Submitted:
16 April 2026
Posted:
17 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
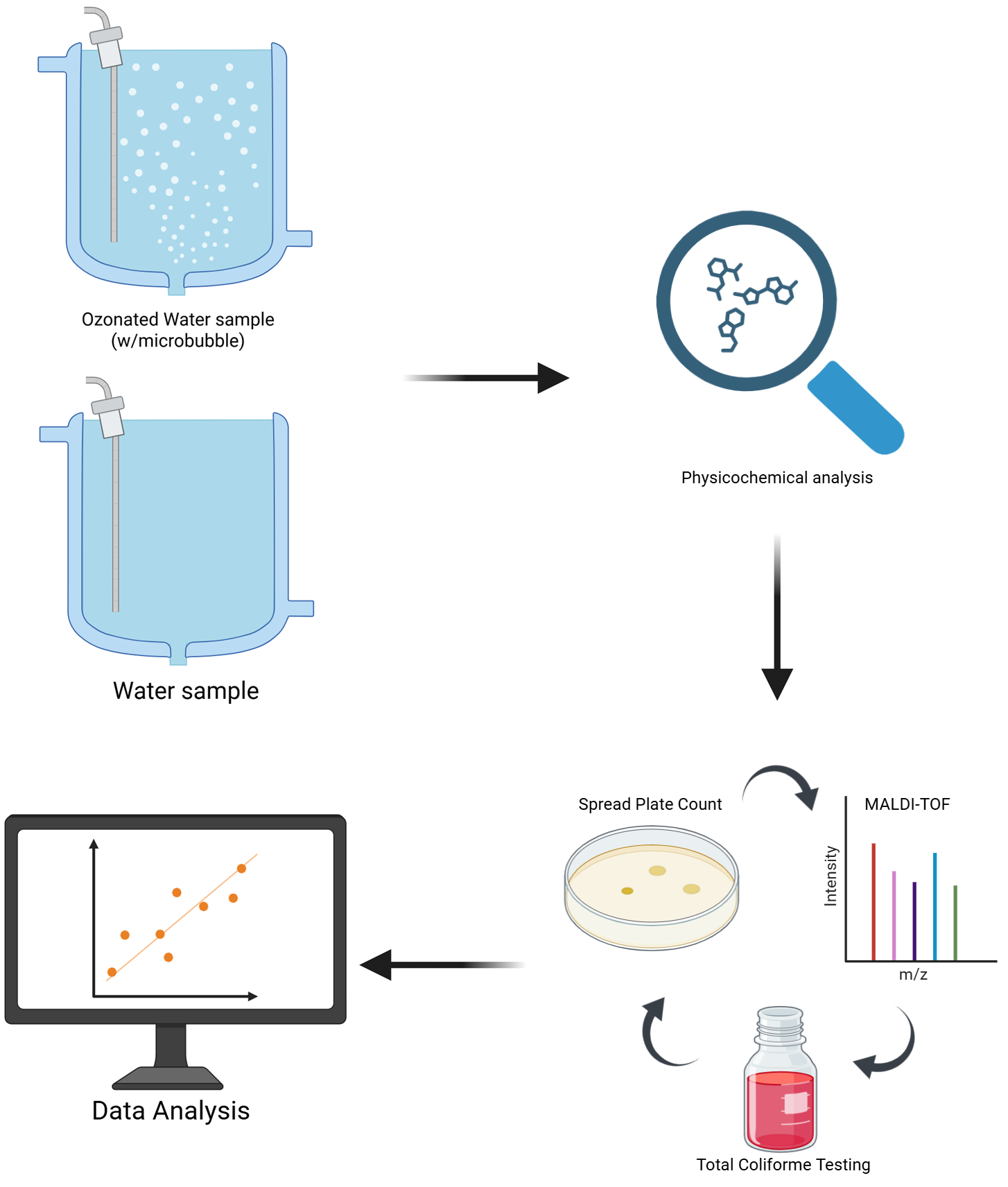
2.1. Ozone Treatment
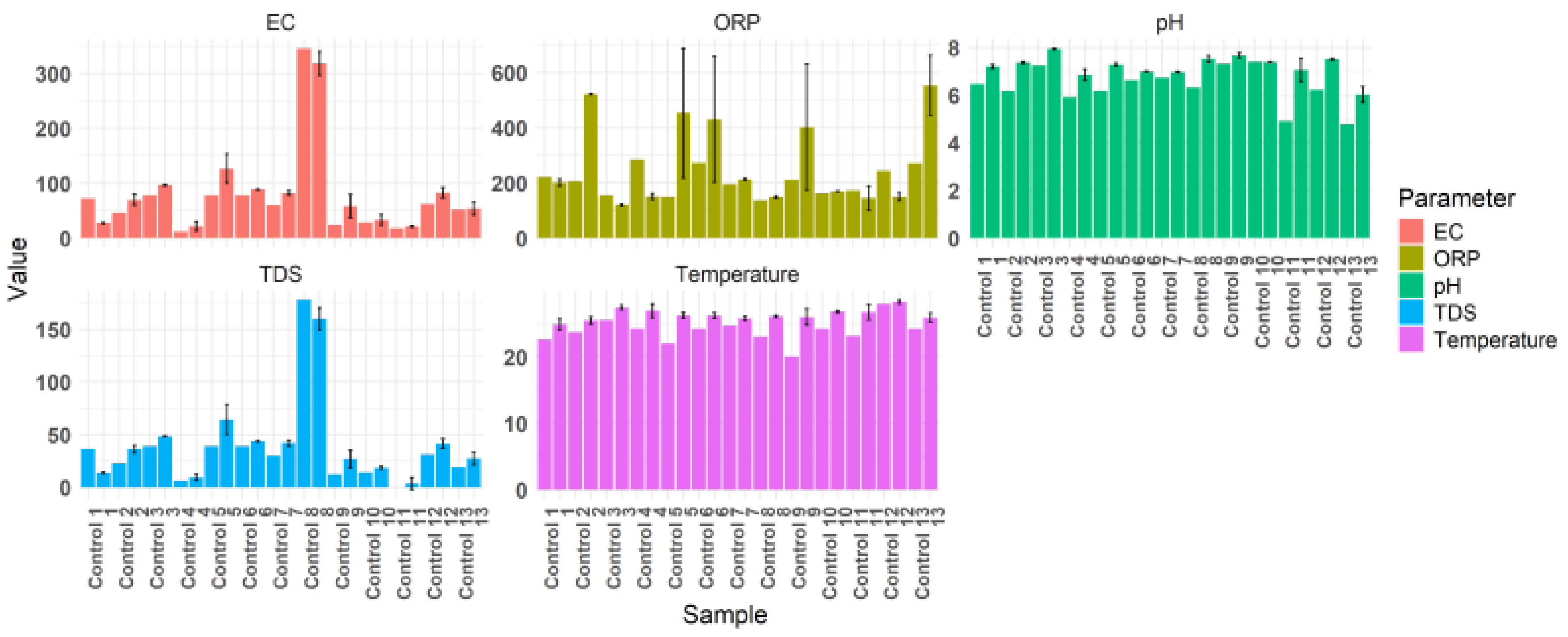
2.2. Monitoring of Physicochemical Parameters (pH, Temperature, TDS, EC, and ORP)
2.3. Total Coliform Testing
2.3.1. Presumptive/Confirmatory Analysis
2.4. Mass Spectrometry (MALDI-TOF)
3. Discussion
4. Materials and Methods
4.1. Bacterial Strain
4.2. Water Sample
| Strain | Sample description |
|---|---|
| 1 | Water collected from a private well in Praia Seca, Região dos Lagos - R.J. |
| 2 | Water used for irrigation supply in a vegetable garden in Corrêas, Petrópolis - R.J. |
| 3 | Water stored in a washing tank used for a vegetable garden in Corrêas, Petrópolis - R.J. |
| 4 | Rainwater collected in a tank with debris and foliage from a private property in Corrêas, Petrópolis - R.J. |
| 5 | Well water collected from a household tap in Corrêas, Petrópolis, RJ. |
| 6 | Water collected from Private Property 1 in Corrêas, Petrópolis - R.J. |
| 7 | Water collected from Private Property 2 in Corrêas, Petrópolis - R.J. |
| 8 | Water collected from a private cistern in Praia Seca, Região dos Lagos - R.J. |
| 9 | Water collected from the Alcobacinha spring in Cascatinha, Petrópolis - R.J. |
| 10 | Well water collected from a tap in Angra dos Reis, RJ. |
| 11 | Water collected from a well in the Kélé region, Middle-Ogooué Province, Gabon - Central Africa |
| 12 | Water collected from a well in the Eyouga region, Haut-Ogooué Province, Gabon - Central Africa |
| 13 | Water collected from a well in the Eyouga region, Haut-Ogooué Province, Gabon - Central Africa |
| 14 | Water collected directly from Marapendi Lagoon, Barra da Tijuca - RJ |
4.3. Ozone Generating System (OGS)
4.4. Ozone Treatment Procedure
4.5. Statistical Analysis
4.6. Total Coliform Testing
4.6.1. Presumptive Analysis
4.6.2. Confirmatory Analysis
4.6.3. Culture Media Controls
4.7. Monitoring of pH, Temperature, TDS, EC, and ORP
4.8. Mass Spectrometry (MALDI-TOF)
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Von Sonntag, C.; Von Gunten, U. Chemistry of Ozone in water and waste-water treatment: from basic principles to applications; IWA Publishing: London, 2012; 306 p, ISBN 9781780400839. [Google Scholar]
- Chen, S.; Qiao, Y.; Jiang, Y.; Qiu, W.; Zang, S.; Zhang, J.; Wang, X.; Ma, J. Trace analysis of 59 halogenated aromatic disinfection by-products through the SPE-LC-MS/MS method and their occurrence and transformation during chlorine disinfection. J Environ Sci (China). 2025, 157, 821–832. [Google Scholar] [CrossRef] [PubMed]
- Dhanda, N.; Kumar, S. Water disinfection and disinfection by-products. Environ Monit Assess. 2025, 197, 461. [Google Scholar] [CrossRef] [PubMed]
- Kruithof, J.C.; Masschelein, W.J. State-of-the-art of the application of ozonation in BENELUX drinking water treatment. Ozone: Science and Engineering 1999, 21, 139–152. [Google Scholar] [CrossRef]
- Sorlini, S.; Collivignarelli, C. Trihalomethane formation during chemical oxidation with chlorine, chlorine dioxide, and ozone of ten Italian natural waters. Desalination. 2005, 176, 103–111. [Google Scholar] [CrossRef]
- Zhang, B.; Xian, Q.; Lu, J.; Gong, T.; Li; A. Feng, J. Evaluation of DBPs formation from SMPs exposed to chlorine, chloramine, and ozone. J Water Health 2017, 15, 185–195. [Google Scholar] [CrossRef]
- Mazhar, M.A.; Khan, N.A.; Ahmed, S.; Khan, A.H.; Hussain, A.; Rahisuddin; Changani, F.; Yousefi, M.; Ahmadi, S.; Vambol, V. Chlorination disinfection by-products in municipal drinking water – A review. J Clean Prod. 2020, 10, 123159. [Google Scholar] [CrossRef]
- Forster, A.L.B.; Wiskur, S.L.; Richardson, S.D. Formation of eight classes of DBPs from chlorine, chloramine, and ozone: Mechanisms and formation Pathways. Environ Sci Technol 202521. [Google Scholar] [CrossRef]
- Karanis, P.; Kourenti, C.; Smith, H. Waterborne transmission of protozoan parasites: a worldwide review of outbreaks and lessons learnt. J Water Health. 2007, 5, 1–38. [Google Scholar] [CrossRef]
- Xu, H.; Yang, A.; Pang, Y.; Pei, H. Advances and challenges in the technologies for cyanobacterial cells removal in drinking water treatment. Chemosphere. 2024, 359, 142338. [Google Scholar] [CrossRef]
- Arana, I.; Santorum, P.; Muela, A.; Barcina, I. Chlorination and ozonation of waste-water: comparative analysis of efficacy through the effect on Escherichia coli membranes. J Appl Microbiol. 1989, 86, 883–8. [Google Scholar] [CrossRef]
- Camel, V.; Bermond, A. The use of ozone and associated oxidation processes in drinking water treatment. Water Res. 1998, 32, 3208–22. [Google Scholar] [CrossRef]
- Hoigné, J. Chemistry of aqueous ozone, and transformation of pollutants by ozonation and advanced oxidation processes. In The handbook of environmental chemistry, quality and treatment of drinking water; Hubrec, J., Ed.; Springer: Berlin, 1998. [Google Scholar]
- Von Gunten, U. Ozonation of drinking water: Part I. Oxidation kinetics and product formation. Water Res. 2003, 37, 1443–67. [Google Scholar] [CrossRef] [PubMed]
- Staehelin, J.; Hoigné, J. Decomposition of ozone in water in the presence of organic solutes acting as promoters and inhibitors of radical chain reactions. Environ Sci Technol. 1985, 19, 1206–13. [Google Scholar] [CrossRef] [PubMed]
- Facile, N.; Barbeau, B.; Prevost, M.; Koudjonou, B. Evaluating bacterial aerobic spores as a surrogate for Giardia and Cryptosporidium inactivation by ozone. Water Res. 2000, 34, 3238–46. [Google Scholar] [CrossRef]
- Mahmoodi, M.; Pishbin, E. Ozone-based advanced oxidation processes in water treatment: recent advances, challenges, and perspective. Environ Sci Pollut Res Int. 2005, 32, 3531–3570. [Google Scholar] [CrossRef]
- Hoigné, J.; Bader, H. Characterization of water quality criteria for ozonation processes. Part II: Lifetime of added ozone. Ozone: Sci Eng. 2013, 1994, 121–134. [Google Scholar] [CrossRef]
- Langlais, B.; Reckhow, D.A.; Brink, D.R. Ozone in water treatment: application and engineering; Lewis Publisher: Paris, 2019. [Google Scholar]
- Richardson, S.D. Water analysis: emerging contaminants and current issues. Anal Chem. 2009, 81, 4645–4677. [Google Scholar] [CrossRef]
- Liang, J.; Fei, Y.; Yin, Y.; Han, Q.; Liu, Y.; Feng, L.; Zhang, L. Advancements in waste-water treatment: A comprehensive review of ozone microbubbles technology. Environ Res. 2025, 266, 120469. [Google Scholar] [CrossRef]
- Seki, M.; Ishikawa, T.; Terada, H.; Nashimoto, M. Microbicidal Effects of stored aqueous ozone solution generated by nano-bubble technology. In Vivo. 2017, 31, 579–583. [Google Scholar] [CrossRef]
- Von Gunten, U. Ozonation of drinking water: Part II. Disinfection and by-product formation in presence of bromide, iodide or chlorine. Water Res. 2003, 37, 1469–87. [Google Scholar] [CrossRef]
- Holcomb, D.A.; Stewart, J.R. Microbial Indicators of Fecal Pollution: Recent Progress and Challenges in Assessing Water Quality. Curr Environ Health Rep. 2020, 7, 311–324. [Google Scholar] [CrossRef] [PubMed]
- Ershov, B.G.; Morozov, P.A. The kinetics of ozone decomposition in water, the influence of pH and temperature. Russ. J Phys Chem. 2009, 83, 1295–1299. [Google Scholar] [CrossRef]
- Galdeano, M.C.; Wilhelm, A.E.; Goulart, I.B.; Tonon, R.V.; Freitas-Silva, O.; Germani, R.; Chávez, D.W.H. Effect of water temperature and pH on the concentration and time of ozone saturation. Braz. J Food Technol. 2018, 21, e2017156. [Google Scholar] [CrossRef]
- Kalandarov, P.; Zhunussov, K.; Kassimov, A.; Baibolov, B.; Junussov, N.; Kaliyeva, K. Changes in pH during the ozonation process of surface water. E3S Web Conf. 2004, 563, 01020. [Google Scholar] [CrossRef]
- BRASIL; Ministério da Saúde. Portaria GM/MS nº 888, de 4 de maio de 2021. Dispõe sobre os padrões de qualidade para água potável. Available online: https://www.in.gov.br (accessed on 15 March 2025).
- Martinelli, M.; Giovannangeli, F.; Rotunno, S.; Trombetta, C.M.; Montomoli, E. Water and air ozone treatment as an alternative sanitizing technology. J Prev Med Hyg. 2017, 58, E48–E52. [Google Scholar] [CrossRef]
- Tanner, B.D.; Kuwahara, S.; Gerba, C.P.; Reynolds, K.A. Evaluation of electrochemically generated ozone for the disinfection of water and waste-water. Water Sci Technol. 2004, 50, 19–25. [Google Scholar] [CrossRef]
- Fox, G.E.; Stackebrandt, E.; Hespell, R.B.; Gibson, J.; Maniloff, J.; Dyer, T.A.; Wolfe, R.S.; Balch, W.E.; Tanner, R.S.; Magrum, L.J.; et al. The phylogeny of prokaryotes. Science 1980, 209, 457–63. [Google Scholar] [CrossRef]
- BRASIL. ANVISA (2002) Resolução RDC nº 275.; de 21 de outubro de 2002. Dispõe sobre o regulamento técnico de boas práticas para serviços de alimentação. Available online: https://www. (accessed on 15 July 2025).
- Manasfi, T. Chapter Four - Ozonation in drinking water treatment: an overview of general and practical aspects, mechanisms, kinetics, and by-product formation. Compr An Chem. 2021, 92, 85–116. [Google Scholar] [CrossRef]
- Feng, P.; Weagant, S.D.; Grant, M. A Enumeration of Escherichia coli and the coliform bacteria: Bacteriological analytical manual (BAM); U.S. Food and Drug Administration, 2022. [Google Scholar]
- Sauer, S.; Kliem, M. Mass spectrometry tools for the classification and identification of bacteria. Nat Rev Microbiol. 2010, 8, 74–82. [Google Scholar] [CrossRef]
- Liu, H.; Du, Z.; Wang, J.; Yang, R. Universal sample preparation method for characterization of bacteria by matrix-assisted laser desorption ionization-time of flight mass spectrometry. Appl Environ Microbiol. 2007, 73, 1899–907. [Google Scholar] [CrossRef]
- Sauer, S.; Freiwald, A.; Maier, T.; Kube, M.; Reinhardt, R.; Kostrzewa, M.; Geider, K. Classification and identification of bacteria by mass spectrometry and computational analysis. PLoS One 2008, 3, e2843. [Google Scholar] [CrossRef]
- Welker, M.; Moore, E.R. Applications of whole-cell matrix-assisted laser-desorption/ionization time-of-flight mass spectrometry in systematic microbiology. Syst Appl Microbiol. 2011, 34, 2–11. [Google Scholar] [CrossRef]
- Ashfaq, M.Y.; Da'na, D.A.; Al-Ghouti, M.A. Application of MALDI-TOF MS for identification of environmental bacteria: A review. J Environ Manage 2022, 305, 114359. [Google Scholar] [CrossRef]
- Suzuki, Y.; Niina, K.; Matsuwaki, T.; Nukazawa, K.; Iguchi, A. Bacterial flora analysis of coliforms in sewage.; river water.; and ground water using MALDI-TOF mass spectrometry. J Environ Sci Health A Tox Hazard Subst Environ Eng. 2018, 53, 160–173. [Google Scholar] [CrossRef]
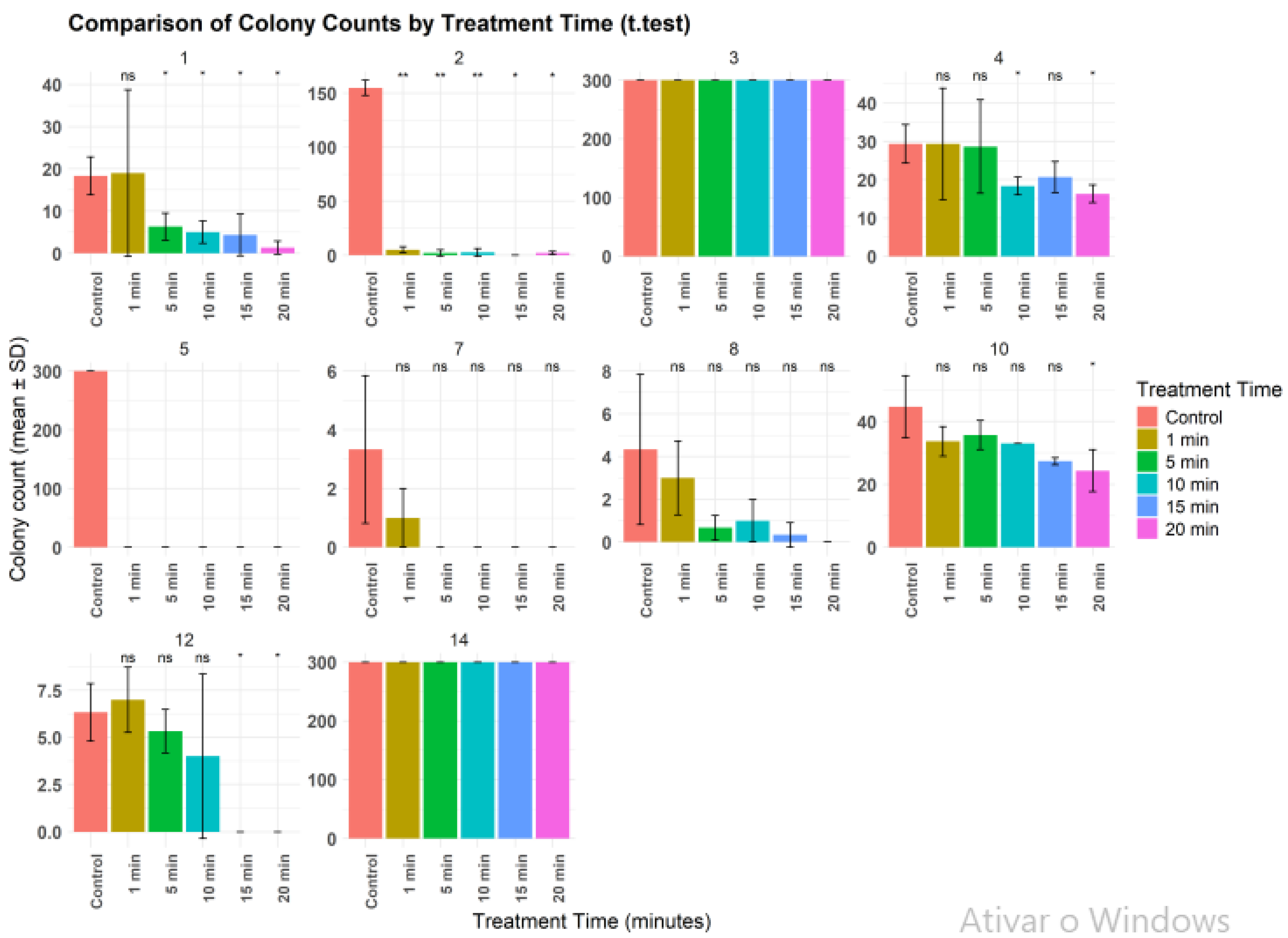
| Strain | Control (CFU/ML) |
Ozone Exposure Time | ||||
|---|---|---|---|---|---|---|
| 1’ | 5’ | 10’ | 15’ | 20’ | ||
| Count nº CFU/mL / % reduction | ||||||
| 1 | 18,3 | 19* | 6,3/65,6 | 5/72,7 | 4,3/76,5 | 1,3/92,9 |
| 2 | 155 | 5/96,8 | 2/98,7 | 2,5/98,4 | 0/100 | 2/98,7 |
| 3* | > 300 | > 300 | > 300 | > 300 | > 300 | > 300 |
| 4 | 29,3 | 29,3* | 28,6/2,4 | 18,3/37,5 | 20,7/29,4 | 16,3/44,4 |
| 5 | > 300 | 0/100 | 0/100 | 0/100 | 0/100 | 0/100 |
| 7 | 3,3 | 1/69,7 | 0/100 | 0/100 | 0/100 | 0/100 |
| 8 | 4,3 | 3/30,2 | 0,7/83,7 | 0/100 | 0/100 | 0/100 |
| 10 | 44,7 | 33,7/24,6 | 35,7/20,13 | 33/26,2 | 27,3/38,9 | 24,33/45,6 |
| 12 | 6,3 | 7* | 5,3/15,9 | 4/36,5 | 0/100 | 0/100 |
| 14* | > 300 | > 300 | > 300 | > 300 | > 300 | > 300 |
| Strain | T (°C) C/T |
pH C/T |
TDS (ppm) C/T |
EC (µS/cm) C/T |
ORP (mV) C/T |
|||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 22,6 | 24,8 | 6,46 | 7,2 | 36 | 13,7 | 72 | 27,3 | 222 | 201,3 |
| 2 | 23,7 | 25,4 | 6,18 | 7,4 | 23 | 36 | 46 | 69,7 | 206 | 388,7 |
| 3 | 25,5 | 27,4 | 7,24 | 7,94 | 39 | 48,3 | 78 | 96,7 | 156 | 119,7 |
| 4 | 24,2 | 26,9 | 5,92 | 6,8 | 6 | 9,3 | 12 | 21,7 | 285 | 150,7 |
| 5 | 22 | 26,2 | 6,18 | 7,27 | 39 | 64,3 | 78 | 126,7 | 149 | 453,3 |
| 6 | 24,2 | 26,1 | 6,62 | 6,99 | 39 | 43,7 | 78 | 88,3 | 273 | 431 |
| 7 | 24,7 | 25,8 | 6,73 | 6,96 | 30 | 42 | 60 | 82 | 195 | 212,3 |
| 8 | 23 | 26 | 6,33 | 7,5 | 178 | 160 | 346 | 318,7 | 136 | 149 |
| 9 | 20 | 25,9 | 7,32 | 7,7 | 12 | 16,1 | 24 | 58 | 211 | 402,3 |
| 10 | 24,2 | 26,8 | 7,39 | 7,37 | 14 | 18,3 | 28 | 31 | 163 | 168,7 |
| 11 | 23,1 | 26,7 | 4,9 | 7,1 | 0,09 | 3,4 | 18 | 21,3 | 172 | 144,3 |
| 12 | 27,9 | 28,2 | 6,23 | 7,5 | 31 | 41,3 | 62 | 82,7 | 244 | 149,3 |
| 13 | 24,2 | 25,8 | 4,78 | 6 | 19 | 27 | 52 | 53,3 | 271 | 554 |
| 14 | 22,8 | 25,4 | 6,3 | 7,7 | 9999 * | 9999 * | 1999x10 * | 1999x10 * | 167 | 127 |
| Average | 23,7 | 26,3 | 6,3 | 7,2 | 35,9 | 41,1 | 73,4 | 83 | 206,4 | 260,8 |
| Total Coliform Research | Strains | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1, 5, 7, 12 | 2 | 3, 4, 10, 14 | 6, 9, 11, 13 | 8 | ||||||
| C | T 1ˢᵗ/2ⁿᵈ/3ʳᵈ |
C | T 1ˢᵗ/2ⁿᵈ/3ʳᵈ |
C | T 1ˢᵗ/2ⁿᵈ/3ʳᵈ |
C | T 1ˢᵗ/2ⁿᵈ/3ʳᵈ |
C | T 1ˢᵗ/2ⁿᵈ/3ʳᵈ |
|
| Presumptive (CPA) | + | −/−/− | + | −/−/+ | + | +/+/+ | − | −/−/− | + | −/−/− |
| Confirmatory (CVBBL) | − | −/−/− | + | −/−/+ | + | +/+/+ | − | −/−/− | + | −/−/− |
| Experiment (Sample 2, 20 min) |
UFC reduction (%) | ORP (mV) |
Coliform result (BGBLB) |
Microbubbles observed |
|---|---|---|---|---|
| 1 | 98.7 | 523 | Negative | Yes |
| 2 | 98.7 | 521 | Negative | Yes |
| 3 | 0 | 122 | Positive | No |
| Sample/ Colony |
Morphotype | ID. MALDI-TOF | Score | Category |
|---|---|---|---|---|
| 1/1 | Small white colony | Bacillus mycoides | 2.40 | A |
| 1/2 | Flat white colony with central depression | Paenibacillus pabuli | 1.77 | B |
| 1/3 | White colony | Bacillus mycoides | 2.22 | A |
| 1/4 | Mucoid white colony | Pseudomonas chlororaphis | 1.94 | B |
| 1/5 | Rough white colony | Bacillus mycoides | 1.82 | B |
| 1/6 | Medium-sized mucoid white colony | Pseudomonas corrugata | 1.95 | B |
| 1/7 | Medium white colony | Pseudomonas chlororaphis | 1.91 | B |
| 1/8 | Medium white colony | Bacillus mycoides | 2.24 | A |
| 2/1 | Very small white colony | Pseudomonas extremorientalis | 2.20 | B |
| 2/2 | Medium-sized mucoid white colony | Pseudomonas koreensis | 1.94 | B |
| 2/3 | Medium white colony | Pseudomonas koreensis | 1.94 | B |
| 2/4 | Small white colony | Pseudomonas rhodesiae | 1.92 | B |
| 2/5 | Medium white colony | Pseudomonas koreensis | 2.00 | A |
| 2/6 | Medium white colony with irregular edge | Pseudomonas extremorientalis | 1.94 | B |
| 2/7 | Yellow colony with irregular, rough margin | Pseudomonas extremorientalis | 1.91 | B |
| 2/8 | Flat, irregular white colony | No Organism Identification Possible | 1.63 | C |
| 2/9 | Medium white colony | Pseudomonas koreensis | 1.97 | B |
| 2/10 | Small yellowish colony | Paenarthrobacter ilicis | 2.21 | A |
| 2/11 | Small white colony | Aeromonas bestiarum | 2,02 | A |
| 2/12 | Small white colony | Pseudomonas rhodesiae | 1.95 | B |
| 3/1 | Medium white colony adhering to the plate | Bacillus pumilus | 1.76 | B |
| 3/2 | Translucent gel-like colony with yellow-edged root-like projections | No Organism Identification Possible | 1.46 | C |
| 4/1 | Yellow colony with irregular, rough margin | Pseudomonas grimontii | 1.93 | B |
| 4/2 | Rough white colony with a cloud-like appearance | Pseudomonas koreensis | 2.13 | A |
| 4/3 | Medium irregular white colony | Bacillus pumilus | 1.90 | B |
| 4/4 | White/translucent irregular colony | Pseudomonas koreensis | 2,09 | A |
| 4/5 | Cracked white colony with cloud effect | Bacillus pumilus | 1.82 | B |
| 4/6 | Irregular translucent colony | Pseudomonas antarctica | 1.99 | B |
| 5/1 | Small translucent colony | No Organism Identification Possible | 1.36 | C |
| 5/2 | Small translucent colony | Cupriavidus basilensis | 1.76 | B |
| 5/4 | Tiny colony | Microbacterium testaceum | 1.73 | B |
| 5/5 | Small translucent colony | Microbacterium testaceum | 1.76 | B |
| 5/6 (PP) | Very small yellowish colony | Rhodococcus erythropolis | 2,03 | A |
| 5/6 (O) | "Fried egg"-like colony | Pseudomonas koreensis | 1.99 | B |
| 5/6 (M) | Medium translucent colony | Bacillus pumilus | 1.94 | B |
| 7/1 | Medium mucoid white colony | Pseudomonas koreensis | 1.88 | B |
| 7/2 | Medium mucoid white colony | Pseudomonas koreensis | 2.00 | A |
| 9/1 | Medium mucoid white colony | Pseudomonas koreensis | 1.84 | B |
| 9/2 | Medium mucoid white colony | Pseudomonas koreensis | 1.95 | B |
| 9/3 | Medium translucent "watery"-appearing colony | Pseudomonas corrugata | 2,01 | A |
| 9/4 | Medium translucent "watery"-appearing colony | Pseudomonas corrugata | 2,02 | A |
| 9/5 | Medium orange colony | Pseudomonas marginalis | 2,09 | B |
| 9/6 | Medium orange colony | Pseudomonas marginalis | 2.00 | A |
| 10/1 | Rough white colony | Bacillus cereus | 2,08 | A |
| 10/2 | Small yellowish colony | Paenarthrobacter nicotinovorans | 2.17 | B |
| 10/3 | Small yellowish colony | Paenarthrobacter nicotinovorans | 2.38 | B |
| 10/4 | Small yellowish colony with a central dot | Pseudomonas libanensis | 2.19 | A |
| 10/5 | Irregular white colony | Micrococcus luteus | 2,08 | A |
| 10/6 | Small yellowish colony | Pseudomonas libanensis | 2,11 | A |
| 12/1 | Small translucent colony | Comamonas testosteroni | 2.17 | A |
| 12/2 | Very small translucent colony | Pseudarthrobacter siccitolerans | 1.95 | B |
| 12/3 | Small translucent colony | Pseudarthrobacter oxydans | 1.92 | B |
| 12/4 | Very small translucent colony | Comamonas testosteroni | 2.11 | A |
| 12/5 | Very small translucent colony | Comamonas testosteroni | 2,07 | A |
| 12/6 | Medium translucent colony with irregular margin | Comamonas testosteroni | 2.15 | A |
| 12/8 | Small translucent colony | Comamonas testosteroni | 2.17 | A |
| 12/9 | Small translucent colony | Pseudarthrobacter oxydans | 1.95 | B |
| 12/10 | Translucent colony with irregular border | Comamonas testosteroni | 2,07 | A |
| 12/12 | Small translucent colony | Acinetobacter junii | 2.33 | A |
| 12/13 | Very small, watery, translucent colony | Pseudarthrobacter oxydans | 1.82 | B |
| 14/1 | Rough yellowish colony | Pseudomonas fluorescens | 1.94 | B |
| 14/2 | Medium white/yellowish colony | Aeromonas eucrenophila | 2,03 | A |
| Parameters | Measure |
|---|---|
| Dimensions | 3,5 x 3,5 x 4 cm |
| Weight | 64g |
| Power | 3W |
| Maximum flow rate | 150L/H |
| Water column (Maximum height) | 60cm |
| Outlet diameter | 8mm |
| Voltage | 110-240V |
| Power cord length | 1.47 meters |
| Range | Interpretation | Symbols | Color |
|---|---|---|---|
| 2.00 - 3.00 | High-confidence identification | (+++) | green |
| 1.7 – 1.99 | Low-confidence identification | (+) | yellow |
| 0.00 – 1.69 | No Organism Identification Possible | (−) | red |
| Category | Interpretation |
|---|---|
| (A) | High consistency: The best match is a high-confidence identification. The second-best match is (1) a high-confidence identification in which the species is identical to the best match, (2) a low-confidence identification in which the species or genus is identical to the best match, or (3) a non-identification. |
| (B) | Low consistency: The requirements for high consistency are not met. The best match is a high- or low- confidence identification. The second-best match is (1) a high- or low-confidence identification in which the genus is identical to the best match or (2) a non-identification. |
| (C) | No consistency: The requirements for high or low consistency are not met. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




