Submitted:
19 April 2026
Posted:
20 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental Procedure
3. Characterization
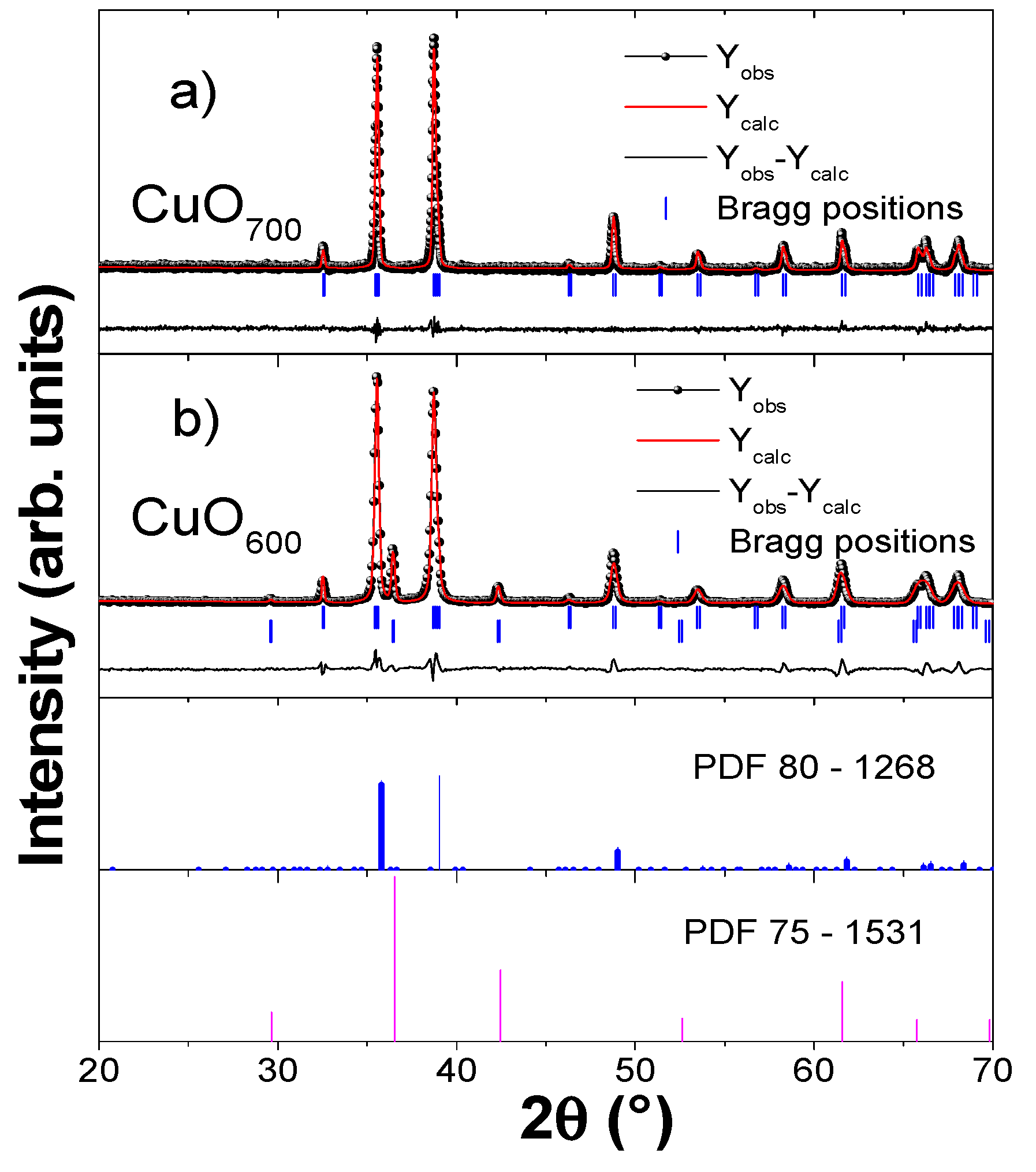
3.1. Structural Characterization by XRD
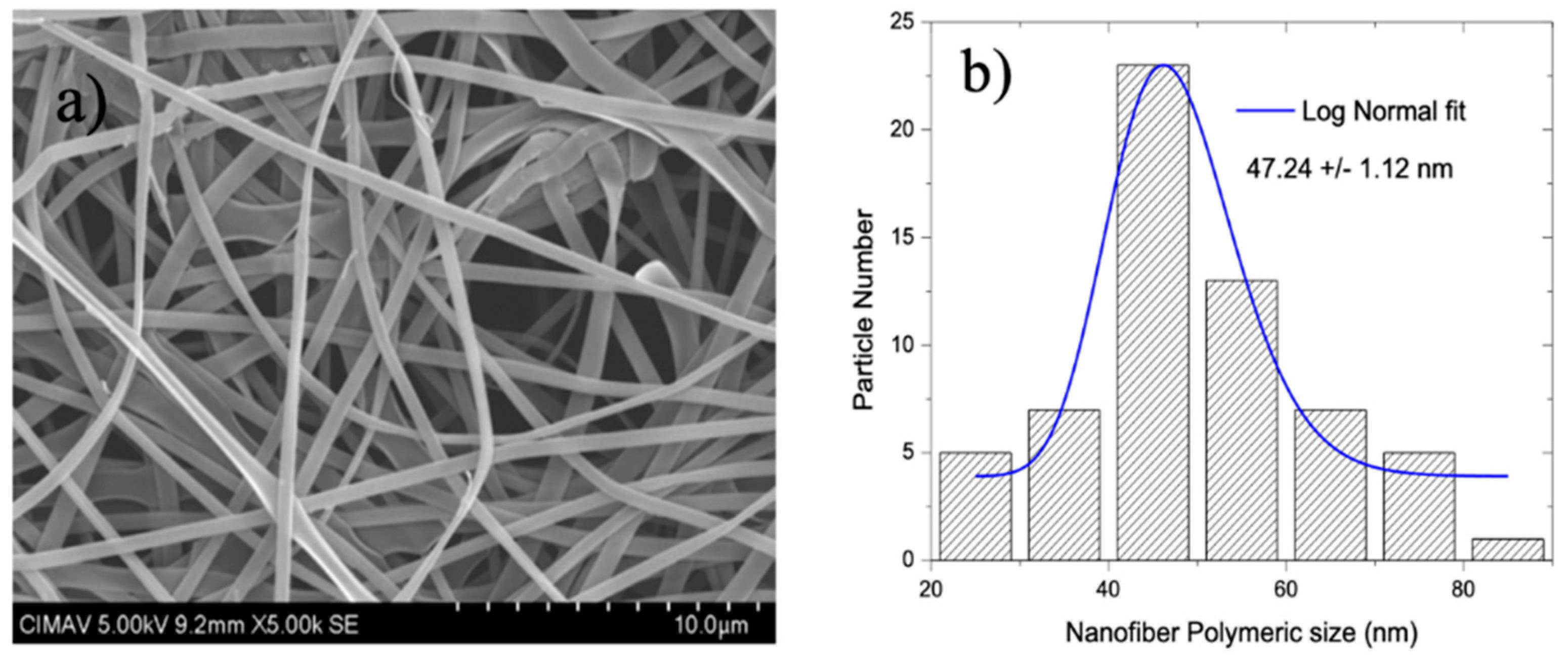
3.2. Scanning Electron Microscopy (SEM)
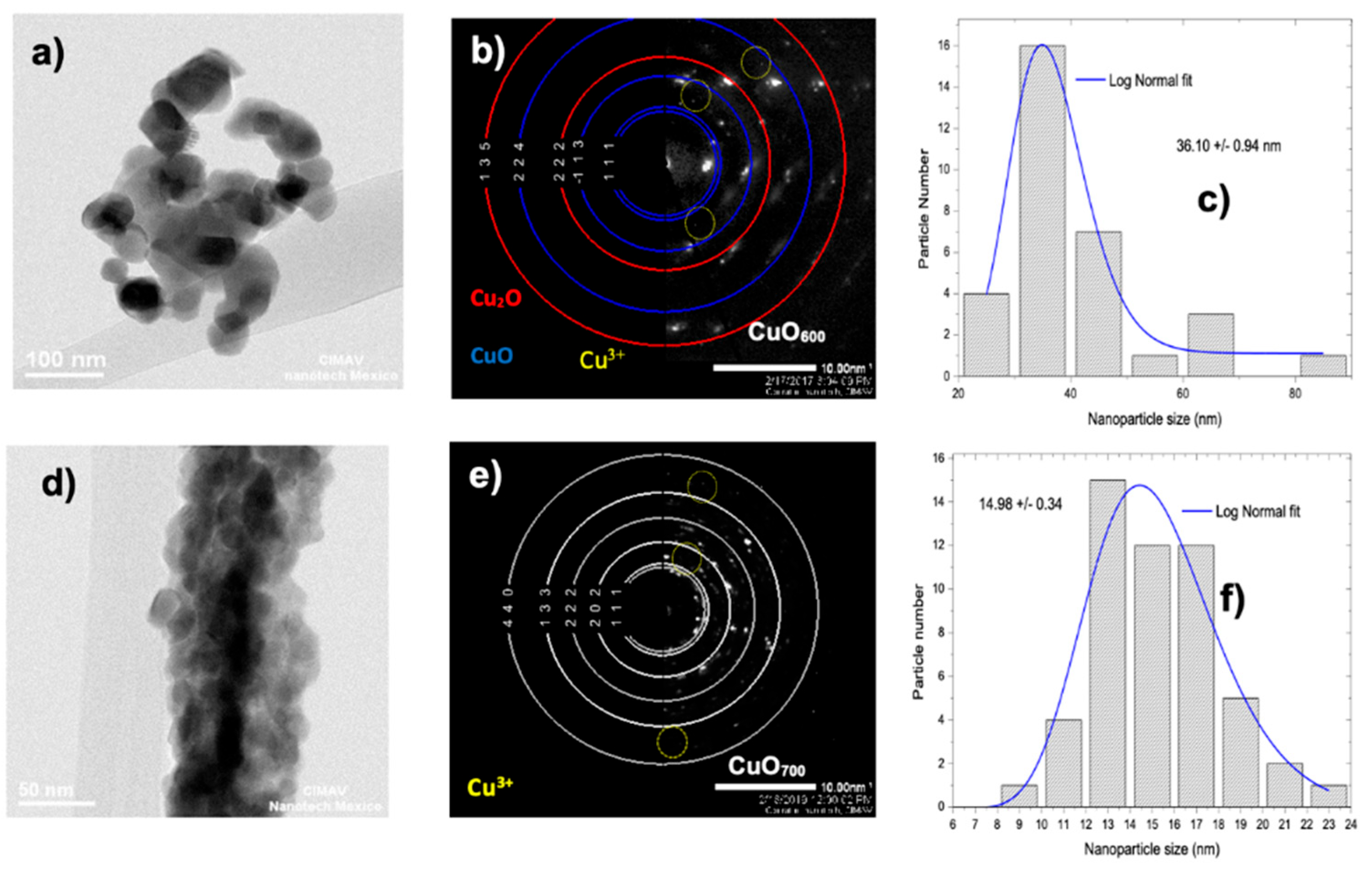
3.3. High-Resolution Transmission Electron Microscopy (HRTEM)
3.4. X-Ray Photoelectron Spectroscopy (XPS)
3.5. Data analysis
4. Results and Discussion
4.1. XRD Analysis
4.2. SEM, HR-TEM, and NBD Analysis
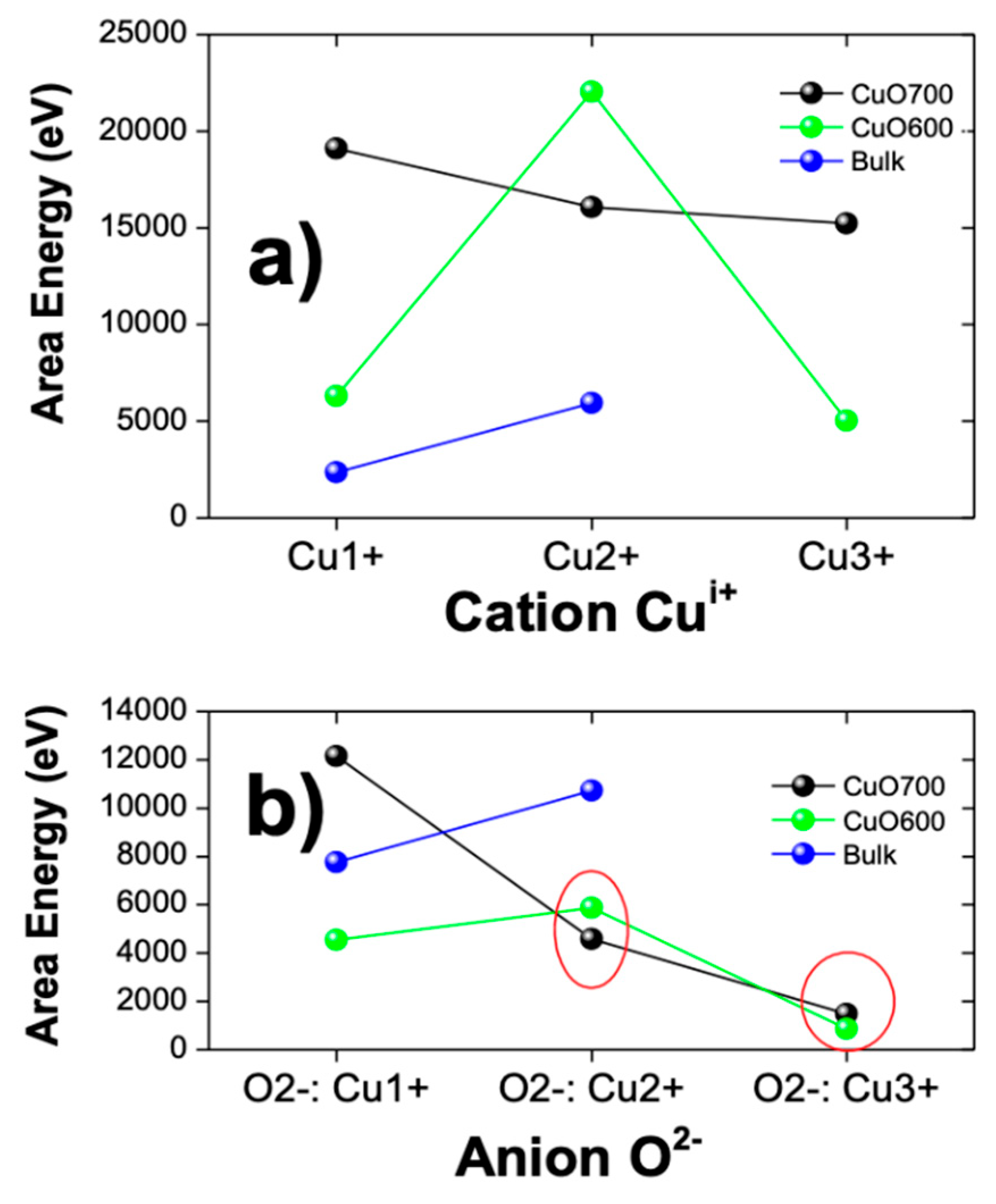
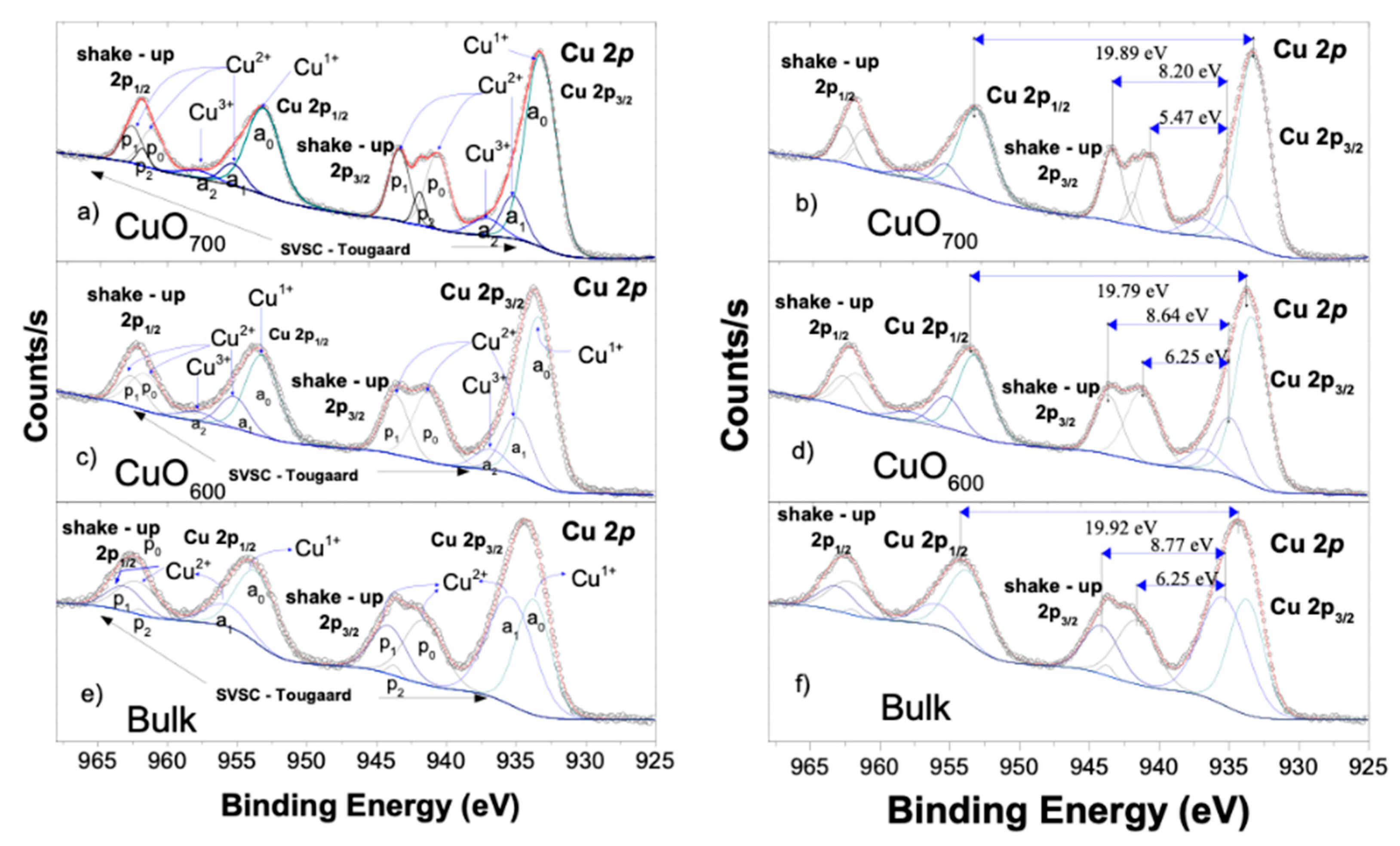
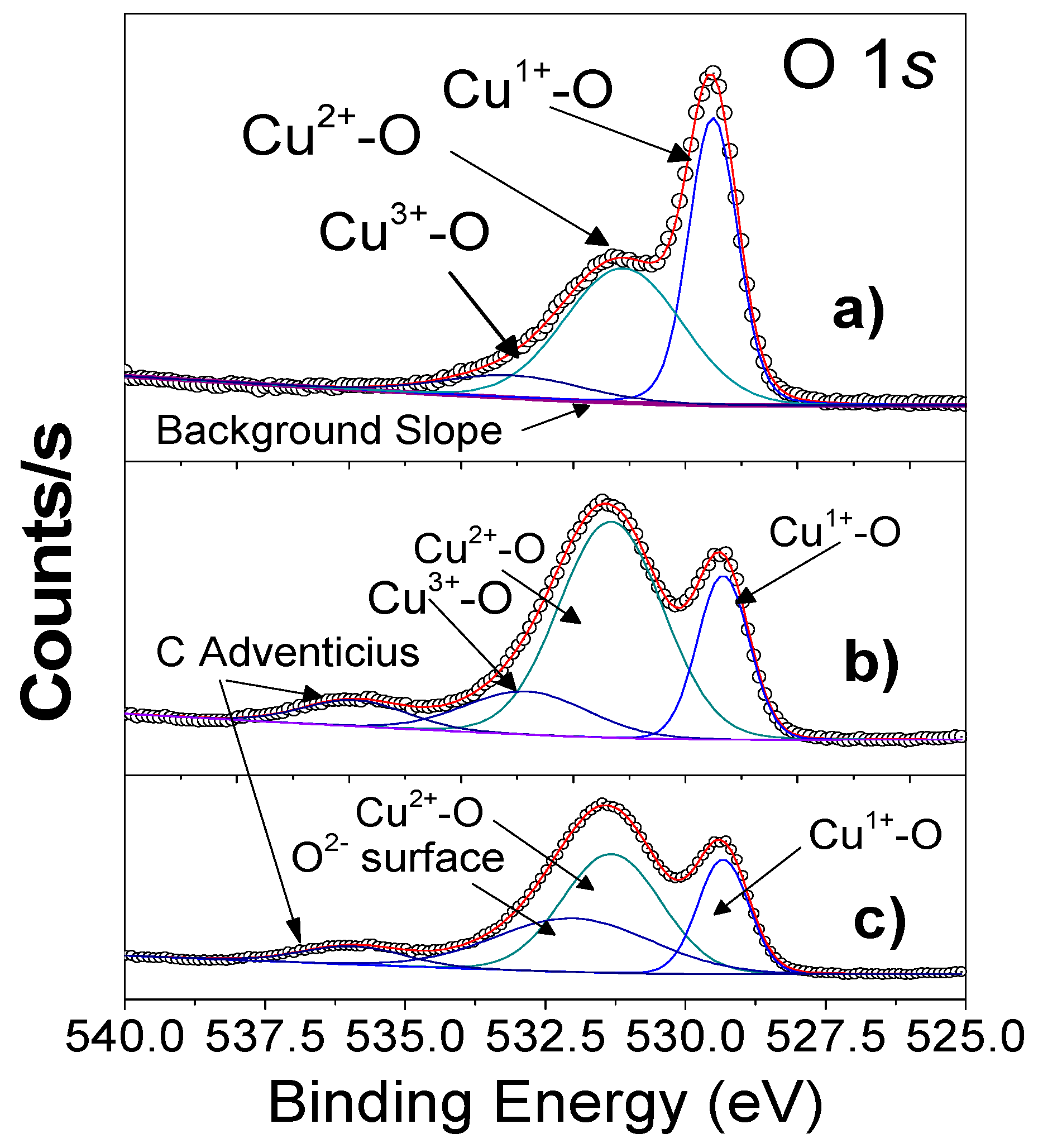
4.3. XPS Analysis and Chemical Composition
5. Conclusions
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- S. Sarkar, P. Kumar Jana, B. K. Chaudhuri, and H. Sakata, “Copper (II) oxide as a giant dielectric material Synthesis of Cu 2 O from CuO thin films: Optical and electrical properties Copper " II… oxide as a giant dielectric material,” Cit. Appl. Phys. Lett. J. Appl. Phys. Appl. Phys. Lett. Appl. Phys. Lett. Nickel Oxide, Their Mix. Cryst. J. Appl. Phys., vol. 8911, no. 10, pp. 212905–2703, 2006. [CrossRef]
- Collins, B.; Desisto, W.; Kershaw, R.; Dwight, K.; Wold, A. Preparation and characterization of Cu(II) oxide. J. Less Common Met. 1989, 156, 341–346. [CrossRef]
- Raj, A.A.; Biju, V. Nanostructured CuO: Facile synthesis, optical absorption and defect dependent electrical conductivity. Mater. Sci. Semicond. Process. 2017, 68, 38–47. [CrossRef]
- Végh, J. The Shirley background revised. J. Electron Spectrosc. Relat. Phenom. 2006, 151, 159–164. [CrossRef]
- Cabrera-German, D.; Gomez-Sosa, G.; Herrera-Gomez, A. Accurate peak fitting and subsequent quantitative composition analysis of the spectrum of Co 2 p obtained with Al Kα radiation: I: cobalt spinel. Surf. Interface Anal. 2016, 48, 252–256. [CrossRef]
- Herrera-Gomez, A.; Bravo-Sanchez, M.; Ceballos-Sanchez, O.; Vazquez-Lepe, M.O. Practical methods for background subtraction in photoemission spectra. Surf. Interface Anal. 2014, 46, 897–905. [CrossRef]
- Salvia, A.; E Castle, J. The intrinsic asymmetry of photoelectron peaks: dependence on chemical state and role in curve fitting. J. Electron Spectrosc. Relat. Phenom. 1998, 95, 45–56. [CrossRef]
- Desimoni, E.; Casella, G.; Cataldi, T.; Malitesta, C. A comparison of some asymmetrical line shapes for XPS data analysis. J. Electron Spectrosc. Relat. Phenom. 1989, 49, 247–261. [CrossRef]
- L. G. M. Herrera-Gomez, Alberto., Dagoberto Cabrera-German., Anthony D. Dutoi., Milton Vazquez Lepe., Servando Aguirre-Tostado., Piero Pianetta., Dennis Nordlund., Orlando Cortazar-Martinez., Alejandro Torres-Ochoa.,Oscar Ceballos-Sanchez., “Modulation of the intensity of the Shirley background of the Cr 3p spectra with photon energies around the Cr 2p edge,” J. Electron Spectros. Relat. Phenomena, 2017.
- A. H.-G. et Al, “Formation of Si1+ in the early stages of the oxidation of the Si [001] 2 x 1 surfaces,” Vaccum Sci. Tecnol. A Vaccum, Surfaces, Film., vol. 34, no. 020601, pp. 1–4, 2016.
- Rodríguez-Carvajal, J. Recent advances in magnetic structure determination by neutron powder diffraction. Phys. B Condens. Matter 1993, 192, 55–69. [CrossRef]
- Rodríguez-Carvajal, J. Recent advances in magnetic structure determination by neutron powder diffraction. Phys. B Condens. Matter 1993, 192, 55–69. [CrossRef]
- K. W. Eliceiri, “Micrograph Digital,” NIH Image to ImageJ 25 years image Anal. Nat. Methods, vol. 9, p. 671, 2009.
- Shard, A.G. A Straightforward Method For Interpreting XPS Data From Core–Shell Nanoparticles. J. Phys. Chem. C 2012, 116, 16806–16813. [CrossRef]
- Shard, A.G.; Wang, J.; Spencer, S.J. XPS topofactors: determining overlayer thickness on particles and fibres. Surf. Interface Anal. 2009, 41, 541–548. [CrossRef]
- Rodríguez-Carvajal, J. Recent advances in magnetic structure determination by neutron powder diffraction. Phys. B Condens. Matter 1993, 192, 55–69. [CrossRef]
- Roisnel, T.; Rodríquez-Carvajal, J. WinPLOTR: A Windows Tool for Powder Diffraction Pattern Analysis. Mater. Sci. Forum 2001, 378-381, 118–123. [CrossRef]
- A. J. Klinger, M., “Crystallographic Tool Box (CrysTBox): automated tools for transmission electron microscopists and crystallographers,” Appl. Crystallogr., 2015.
- Zheng, X.G.; Xu, C.N.; Tomokiyo, Y.; Tanaka, E.; Yamada, H.; Soejima, Y. Observation of Charge Stripes in Cupric Oxide. Phys. Rev. Lett. 2000, 85, 5170–5173. [CrossRef]
- Dongliang, Y.; Chuannan, G.; Youwei, D. Preparation and characterization of CuO nanowire arrays. J. Semicond. 2009, 30. [CrossRef]
- Chusuei, C.C.; Brookshier, M.A.; Goodman, D.W. Correlation of Relative X-ray Photoelectron Spectroscopy Shake-up Intensity with CuO Particle Size. Langmuir 1999, 15, 2806–2808. [CrossRef]
- F. Y. N. Pauly, S. Tougaard, “Determination of the Cu 2p primary excitation spectra for Cu, Cu2O and CuO,” Surf. Sci., vol. 620, pp. 17–22, 2014.
- M. B.-S. J. A. Torres - Ochoa, Diego Fernando Mulato - Gòmez, Dagoberto Cabrera - Germàn, “Detailed analysis of the photoemission spectra of copper films with coexistent Cu1+, Cu2+ and Cu3+ oxidation states,” in Detailed analysis of the photoemission spectra of copper films with coexistent Cu1+, Cu2+ and Cu3+ oxidation states, 2017, pp. 125–126.
- Hauschild, D.; Handick, E.; Göhl-Gusenleitner, S.; Meyer, F.; Schwab, H.; Benkert, A.; Pohlner, S.; Palm, J.; Tougaard, S.M.; Heske, C.; et al. Band-Gap Widening at the Cu(In,Ga)(S,Se)2Surface: A Novel Determination Approach Using Reflection Electron Energy Loss Spectroscopy. ACS Appl. Mater. Interfaces 2016, 8, 21101–21105. [CrossRef]
- Leapman, R.D.; Grunes, L.A.; Fejes, P.L. Study of the L23 edges in the 3d transition metals and their oxides by electron-energy-loss spectroscopy with comparisons to theory. Phys. Rev. B 1982, 26, 614–635. [CrossRef]
- Tougaard, S.; Sigmund, P. Influence of elastic and inelastic scattering on energy spectra of electrons emitted from solids. Phys. Rev. B 1982, 25, 4452–4466. [CrossRef]
- A. Herrera-Gomez, “The Peak-Shirley Background (Shirley background in overlapping peaks).”.
- Castle, J.; Chapman-Kpodo, H.; Proctor, A.; Salvi, A. Curve-fitting in XPS using extrinsic and intrinsic background structure. J. Electron Spectrosc. Relat. Phenom. 2000, 106, 65–80. [CrossRef]
- Gao, D.; Yang, G.; Li, J.; Zhang, J.; Zhang, J.; Xue, D. Room-Temperature Ferromagnetism of Flowerlike CuO Nanostructures. J. Phys. Chem. C 2010, 114, 18347–18351. [CrossRef]
- Baer, D.R.; Engelhard, M.H.; Johnson, G.E.; Laskin, J.; Lai, J.; Mueller, K.; Munusamy, P.; Thevuthasan, S.; Wang, H.; Washton, N.; et al. Surface characterization of nanomaterials and nanoparticles: Important needs and challenging opportunities. J. Vac. Sci. Technol. A 2013, 31, 050820. [CrossRef]
- Kim, S.Y.; Khanal, D.; Tharkar, P.; Kalionis, B.; Chrzanowski, W. None of us is the same as all of us: Resolving heterogeneity of stem cell-derived extracellular vesicles using single-vesicle, nanoscale characterization with highresolution resonance enhanced atomic force microscope infrared spectroscopy (AFM-IR). Nanoscale Horiz. 2018, 3, 430–438. [CrossRef]
- Debbichi, L.; de Lucas, M.C.M.; Pierson, J.F.; Krüger, P. Vibrational Properties of CuO and Cu4O3 from First-Principles Calculations, and Raman and Infrared Spectroscopy. J. Phys. Chem. C 2012, 116, 10232–10237. [CrossRef]
- Zeng, J.; Xu, J.; Wang, S.; Tao, P.; Hua, W. Ferromagnetic behavior of copper oxide-nanowire-covered carbon fibre synthesized by thermal oxidation. Mater. Charact. 2009, 60, 1068–1070. [CrossRef]
- Debbichi, L.; de Lucas, M.C.M.; Pierson, J.F.; Krüger, P. Vibrational Properties of CuO and Cu4O3 from First-Principles Calculations, and Raman and Infrared Spectroscopy. J. Phys. Chem. C 2012, 116, 10232–10237. [CrossRef]
- Iqbal, M.; Thebo, A.A.; Shah, A.H.; Iqbal, A.; Thebo, K.H.; Phulpoto, S.; Mohsin, M.A. Influence of Mn-doping on the photocatalytic and solar cell efficiency of CuO nanowires. Inorg. Chem. Commun. 2017, 76, 71–76. [CrossRef]
| nanostructure |
A (Å) |
b (Å) |
c (Å) |
V (Å3) |
Rp (%) |
Rwp (%) |
Rexp (%) |
χ2 | Y |
| 80-1268 | 4.6837 | 3.4226 | 5.1288 | 82.2830 | - | - | - | - | - |
| 75-1531 | 4.2678 | 4.2678 | 4.2678 | 77.31 | - | - | - | - | - |
| CuO700 | 4.6858 | 3.4230 | 5.1298 | 82.2828 | 4.73 | 6.35 | 5.29 | 1.44 | 0.4184 |
| CuO600 | 4.6860 | 3.4250 | 5.1343 | 81.297 | 12.8 | 18.2 | 17.70 | 1.05 | 0.41354 |
| 4.2699 | 4.2699 | 4.2699 | 77.774 |
| sample | TakeOff Angle | Cu1+ 2p C (eV) | O 1s Cu1+ A (eV) | Cu 2+ 2p A (eV) | O 1s Cu2+ A (eV) | Cu3+ 2p C (eV) | O 1s Cu3+ A (eV) |
| CuO600 | 90 | 19104.6 | 12159 | 16315.5 | 4590.52 | 4357.15 | 1474.3 |
| CuO700 | 90 | 6063.7 | 4543.56 | 22029.7 | 5865.18 | 15944.72 | 869.603 |
| Bulk | 90 | 5228 | 7766.1 | 5035.3 | 10721 | - | - |
| Sample | Date | Peak | 2p3/2 BE (eV) | 2p1/2 BE (eV) | Shake-up 2p3/2 BE (eV) | Peak Width FWHM | |
| Gaussian (eV) | Lorentzian (eV) | ||||||
| Bulk | Cu 2p | a1 | 933.72 | 953.58 | 3.78a-3.30b | 0.085 | |
| a2 | 935.35 | 955.8 | 2.79a-3.41b | 0.085 | |||
| p1 | 941.59 | 3.22 | 0.085 | ||||
| p2 | 944.11 | 2.86 | 0.085 | ||||
| p3 | 943.72 | 1.00 | 0.085 | ||||
| CuO600 | Cu 2p | a1 | 933.37 | 953.19 | 2.48 | 0.270 | |
| a2 | 934.91 | 955.21 | 1.86 | 0.270 | |||
| a3 | 936.81 | 957.91 | 2.48 | 0.270 | |||
| p1 | 941.19 | 1.97 | 0.270 | ||||
| p2 | 943.66 | 1.74 | 0.270 | ||||
| CuO700 | Cu 2p | a1 | 933.19 | 953.09 | 2.48 | 0.270a–0.514b | |
| a2 | 935.17 | 955.28 | 1.86 | 0.270a–0.059b | |||
| a3 | 937.1 | 957.67 | 2.48 | 0.270a–0.514b | |||
| p1 | 940.64 | 1.97 | 0.27 | ||||
| p2 | 943.37 | 1.74 | 0.27 | ||||
| p3 | 941.91 | 1.00 | 0.27 | ||||
| Sample | Shirley type SVSC (eV-1) | Shirley type Tougaard (eV-1) | Shirley type Slope (eV-1) |
| Bulk | 0.028, 0.045 | 2000 | 0.0040 |
| CuO600 | 0.035, 0.048, 0.035 | 2000 | 0.0025 |
| CuO700 | 0.054, 0.068, 0.75 | 2000 | 0.0038 |
| Fitting peaks for the Chemical species Cu1+, Cu2+, Cu3+ and the variation with the calcination temperatures. | ||||||||||
| Heat Treatment | Chemical Species | Peak Binding Energy BE (eV) | Peak Binding Energy BE (eV) | CuO | XCuO | XC | I1/I2 | A | SF | |
| 700 ºC | Cu+1 | O-2 | 933.19 | 529.50 | Cu+1–O-2 | 0.33–0.54 | 0.35–0.64 | 0.98 | Cu1.02O1.16 | |
| Cu+2 | O-2 | 935.17. | 531.15 | Cu+2–O-2 | 0.34–0.65 | 0.75–1.29 | 0.98 | Cu2.22O2.15 | ||
| Cu+3 | O-2 | 937.1 | 533.13 | Cu+3–O-2 | 0.33–0.58 | 0.75–0.46 | 0.98 | Cu2.22O3.03 | ||
| 600 ºC | Cu+1 | O-2 | 933.36 | 529.3 | Cu+1–O-2 | 0.33–0.54 | 0.35–0.64 | 0.98 | Cu1.02O1.15 | |
| Cu+2 | O-2 | 934.91 | 531.33 | Cu+2–O-2 | 0.33–0.57 | 1.05–0.64 | 0.98 | Cu2.06O2.03 | ||
| Cu+3 | O-2 | 936.81 | 532.84 | Cu+3–O-2 | 0.33–0.14 | 1.05–2.37 | 0.98 | Cu2.05O3.19 | ||
| C -O | O-2 | 284.84 | 536.01 | C–O | 1.00 | 0.35 | 0.98 | |||
| Bulk | Cu+1 | O-2 | 933.33 | 529.76 | Cu+1–O-2 | 0.35–0.48 | 0.35–0.51 | 0.98 | Cu0.97O1.02 | |
| Cu+2 | O-2 | 936.06 | 530.66 | Cu+2–O-2 | 0.81–0.54 | 1.66–1.36 | 0.98 | Cu2.43O2.01 | ||
| C-O | O-2 | 284.96 | 536.1 | C–O | 1.00 | 0.45 | 0.98 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




