3. Results
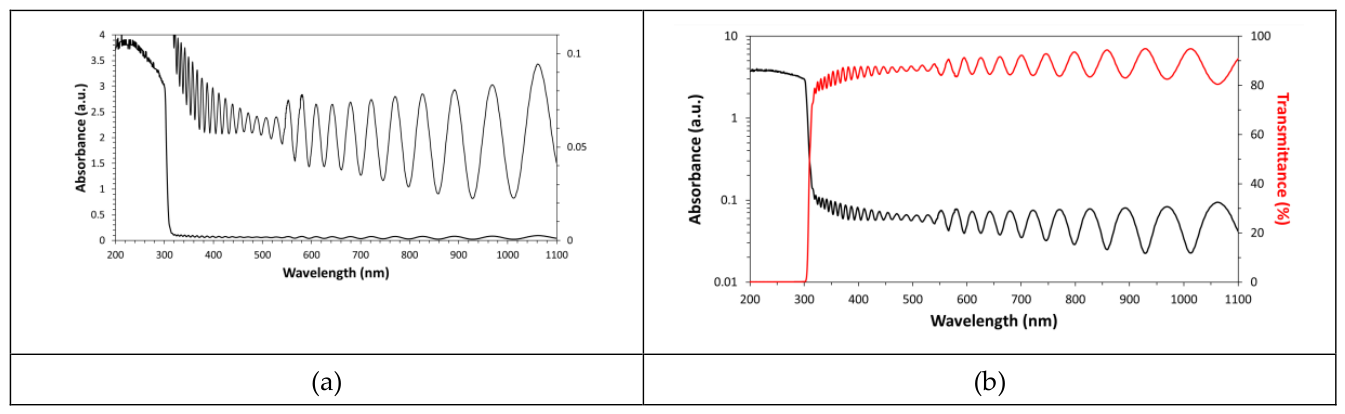
The interference phenomenon can be clearly detected in the high transparence region of the UV-Vis-NIR spectrum of ultrathin Mylar windows. This physical phenomenon can be used for accurately measuring the film thickness (d is of the same order as the light wavelength). In particular, the following equation, derived from the interference maxima condition (i.e., 2n·d=m·λ
m) [
9] in the case of a normally incident light beam, can be adopted [
10]:
where d is the film thickness (in nm), Δm is the number of fringes (oscillations maxima) contained in the measurement interval, n is the average polymer refractive index, and λ
1, λ
2 are the wavelength limits of the measurement interval. According to the spectral oscillations in the ultrathin PET film optical spectrum shown in
Figure 2, the film thickness obtained by using the above equation is 3.665µm (an average refractive index value of 1.575 has been used for calculation [
11]).
Since the PET films have a regular shape (i.e., thin circular dishes), this optically measured thickness value has been confirmed by a simple geometrical calculation based on density [
12]. In particular, the following expression has been used: d=m/(π·R
2·ρ), where m is the film weight, R the dish radius and ρ the PET density. A value of 3.59 µm has been found by using a PET density value of 1.3g/cm
3 [
13]. This thickness value perfectly agrees with value obtained by the interferometric measurement.
The interferometrically measured film thickness has been used for calculating the absorption coefficient values. It must be pointed out that the absorbance data in the fundamental absorption edge (required for both Urbach and Tauc analysis) are readily accessible for the absorption coefficient calculation because spectral oscillations are present only in the high transparency region of the UV-Vis spectrum. In addition, the UV-Vis Analyst software allows also the application of spectral smoothing and such operation can be conveniently used to reduce fringes in the spectrum and obtain the average absorbance in this spectral region [
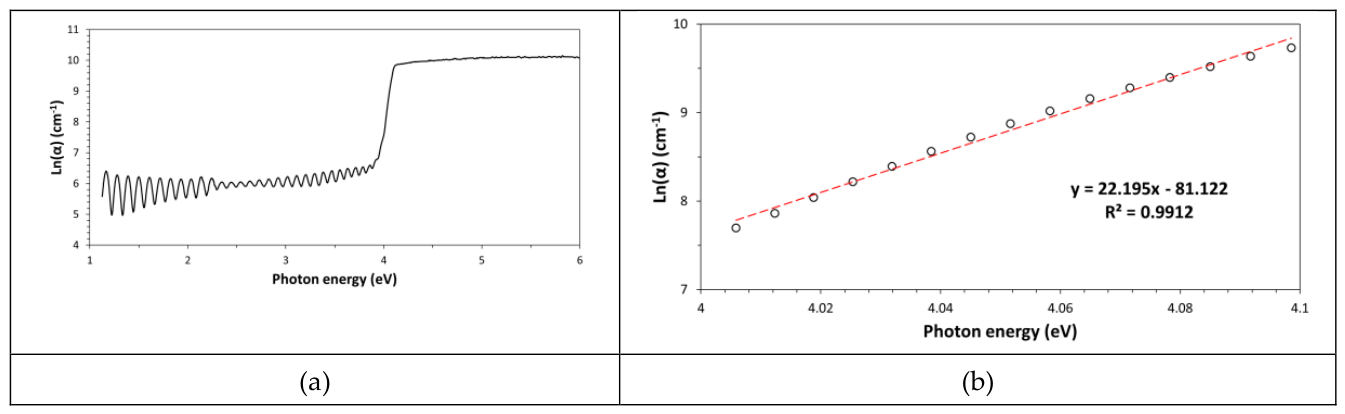
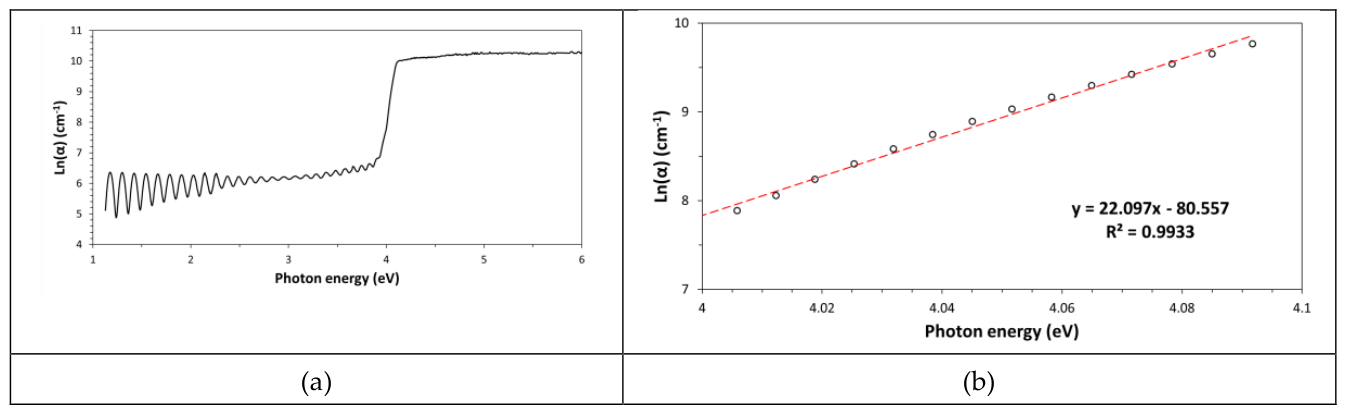
14]. A graph of ln(α) vs. the photon energy is named Urbach plot (see
Figure 3a) and this special graph can be used to know the amount of structural disorder in this ultrathin PET film [
15]. Structural disorder is proportional to the Urbach energy, E
U, which is given by the inverse of the slope of the linear part of curve in the Urbach plot. In particular, this quantity can be obtained by linear regression analysis of this strait part of the graph (see
Figure 3b). In general, the structural disorder obtained by the Urbach plot depends on all types of defects contained in this thin PET film like for example air bubbles (porosity), surface roughness, chemical impurities (molecular additives like plasticizers, UV-stabilizers, etc.) dissolved in the polymer, percentage of the amorphous phase, etc. However, with respect to perfectly processed films, most contributions can be neglected and the measured structural disorder corresponds exactly to the content of amorphous phase and to the degree of chain entanglement characterizing this amorphous phase. In the case of the ultrathin PET film such calculation leaded to a value of 45.0meV (with R
2=0.9912), which indicates a quite low disorder, probably due to the semi-crystalline nature of this polymer.
The small value of E
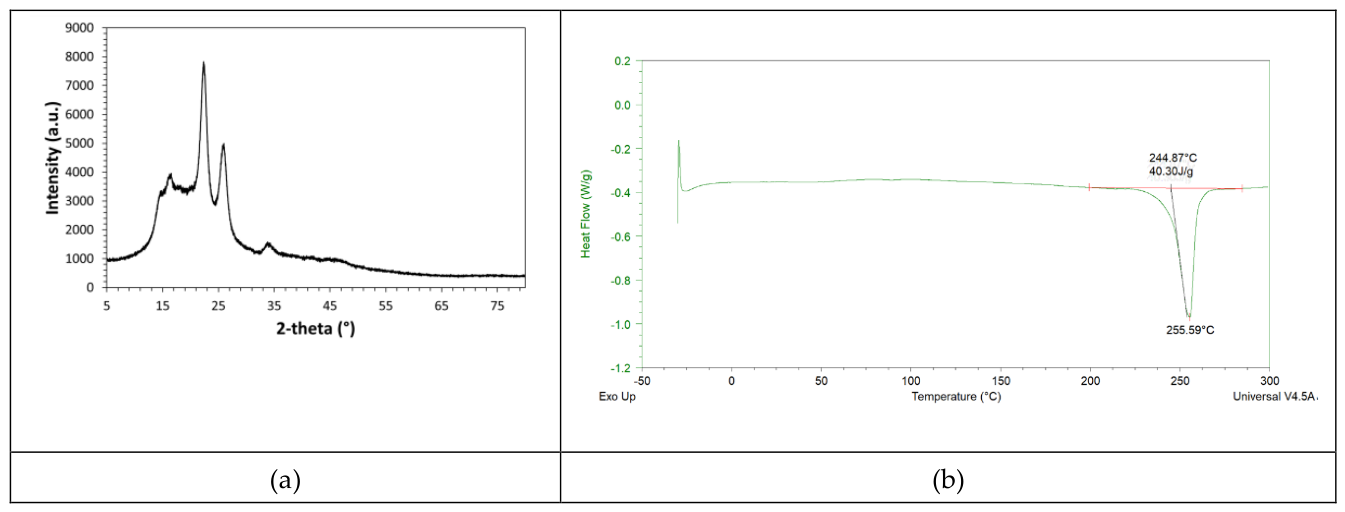
U that has been found by applying the Urbach approach to the spectral data of PET films can be well justified on the basis of the semi-crystalline nature of this thermoplastic polymer. Indeed, according to the XRD diffrattogram shown in
Figure 4a, the polymer contains an amorphous fraction, which causes the presence of a broad diffuse halo in the diffractogram, and a crystalline part, which causes the presence of three main peaks overlapped to the diffuse halo. These peaks are located at 2θ values of 16.5°, 22.4°, and 25.8°; such diffraction peak positioning is in good accordance with literature information [
16]. The FWHM of diffraction peaks is inversely proportional to the crystallite size (Scherrer equation [
16]); consequently, the observed broad diffraction peaks suggest the presence of very small sized crystallites in the polymer, which justifies to observed high optical transparence in the visible spectral region.
The semi-crystalline nature of the ultrathin PET films has been further confirmed by Differential Scanning Calorimetry (DSC). According to the DSC-thermogram shown in
Figure 4b, the polymer presents an endothermic signal due to the melting of the crystalline phase and such melting signal appears above the glass transition temperature (T
g), which is located at ca. 80.91°C. The PET crystallites melting signal has an onset temperature at ca. 240°C and minimum at 255.59°C. The DSC characterization of the polymer allows to measure the melting enthalpy, which resulted of ca. 40.30J/g. The calorimetric crystallinity degree, X
C, of the polymeric sample can be determined by comparing the specimen melting enthalpy with the theoretical melting enthalpy of an hypothetical fully crystalline PET sample (ca. 140J/g [
17]) and such ratio resulted of ca. 28.8%. This crystallinity value agrees with the value characteristic of bi-axially oriented PET films [
18]. Therefore, the small E
U value experimentally found for the PET sample by the Urbach approach seems to be completely justified by its semi-crystalline nature.
The same type of analysis (Urbach plot) has been applied to a slightly mechanically deformed (manually stretched) ultrathin PET film (see
Figure 5a,b). Although, mechanical deformation had the effect to modify the film thickness, the resulting Urbach energy value (E
U=45.2meV, with R
2=0.9933) remained practically unchanged (see
Figure 5b). Therefore, structural disorder did not increase appreciably in this system as a result of the applied stress.
Ultraviolet radiation interacts with dielectric polymers causing photoexcitation (i.e., optical absorption due to electronic transitions). For some polymers like Kapton-H and polyetherimide (PEI), even visible photons may cause photoexcitations [
19]. The cutoff wavelength has been observed for these ultrathin PET films at ca. 300nm. Consequently, photons with a wavelength lower than 300nm are completely absorbed by the film, while transparency is close to 87.4% at wavelengths higher than 300nm. The type of photoexcitation model and bandgap energy value (E
g) can be established optically by using the Tauc plot method. E
g is an important parameter for polymers since it is related to other physical properties like for example the permittivity (E
g is inversely proportional to material permittivity, which is the capability of electric field instauration in a material and corresponds to the material polarizability [
7]). In addition, accurate E
g determination allows dielectric polymer identifying (however, identification is not possible with conjugated polymers like polyacetylene because of the E
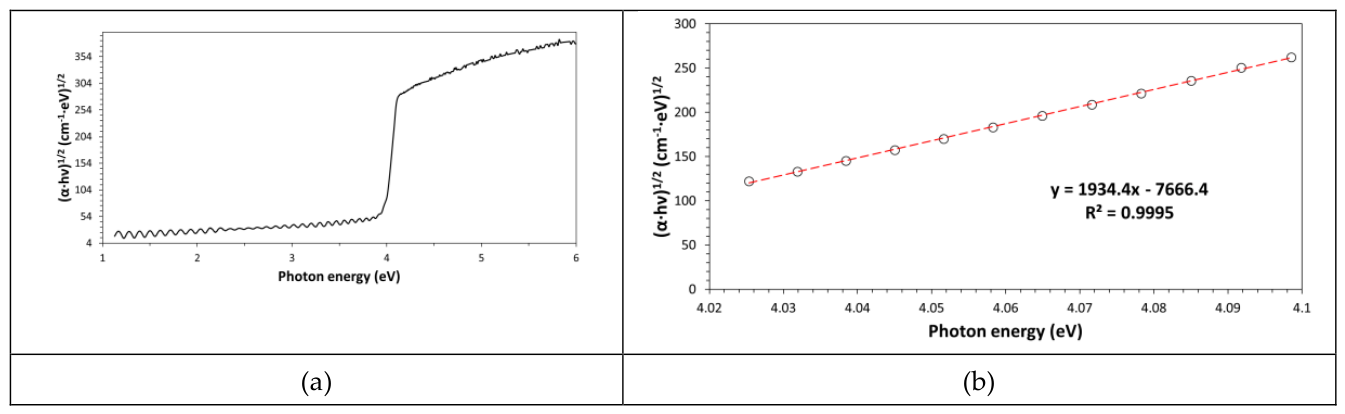
g dependence on the conjugation extension and therefore on molecular weight). The bandgap energy has been easily and accurately calculated by using the Tauc plot (see
Figure 6a,b), which is a special graphical representation obtained by graphing the quantity (α·E)
n as a function of the photon energy, E. The n value must be selected to allow the linearization of the (α·E)
n function and n is 0.5 for an indirect electron transition model and 2 for a direct electron transition model (both models are for allowed electron transitions). Depending on the most convenient value found for n (the model with best correlation factor), the type of electron transition model can be established. According to the following equation:
the value of the intercept with the energy axis corresponds to E
g (indeed, for (α·E)
n = 0, it results E = E
g). The intercept value is obtained from the best fitting equation of the linear portion of curve by using the following expression: -intercept/slope. In our case, an E
g value of 3.96eV (with R
2=0.9995) has been found for an indirect allowed electron transition model (n=0.5), which perfectly agrees with information provided in the literature [
20,
21].
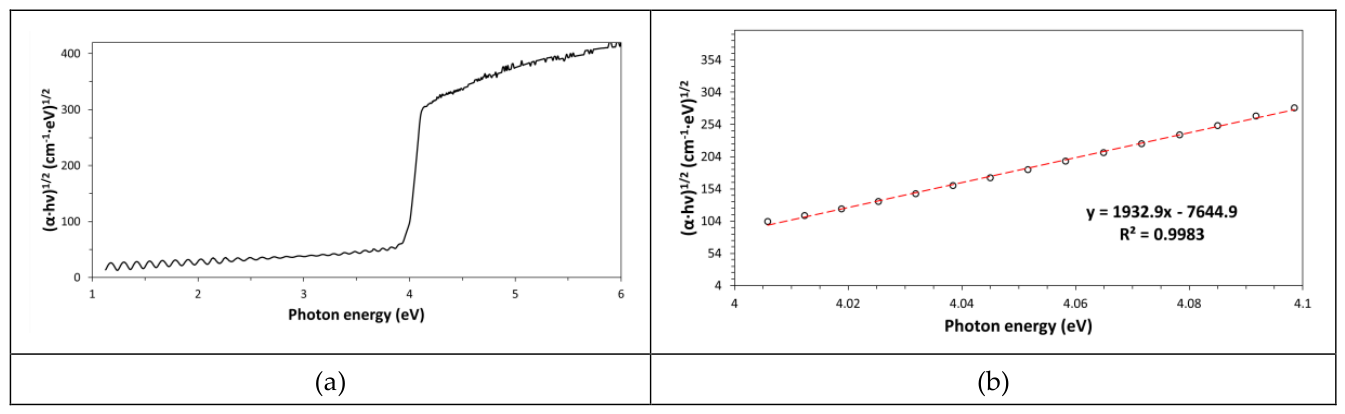
Manual stretching allowed simulating mild uniaxial mechanical stresses that polymeric films typically suffer in service or during industrial uses. This investigation has shown that E
g is a polymer physical characteristic, which remains practically unchanged after mild mechanical damage undergone by this material (see
Figure 7a,b). Indeed, E
g depends mainly on atom types (composition) and the way atoms are chemically bonded together (constitution) in the repeating unit.
According to the achieved experimental results, dielectric polymers at solid-state have a band structure principally depending on the polymer chemical composition/constitution and only marginally on crystallinity, molecular weight and texture (i.e., fibrous/spherulitic morphology). Such behavior allows using the characteristic bandgap energy numerical value to establish polymer nature. Since the very dawn of analytical chemistry, the identification of inorganic compounds (e.g., elemental metals, metal salts) has been based on recognizing the electronic transitions (emission or absorption lines) involving frontier orbitals, that is the valence shell orbitals (the Lewis electron shell). One need only think about the classical ‘flame-tests’ used by chemists more than one hundred years ago, or the early quartz-prism spectroscopes (Kirchhoff-Bunsen spectroscope), now replaced by Atomic Absorption Spectroscopy (AAS). The yellow color imparted by sodium to the flame, the violet of potassium, the red of calcium, and so on, are well-known phenomena. Thermally excited metals emit radiations that they are capable of absorbing too in the analysis by spectroscopes. Similarly, the precise assignment of the energy of the optical absorption edge (HOMO-LUMO transition energy, Eg) of dielectric polymers by UV-Vis spectrometric analysis can enable their reliable identification. This concept cannot be applied to inorganic semiconductors (consider, for instance, how silicon is used - through doping - to fabricate multiple types of semiconductor devices), nor to conductive/semiconductive polymers, due to their electronic configuration being dependent on the extent of conjugation (i.e., Eg = f(Mw)); however, it works perfectly with dielectric polymers.
This chemical analysis approach can be conveniently adopted in fields like microplastics since these types of waste belong to only few, very different plastic classes that can be easily and with safety distinguished on the basis of the Eg parameter (identifying microplastics is an important aspect, since it allows adequate selection of suitable recycling/remediation processes). UV-Vis spectrophotometers could be very convenient in this research area because they have a very small beam spot and therefore sampling requires only little polymer amounts. Furthermore, these devices usually can have small size, lightweight, and are easily transportable; they are structurally simple and consequently inexpensive, mechanically robust (diode-array devices have no moving parts) and capable of resisting in various environmental conditions. Therefore, optical spectrophotometers are suitable for building field/offshore laboratories to be placed, for example, on boats, on beaches, in landfills, etc. Yet, the chemical analysis of microplastics can be conducted directly in water (aqueous suspensions of microplastics) since this type of molecule is perfectly transparent to visible and ultraviolet light (up to approximately 100 nm).