetallization catalyst) |
T = 330 ºC, Pr. = 70 bar LHSV = 1 h-1 |
0.39 wt. % O2 in stabilized biocrude | [68] |
| Model compounds of HTL biocrude, derived from black liquor | Catalytic HDO in subcritical/supercritical water | Activated Carbon supported NiMoSx (6.5 wt.%) |
T = 380 ºC, Pr. = 15 bar t = 2 h |
Highest selectivity to phenols. 40% degree of deoxygenation for both conditions | [69] |
| HTL biocrude mixture of wheat straw and waste cooking oil | Single-step HDO | Sulfided NiMo/γAl2O3 (13 wt.%) |
T = 350 ºC, Pr. = 103 bar t = 8 h |
Final O2 content 0.6 wt.%, HHV of 46 MJ/kg, and low acidity | [70] |
| Category of Study | Primary Objective | AI/ML model | Key Observations | Limitations Observed | Reference |
|---|---|---|---|---|---|
| Syngas Production | Optimize hydrogen-rich syngas from aqueous phase reforming (APR). | Artificial Neural Networks (ANN) | Catalyst type and temperature are the dominant variables for H2 selectivity. 90% of predictions lay within ±5% error of experimental values. | ANN performance fluctuates with extreme pressure variations. | [134] |
| Algal Biofuels production | Optimize HTL and HTG for bio-oil and hydrogen production. | Combined machine learning based Tunable Decision Support System and Tunable Recommendation System | AI-optimized parameters deviated by < 3% from experimental optima. Required experimental runs reduced by 65% compared to manual trial-and-error optimization | Nonlinearity in supercritical water gasification. Prediction confidence intervals and robustness under noisy data were not formally assessed | [138] |
| Feedstock Characterization | Predict HHV of biomass fuels through ML models | Linear regression, Random Forest, Extreme gradient boosting (XGBoost), adaptive boosting (AdaBoost | ML outperforms linear regression, XGBoost gives best performance across training datasets. | The model functions as a screening tool rather than offering dynamic optimization. Variability due to experimental protocols is not normalized. | [139] |
| Pyrolysis Kinetics | Develop ML-models to predict biomass pyrolysis kinetic parameters | ANN models and a hybrid Particle Swarm Optimization (PSO-ANN) approach | PSO significantly improved ANN training stability. Maximum relative deviation decreased from 12.85% (ANN-3) to 6.72% (PSO-ANN) | Model accuracy drops for Continuous pyrolysis systems, limiting its application at industrial scales. | [140] |
| Bioethanol production | Establish a data-driven ANN model capable of predicting bioethanol yield in a system | Multilayer Perceptron (MLP) through back propagation. | ANN model demonstrated high predictive accuracy, with reported R² values. The model effectively captured strong nonlinear coupling between reaction parameters | Prediction confidence intervals were not reported, limiting industrial applicability. Optimization was conducted solely on yield, without integration of cost, energy efficiency, or emissions | [141] |
| Hydrogen production | Compared different ML models to predict the yield of hydrogen | Hyper-parameter through Genetic algorithm and PSO | PSO-optimized Gradient Boosting Regression (Test R² = 0.96; cross-validation R² = 0.92). SCWG had more influence on predictions (61%) than feed properties | A mixed and limited dataset, with varying experimental conditions, led to underperformance of the ANN model | [142] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




