Submitted:
17 April 2026
Posted:
17 April 2026
You are already at the latest version
Abstract
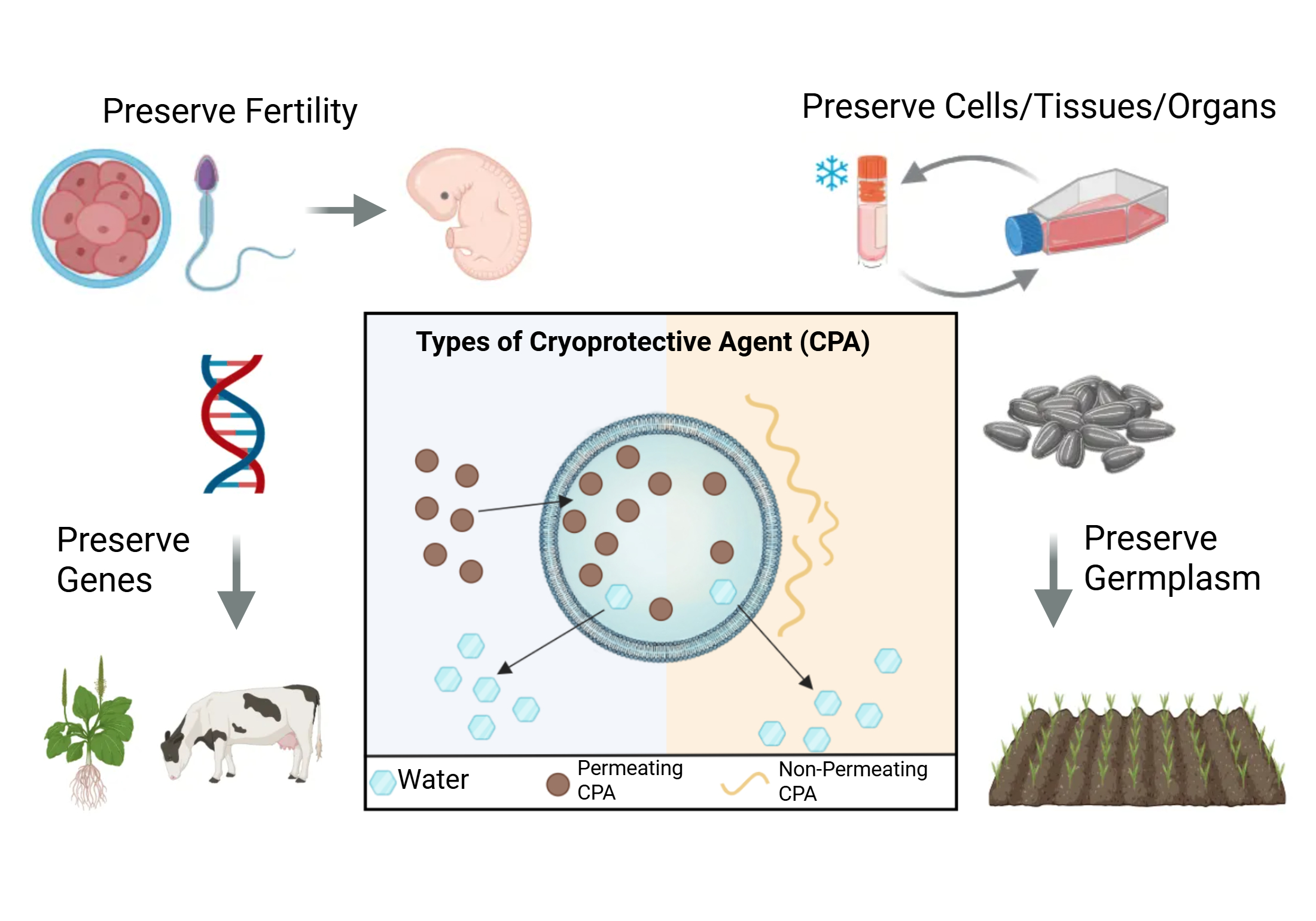
Keywords:
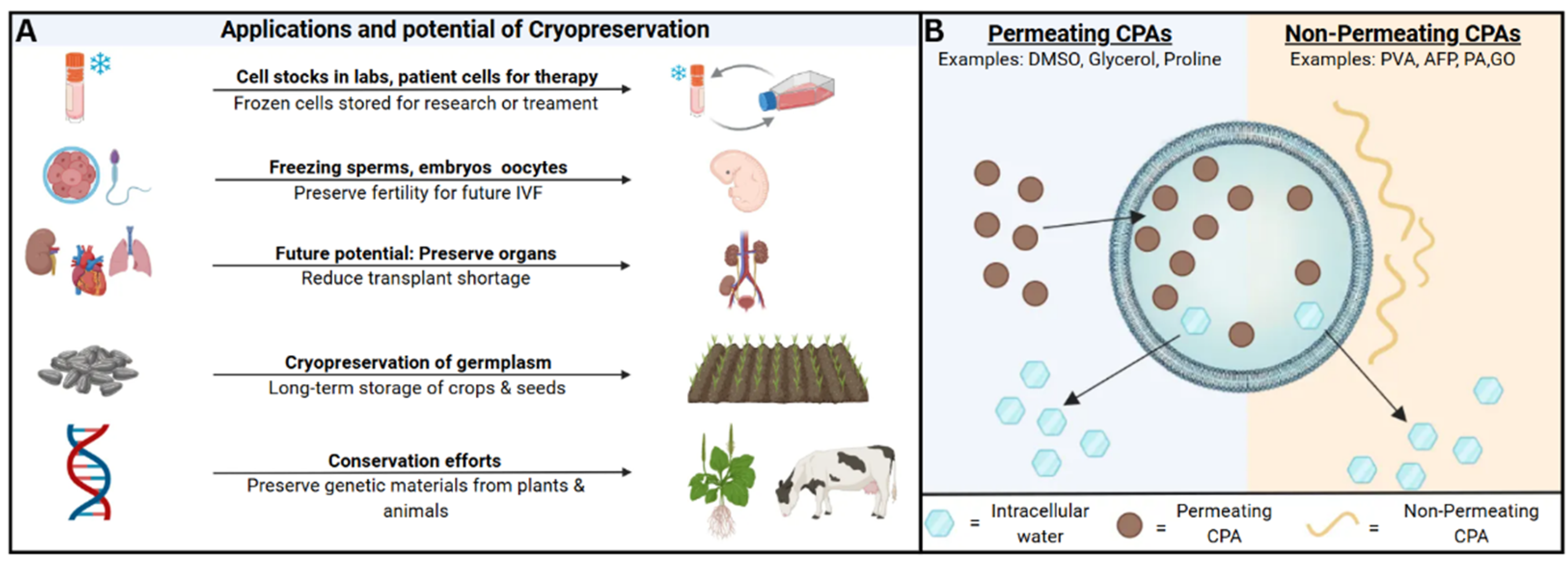
1. Importance of Cryopreservation
2. Mechanism of Cryopreservation
3. CPA Classification: Permeating vs. Non-Permeating Strategies
4. Novel Cryoprotectant Development
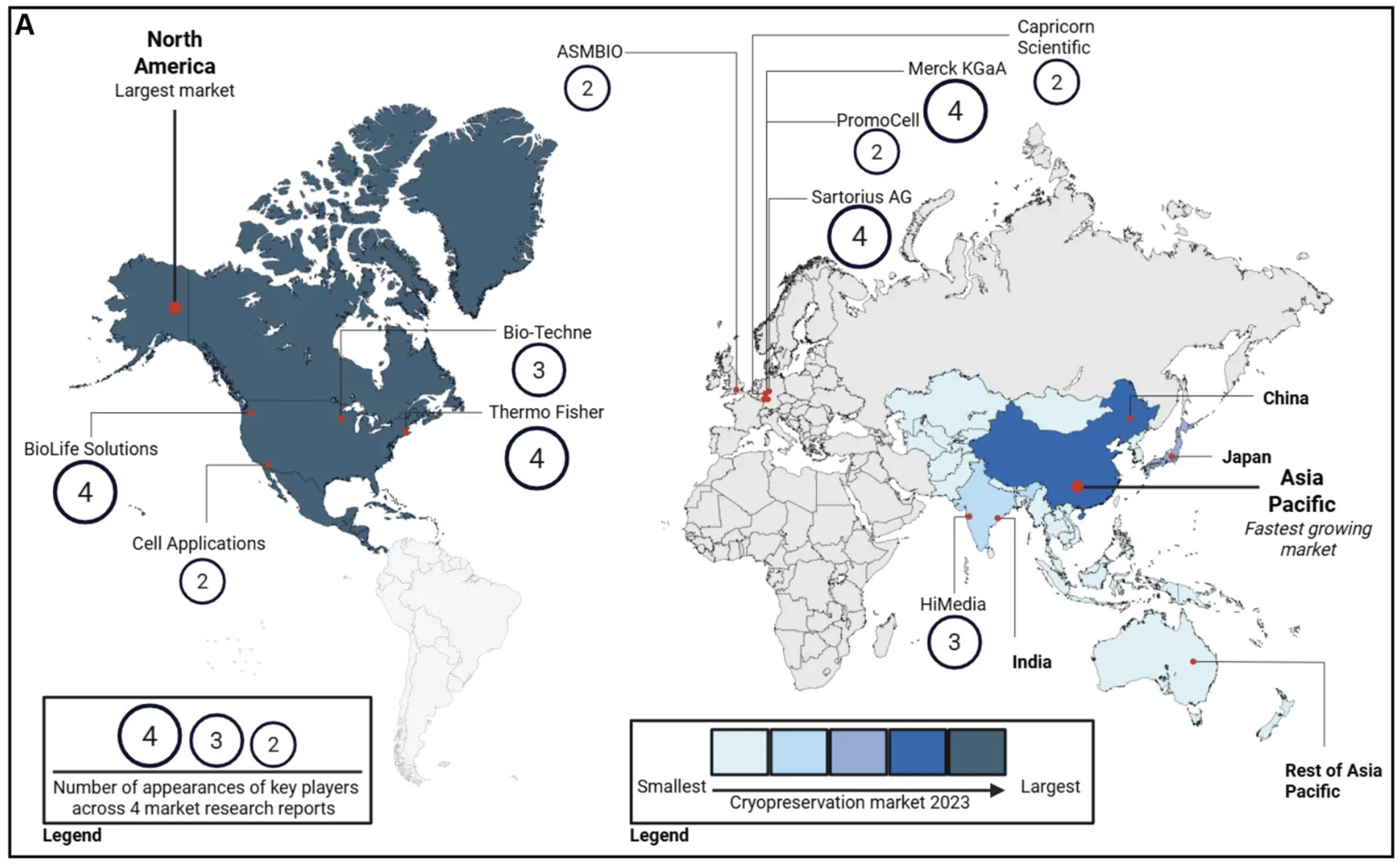
5. Commercial Freezing Media Market
6. Current Progress in Tissue and Organ Cryopreservation
7. Regulatory Challenges to Commercialization
8. The Gaps in Current Cryopreservation Media Market and Future Potential
9. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Love, R. Chillin’at the Symposium with Plato: Refrigeration in the Ancient World. Ashrae Transactions 2009, vol. 115(no. 1). [Google Scholar]
- Ernest John Christopher Polge. [Online]. Available online: https://keep.lib.asu.edu/items/173448.
- Bojic, S. Winter is coming: the future of cryopreservation. BMC Biol 2021, vol. 19(no. 1), 56. [Google Scholar] [CrossRef]
- Aarattuthodi, S.; et al. Cryopreservation of biological materials: applications and economic perspectives. In Vitro Cell Dev Biol Anim; 23 Apr 2025. [Google Scholar] [CrossRef]
- Geraghty, R. J. Guidelines for the use of cell lines in biomedical research. Br J Cancer 2014, vol. 111(no. 6), 1021–46. [Google Scholar] [CrossRef] [PubMed]
- Murray, K. A.; Gibson, M. I. Chemical approaches to cryopreservation. Nat Rev Chem 2022, vol. 6(no. 8), 579–593. [Google Scholar] [CrossRef]
- Parihar; Kumar, A.; Panda, U.; Khan, R.; Parihar, D. S.; Khan, R. Cryopreservation: A Comprehensive Overview, Challenges, and Future Perspectives. Adv Biol (Weinh) 2023, vol. 7(no. 6), e2200285. [Google Scholar] [CrossRef]
- F. Engelmann, “Plant cryopreservation: Progress and prospects,” In Vitro Cellular & Developmental Biology - Plant, vol. 40, no. 5, pp. 427-433, 2004/09/01 2004. [CrossRef]
- Pan, J.; Zeng, Q.; Peng, K.; Zhou, Y.; Shu, Z. Review of Rewarming Methods for Cryopreservation. Biopreserv Biobank 2024, vol. 22(no. 4), 304–311. [Google Scholar] [CrossRef]
- Barrett, D.; Digaudio, D. The landscape of cell and gene therapy today. Mol Ther 2025, vol. 33(no. 6), 2316–2323. [Google Scholar] [CrossRef]
- Huang, M. Overcoming ice: cutting-edge materials and advanced strategies for effective cryopreservation of biosample. J Nanobiotechnology 2025, vol. 23(no. 1), 187. [Google Scholar] [CrossRef]
- Whaley, D.; Damyar, K.; Witek, R. P.; Mendoza, A.; Alexander, M.; Lakey, J. R. Cryopreservation: An Overview of Principles and Cell-Specific Considerations. Cell Transplantation 2021, vol. 30, 0963689721999617. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Pan, F. Advances in cryopreservation of organs. J Huazhong Univ Sci Technolog Med Sci 2016, vol. 36(no. 2), 153–161. [Google Scholar] [CrossRef] [PubMed]
- Brinkkoetter, P. T.; et al. Hypothermic injury: the mitochondrial calcium, ATP and ROS love-hate triangle out of balance. Cell Physiol Biochem 2008, vol. 22(no. 1-4), 195–204. [Google Scholar] [CrossRef]
- Best, B. P. Cryoprotectant Toxicity: Facts, Issues, and Questions. Rejuvenation Research 2015, vol. 18(no. 5), 422–436. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Zhang, X.; Cui, Z.; Zhao, M.; Zhang, L.; Qi, H. Investigation into antifreeze performances of natural amino acids for novel CPA development. Journal of Materials Chemistry B, 10.1039/D3TB00131H 2023, vol. 11(no. 18), 4042–4049. [Google Scholar] [CrossRef] [PubMed]
- X. Zhang, H. Qi, J. Yang, X. Chen, and L. Zhang, “Development of Low Immunogenic Antifreeze Peptides for Cryopreservation,” Industrial & Engineering Chemistry Research, vol. 62, no. 31, pp. 12063-12072, 2023/08/09 2023. [CrossRef]
- Correia, L. F. L.; Alves, B. R. C.; Batista, R.; Mermillod, P.; Souza-Fabjan, J. M. G. Antifreeze proteins for low-temperature preservation in reproductive medicine: A systematic review over the last three decades. Theriogenology 2021, vol. 176, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Stolzing. New Cryoprotectants for Cell Therapies. Adv Exp Med Biol 2025, vol. 1486, 283–293. [Google Scholar] [CrossRef]
- H. Han et al., “Natural Deep Eutectic Solvents Enable Programmable Cooling-Free Cryopreservation,” Langmuir, vol. 41, no. 47, pp. 32099-32109, 2025/12/02 2025. [CrossRef]
- G. V. Research, “Cell Cryopreservation Market Size, Share & Trends Analysis Report By Product (Cell Freezing Media, Equipment, Consumables), By Application (Stem Cells, Bioproduction Cell Lines, Primary Cells), By End-use (Biobanks, IVF Clinics), By Region, And Segment Forecasts, 2025 - 2033,” in “Grand View Research,” GVR-4-68040-147-0, 2024. [Online]. Available: https://www.grandviewresearch.com/industry-analysis/cell-cryopreservation-market-report.
- S. Research, “Cell Cryopreservation Market Size, Share & Trends Analysis Report: Forecasts 2026-2034,” SRBI57741DR, 2025. [Online]. Available: https://straitsresearch.com/report/cell-cryopreservation-market.
- MarketsandMarkets, “Biopreservation Market by Product (Media [Cryopreservation, Hypothermic]), Equipment (Freezers, Refrigerators, Incubators, Centrifuge, Accessories), Biospecimen (Tissues, Organs, Stem Cells), Application (Research, Therapeutic) - Global Forecast to 2029,” BT 1305, 2024. [Online]. Available: https://www.marketsandmarkets.com/Market-Reports/biopreservation-media-storage-equipment-market-842.html.
- R. a. Markets, “Cell Freezing Media Market Size, Share & Trends Analysis Report By Product (DMSO, Glycerol), By Application (Stem Cell Lines, Cancer Cell Lines), By End-use, By Region, And Segment Forecasts, 2023-2030,” 2023. [Online]. Available: https://www.researchandmarkets.com/report/cell-freezing-media.
- Group, “Cell Freezing Media Market: Global Industry Trends, Share, Size, Growth, Opportunity and Forecast 2025-2033,” SR112026A13044, 2024. [Online]. Available: https://www.imarcgroup.com/cell-freezing-media-market.
- FMI. [Online]. Available: https://www.futuremarketinsights.com/reports/in-vitro-fertilization-market.
- Merck Group. [Online]. Available: https://www.merckgroup.com/en/expertise/fertility.html.
- Ashok; Choudhury, D.; Fang, Y.; Hunziker, W. Towards manufacturing of human organoids. Biotechnol Adv 2020, vol. 39, 107460. [Google Scholar] [CrossRef]
- Chen; Liu, X.; Hu, Y.; Chen, X.; Tan, S. Cryopreservation of tissues and organs: present, bottlenecks, and future. Front Vet Sci 2023, vol. 10, 1201794. [Google Scholar] [CrossRef]
- T. Ishizaki, Y. Takeuchi, K. Ishibashi, N. Gotoh, E. Hirata, and K. Kuroda, “Cryopreservation of tissues by slow-freezing using an emerging zwitterionic cryoprotectant,” Scientific Reports, vol. 13, no. 1, p. 37, 2023/01/02 2023. [CrossRef]
- Xue, W. Effective cryopreservation of human brain tissue and neural organoids. Cell Reports Methods 2024, vol. 4(no. 5). [Google Scholar] [CrossRef]
- Han, Z.; et al. Vitrification and nanowarming enable long-term organ cryopreservation and life-sustaining kidney transplantation in a rat model. Nat Commun 2023, vol. 14(no. 1), 3407. [Google Scholar] [CrossRef]
- Ozgur, S. Optimized partial freezing protocol enables 10-day storage of rat livers. Sci Rep 2024, vol. 14(no. 1), 25260. [Google Scholar] [CrossRef]
- Jing; Yao, L.; Zhao, M.; Peng, L. P.; Liu, M. Organ preservation: from the past to the future. Acta Pharmacol Sin 2018, vol. 39(no. 5), 845–857. [Google Scholar] [CrossRef] [PubMed]
- Sharma. Vitrification and Nanowarming of Kidneys. Adv Sci (Weinh) 2021, vol. 8(no. 19), e2101691. [Google Scholar] [CrossRef]
- Wang, Z. Replantation of Cryopreserved Fingers: An Organ Banking Breakthrough. Plastic and Reconstructive Surgery 2019, vol. 144(no. 3), 679–683. [Google Scholar] [CrossRef]
- Khaydukova, V. Current State and Challenges of Tissue and Organ Cryopreservation in Biobanking. Int J Mol Sci 2024, vol. 25(no. 20). [Google Scholar] [CrossRef]
- Elliott, G. D.; Wang, S.; Fuller, B. J. Cryoprotectants: A review of the actions and applications of cryoprotective solutes that modulate cell recovery from ultra-low temperatures. Cryobiology 2017, vol. 76, 74–91. [Google Scholar] [CrossRef]
- Giwa, S. The promise of organ and tissue preservation to transform medicine. Nat Biotechnol 2017, vol. 35(no. 6), 530–542. [Google Scholar] [CrossRef]
- Karlsson; Toner, M. Long-term storage of tissues by cryopreservation: critical issues. Biomaterials 1996, vol. 17(no. 3), 243–56. [Google Scholar] [CrossRef]
- Warner, R. M.; Shuttleworth, R.; Benson, J. D.; Eroglu, A.; Higgins, A. Z. General tissue mass transfer model for cryopreservation applications. Biophys J 2021, vol. 120(no. 22), 4980–4991. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, S. K.; Coger, R. N. Heat and mass transfer during the cryopreservation of a bioartificial liver device: a computational model. ASAIO J 2005, vol. 51(no. 3), 184–93. [Google Scholar] [CrossRef]
- Eisenberg, D. P.; Steif, P. S.; Rabin, Y. On the Effects of Thermal History on the Development and Relaxation of Thermo-Mechanical Stress in Cryopreservation. Cryogenics (Guildf) 2014, vol. 64, 86–94. [Google Scholar] [CrossRef] [PubMed]
- “ThermoFisher Scientific.” https://www.thermofisher.com/order/catalog/product/A1254201, (accessed.
- “Sartorius.” https://shop.sartorius.com/ww/p/nutrifreez-d10-cryopreservation-medium/NutriFreez_D10_Cryopreservation_Medium, (accessed.
- “BioLife Solutions.” stemcell.com/products/cryostor-cs10.html (accessed.
- “Merck.” https://www.sigmaaldrich.com/SG/en/product/sigma/c9249?srsltid=AfmBOoonDGYtXPLsdnMGE0-bLWfh5H2oL_mwQNWMhruTSYdTuwpKxivM (accessed.
- “Biotechne.” https://www.bio-techne.com/p/cell-culture/cryodefend-cell-lines-5-x-10-ml_ccm019 (accessed.
- “HIMEDIA.” https://www.himedialabs.com/us/tcl093-cryoxl-cell-freezing-medium-dmso-1x-w-fbs-and-dmso-w-o-antibiotics-sterile-filtered.html (accessed.
- “VitroLife.” https://www.vitrolife.com/products/cryopreservation/spermfreeze-solution/ (accessed.
- “Cooper Surgical.” https://www.coopersurgical.com/products/?product_type=art-media&procedure=cryopreservation (accessed.
- Solomon. Current perspectives on the use of ancillary materials for the manufacture of cellular therapies. Cytotherapy 2016, vol. 18(no. 1), 1–12. [Google Scholar] [CrossRef] [PubMed]
- “CFR 312.”.
- CMC. [Online] Available: https://www.fda.gov/media/113760/download#page=9.31.
- USP. [Online]. Available: https://www.usp.org/sites/default/files/usp/document/our-work/biologics/asgct-poster.pdf.
- Considerations for the Use of Humanand Animal-Derived Materials in the Manufacture of Cellular and Gene Therapy and Tissue-Engineered Medical Products. [Online] Available: https://www.fda.gov/media/178022/download.
- CFR 210. [Online] Available: https://www.ecfr.gov/current/title-21/chapter-I/subchapter-C/part-210.
- CFR 211. [Online] Available: https://www.ecfr.gov/current/title-21/chapter-I/subchapter-C/part-211.
- Warren, T.; Biggs, C. I.; Bissoyi, A.; Gibson, M. I.; Sosso, G. C. Data-driven discovery of potent small molecule ice recrystallisation inhibitors. Nat Commun 2024, vol. 15(no. 1), 8082. [Google Scholar] [CrossRef] [PubMed]
- Sambu, S. A Bayesian approach to optimizing cryopreservation protocols. PeerJ 2015, vol. 3, e1039. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Orimi, H. E.; Pedabaliyarasimhuni, P. K. R.; Hoesli, C. A.; Chioua, M. AI-Driven Quality Monitoring and Control in Stem Cell Cultures: A Comprehensive Review. Biotechnol J 2025, vol. 20(no. 8), e70100. [Google Scholar] [CrossRef]
- EMA. “Guiding Principles of Good AI Practice in Drug Development.” https://www.fda.gov/about-fda/artificial-intelligence-drug-development/guiding-principles-good-ai-practice-drug-development (accessed March, 2026). (accessed on March 2026).
| Company | Medium name | Composition | Target cell types | Grade |
|
Thermo Fisher Scientific |
(CTS)Synth-a-Freeze [44] | 10% DMSO, antibiotic free, serum free, protein-free | Various mammalian cells (except melanocytes) | RUO/ cGMP |
| Recovery Cell Culture Freezing Medium | 10% DMSO, high glucose, phenol red, with FBS | Mammalian cells | RUO | |
| Sartorius | NutriFreez D5/D10 [45] | 5%/ 10% DMSO, animal component free, serum free, protein free | Various | cGMP |
|
BioLife solutions |
CryoStor CS5/CS10 [46] | 5%/ 10% DMSO, serum free, protein free | Various | cGMP, USP |
| BloodStor 27/55/100 | 27%/ 55%/ 100% DMSO in saline, animal component free, serum free | Stem cells, hematopoietic stem and progenitor cells | cGMP, USP | |
| Merck | C9249 CryoSOfree DMSO-free [47] | DMSO free, animal component free | Various | RUO |
| C6295 Cell Freezing Medium | 8.7% DMSO, serum free | Various | RUO | |
| Biotechne | CryoDefend-Cell Lines [48] | Contain DMSO, protein free | Multiple cell lines | RUO |
| StemXVivo® Serum-Free MSC Freezing Media | 10% DMSO, serum free, antibiotic free | Human/mouse/rat MSC | RUO | |
| HIMEDIA | CryoXL™ Cell Freezing Medium [49] | DMSO, FBS | Mammalian cells | RUO |
| Vitrolife | SpermFreeze Solution [50] | MOPS, HSA (Human Serum Albumin), glycerol, cholesterol | Human sperm | Regulatory approved |
| RapidVit & RapidWarm | MOPS, HSA, cryoprotectants | Oocytes, embryos of various stages | Regulatory approved | |
|
Cooper Surgical |
Quinn’s Advantage Sperm Freezing [51] | HEPES, HSA, phenol red, gentamicin | Human sperm | |
| Quinn’s Advantage Embryo Thaw Kit | HSA | Embryos and blastocysts | Regulatory approved | |
| SAGE Vitrification Solutions | NA | Oocyte and embryo | Regulatory approved | |
| Global Blastocyst Fast Freeze Thawing Kit | HSA, DMSO free | Human blastocyst | ||
| Global DMSO Blastocyst Vitrification Kit | DMSO, HEPES, HSA | Human blastocysts | ||
| Sperm Freezing Medium / CryoSperm | Insulin, sucrose, egg yolk-free, HSA-free, HEPES free | Human sperm | ||
| Embryo Freezing/ Thawing Pack | HSA, propylene glycol, sucrose | Human zygotes and cleavage stage embryos | ||
| MediCult Vitrification Cooling/ Warming | DMSO-free, HEPES, HSA | Human oocytes, embryos |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




