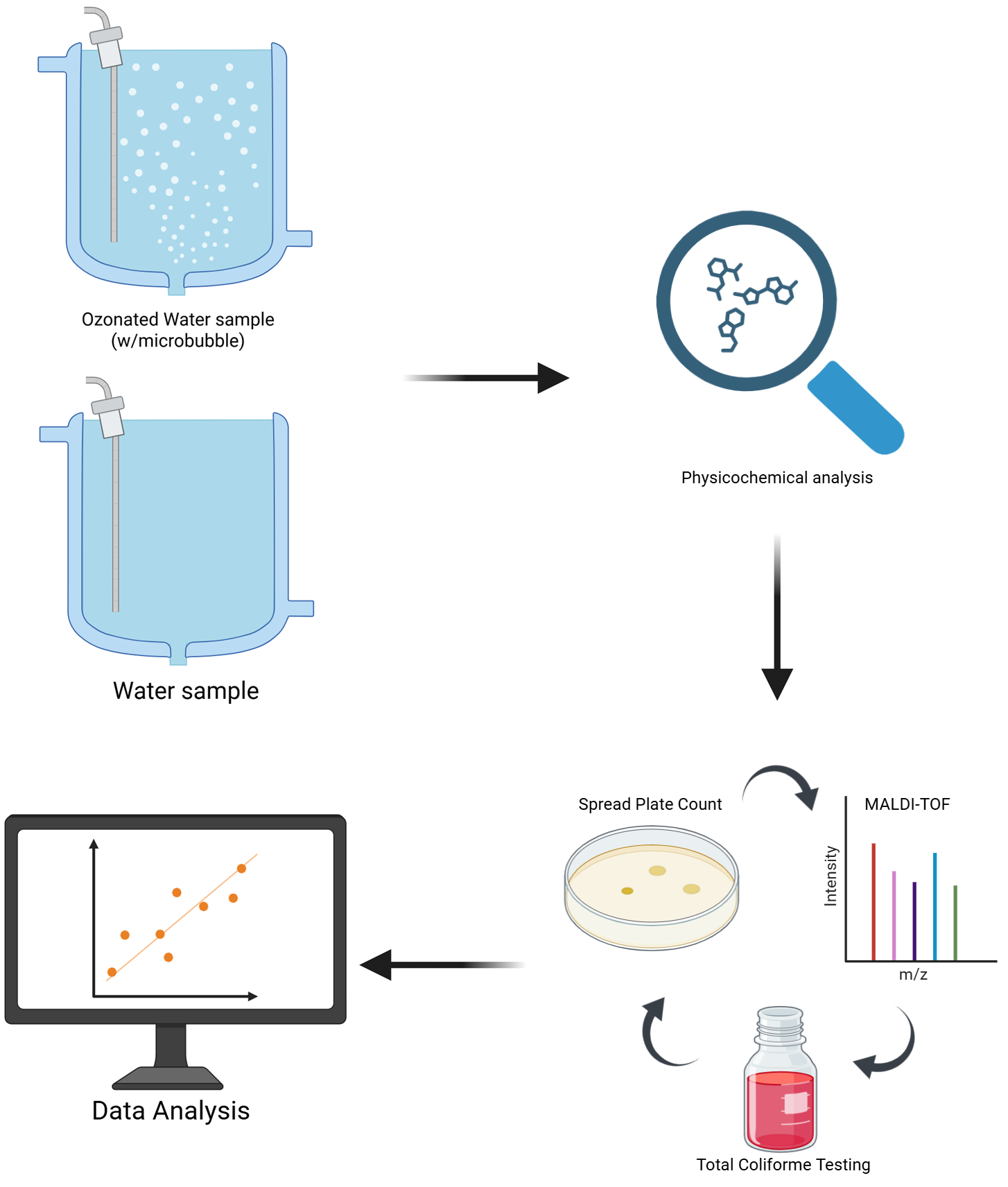
Ozone (O₃) is a potent disinfectant, yet its efficacy in environmentally complex waters can be inconsistent. This study investigated the effectiveness of ozonation across 14 water samples (wells, rainwater, ponds) and identified key success factors. Ozone exposure (1-20 min) significantly reduced colony-forming units (CFU) in most samples, achieving complete elimination in four. However, two high-organic-load samples showed minimal change. Physicochemical analyses revealed a strong correlation between increased Oxidation-Reduction Potential (ORP) and disinfection success. We observed that the efficient generation of ozone microbubbles was the critical factor in raising the ORP to lethal levels (> ~450 mV). A paradigmatic case was sample 2, where the elimination of total coliforms (98.7%) only occurred in replicates where microbubbles formed (ORP >520 mV), failing in the replicate without microbubbles (ORP 122 mV). Mass spectrometry (MALDI-TOF) identified genera such as Pseudomonas and Bacillus, indicating the presence of microbial diversity. We conclude that ozo-nation is highly effective, but its success depends on optimizing mass transfer via microbubbles, with ORP as a fundamental real-time indicator to ensure process reliability. This study offers a practical guideline for implementing more robust and safer ozone systems, overcoming limitations observed in waters with complex contamination.