Submitted:
14 April 2026
Posted:
16 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
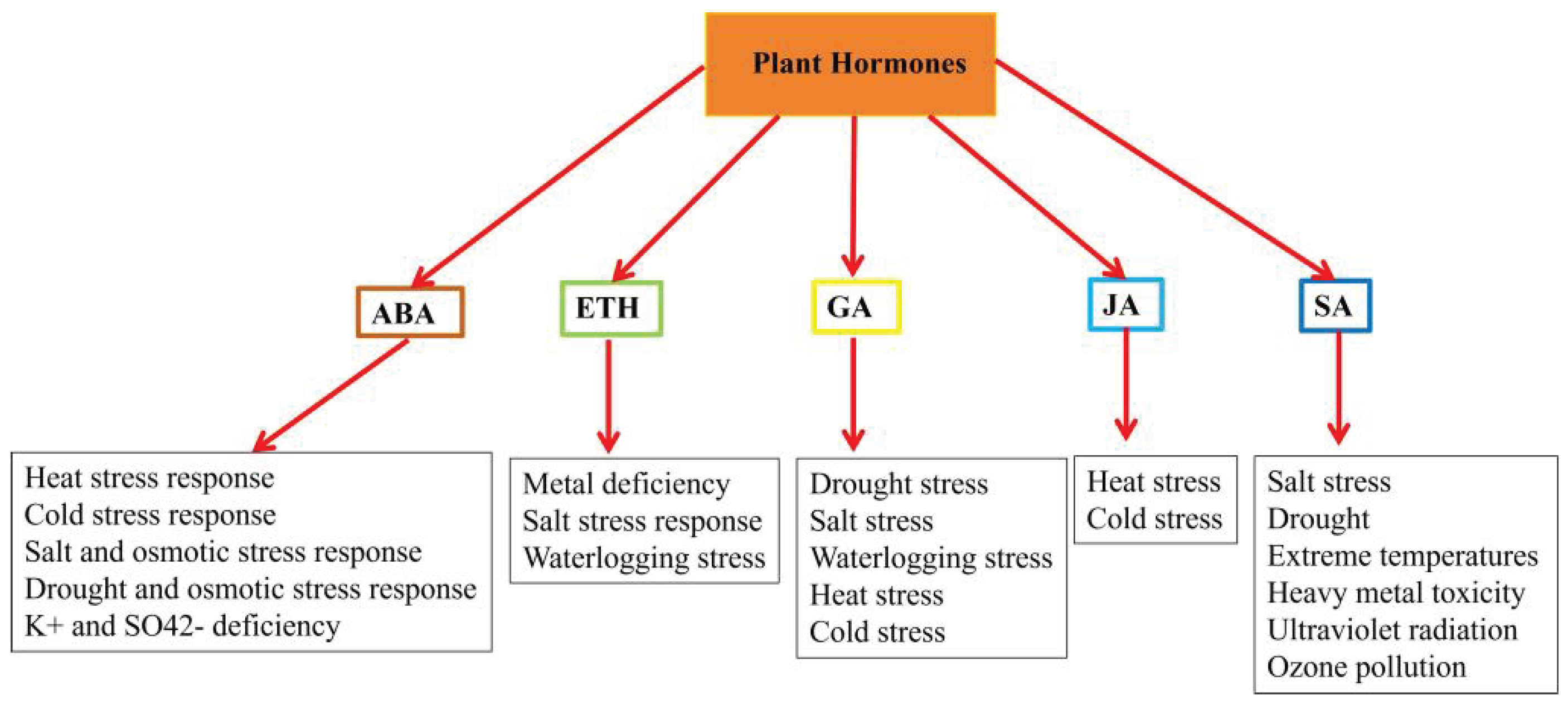
2. Mechanisms of Phytohormones Resistance to Abiotic Stress in Plants
2.1. The Role of ABA in Plant Responses to Abiotic Stress
2.2. The Role of ETH in Plant Responses to Abiotic Stress
2.3. The Role of GA in Plant Responses to Abiotic Stress
2.4. The Role of JA in Plant Responses to Abiotic Stress
2.5. The Role of SA in Plant Responses to Abiotic Stress
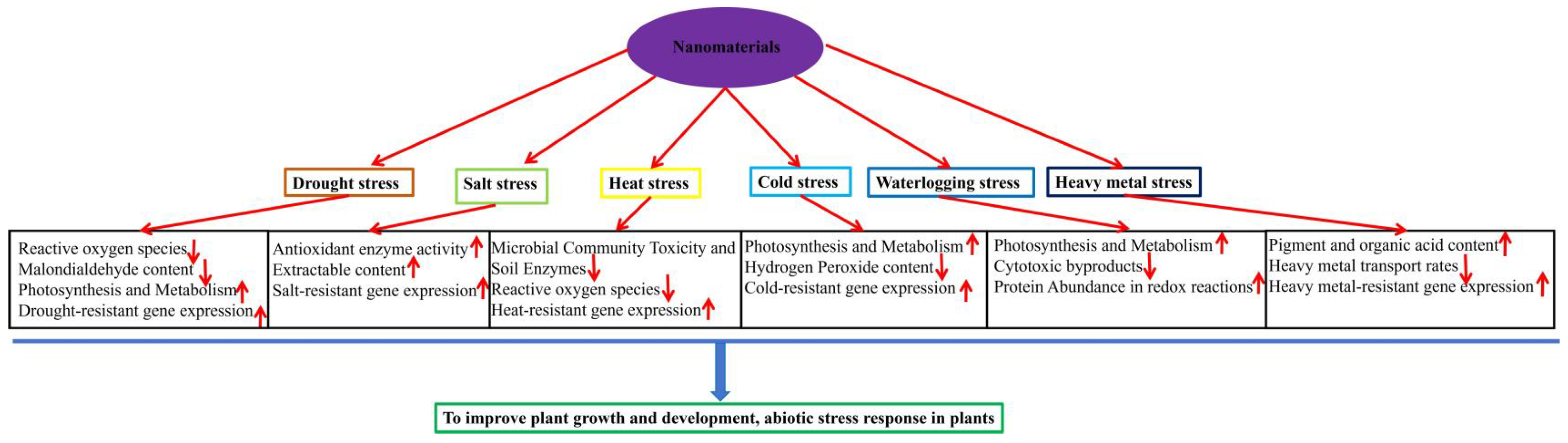
3. Mechanisms of Nanomaterials Resistance to Abiotic Stress in Plants
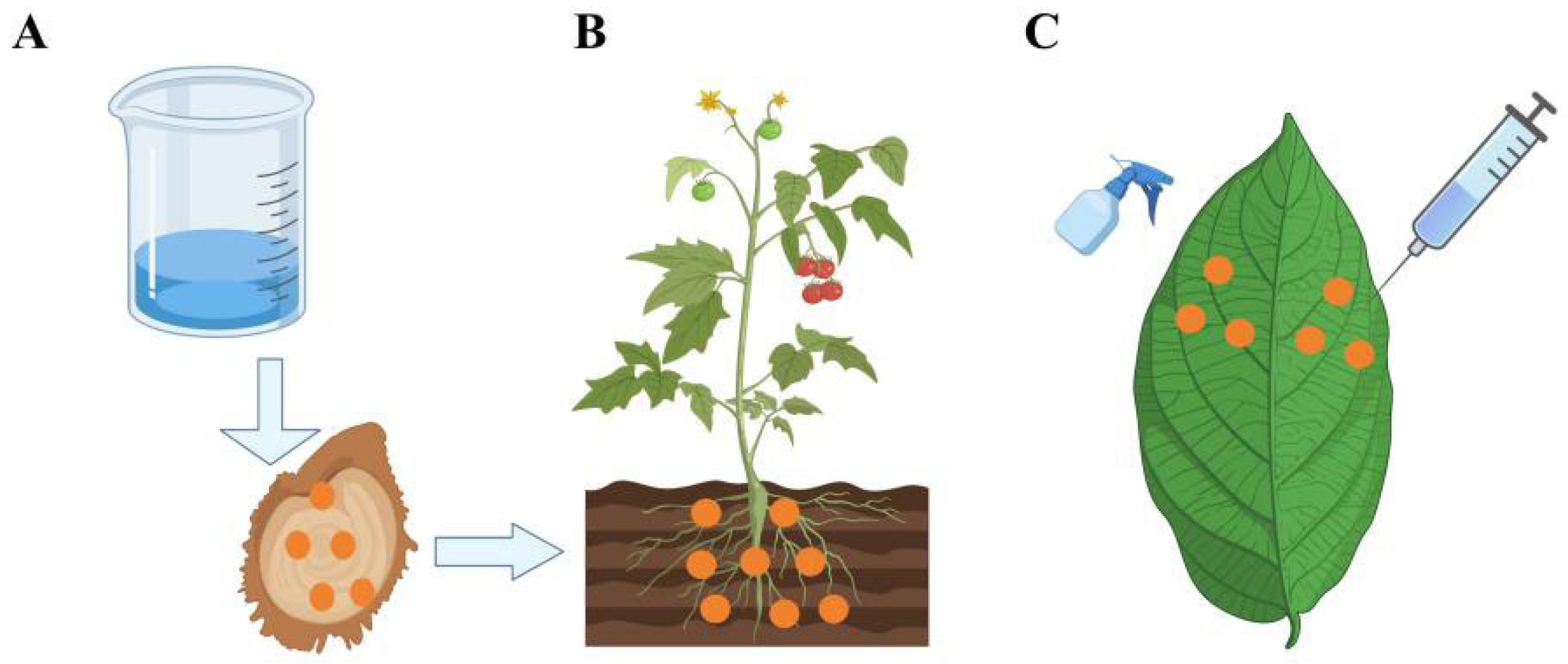
3.1. Plants Absorb Nanomaterials Through Their Root Systems
3.2. Plants Absorb Nanomaterials Through Their Leaves
3.3. Mechanisms of Which Nanomaterials Enhance Plant Tolerance to Abiotic Stress
3.3.1. The Role of Nanomaterials Under Drought Stress
3.3.2. The Role of Nanomaterials Under Temperature Stress
3.3.3. The Role of Nanomaterials Under Salt Stress
3.3.4. The Role of Nanomaterials Under Heavy Metals Stress
4. Conclusions and Prospects
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bari, R.; Jones, J.D. Role of plant hormones in plant defence responses. Plant Mol Biol. 2009, 69, 473–88. [Google Scholar] [CrossRef]
- Nakashima, K.; Yamaguchi-Shinozaki, K. ABA signaling in stress-response and seed development. Plant Cell Rep. 2013, 32, 959–70. [Google Scholar] [CrossRef]
- Lata, C.; Prasad, M. Role of DREBs in regulation of abiotic stress responses in plants. J Exp Bot. 2011, 62, 4731–48. [Google Scholar] [CrossRef]
- Navarro, L.; Bari, R.; Achard, P.; Lisón, P.; Nemri, A.; Harberd, N.P.; Jones, J.D. DELLAs control plant immune responses by modulating the balance of jasmonic acid and salicylic acid signaling. Curr Biol. 2008, 18, 650–5. [Google Scholar] [CrossRef]
- Nishiyama, R.; Watanabe, Y.; Leyva-Gonzalez, M.A.; Ha, C.V.; Fujita, Y.; Tanaka, M.; Seki, M.; Yamaguchi-Shinozaki, K.; Shinozaki, K.; Herrera-Estrella, L.; Tran, L.S. Arabidopsis AHP2, AHP3, and AHP5 histidine phosphotransfer proteins function as redundant negative regulators of drought stress response. Proc Natl Acad Sci U S A 2013, 110, 4840–5. [Google Scholar] [CrossRef]
- Verma, V.; Ravindran, P.; Kumar, P.P. Plant hormone-mediated regulation of stress responses. BMC Plant Biol. 2016, 16, 86. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Hu, L.; Zhong, Y. Structure, evolution, and roles of MYB transcription factors proteins in secondary metabolite biosynthetic pathways and abiotic stresses responses in plants: a comprehensive review. Front Plant Sci. 2025, 16, 1626844. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Wu, T.; Huang, K.; Jin, Y.M.; Li, Z.; Chen, M.; Yun, S.; Zhang, H.; Yang, X.; Chen, H.; Bai, H.; Du, L.; Ju, S.; Guo, L.; Bian, M.; Hu, L.; Du, X.; Jiang, W. A Novel AP2/ERF Transcription Factor, OsRPH1, Negatively Regulates Plant Height in Rice. Front Plant Sci. 2020, 11, 709. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Jin, Y.M.; Wu, T.; Hu, L.; Zhang, Y.; Jiang, W.; Du, X. OsDREB2B, an AP2/ERF transcription factor, negatively regulates plant height by conferring GA metabolism in rice. Front Plant Sci. 2022, 13, 1007811. [Google Scholar] [CrossRef]
- Waadt, R.; Seller, C.A.; Hsu, P.K.; Takahashi, Y.; Munemasa, S.; Schroeder, J.I. Plant hormone regulation of abiotic stress responses. Nat Rev Mol Cell Biol. 2022, 23, 680–694. [Google Scholar] [CrossRef]
- Chakraborty, R.; Rehman, R.U.; Siddiqui, M.W.; Liu, H.; Seth, C.S. Phytohormones: Heart of plants' signaling network under biotic, abiotic, and climate change stresses. Plant Physiol Biochem. 2025, 223, 109839. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Ma, Z.; Hu, L.; Huang, K.; Zhang, M.; Zhang, S.; Jiang, W.; Wu, T.; Du, X. Involvement of rice transcription factor OsERF19 in response to ABA and salt stress responses. Plant Physiol Biochem. 2021, 167, 22–30. [Google Scholar] [CrossRef]
- Ma, Z.; Hu, L. MicroRNA: A Dynamic Player from Signalling to Abiotic Tolerance in Plants. Int J Mol Sci. 2023, 24, 11364. [Google Scholar] [CrossRef]
- Ma, Z.; Hu, L.; Jiang, W. Understanding AP2/ERF Transcription Factor Responses and Tolerance to Various Abiotic Stresses in Plants: A Comprehensive Review. Int J Mol Sci. 2024, 25, 893. [Google Scholar] [CrossRef]
- Ma, Z.; Hu, L. WRKY Transcription Factor Responses and Tolerance to Abiotic Stresses in Plants. Int J Mol Sci. 2024, 25, 6845. [Google Scholar] [CrossRef]
- Pérez-Llorca, M.; Pollmann, S.; Müller, M. Ethylene and Jasmonates Signaling Network Mediating Secondary Metabolites under Abiotic Stress. Int J Mol Sci. 2023, 24, 5990. [Google Scholar] [CrossRef]
- Xie, Z.; Nolan, T.M.; Jiang, H.; Yin, Y. AP2/ERF Transcription Factor Regulatory Networks in Hormone and Abiotic Stress Responses in Arabidopsis. Front Plant Sci. 2019, 10, 228. [Google Scholar] [CrossRef]
- Yang, X.; Jia, Z.; Pu, Q.; Tian, Y.; Zhu, F.; Liu, Y. ABA Mediates Plant Development and Abiotic Stress via Alternative Splicing. Int J Mol Sci. 2022, 23, 3796. [Google Scholar] [CrossRef]
- Bianchetti, R.; Ali, A.; Gururani, M. Abscisic acid and ethylene coordinating fruit ripening under abiotic stress. Plant Sci. 2024, 349, 112243. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.A.; Asaf, S.; Ahmad, W.; Jan, R.; Bilal, S.; Khan, I.; Khan, A.L.; Kim, K.M.; Al-Harrasi, A. Diversity, Lifestyle, Genomics, and Their Functional Role of Cochliobolus, Bipolaris, and Curvularia Species in Environmental Remediation and Plant Growth Promotion under Biotic and Abiotic Stressors. J Fungi (Basel) 2023, 9, 254. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Song, L.; Gong, X.; Xu, J.; Li, M. Functions of Jasmonic Acid in Plant Regulation and Response to Abiotic Stress. Int J Mol Sci. 2020, 21, 1446. [Google Scholar] [CrossRef] [PubMed]
- Nadarajah, K.; Abdul, H.N.W.; Abdul, R.N.S.N. SA-Mediated Regulation and Control of Abiotic Stress Tolerance in Rice. Int J Mol Sci. 2021, 22, 5591. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Lu, L.; Wang, A.; Zhang, H.; Huang, M.; Wu, H.; Xing, B.; Wang, Z.; Ji, R. Nano-Biotechnology in Agriculture: Use of Nanomaterials to Promote Plant Growth and Stress Tolerance. J Agric Food Chem. 2020, 68, 1935–1947. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Bai, T.; Wei, H.; Gardea-Torresdey, J.L.; Keller, A.; White, J.C. Nanobiotechnology-based strategies for enhanced crop stress resilience. Nat Food 2022, 3, 829–836. [Google Scholar] [CrossRef]
- Zhou, H.; Wu, H.; Zhang, F.; Su, Y.; Guan, W.; Xie, Y.; Shen, W. Molecular basis of cerium oxide nanoparticle enhancement of rice salt tolerance and yield. Environmental Science: Nano 2021, 8, 3294–3311. [Google Scholar] [CrossRef]
- Arora, S.; Murmu, G.; Mukherjee, K.; Saha, S.; Maity, D. A comprehensive overview of nanotechnology in sustainable agriculture. J Biotechnol. 2022, 355, 21–41. [Google Scholar] [CrossRef]
- Auffan, M.; Rose, J.; Bottero, J.Y.; Lowry, G.V.; Jolivet, J.P.; Wiesner, M.R. Towards a definition of inorganic nanoparticles from an environmental, health and safety perspective. Nat Nanotechnol. 2009, 4, 634–41. [Google Scholar] [CrossRef]
- Pramanik, B.; Sar, P.; Bharti, R.; Gupta, R.K.; Purkayastha, S.; Sinha, S.; Chattaraj, S.; Mitra, D. Multifactorial role of nanoparticles in alleviating environmental stresses for sustainable crop production and protection. Plant Physiol Biochem. 2023, 201, 107831. [Google Scholar] [CrossRef]
- Paulami, D.A.M.; Paret, M.L.; Mondal, R.; Mandal, A.K. Advancement of noble metallic nanoparticles in agriculture: a promising future. Pedosphere 2023, 33, 116–128. [Google Scholar]
- Shaw, D.S.; Honeychurch, K.C. Nanosensor Applications in Plant Science. Biosensors (Basel) 2022, 12, 675. [Google Scholar] [CrossRef]
- Gao, L.; Zhuang, J.; Nie, L.; Zhang, J.; Zhang, Y.; Gu, N.; Wang, T.; Feng, J.; Yang, D.; Perrett, S.; Yan, X. Intrinsic peroxidase-like activity of ferromagnetic nanoparticles. Nat Nanotechnol. 2007, 2, 577–83. [Google Scholar] [CrossRef]
- Huang, Y.; Ren, J.; Qu, X. Nanozymes: Classification, Catalytic Mechanisms, Activity Regulation, and Applications. Chem Rev. 2019, 119, 4357–4412. [Google Scholar] [CrossRef]
- Zhang, R.; Yan, X.; Fan, K. Nanozymes inspired by natural enzymes. Accounts of Materials Research 2021, 2, 534–547. [Google Scholar] [CrossRef]
- Xu, Z.; Zhang, T.; Xu, Z.; Ma, Y.; Niu, Z.; Chen, J.; Zhang, M.; Shi, F. Research Progress and Prospects of Nanozymes in Alleviating Abiotic Stress of Crops. J Agric Food Chem. 2025, 73, 8694–8714. [Google Scholar] [CrossRef] [PubMed]
- Imtiaz, H.; Shiraz, M.; Mir, A.R.; Siddiqui, H.; Hayat, S. Nano-priming techniques for plant physio-biochemistry and stress tolerance. Journal of Plant Growth Regulation 2023, 42, 6870–6890. [Google Scholar] [CrossRef]
- Imran, O.I. Nano biotechnology: exploring the potential of nano materials in enhancing crop health and combating abiotic stress. Communications in Soil Science and Plant Analysis 2024, 55, 2509–2528. [Google Scholar] [CrossRef]
- Adeleke, B.S.; Akinola, S.A.; Adedayo, A.A.; Glick, B.R.; Babalola, O.O. Synergistic relationship of endophyte-nanomaterials to alleviate abiotic stress in plants. Frontiers in Environmental Science 2022, 10, 1015897. [Google Scholar] [CrossRef]
- Khalid, F.; Asif, K.; Rasheed, Y.; Ashraf, H.; Maqsood, M. F.; Rana, S. Nano priming for boosting growth and resilience in crops under abiotic stresses. Biocatal Agric Biotechnol. 2023, 53, 102892. [Google Scholar] [CrossRef]
- Wu, H.; Tito, N.; Giraldo, J.P. Anionic Cerium Oxide Nanoparticles Protect Plant Photosynthesis from Abiotic Stress by Scavenging Reactive Oxygen Species. ACS Nano 2017, 11, 11283–11297. [Google Scholar] [CrossRef]
- Khan, M.N.; Li, Y.; Fu, C.; Hu, J.; Chen, L.; Yan, J.; Khan, Z.; Wu, H.; Li, Z. CeO2 Nanoparticles Seed Priming Increases Salicylic Acid Level and ROS Scavenging Ability to Improve Rapeseed Salt Tolerance. Glob Chall. 2022, 6, 2200025. [Google Scholar] [CrossRef]
- Mittler, R.; Zandalinas, S.I.; Fichman, Y.; Van, B.F. Reactive oxygen species signalling in plant stress responses. Nat Rev Mol Cell Biol. 2022, 23, 663–679. [Google Scholar] [CrossRef]
- Banerjee, A.; Roychoudhury, A. Maghemite nano-fertilization promotes fluoride tolerance in rice by restoring grain yield and modulating the ionome and physiome. Ecotoxicol Environ Saf. 2021, 215, 112055. [Google Scholar] [CrossRef]
- Xu, L.; Zhu, Z.; Sun, D.W. Bioinspired Nanomodification Strategies: Moving from Chemical-Based Agrosystems to Sustainable Agriculture. ACS Nano 2021, 15, 12655–12686. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xu, J.; Li, R.; Ge, Y.; Li, Y.; Li, R. Plants' Response to Abiotic Stress: Mechanisms and Strategies. Int J Mol Sci. 2023, 24, 10915. [Google Scholar] [CrossRef] [PubMed]
- Handayani, T.; Watanabe, K. The combination of drought and heat stress has a greater effect on potato plants than single stresses. Plant, Soil & Environment 2020, 66. [Google Scholar]
- Gupta, B.; Shrestha, J. Editorial: Abiotic stress adaptation and tolerance mechanisms in crop plants. Front Plant Sci. 2023, 14, 1278895. [Google Scholar] [CrossRef]
- Ndikuryayo, C.; Ndayiragije, A.; Kilasi, N.; et al. Breeding for rice aroma and drought tolerance: a review. Agronomy 2022, 12, 1726. [Google Scholar] [CrossRef]
- Wan, Q.; Hongbo, S.; Zhaolong, X.; Jia, L.; Dayong, Z.; Yihong, H. Salinity Tolerance Mechanism of Osmotin and Osmotin-like Proteins: A Promising Candidate for Enhancing Plant Salt Tolerance. Curr Genomics 2017, 18, 553–556. [Google Scholar] [CrossRef]
- Almeida, D.M.; Oliveira, M.M.; Saibo, N.J.M. Regulation of Na+ and K+ homeostasis in plants: towards improved salt stress tolerance in crop plants. Genet Mol Biol. 2017, 40, 326–345. [Google Scholar] [CrossRef]
- Ali, A.; Maggio, A.; Bressan, R.A.; Yun, D.J. Role and Functional Differences of HKT1-Type Transporters in Plants under Salt Stress. Int J Mol Sci. 2019, 20, 1059. [Google Scholar] [CrossRef]
- Napieraj, N.; Reda, M.G.; Janicka, M.G. The role of NO in plant response to salt stress: interactions with polyamines. Funct Plant Biol. 2020, 47, 865–879. [Google Scholar] [CrossRef]
- Jin, X.; Long, Y.; Xiong, S.; Yang, Z.; Chen, W.; Hawar, A.; Sun, B. SbNAC2 enhances abiotic stress tolerance by upregulating ROS scavenging activities and inducing stress-response genes in sorghum. Environmental and Experimental Botany 2021, 192, 104664. [Google Scholar] [CrossRef]
- Luo, P.; Chen, L.; Chen, Y.; Shen, Y.; Cui, Y. RmZAT10, a novel Cys2/His2 zinc finger transcription factor of Rosa multiflora, functions in cold tolerance through modulation of proline biosynthesis and ROS homeostasis. Environmental and Experimental Botany 2022, 198, 104845. [Google Scholar] [CrossRef]
- Kurosawa, E. Experimental studies on the nature of the substance secreted by the “bakanae”fungus. Nat Hist Soc Formosa 1926, 16, 213–227. [Google Scholar]
- Rankenberg, T.; Geldhof, B.; van, V.H.; Holsteens, K.; Van de, P.B.; Sasidharan, R. Age-Dependent Abiotic Stress Resilience in Plants. Trends Plant Sci. 2021, 26, 692–705. [Google Scholar] [CrossRef]
- Mukherjee, A.; Dwivedi, S.; Bhagavatula, L.; Datta, S. Integration of light and ABA signaling pathways to combat drought stress in plants. Plant Cell Rep. 2023, 42, 829–841. [Google Scholar] [CrossRef]
- Zha, D.; He, Y.; Song, J. Regulatory role of ABA-responsive element binding factors in plant abiotic stress response. Physiol Plant. 2025, 177, e70233. [Google Scholar] [CrossRef]
- Li, Q.; Zhu, P.; Yu, X.; Xu, J.; Liu, G. Physiological and Molecular Mechanisms of Rice Tolerance to Salt and Drought Stress: Advances and Future Directions. Int J Mol Sci. 2024, 25, 9404. [Google Scholar] [CrossRef] [PubMed]
- Perin, E.C.; da Silva, M.R.; Borowski, J.M.; Crizel, R.L.; Schott, I.B.; Carvalho, I.R.; Rombaldi, C.V.; Galli, V. ABA-dependent salt and drought stress improve strawberry fruit quality. Food Chem. 2019, 271, 516–526. [Google Scholar] [CrossRef]
- Zhu, J.K. Salt and drought stress signal transduction in plants. Annu Rev Plant Biol. 2002, 53, 247–73. [Google Scholar] [CrossRef]
- Lim, C.; Kang, K.; Shim, Y.; Yoo, S.C.; Paek, N.C. Inactivating transcription factor OsWRKY5 enhances drought tolerance through abscisic acid signaling pathways. Plant Physiol. 2022, 188, 1900–1916. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Xu, H.; Su, C.; Wang, X.; Wang, L. Rice CIRCADIAN CLOCK ASSOCIATED 1 transcriptionally regulates ABA signaling to confer multiple abiotic stress tolerance. Plant Physiol. 2022, 190, 1057–1073. [Google Scholar] [CrossRef]
- Van, de.P.B.; de, V.J. Evolution of ethylene as an abiotic stress hormone in streptophytes. Environ Exp Bot. 2023, 214, 105456. [Google Scholar]
- Chen, H.; Bullock, D.A., Jr.; Alonso, J.M.; Stepanova, A.N. To Fight or to Grow: The Balancing Role of Ethylene in Plant Abiotic Stress Responses. Plants (Basel) 2021, 11, 33. [Google Scholar] [CrossRef]
- Djemal, R.; Khoudi, H. TdSHN1, a WIN1/SHN1-type transcription factor, imparts multiple abiotic stress tolerance in transgenic tobacco. Environ. Exp. Bot. 2016, 131, 89–100. [Google Scholar] [CrossRef]
- Cheng, M.C.; Liao, P.M.; Kuo, W.W.; Lin, T.P. The Arabidopsis ETHYLENE RESPONSE FACTOR1 regulates abiotic stress-responsive gene expression by binding to different cis-acting elements in response to different stress signals. Plant Physiol. 2013, 162, 1566–82. [Google Scholar] [CrossRef]
- An, J.P.; Zhang, X.W.; Xu, R.R.; You, C.X.; Wang, X.F.; Hao, Y.J. Apple MdERF4 negatively regulates salinity tolerance by inhibiting MdERF3 transcription. Plant Sci. 2018, 276, 181–188. [Google Scholar] [CrossRef]
- Sandhu, N.; Aggarwal, H.; Kumar, A.; Augustine, G.; Vishnoi, R.; Pandey, A.K.; Chauhan, H.; Chhuneja, P. Regulating Plant Architecture to Enhance the Future of Cereal Crop Production. Physiol Plant. 2025, 177, e70367. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wu, Y.; Dong, G.; Zhu, G.; Zhou, G. Progress of Research on the Physiology and Molecular Regulation of Sorghum Growth under Salt Stress by Gibberellin. Int J Mol Sci. 2023, 24, 6777. [Google Scholar] [CrossRef] [PubMed]
- Lu, K.; Guo, Z.; Di, S.; Lu, Y.; Muhammad, I.A.R.; Rong, C.; Ding, Y.; Li, W.; Ding, C. OsMFT1 Inhibits Seed Germination by Modulating Abscisic Acid Signaling and Gibberellin Biosynthesis under Salt Stress in Rice. Plant Cell Physiol. 2023, 64, 674–685. [Google Scholar] [CrossRef]
- Li, J.; Chen, Y.; Zhang, R.; Wu, B.; Xiao, G. Expression identification of three OsWRKY genes in response to abiotic stress and hormone treatments in rice. Plant Signal Behav. 2023, 18, 2292844. [Google Scholar] [CrossRef]
- Ruan, J.; Zhou, Y.; Zhou, M.; Yan, J.; Khurshid, M.; Weng, W.; Cheng, J.; Zhang, K. Jasmonic Acid Signaling Pathway in Plants. Int J Mol Sci. 2019, 20, 2479. [Google Scholar] [CrossRef]
- Wang, Y.; Mostafa, S.; Zeng, W.; Jin, B. Function and Mechanism of Jasmonic Acid in Plant Responses to Abiotic and Biotic Stresses. Int J Mol Sci. 2021, 22, 8568. [Google Scholar] [CrossRef]
- Kong, L.; Song, Q.; Wei, H.; Wang, Y.; Lin, M.; Sun, K.; Zhang, Y.; Yang, J.; Li, C.; Luo, K. The AP2/ERF transcription factor PtoERF15 confers drought tolerance via JA-mediated signaling in Populus. New Phytol. 2023, 240, 1848–1867. [Google Scholar] [CrossRef]
- Wang, X.; Li, J.; Guo, X.; Ma, Y.; Qiao, Q.; Guo, J. PlWRKY13: A Transcription Factor Involved in Abiotic and Biotic Stress Responses in Paeonia lactiflora. Int J Mol Sci. 2019, 20, 5953. [Google Scholar] [CrossRef] [PubMed]
- Prakash, V.; Singh, V.P.; Tripathi, D.K.; Sharma, S.; Corpas, F.J. Nitric oxide (NO) and salicylic acid (SA): A framework for their relationship in plant development under abiotic stress. Plant Biol (Stuttg) 2021, 1, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Jin, Y.; Yang, W. ERF3 represses SA pathway defense to modulate tradeoffs between biotic and abiotic stress responses in Arabidopsis. Plant Cell Rep. 2025, 45, 7. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Zhu, L.; Xu, L.; Guo, W.; Zhang, X. GhATAF1, a NAC transcription factor, confers abiotic and biotic stress responses by regulating phytohormonal signaling networks. Plant Cell Rep. 2016, 35, 2167–79. [Google Scholar] [CrossRef]
- Wu, J.; Su, Y.; Pan, Z.; Wang, Y.; Zhang, Y.; Li, L.; Jiang, J.; Cao, X. Identification of WRKY transcription factors in Ipomoea pes-caprae and functional role of IpWRKY16 in sweet potato salt stress response. BMC Plant Biol. 2024, 24, 1190. [Google Scholar] [CrossRef]
- Šola, I.; Poljuha, D.; Pavičić, I.; Jurinjak, T.A.; Šamec, D. Climate Change and Plant Foods: The Influence of Environmental Stressors on Plant Metabolites and Future Food Sources. Foods 2025, 14, 416. [Google Scholar] [CrossRef]
- Eckardt, N.A.; Ainsworth, E.A.; Bahuguna, R.N.; Broadley, M.R.; Busch, W.; Carpita, N.C.; Castrillo, G.; Chory, J.; DeHaan, L.R.; Duarte, C.M.; Henry, A.; Jagadish, S.V.K.; Langdale, J.A.; Leakey, A.D.B.; Liao, J.C.; Lu, K.J.; McCann, M.C.; McKay, J.K.; Odeny, D.A.; Jorge de, O.E.; Platten, J.D.; Rabbi, I.; Rim, E.Y.; Ronald, P.C.; Salt, D.E.; Shigenaga, A.M.; Wang, E.; Wolfe, M.; Zhang, X. Climate change challenges, plant science solutions. Plant Cell. 2023, 35, 24–66. [Google Scholar] [CrossRef]
- Imran, Q.M.; Falak, N.; Hussain, A.; et al. Abiotic stress in plants; Stress perception to molecular response and role of biotechnological tools in stress resistance. Agronomy 2021, 11, 1579. [Google Scholar] [CrossRef]
- Jiang, S.; Li, H.; Zhang, L.; Mu, W.; Zhang, Y.; Chen, T.; Wu, J.; Tang, H.; Zheng, S.; Liu, Y.; Wu, Y.; Luo, X.; Xie, Y.; Ren, J. Generic Diagramming Platform (GDP): a comprehensive database of high-quality biomedical graphics. Nucleic acids research 2025, 53, D1670–D1676. [Google Scholar] [CrossRef] [PubMed]
- Avellan, A.; Schwab, F.; Masion, A.; Chaurand, P.; Borschneck, D.; Vidal, V.; Rose, J.; Santaella, C.; Levard, C. Nanoparticle Uptake in Plants: Gold Nanomaterial Localized in Roots of Arabidopsis thaliana by X-ray Computed Nanotomography and Hyperspectral Imaging. Environ Sci Technol. 2017, 51, 8682–8691. [Google Scholar] [CrossRef]
- Dutta, P.; Kumari, A.; Mahanta, M.; Upamanya, G.K.; Heisnam, P.; Borua, S.; Kaman, P.K.; Mishra, A.K.; Mallik, M.; Muthukrishnan, G.; Sabarinathan, K.G.; Puzari, K.R.; Vijayreddy, D. Nanotechnological approaches for management of soil-borne plant pathogens. Front Plant Sci. 2023, 14, 1136233. [Google Scholar] [CrossRef]
- Vishwakarma, V.; Ogunkunle, C.O.; Rufai, A.B.; Okunlola, G.O.; Olatunji, O.A.; Jimoh, M.A. Nanoengineered particles for sustainable crop production: potentials and challenges. 3 Biotech. 2023, 13, 163. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.; Awan, S.A.; Rizwan, M.; Hassan, Z.U.; Akram, M.A.; Tariq, R.; Brestic, M.; Xie, W. Nanoparticle's uptake and translocation mechanisms in plants via seed priming, foliar treatment, and root exposure: a review. Environ Sci Pollut Res Int. 2022, 29, 89823–89833. [Google Scholar] [CrossRef] [PubMed]
- Butova, V.V.; Bauer, T.V.; Polyakov, V.A.; Minkina, T.M. Advances in nanoparticle and organic formulations for prolonged controlled release of auxins. Plant Physiol Biochem. 2023, 201, 107808. [Google Scholar] [CrossRef]
- Sabo-Attwood, T.; Unrine, J.M.; Stone, J.W.; Murphy, C.J.; Ghoshroy, S.; Blom, D.; Bertsch, P.M.; Newman, L.A. Uptake, distribution and toxicity of gold nanoparticles in tobacco (Nicotiana xanthi) seedlings. Nanotoxicology 2012, 6(4), 353–60. [Google Scholar] [CrossRef]
- Stolte Bezerra Lisboa Oliveira, L.; Ristroph, K.D. Critical Review: Uptake and Translocation of Organic Nanodelivery Vehicles in Plants. Environ Sci Technol. 2024, 58, 5646–5669. [Google Scholar] [CrossRef]
- Taylor, A.F.; Rylott, E.L.; Anderson, C.W.; Bruce, N.C. Investigating the toxicity, uptake, nanoparticle formation and genetic response of plants to gold. PLoS One 2014, 9, e93793. [Google Scholar] [CrossRef]
- Guleria, G.; Thakur, S.; Shandilya, M.; Sharma, S.; Thakur, S.; Kalia, S. Nanotechnology for sustainable agro-food systems: The need and role of nanoparticles in protecting plants and improving crop productivity. Plant Physiol Biochem. 2023, 194, 533–549. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, L.; Ma, C.; Wang, K.; Hao, Y.; Chen, Q.; Mo, Y.; Rui, Y. Effects of cerium oxide on rice seedlings as affected by co-exposure of cadmium and salt. Environ Pollut. 2019, 252, 1087–1096. [Google Scholar] [CrossRef]
- Kulikova, N.A.; Volkov, D.S.; Volikov, A.B.; et al. Silver nanoparticles stabilized by humic substances adversely affect wheat plants and soil. Journal of Nanoparticle Research 2020, 22, 100. [Google Scholar] [CrossRef]
- Liu, J.; Williams, P.C.; Goodson, B.M.; Geisler-Lee, J.; Fakharifar, M.; Gemeinhardt, M.E. TiO2 nanoparticles in irrigation water mitigate impacts of aged Ag nanoparticles on soil microorganisms, Arabidopsis thaliana plants, and Eisenia fetida earthworms. Environ Res. 2019, 172, 202–215. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Tito, N.; Giraldo, J.P. Anionic Cerium Oxide Nanoparticles Protect Plant Photosynthesis from Abiotic Stress by Scavenging Reactive Oxygen Species. ACS Nano 2017, 11, 11283–11297. [Google Scholar] [CrossRef]
- Hu, P.; An, J.; Faulkner, M.M.; Wu, H.; Li, Z.; Tian, X.; Giraldo, J.P. Nanoparticle Charge and Size Control Foliar Delivery Efficiency to Plant Cells and Organelles. ACS Nano 2020, 14, 7970–7986. [Google Scholar] [CrossRef]
- Ali, S.; Mehmood, A.; Khan, N. Uptake, translocation, and consequences of nanomaterials on plant growth and stress adaptation. Journal of Nanomaterials 2021, 2021, 6677616. [Google Scholar] [CrossRef]
- Sembada, A.A.; Lenggoro, I.W. Transport of Nanoparticles into Plants and Their Detection Methods. Nanomaterials (Basel) 2024, 14, 131. [Google Scholar] [CrossRef] [PubMed]
- Avellan, A.; Yun, J.; Zhang, Y.; Spielman-Sun, E.; Unrine, J.M.; Thieme, J.; Li, J.; Lombi, E.; Bland, G.; Lowry, G.V. Nanoparticle Size and Coating Chemistry Control Foliar Uptake Pathways, Translocation, and Leaf-to-Rhizosphere Transport in Wheat. ACS Nano 2019, 13, 5291–5305. [Google Scholar] [CrossRef]
- Zhu, J.K. Abiotic Stress Signaling and Responses in Plants. Cell. 2016, 167, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, A.M.; Waseem, M.; Jakada, B.H.; Okal, E.J.; Lei, Z.; Saqib, H.S.A.; Yuan, W.; Xu, W.; Zhang, Q. Mechanisms of Abscisic Acid-Mediated Drought Stress Responses in Plants. Int J Mol Sci. 2022, 23, 1084. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Yang, W.; Ma, J.; Cheng, Z.; Zhang, X.; Liu, X.; Wu, X.; Zhang, J. An Integrated Framework for Drought Stress in Plants. Int J Mol Sci. 2024, 25, 9347. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wu, H.; Wang, Y.; Ye, W.; Kong, X.; Yin, Z. Small particles, big effects: How nanoparticles can enhance plant growth in favorable and harsh conditions. J Integr Plant Biol. 2024, 66, 1274–1294. [Google Scholar] [CrossRef]
- Rezayian, M.; Niknam, V.; Arabloo, M. Iron nanoparticle regulate succinate dehydrogenase activity in canola plants under drought stress. Sci Rep. 2023, 13, 9628. [Google Scholar] [CrossRef]
- Shirvani-Naghani, S.; Fallah, S.; Pokhrel, L.R.; Rostamnejadi, A. Drought stress mitigation and improved yield in Glycine max through foliar application of zinc oxide nanoparticles. Sci Rep. 2024, 14, 27898. [Google Scholar] [CrossRef]
- Zhao, L.; Wang, W.; Fu, X.; Liu, A.; Cao, J.; Liu, J. Graphene Oxide, a Novel Nanomaterial as Soil Water Retention Agent, Dramatically Enhances Drought Stress Tolerance in Soybean Plants. Front Plant Sci. 2022, 13, 810905. [Google Scholar] [CrossRef]
- Ding, Y.; Yang, S. Surviving and thriving: How plants perceive and respond to temperature stress. Dev Cell. 2022, 57, 947–958. [Google Scholar] [CrossRef]
- Cvetkovska, M.; Vakulenko, G.; Smith, D.R.; Zhang, X.; Hüner, N.P.A. Temperature stress in psychrophilic green microalgae: Minireview. Physiol Plant. 2022, 174, e13811. [Google Scholar] [CrossRef]
- Guy, C.; Kaplan, F.; Kopka, J.; Selbig, J.; Hincha, D.K. Metabolomics of temperature stress. Physiol Plant. 2008, 132, 220–35. [Google Scholar] [CrossRef]
- Yadav, A.; Bhatia, A.; Bana, R.S.; Ranjan, R.; Dhakar, R.; Shivay, Y.S.; Meena, S.L.; Yadav, S.; Singh, T.; Raj, R.; Kumar, M.; Kumar, V.; Sharan, S.P.; Kumar, A. Green zinc oxide nanoparticles improve zinc bioavailability and mitigate high temperature stress in rice. Sci Rep. 2026, 16, 6573. [Google Scholar] [CrossRef]
- Mahmoudi, R.; Razavi, F.; Rabiei, V.; Gohari, G.; Palou, L. Application of Glycine betaine coated chitosan nanoparticles alleviate chilling injury and maintain quality of plum (Prunus domestica L.) fruit. Int J Biol Macromol. 2022, 207, 965–977. [Google Scholar] [CrossRef]
- Zhao, S.; Zhang, Q.; Liu, M.; Zhou, H.; Ma, C.; Wang, P. Regulation of Plant Responses to Salt Stress. Int J Mol Sci. 2021, 22, 4609. [Google Scholar] [CrossRef]
- Zhou, H.; Shi, H.; Yang, Y.; Feng, X.; Chen, X.; Xiao, F.; Lin, H.; Guo, Y. Insights into plant salt stress signaling and tolerance. J Genet Genomics 2024, 51, 16–34. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Guo, Y. Unraveling salt stress signaling in plants. J Integr Plant Biol. 2018, 60, 796–804. [Google Scholar] [CrossRef] [PubMed]
- Ghassemi-Golezani, K.; Farhangi-Abriz, S.; Abdoli, S. How can biochar-based metal oxide nanocomposites counter salt toxicity in plants? Environ Geochem Health 2021, 43, 2007–2023. [Google Scholar] [CrossRef] [PubMed]
- Sheikhalipour, M.; Mohammadi, S.A.; Esmaielpour, B.; Spanos, A.; Mahmoudi, R.; Mahdavinia, G.R.; Milani, M.H.; Kahnamoei, A.; Nouraein, M.; Antoniou, C.; Kulak, M.; Gohari, G.; Fotopoulos, V. Seedling nanopriming with selenium-chitosan nanoparticles mitigates the adverse effects of salt stress by inducing multiple defence pathways in bitter melon plants. Int J Biol Macromol. 2023, 242, 124923. [Google Scholar] [CrossRef]
- Guo, Z.; Gao, Y.; Yuan, X.; Yuan, M.; Huang, L.; Wang, S.; Liu, C.; Duan, C. Effects of Heavy Metals on Stomata in Plants: A Review. Int J Mol Sci. 2023, 24, 9302. [Google Scholar] [CrossRef]
- Asgher, M.; Rehaman, A.; Nazar, Ul; Islam, S.; Khan, N.A. Multifaceted roles of silicon nano particles in heavy metals-stressed plants. Environ Pollut. 2024, 341, 122886. [Google Scholar] [CrossRef]
- Panda, A.; Fatnani, D.; Parida, A.K. Uptake, impact, adaptive mechanisms, and phytoremediation of heavy metals by plants: Role of transporters in heavy metal sequestration. Plant Physiol Biochem. 2025, 221, 109578. [Google Scholar] [CrossRef]
- Wang, S.; Fu, Y.; Zheng, S.; Xu, Y.; Sun, Y. Phytotoxicity and Accumulation of Copper-Based Nanoparticles in Brassica under Cadmium Stress. Nanomaterials (Basel) 2022, 12, 1497. [Google Scholar] [CrossRef]
- Yan, G.; Jin, H.; Yin, C.; Hua, Y.; Huang, Q.; Zhou, G.; Xu, Y.; He, Y.; Liang, Y.; Zhu, Z. Comparative effects of silicon and silicon nanoparticles on the antioxidant system and cadmium uptake in tomato under cadmium stress. Sci Total Environ. 2023, 904, 166819. [Google Scholar] [CrossRef]
- Khan, T.A.; Kappachery, S.; Karumannil, S.; AlHosani, M.; Almansoori, N.; Almansoori, H.; Yusuf, M.; Tran, L.P.; Gururani, M.A. Brassinosteroid Signaling Pathways: Insights into Plant Responses under Abiotic Stress. Int J Mol Sci. 2023, 24, 17246. [Google Scholar] [CrossRef]
- Wang, C.; Qu, K.; Wang, J.; Qin, R.; Li, B.; Qiu, J.; Wang, G. Biomechanical regulation of planar cell polarity in endothelial cells. Biochim Biophys Acta Mol Basis Dis. 2022, 1868, 166495. [Google Scholar] [CrossRef]
- Mahakham, W.; Sarmah, A.K.; Maensiri, S.; Theerakulpisut, P. Nanopriming technology for enhancing germination and starch metabolism of aged rice seeds using phytosynthesized silver nanoparticles. Sci Rep. 2017, 7, 8263. [Google Scholar] [CrossRef]
- Mazhar, M.W.; Ishtiaq, M.; Maqbool, M. Seed priming with calcium oxide nanoparticles improves germination, biomass, antioxidant defence and yield traits of canola plants under drought stress. South African Journal of Botany 2022, 151, 889–899. [Google Scholar] [CrossRef]
- Omar, A.A.; Heikal, Y.M.; Zayed, E.M.; Shamseldin, S.A.M.; Salama, Y.E.; Amer, K.E.; Basuoni, M.M.; Abd, E.S.; Mohamed, A.H. Conferring of Drought and Heat Stress Tolerance in Wheat (Triticum aestivum L.) Genotypes and Their Response to Selenium Nanoparticles Application. Nanomaterials (Basel) 2023, 13, 998. [Google Scholar] [CrossRef] [PubMed]
- Zahedi, S.M.; Hosseini, M.S.; Daneshvar, H.M.N.; Peijnenburg, W. Mitigation of the effect of drought on growth and yield of pomegranates by foliar spraying of different sizes of selenium nanoparticles. J Sci Food Agric. 2021, 101, 5202–5213. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.Y.; Song, F.B.; Li, X.N.; et al. Nano-ZnO alleviates drought stress via modulating plant water use and carbohydrate metabolism in maize. Archives of Agronomy and Soil Science 2021, 67, 245–259. [Google Scholar] [CrossRef]
- Iqbal, M.; Raja, N.I.; Mashwani, Z.; et al. Effect of silver nanoparticles on growth of wheat under heat stress. Iranian Journal of Science and Technology. Transactions A: Science 2019, 43, 387–395. [Google Scholar] [CrossRef]
- Mahmoud, N.E.; Abdelhameed, R.M. Use of titanium dioxide doped multi-wall carbon nanotubes as promoter for the growth, biochemical indices of Sesamum indicum L. under heat stress conditions. Plant Physiol Biochem. 2023, 201, 107844. [Google Scholar] [CrossRef]
- Hasanpour, H.; Maali-Amir, R.; Zeinali, H. Effect of TiO2 nanoparticles on metabolic limitations to photosynthesis under cold in chickpea. Russian Journal of Plant Physiology 2015, 62, 779–787. [Google Scholar] [CrossRef]
- Mogazy, A.M.; Hanafy, R.S. Foliar spray of biosynthesized zinc oxide nanoparticles alleviate salinity stress effect on Vicia faba plants. Journal of Soil Science and Plant Nutrition 2022, 22, 2647–2662. [Google Scholar] [CrossRef]
- Lalarukh, I.; Zahra, N.; Al, H.A.A.; et al. Exogenously applied ZnO nanoparticles induced salt tolerance in potentially high yielding modern wheat (Triticum aestivum L.) cultivars. Environmental Technology & Innovation 2022, 27, 102799. [Google Scholar] [CrossRef]
- Liu, J.; Li, G.; Chen, L.; Gu, J.; Wu, H.; Li, Z. Cerium oxide nanoparticles improve cotton salt tolerance by enabling better ability to maintain cytosolic K+/Na+ ratio. J Nanobiotechnology 2021, 19, 153. [Google Scholar] [CrossRef] [PubMed]
- Rossi, L.; Zhang, W.; Ma, X. Cerium oxide nanoparticles alter the salt stress tolerance of Brassica napus L. by modifying the formation of root apoplastic barriers. Environ Pollut. 2017, 229, 132–138. [Google Scholar] [CrossRef]
- Torabian, S.; Farhangi-Abriz, S.; Zahedi, M. Efficacy of FeSO4 nano formulations on osmolytes and antioxidative enzymes of sunflower under salt stress. Indian Journal of Plant Physiology 2018, 23, 305–315. [Google Scholar] [CrossRef]
- Wang, K.; Wang, Y.; Wan, Y.; Mi, Z.; Wang, Q.; Wang, Q.; Li, H. The fate of arsenic in rice plants (Oryza sativa L.): Influence of different forms of selenium. Chemosphere 2021, 264, 128417. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.; Rizwan, M.; Noureen, S.; Anwar, S.; Ali, B.; Naveed, M.; Abd, A.E.F.; Alqarawi, A.A.; Ahmad, P. Combined use of biochar and zinc oxide nanoparticle foliar spray improved the plant growth and decreased the cadmium accumulation in rice (Oryza sativa L.) plant. Environ Sci Pollut Res Int. 2019, 26, 11288–11299. [Google Scholar] [CrossRef]
- Sebastian, A.; Nangia, A.; Prasad, M.N.V. Cadmium and sodium adsorption properties of magnetite nanoparticles synthesized from Hevea brasiliensis Muell. Arg. bark: Relevance in amelioration of metal stress in rice. J Hazard Mater. 2019, 371, 261–272. [Google Scholar] [CrossRef]
- Ma, J.; Cai, H.; He, C.; Zhang, W.; Wang, L. A hemicellulose-bound form of silicon inhibits cadmium ion uptake in rice (Oryza sativa) cells. New Phytol. 2015, 206, 1063–1074. [Google Scholar] [CrossRef]
- Koleva, L.; Umar, A.; Yasin, N.A.; Shah, A.A.; Siddiqui, M.H.; Alamri, S.; Riaz, L.; Raza, A.; Javed, T.; Shabbir, Z. Iron Oxide and Silicon Nanoparticles Modulate Mineral Nutrient Homeostasis and Metabolism in Cadmium-Stressed Phaseolus vulgaris. Front Plant Sci. 2022, 13, 806781. [Google Scholar] [CrossRef]
- Sun, H.Y.; He, S.J.; Liu, T.L.; et al. Alleviation of cadmium toxicity by nano-silicon dioxide in Momordica charantia L. seedlings. Journal of Soil Science and Plant Nutrition 2023, 23, 1060–1069. [Google Scholar] [CrossRef]
- Noman, M.; Shahid, M.; Ahmed, T.; Tahir, M.; Naqqash, T.; Muhammad, S.; Song, F.; Abid, H.M.A.; Aslam, Z. Green copper nanoparticles from a native Klebsiella pneumoniae strain alleviated oxidative stress impairment of wheat plants by reducing the chromium bioavailability and increasing the growth. Ecotoxicol Environ Saf. 2020, 192, 110303. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, T.; Mustafa, G.; Nishiuchi, T.; Komatsu, S. Comparative Analysis of the Effect of Inorganic and Organic Chemicals with Silver Nanoparticles on Soybean under Flooding Stress. Int J Mol Sci. 2020, 21, 1300. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Niu, J.; Guo, Z.; Sui, X.; Xu, N.; Kareem, H.A.; et al. Graphene enhances photosynthesis and the antioxidative defense system and alleviates salinity and alkalinity stresses in alfalfa (Medicago sativa L.) by regulating gene expression. Environmental Science: Nano 2021, 8, 2731–2748. [Google Scholar] [CrossRef]
| Nanoparticles | Abiotic stresses | Usage | Crop species | Impact | Reference |
| AgNPs | Drought stress | Seed-induced | Oryza sativa | To enhance water absorption and promote the germination of aged seeds | [125] |
| CaO-NPs | Drought stress | Seed-induced | Brassica napus | To increase seedling fresh weight, number of leaves, chlorophyll content, and yield, as well as antioxidant enzyme levels, while reducing MDA content | [126] |
| SeNPs | Drought stress | Leaf application | Triticum aestivum | To increases CAT, SOD, and APX activity; improves photosynthetic rate, gas exchange, and transpiration rate; regulates the expression of certain stress-related genes; and enhances heat tolerance | [127] |
| SeNPs | Drought stress | Leaf application | Punica granatum | To increase antioxidant enzyme activity and the biosynthesis of photosynthetic pigments, and reduce levels of H₂O₂ and malondialdehyde | [128] |
| ZnO-NPs | Drought stress | Seed-induced | Zea mays | To increase net photosynthetic rate, water use efficiency, and the activity of key enzymes involved in carbon metabolism; enhance sucrose and starch synthesis in leaves, as well as glycolytic metabolism | [129] |
| GO | Drought stress | Soil-root uptake | Glycine max | To increase the levels of defense enzymes and hormones, as well as the expression of certain drought-stress genes, thereby enhancing the plant's drought tolerance | [130] |
| AgNPs | High temperature stress | Soil-root uptake | Triticum aestivum | To increase the root-to-shoot ratio, fresh and dry plant weight, and leaf area, and promote a decrease in ROS levels | [107] |
| TiO2-NPs | High temperature stress | Leaf application | Sesamum indicum | To increase peroxidase activity and the proportion of unsaturated fatty acids, and reduce the concentrations of malondialdehyde and H₂O₂ | [131] |
| SeNPs | High temperature stress | Leaf application | Triticum aestivum | To increases CAT, SOD, and APX activity; improves photosynthetic rate, gas exchange, and transpiration rate; regulates the expression of certain stress-related genes; and enhances heat tolerance | [132] |
| CTS-GB-NPs | Cold temperature stress | Fruit spread | Prunus salicina | To increase antioxidant enzyme activity while reducing weight loss and tissue softening during storage | [128] |
| TiO2-NPs | Cold temperature stress | Seed-induced | Cicer arietinum | To increase the expression of genes encoding chlorophyll-binding proteins and the activity of phosphoenolpyruvate carboxylase to promote photosynthesis | [112] |
| CeO2-NPs | Salt stress | Root uptake | Oryza sativa | Regulate the enzymatic activity of the antioxidant system and reduce 8-OHdG levels | [93] |
| ZnO-NPs | Salt stress | Leaf application | Vicia faba | To increase the content of proline and total soluble sugars | [133] |
| ZnO-NPs | Salt stress | Leaf application | Triticum aestivum | To promote the formation of plant sap and nutrient absorption | [134] |
| CeO2-NPs | Salt stress | Leaf injection | Gossypium hirsutum Linn | To regulate the expression of KOR, SOS, and other ion transport genes to minimize Na+ absorption | [135] |
| CeO2-NPs | Salt stress | Soil-root uptake | Brassica napus | To reduce the barrier in the plant's plastids, promoting the transport of more Na+ from the roots to the stems | [136] |
| FeSO4-NPs | Salt stress | Leaf application | Helianthus annuus | To increase CAT, POX, and PPO activity and reduces hydroxyl radical production | [137] |
| SeNPs | Heavy Metals Stress | Root uptake | Oryza sativa | To form complexes with As, reducing the transport of heavy metals from roots to stems | [138] |
| CeO2-NPs | Heavy Metals Stress | Root uptake | Oryza sativa | To increase chlorophyll content in seedlings and reduce proline content | [93] |
| ZnO-NPs | Heavy Metals Stress | Leaf application | Oryza sativa | To reduce Cd concentrations in plant roots and stems, increased soil pH, and significantly reduced soil-available Cd | [139] |
| Fe3O4-NPs | Heavy Metals Stress | Root uptake | Oryza sativa | To reduce the accumulation of Cd in plants and its mobility in soil | [140] |
| SiNPs | Heavy Metals Stress | Root uptake | Oryza sativa | To form complexes with Cd, reduces the translocation of heavy metals from roots to stems, stimulates the expression of the Si-absorption gene OsLsi1, and enhances resistance to Cd stress | [141] |
| Fe3O4-NP | Heavy Metals Stress | Seed-induced | Phaseolus vulgaris | To increase K+ levels, promote polyamine biosynthesis, and reduce MDA levels and electrolyte leakage | [142] |
| SiNPs | Heavy Metals Stress | Root uptake | Momordica charantia | To increase chlorophyll content, photosynthetic rate, transpiration rate, and stomatal conductance; enhance antioxidant enzyme activity; reduce Cd concentrations in plant stems and roots; and decrease flavonoid and soluble sugar levels to enhance Cd tolerance | [143] |
| CuNPs | Heavy Metals Stress | Root uptake | Triticum aestivum | To increase root length and raised levels of antioxidants in the cells | [144] |
| AgNPs | Waterlogging stress | Root uptake | Glycine max | To increase levels of soybean calmodulin, calreticulin, and glycoproteins to regulate misfolded proteins or severely damaged proteins | [145] |
| Graphene-NPs | Salt stress | Root fertilization | Medicago sativa | Biomass increased significantly under stress | [146] |
| Graphene-NPs | Alkali stress | Root fertilization | Medicago sativa | Biomass increased significantly under stress | [146] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




