Submitted:
14 April 2026
Posted:
16 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Protein Metabolism: Beyond Energy Provision
3. Fate of Proteins in Caloric Deficit
3.1. Protein Sparing Effect of Calories
4. Protein Turnover and Nitrogen Balance
5. More than Just Building Blocks: Essential, Non-Essential and Conditionally Essential Amino Acids
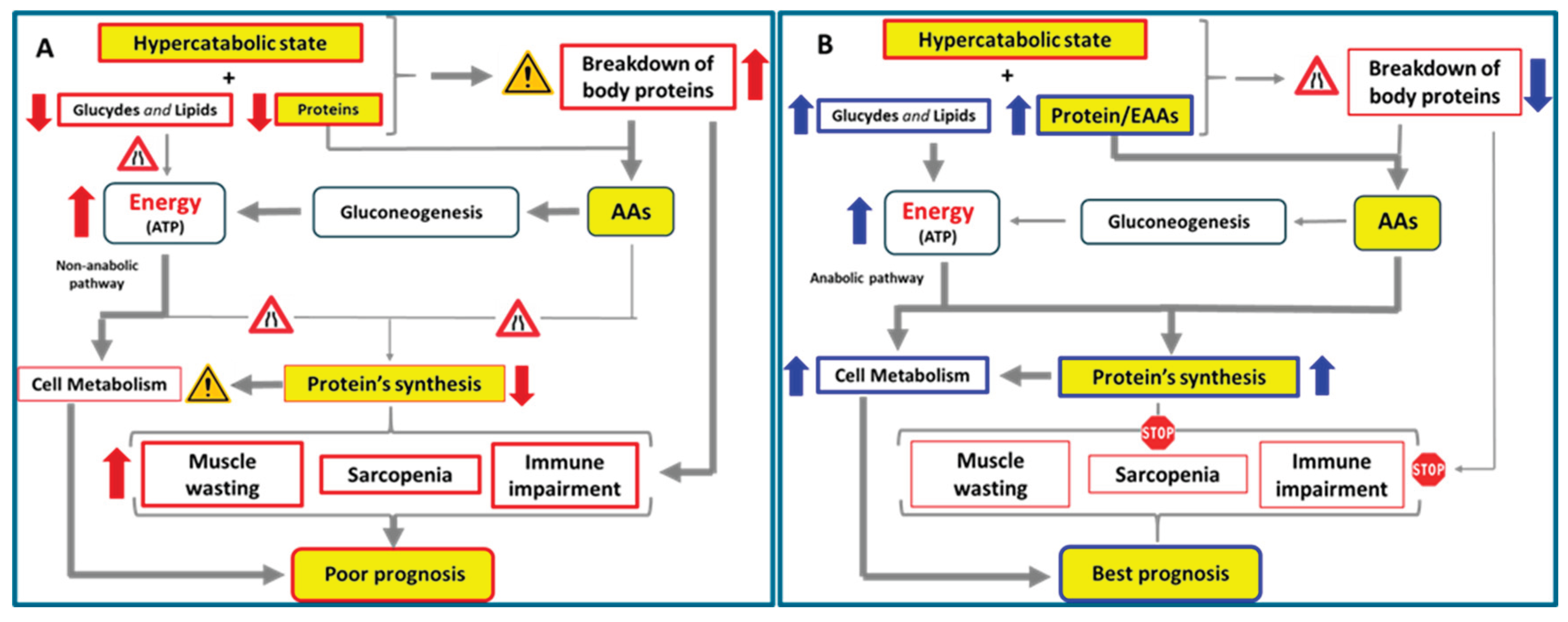
6. Hypercatabolic States and Nutritional Implications
7. Nutritional Supplementation of Proteins and EAA
8. Ultra-Processed Foods and Chronic Caloric Overfeeding: Health Implications
9. Clinical Implications and Future Directions
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AAs | Amino acids |
| CEAAs | Conditionally essential amino acids |
| DIAAS | Digestible indispensable amino acid score |
| EAAs | Essential amino acids |
| eNOS | Endothelial nitric oxide synthase |
| GLP-1 | Glucagon-like peptide-1 |
| HCS | Hyper catabolic state |
| ICU | Intensive care unit |
| LBM | Lean body mass |
| MPS | Muscle protein synthesis |
| mTORC1 | Mechanistic target of rapamycin complex-1 |
| NB | Nitrogen balance |
| NEAAs | Non-essential amino acids |
| PSMF | protein-sparing modified fast |
| TLR | Toll-like receptors |
| UPS | Ultra-processed foods |
References
- Pasini, E.; Aquilani, R.; Dioguardi, F.S.; D’Antona, G.; Gheorghiade, M.; Taegtmeyer, H. Hypercatabolic syndrome: molecular basis and effects of nutritional supplements with amino acids. Am J Cardiol. 2008, 101(11A), 11E–15E. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.C.; Sun, C.Y.; Tsai, C.Y.; Chen, M.Y.; Wang, S.Y.; Hsu, J.T.; Yeh, C.N.; Yeh, T.S. Metabolism of Proteins and Amino Acids in Critical Illness: From Physiological Alterations to Relevant Clinical Practice. J Multidiscip Healthc 2021, 14, 1107–1117. [Google Scholar] [CrossRef]
- Zhang, J.; Luo, W.; Miao, C.; Zhong, J. Hypercatabolism and Anti-catabolic Therapies in the Persistent Inflammation, Immunosuppression, and Catabolism Syndrome. Front Nutr. 2022, 9, 941097. [Google Scholar] [CrossRef]
- Chadda, K.R.; Puthucheary, Z. Persistent inflammation, immunosuppression, and catabolism syndrome (PICS): a review of definitions, potential therapies, and research priorities. Br J Anaesth 2024, 132(3), 507–518. [Google Scholar] [CrossRef] [PubMed]
- Burstad, K.M.; Lamina, T.; Erickson, A.; Gholizadeh, E.; Namigga, H.; Claussen, A.M.; Slavin, J.L.; Teigen, L.; Hill Gallant, K.M.; Stang, J.; Steffen, L.M.; Harindhanavudhi, T.; Kouri, A.; Duval, S.; Butler, M. Evaluation of dietary protein and amino acid requirements: a systematic review. Am J Clin Nutr 2025, 122(1), 285–305. [Google Scholar] [CrossRef]
- Pasini, E.; Corsetti, G.; Dioguardi, F.S. Behind Protein Synthesis: Amino Acids-Metabokine Regulators of Both Systemic and Cellular Metabolism. Nutrients 2023, 15(13), 2892. [Google Scholar] [CrossRef]
- Burrin, D.G.; Davis, T.A. Proteins and amino acids in enteral nutrition. Curr Opin Clin Nutr Metab Care 2004, 7(1), 79–87. [Google Scholar] [CrossRef]
- Corsetti, G.; Pasini, E.; Scarabelli, T.M.; Romano, C.; Singh, A.; Scarabelli, C.C.; Dioguardi, F.S. Importance of Energy, Dietary Protein Sources, and Amino Acid Composition in the Regulation of Metabolism: An Indissoluble Dynamic Combination for Life. Nutrients 2024, 16, 2417. [Google Scholar] [CrossRef]
- Petkova, D.; Stoyanova, S.; Dinkov, G.; Bogdanov, M.G. Beyond Protein Building Blocks: A Review of Biological Roles and Therapeutic Potential of Free Amino Acids. Int J Mol Sci 2025, 26, 11264. [Google Scholar] [CrossRef] [PubMed]
- Theodorakis, N.; Kreouzi, M.; Pappas, A.; Nikolaou, M. Beyond Calories: Individual Metabolic and Hormonal Adaptations Driving Variability in Weight Management-A State-of-the-Art Narrative Review. Int J Mol Sci 2024, 25(24), 13438. [Google Scholar] [CrossRef]
- Hall, K.D.; Farooqi, I.S.; Friedman, J.M.; Klein, S.; Loos, R.J.F.; Mangelsdorf, D.J.; O’Rahilly, S.; Ravussin, E.; Redman, L.M.; Ryan, D.H.; Speakman, J.R.; Tobias, D.K. The energy balance model of obesity: beyond calories in, calories out. Am J Clin Nutr 2022, 115(5), 1243–1254. [Google Scholar] [CrossRef] [PubMed]
- Lucan, S.C.; DiNicolantonio, J.J. How calorie-focused thinking about obesity and related diseases may mislead and harm public health. An alternative. Public Health Nutr. 2015, 18(4), 571–81. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, B.C.; Harris, I.B.; Beckman, T.J.; Reed, D.A.; Cook, D.A. Standards for reporting qualitative research: A synthesis of recommendations. Acad Med 2014, 89, 1245–1251. [Google Scholar] [CrossRef]
- Wu, G. Amino acids: metabolism, functions, and nutrition. Amino Acids 2009, 37(1), 1–17. [Google Scholar] [CrossRef]
- Gorissen, S.H.M.; Crombag, J.J.R.; Senden, J.M.G.; Waterval, W.A.H.; Bierau, J.; Verdijk, L.B.; van Loon, L.J.C. Protein content and amino acid composition of commercially available plant-based protein isolates. Amino Acids 2018, 50(12), 1685–1695. [Google Scholar] [CrossRef]
- FAO. Dietary protein quality evaluation in human nutrition. Report of an FAO Expert Consultation. FAO Food and Nutrition Paper 92. 2013. Available online: https://www.fao.org/ag/humannutrition/35978-02317b979a686a57aa4593304ffc17f06.pdf.
- Cruz-Pierard, S.; Iñiguez-Jiménez, S. Synergistic Effects of Protein Intake and Exercise on Biomarkers of Sarcopenia: A Systematic Review. Biomolecules 2026, 16, 195. [Google Scholar] [CrossRef]
- Prokopidis, K.; Brouer, I.C.; Lett, A.M. Food matrix in the context of muscle and whole-body protein synthesis: a scoping review. Nutr Metab (Lond) 2025, 22(1), 151. [Google Scholar] [CrossRef]
- Coker, M.S.; Coker, R.H. Dietary proteins, amino acids and insulin resistance: mini-review. Front Nutr 2025, 12, 1671286. [Google Scholar] [CrossRef] [PubMed]
- Yimam, M.A.; Andreini, M.; Carnevale, S.; Muscaritoli, M. Postprandial aminoacidemia following the ingestion of alternative and sustainable proteins in humans: a narrative review. Nutrients 2025, 17(2), 211. [Google Scholar] [CrossRef] [PubMed]
- Matthews, J.J.; Arentson-Lantz, E.J.; Moughan, P.J.; Wolfe, R.R.; Ferrando, A.A.; Church, D.D. Understanding Dietary Protein Quality: Digestible Indispensable Amino Acid Scores and Beyond. J Nutr 2025, 155(10), 3152–3167. [Google Scholar] [CrossRef] [PubMed]
- Tomé, D.; Benoit, S.; Azzout-Marniche, D. Protein metabolism and related body function: mechanistic approaches and health consequences. Proc Nutr Soc 2021, 80(2), 243–251. [Google Scholar] [CrossRef]
- Discher, D. E.; Janmey, P.; Wang, Y.-L. Tissue cells feel and respond to the stiffness of their substrate. Science 2025, 310(5751), 1139–1143. [Google Scholar] [CrossRef] [PubMed]
- Ingber, D.E.; Tensegrity, I. Cell structure and hierarchical systems biology. Journal of Cell Science 2023, 116(7), 1157–1173. [Google Scholar] [CrossRef] [PubMed]
- Janmey, P.A.; McCulloch, C.A. Cell mechanics: integrating cell responses to mechanical stimuli. Annual Review of Biomedical Engineering 2007, 9, 1–34. [Google Scholar] [CrossRef]
- Rief, M.; Gautel, M.; Oesterhelt, F.; Fernandez, J.M.; Gaub, H.E. Reversible unfolding of individual titin immunoglobulin domains by AFM. Science 1997, 276(5315), 1109–1112. [Google Scholar] [CrossRef]
- Chakrabarti, S.; Jahandideh, F.; Wu, J. Food-derived bioactive peptides on inflammation and oxidative stress. Biomed Res Int 2014, 2014, 608979. [Google Scholar] [CrossRef] [PubMed]
- Machado, M.; Bautista-Hérnandez, I.; Gómez-García, R.; Silva, S.; Costa, E.M. Bioactive Food Proteins: Bridging Nutritional and Functional Benefits with Sustainable Protein Sources. Foods 2025, 14, 3035. [Google Scholar] [CrossRef]
- Saltiel, A.R.; Kahn, C.R. Insulin signaling and the regulation of glucose and lipid metabolism. Nature 2001, 414(6865), 799–806. [Google Scholar] [CrossRef]
- D’Antona, G.; Ragni, M.; Cardile, A.; Tedesco, L.; Dossena, M.; Bruttini, F.; Caliaro, F.; Corsetti, G.; Bottinelli, R.; Carruba, M.O.; et al. Branched-chain amino acid supplementation promotes survival and supports cardiac and skeletal muscle mitochondrial biogenesis in middle-aged mice. Cell Metab 2010, 12, 362–372. [Google Scholar] [CrossRef]
- Laplante, M.; Sabatini, D.M. mTOR signaling in growth control and disease. Cell 2012, 149(2), 274–93. [Google Scholar] [CrossRef]
- Saxton, R.A.; Sabatini, D.M. mTOR Signaling in Growth, Metabolism, and Disease. Cell Erratum in: Cell 2017, 169(2), 361-371. doi: 10.1016/j.cell.2017.03.035.. 2017, 168(6), 960–976. [Google Scholar] [CrossRef] [PubMed]
- Wolfson, R.L.; Chantranupong, L.; Saxton, R.A.; Shen, K.; Scaria, S.M.; Cantor, J.R.; Sabatini, D.M. Sestrin2 is a leucine sensor for the mTORC1 pathway. Science 2016, 351(6268), 43–8. [Google Scholar] [CrossRef] [PubMed]
- Hardie, D.G.; Ross, F.A.; Hawley, S.A. AMPK: a nutrient and energy sensor that maintains energy homeostasis. Nat Rev Mol Cell Biol. 2012, 13(4), 251–62. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, C.; Weinert, B.T.; Nishida, Y.; Verdin, E.; Mann, M. The growing landscape of lysine acetylation links metabolism and cell signalling. Nat Rev Mol Cell Biol 2014, 15(8), 536–50. [Google Scholar] [CrossRef] [PubMed]
- Kharitonenkov, A.; Adams, A.C. Inventing new medicines: the FGF21 story. Molecular metabolism 2014, 3(3), 221–229. [Google Scholar] [CrossRef] [PubMed]
- O’Shea, J.J.; Plenge, R. JAK and STAT signaling molecules in immunoregulation and immune-mediated disease. Immunity 2012, 36(4), 542–50. [Google Scholar] [CrossRef]
- Tanaka, T.; Narazaki, M.; Kishimoto, T. IL-6 in inflammation, immunity, and disease. Cold Spring Harb Perspect Biol 2014, 6(10), a016295. [Google Scholar] [CrossRef]
- Akira, S.; Uematsu, S.; Takeuchi, O. Pathogen recognition and innate immunity. Cell 2006, 124(4), 783–801. [Google Scholar] [CrossRef] [PubMed]
- Sharpe, A.H.; Pauken, K.E. The diverse functions of the PD1 inhibitory pathway. Nat Rev Immunol 2018, 18(3), 153–167. [Google Scholar] [CrossRef] [PubMed]
- Vila-Casahonda, R.G.; Lozano-Aponte, J.; Fragoso-Medina, J.A.; et al. Integrative in vitro and in silico evaluation of HSP60-derived peptides as immunomodulators of the TLR4/MD-2 complex. Sci Rep 2025, 15, 44897. [Google Scholar] [CrossRef]
- Omeje, A.; Ogbu, C.P.; Ngozi Aguchem, R.; Uzochukwu Okagu, I. Food proteins and bioactive peptides as nutraceutical intervention against immune system disorders: a review. Food Nutrition 2026, 2(1), 100051. [Google Scholar] [CrossRef]
- Wolfe, R.R. The underappreciated role of muscle in health and disease. Am J Clin Nutr 2006, 84(3), 475–82. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.M.; Van Loon, L.J. Dietary protein for athletes: from requirements to optimum adaptation. J Sports Sci 2011, 29 Suppl 1, S29–38. [Google Scholar] [CrossRef] [PubMed]
- Pasiakos, S.M.; Cao, J.J.; Margolis, L.M.; Sauter, E.R.; Whigham, L.D.; McClung, J.P.; Rood, J.C.; Carbone, J.W.; Combs, G.F., Jr.; Young, A.J. Effects of high-protein diets on fat-free mass and muscle protein synthesis following weight loss: a randomized controlled trial. FASEB J 2013, 27(9), 3837–47. [Google Scholar] [CrossRef] [PubMed]
- Hector, A.J.; Phillips, S.M. Protein Recommendations for Weight Loss in Elite Athletes: A Focus on Body Composition and Performance. Int J Sport Nutr Exerc Metab. 2018, 28(2), 170–177. [Google Scholar] [CrossRef] [PubMed]
- Martin-Rincon, M.; Perez-Suarez, I.; Pérez-López, A.; et al. Protein synthesis signaling in skeletal muscle is refractory to whey protein ingestion during a severe energy deficit evoked by prolonged exercise and caloric restriction. Int J Obes 2019, 43, 872–882. [Google Scholar] [CrossRef]
- Roth, C.; Rettenmaier, L.; Behringer, M. High-Protein Energy-Restriction: Effects on Body Composition, Contractile Properties, Mood, and Sleep in Active Young College Students. Front Sports Act Living 2021, 3, 683327. [Google Scholar] [CrossRef]
- Wolfe, R.R.; Church, D.D.; Ferrando, A.A.; Moughan, P.J. Consideration of the role of protein quality in determining dietary protein recommendations. Front Nutr. 2024, 11, 1389664. [Google Scholar] [CrossRef]
- Weijs, P.J.; Cynober, L.; DeLegge, M.; Kreymann, G.; Wernerman, J.; Wolfe, R.R. Proteins and amino acids are fundamental to optimal nutrition support in critically ill patients. Crit Care 2014, 18(6), 591. [Google Scholar] [CrossRef]
- Fuller, M.F.; Crofts, R.M.J. The protein-sparing effect of carbohydrate: 1. Nitrogen retention of growing pigs in relation to diet. British Journal of Nutrition 1977, 38(3), 479–488. [Google Scholar] [CrossRef]
- Vazquez, J.A.; Kazi, U.; Madani, N. Protein metabolism during weight reduction with very-low-energy diets: evaluation of the independent effects of protein and carbohydrate on protein sparing. The American Journal of Clinical Nutrition 1995, 62(1), 93–103. [Google Scholar] [CrossRef]
- Thomas, D.D.; Istfan, N.W.; Bistrian, B.R.; Apovian, C.M. Protein sparing therapies in acute illness and obesity: a review of George Blackburn’s contributions to nutrition science. Metabolism 2018, 79, 83–96. [Google Scholar] [CrossRef]
- Bakhach, M.; Shah, V.; Harwood, T.; Lappe, S.; Bhesania, N.; Mansoor, S.; Alkhouri, N. The Protein-Sparing Modified Fast Diet: An Effective and Safe Approach to Induce Rapid Weight Loss in Severely Obese Adolescents. Glob Pediatr Health 2016, 22(3), 2333794 × 15623245. [Google Scholar] [CrossRef]
- Formisano, E.; Schiavetti, I.; Gradaschi, R.; Gardella, P.; Romeo, C.; Pisciotta, L.; Sukkar, S.G. The Real-Life Use of a Protein-Sparing Modified Fast Diet by Nasogastric Tube (ProMoFasT) in Adults with Obesity: An Open-Label Randomized Controlled Trial. Nutrients 2023, 15, 4822. [Google Scholar] [CrossRef]
- Hartl, W.H.; Kopper, P.; Bender, A.; Scheipl, F.; Day, A.G.; Elke, G.; Küchenhoff, H. Protein intake and outcome of critically ill patients: analysis of a large international database using piece-wise exponential additive mixed models. Crit Care 2022, 26(1), 7. [Google Scholar] [CrossRef]
- Bels, J.L.M.; Ali Abdelhamid, Y.; van de Poll, M.C.G. Protein supplementation in critical illness: why, when and how? Curr Opin Clin Nutr Metab Care 2023, 26(2), 146–153. [Google Scholar] [CrossRef] [PubMed]
- Basisty, N.; Meyer, J.G.; Schilling, B. Protein Turnover in Aging and Longevity. Proteomics 2018, 18(5-6), e1700108. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, R.R.; Miller, S.L. The recommended dietary allowance of protein: a misunderstood concept. JAMA Erratum in: JAMA. 2008 Oct 15;300(15):1763. 2008, 299(24), 2891–3. [Google Scholar] [CrossRef]
- Biolo, G.; Tipton, K.D.; Klein, S.; Wolfe, R.R. An abundant supply of amino acids enhances the metabolic effect of exercise on muscle protein. Am J Physiol 1997, 273 1 Pt 1, E122-9. [Google Scholar] [CrossRef] [PubMed]
- Rennie, M.J.; Tipton, K.D. Protein and amino acid metabolism during and after exercise and the effects of nutrition. Annu Rev Nutr 2000, 20, 457–83. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.M. Dietary protein requirements and adaptive advantages in athletes. Br J Nutr 2012, 108(2), S158–67. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, R.R. Branched-chain amino acids and muscle protein synthesis in humans: Myth or reality? J Int Soc Sports Nutr 2017, 14, 30. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, D.; Hayamizu, K.; Uno, C.; Hasegawa, Y.; Kuwahata, M.; Kido, Y.; Suzuki, Y. Nitrogen Requirements in Healthy Adults: A Systematic Review and Meta-Analysis of Nitrogen Balance Studies. Nutrients 2025, 17, 2615. [Google Scholar] [CrossRef] [PubMed]
- Clauss, M.; Puissant, C.; Bastani, N.E.; Nielsen, L.L.K.; Skålhegg, B.S.; Jeppesen, P.B.; Jensen, J. Effect of high intakes of protein-only and carbohydrate-only on plasma metabolites and hormones, in addition to nitrogen excretion. Front Endocrinol 2025, 16, 1618142. [Google Scholar] [CrossRef]
- Tagawa, R.; Watanabe, D.; Inoue, Y.; Takaragawa, M.; Jin, Q.; Ito, K.; Yamazaki, K.; Sanbongi, C.; Hatamoto, Y.; Miyachi, M. Comparison of Protein Requirements Based on the Nitrogen Balance and Indicator Amino Acid Oxidation Methods: An Umbrella Review and Meta-analysis. J Nutr 2025, 155(11), 3877–3885. [Google Scholar] [CrossRef]
- Romano, C.; Corsetti, G.; Flati, V.; Pasini, E.; Picca, A.; Calvani, R.; Marzetti, E.; Dioguardi, F.S. Influence of diets with varying essential/nonessential amino acid ratios on mouse lifespan. Nutrients 2019, 11(6), 1367. [Google Scholar] [CrossRef]
- Paoletti, A.; Courtney-Martin, G.; Elango, R. Determining amino acid requirements in humans. Front Nutr 2024, 11, 1400719. [Google Scholar] [CrossRef] [PubMed]
- Pasini, E.; Corsetti, G.; Aquilani, R.; Romano, C.; Picca, A.; Calvani, R.; Dioguardi, F.S. Protein-amino acid metabolism disarrangements: The hidden enemy of chronic age-related conditions. Nutrients 2018, 10, 391. [Google Scholar] [CrossRef]
- Visvanathan, R.; Chapman, I.M. Undernutrition and anorexia in the older person. Gastroenterol Clin North Am 2009, 38(3), 393–409. [Google Scholar] [CrossRef]
- Heber, D.; Tchekmedyian, N.S. Cancer cachexia and anorexia; Heber, Nutritional oncology D., Blackburn, G.L., Go, V.L.W., Eds.; Academic Press: California, 1999; pp. 537–546. [Google Scholar]
- Hall, D.T.; Ma, J.F.; Marco, S.D.; Gallouzi, I.E. Inducible nitric oxide synthase (iNOS) in muscle wasting syndrome, sarcopenia, and cachexia. Aging 2011, 3(8), 702–715. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Chan, C.P.; Yan, B.P.; Zhang, Q.; Lam, Y.Y.; Li, R.J.; Sanderson, J.E.; Coats, A.J.; Sun, J.P.; Yip, G.W.; Yu, C.M. Albumin levels predict survival in patients with heart failure and preserved ejection fraction. Eur J Heart Fail 2012, 14, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Aquilani, R.; Opasic, C.; Dossena, M.; Iadarola, P.; Gualco, A.; Arcidiaco, P.; Viglio, S.; Boschi, F.; Verri, M.; Pasini, E. Increased skeletal muscle amino acid release with light exercise in deconditioned patients with heart failure. J Am Coll Cardiol 2005, 45, 154–164. [Google Scholar] [CrossRef]
- Anker, S.D.; Ponikowski, P.; Varney, S.; Chua, T.P.; Clark, A.L.; Webb-Peploe, K.M.; Harrington, D.; Kox, W.J.; Poole-Wilson, P.A.; Coats, A.J. Wasting as independent risk factor for mortality in chronic heart failure. Lancet 1997, 349, 1050–1053. [Google Scholar] [CrossRef]
- Gwin, J.A.; Church, D.D.; Allen, J.T.; Wilson, M.A.; Carrigan, C.T.; Murphy, N.E.; Varanoske, A.N.; Margolis, L.M.; Wolfe, R.R.; Ferrando, A.A.; Pasiakos, S.M. Consuming Whey Protein with Added Essential Amino Acids, Not Carbohydrate, Maintains Postexercise Anabolism While Underfed. Med Sci Sports Exerc. 2025, 57(1), 70–80. [Google Scholar] [CrossRef]
- Church, D.D.; Ferrando, A.A.; Wolfe, R.R. Stimulation of muscle protein synthesis with low-dose amino acid composition in older individuals. Front Nutr 2024, 11, 1360312. [Google Scholar] [CrossRef] [PubMed]
- Annunziata, G.; Verde, L.; D’Orsi, V.; Caprio, M.; Gorini, S.; Savastano, S.; Colao, A.; Muscogiuri, G.; Barrea, L. Supplementation with essential amino acids in the early stage of carbohydrate reintroduction after a very-low energy ketogenic therapy (VLEKT) improves body cell mass, muscle strength and inflammation. J Transl Med 2025, 23(1), 111. [Google Scholar] [CrossRef]
- Cereda, E.; Borioli, V.; Caraccia, M.; Uggè, A.; De Simeis, F.; Bruno, R.; Corsico, A.G.; Di Sabatino, A.; Pedrazzoli, P.; Caccialanza, R. Systematic high-calorie, high-protein oral nutritional support in hospitalized, moderately hypophagic patients at nutritional risk: A randomized-controlled trial. Clin Nutr 2026, 56, 106532. [Google Scholar] [CrossRef]
- Chapela, S.P.; Simancas-Racines, D.; Montalvan, M.; Frias-Toral, E.; Simancas-Racines, A.; Muscogiuri, G.; Barrea, L.; Sarno, G.; Martínez, P.I.; Reberendo, M.J.; Llobera, N.D.; Stella, C.A. Signals for Muscular Protein Turnover and Insulin Resistance in Critically Ill Patients: A Narrative Review. Nutrients 2023, 15(5), 1071. [Google Scholar] [CrossRef]
- Weijs, P.J.; Looijaard, W.G.; Beishuizen, A.; Girbes, A.R.; Oudemans-van Straaten, H.M. Early high protein intake is associated with low mortality and energy overfeeding with high mortality in non-septic mechanically ventilated critically ill patients. Crit Care 2014, 18(6), 701. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, A.; Doleman, B.; Phillips, B.E.; Williams, J.P. The effects of protein nutrition on muscle function in critical illness: a systematic review and meta-analysis. Nutrients 2025, 17(16), 2613. [Google Scholar] [CrossRef] [PubMed]
- Casaer, M.P.; Mesotten, D.; Hermans, G.; Wouters, P.J.; Schetz, M.; Meyfroidt, G.; Van Cromphaut, S.; Ingels, C.; Meersseman, P.; Muller, J.; et al. Early versus late parenteral nutrition in critically ill adults. N Engl J Med 2011, 365(6), 506–17. [Google Scholar] [CrossRef]
- ESPEN Expert Group. ESPEN guideline on clinical nutrition in the intensive care unit. Clin Nutr 2019, 38, 48–79. [Google Scholar] [CrossRef]
- Hagiyama, A.; Yamamoto, N.; Watanabe, J.; Tsuge, T.; Nakashima, Y.; Nakao, S.; Sato, H.; Yorifuji, T. Efficacy of Protein and Essential Amino Acid Supplementation in Lower Limb Surgeries: A Systematic Review and Meta-Analysis. Cureus 2024, 16(9), e69212. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.; Kong, J.; Underwood, C.; Petocz, P.; Hirani, V.; Dawson, B.; O’Leary, F. Systematic review and meta-analysis of the effect of protein and amino acid supplements in older adults with acute or chronic conditions. Br J Nutr 2018, 119(5), 527–542. [Google Scholar] [CrossRef]
- Stein, W.; Sauer, H.J.; Oberänder, N.; Weimann, A.; Fischer, M. Effects of Additional Protein Intake on Lean Body Mass in Patients Undergoing Multimodal Treatment for Morbid Obesity. Nutrients 2024, 16(6), 864. [Google Scholar] [CrossRef] [PubMed]
- Nunes, E.A.; Colenso-Semple, L.; McKellar, S.R.; Yau, T.; Ali, M.U.; Fitzpatrick-Lewis, D.; Sherifali, D.; Gaudichon, C.; Tomé, D.; Atherton, P.J.; et al. Systematic review and meta-analysis of protein intake to support muscle mass and function in healthy adults. J Cachexia Sarcopenia Muscle 2022, 13(2), 795–810. [Google Scholar] [CrossRef] [PubMed]
- Arends, J.; Bachmann, P.; Baracos, V.; et al. ESPEN guidelines on nutrition in cancer patients. Clinical Nutrition 2017, 36(1), 11–48. [Google Scholar] [CrossRef] [PubMed]
- Longland, T.M.; Oikawa, S.Y.; Mitchell, C.J.; Devries, M.C.; Phillips, S.M. Higher compared with lower dietary protein during an energy deficit combined with intense exercise promotes greater lean mass gain and fat mass loss: a randomized trial. Am J Clin Nutr 2016, 103(3), 738–46. [Google Scholar] [CrossRef]
- Villet, S.; Chiolero, R.L.; Bollmann, M.D.; Revelly, J.P.; Cayeux, R.N.M.C.; Delarue, J.; Berger, M.M. Negative impact of hypocaloric feeding and energy balance on clinical outcome in ICU patients. Clin Nutr 2005, 24(4), 502–9. [Google Scholar] [CrossRef]
- Paddon-Jones, D.; Sheffield-Moore, M.; Zhang, X.J.; Volpi, E.; Wolf, S.E.; Aarsland, A.; Ferrando, A.A.; Wolfe, R.R. Amino acid ingestion improves muscle protein synthesis in the young and elderly. Am J Physiol Endocrinol Metab 2004, 286(3), E321-8. [Google Scholar] [CrossRef]
- Wolfe, R.R. Protein metabolism in critical illness and injury. Annual Review of Nutrition 2000, 20, 105–130. [Google Scholar] [CrossRef]
- Wolfe, R.R.; Goodenough, R.D.; Burke, J.F.; Wolfe, M.H. Response of protein and urea kinetics in burn patients to different levels of protein intake. Ann Surg 1983, 197(2), 163–71. [Google Scholar] [CrossRef]
- Hu, Y.; Xu, T.; Wei, J.; Zhu, P.; Shi, D.; Liu, J. The impact of protein intake on kidney adverse events in critically Ill patients: a systematic review and meta-analysis. Int Urol Nephrol 2025. [Google Scholar] [CrossRef]
- Castro, S.; Tomé, A.; Granja, C.; Macedo, A.; Binnie, A. High vs low protein intake in chronic critical illness: A systematic review and meta-analysis. Clin Nutr ESPEN 2025, 65, 249–255. [Google Scholar] [CrossRef]
- Badpeyma, M.; Ghalichi, F.; Molani-Gol, R.; Valizadeh, H.; Javadzadeh, Y.; Rasouli, A.; Alizadeh, M.; Kheirouri, S. Protein intake and clinical outcomes in critically ill patients: A dose-response and pairwise meta-analysis of randomized controlled trials. Health Promot Perspect 2025 (1), 15(2), 98–109. [Google Scholar] [CrossRef]
- Badpeyma, M.; Sedaghat, A.; Moghaddam, A.B.; et al. The efficacy of high-protein nutritional support on mortality, clinical outcomes, and nutritional adequacy in critically ill patients: a double-center randomized controlled trial. Nutr Metab (Lond) 2025 (2), 22, 116. [Google Scholar] [CrossRef]
- O’Keefe, G.E.; Brown, S.P.; Shelton, M.M.; Qiu, Q.; Bisgaard, E.K.; Wilson, I.M.; Robinson, J.L.; Roubik, D.J.; Malloy, A.; May, S. Enteral protein supplementation in critically ill trauma and surgical patients: A single-center randomized clinical trial. J Trauma Acute Care Surg 2025, 99(4), 635–642. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ye, Y.; Xuan, L.; Xu, L.; Wang, P.; Ma, J.; Wang, Y.; Chen, Y.; Miao, J.; Wang, W.; Zhou, L. Impact of early high protein intake in critically ill patients: a randomized controlled trial. Nutr Metab (Lond) 2024, 21(1), 39. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Huang, J.; Ping, X.; Zheng, H.; Zhang, K.; Xu, X.; Yu, J. No benefit of higher protein dosing in critically ill patients: a systematic review and meta-analysis of randomized controlled trials. Peer J 2024, 12, e17433. [Google Scholar] [CrossRef] [PubMed]
- Blaauw, L.; Schoonees, A.; Robertson, N.; Visser, J. The impact of guideline recommended protein intake on mortality and length of intensive care unit and hospital stay in critically ill adults: A systematic review. Clin Nutr ESPEN 2024, 61, 356–368. [Google Scholar] [CrossRef]
- Rondanelli, M.; Opizzi, A.; Antoniello, N.; Boschi, F.; Iadarola, P.; Pasini, E.; Aquilani, R.; Dioguardi, F.S. Effect of essential amino acid supplementation on quality of life, amino acid profile and strength in institutionalized elderly patients. Clin Nutr 2011, 30(5), 571–577. [Google Scholar] [CrossRef]
- Aquilani, R.; D’Antona, G.; Baiardi, P.; Gambino, A.; Iadarola, P.; Viglio, S.; Pasini, E.; Verri, M.; Barbieri, A.; Boschi, F. Essential amino acids and exercise tolerance in elderly muscle-depleted subjects with chronic diseases: a rehabilitation without rehabilitation? Biomed Res Int 2014, 341603. [Google Scholar] [CrossRef] [PubMed]
- Negro, M.; Crisafulli, O.; D’Antona, G. Effects of essential amino acid (EAA) and glutamine supplementation on skeletal muscle wasting in acute, subacute, and postacute conditions. Clin Nutr ESPEN 2024, 62, 224–233. [Google Scholar] [CrossRef]
- Mitchell, W.K.; Wilkinson, D.J.; Phillips, B.E.; Lund, J.N.; Smith, K.; Atherton, P.J. Human skeletal muscle protein metabolism responses to amino acid nutrition. Adv Nutr 2016, 7, 828S–838S. [Google Scholar] [CrossRef]
- Corsetti, G.; Pasini, E.; Romano, C.; Calvani, R.; Picca, A.; Marzetti, E.; Flati, V.; Dioguardi, F.S. Body weight loss and tissue wasting in late middle-aged mice on slightly imbalanced essential/non-essential amino acids diet. Front Med 2018, 5, 136. [Google Scholar] [CrossRef]
- Corsetti, G.; Stacchiotti, A.; D’Antona, G.; Nisoli, E.; Dioguardi, F.S.; Rezzani, R. Supplementation with essential amino acids in middle age maintains the health of rat kidney. Int J Immunopathol Pharmacol 2010, 23, 523–533. [Google Scholar] [CrossRef]
- Corsetti, G.; Stacchiotti, A.; Tedesco, L.; D’Antona, G.; Pasini, E.; Dioguardi, F.S.; Nisoli, E.; Rezzani, R. Essential amino acid supplementation decreases liver damage induced by chronic ethanol consumption in rats. Int J Immunopathol Pharmacol 2011, 24, 611–619. [Google Scholar] [CrossRef] [PubMed]
- Corsetti, G.; D’Antona, G.; Ruocco, C.; Stacchiotti, A.; Romano, C.; Tedesco, L.; Dioguardi, F.; Rezzani, R.; Nisoli, E. Dietary supplementation with essential amino acids boosts the beneficial effects of rosuvastatin on mouse kidney. Amino Acids 2014, 46, 2189–2203. [Google Scholar] [CrossRef]
- Corsetti, G.; Romano, C.; Pasini, E.; Scarabelli, T.; Chen-Scarabelli, C.; Dioguardi, F.S. Essential amino acids-rich diet increases cardiomyocytes protection in Doxorubicin-treated mice. Nutrients 2023, 15, 2287. [Google Scholar] [CrossRef] [PubMed]
- Tedesco, L.; Rossi, F.; Ragni, M.; Ruocco, C.; Brunetti, D.; Carruba, M.O.; Torrente, Y.; Valerio, A.; Nisoli, E. A special amino-acid formula tailored to boosting cell respiration prevents mitochondrial dysfunction and oxidative stress caused by Doxorubicin in mouse cardiomyocytes. Nutrients 2020, 12, 282. [Google Scholar] [CrossRef]
- Ragni, M.; Fornelli, C.; Nisoli, E.; Penna, F. Amino Acids in Cancer and Cachexia: An Integrated View. Cancers (Basel) 2022, 14(22), 5691. [Google Scholar] [CrossRef] [PubMed]
- Corsetti, G.; Romano, C.; Codenotti, S.; Pasini, E.; Fanzani, A.; Scarabelli, T.; Dioguardi, F.S. A Diet Rich in Essential Amino Acids Inhibits the Growth of HCT116 Human Colon Cancer Cell In Vitro and In Vivo. Int J Mol Sci 2025, 26, 7014. [Google Scholar] [CrossRef]
- Stacchiotti, A.; Corsetti, G.; Lavazza, A.; Rezzani, R. Microscopic features of mitochondria rejuvenation by aminoacids. In Current Microscopy Contributions to Advances in Science and Technology; Mendez-Vilas, A., Ed.; 2012; Volume 1, pp. 286–294. [Google Scholar]
- Neubauer, S. The failing heart--an engine out of fuel. N Engl J Med 2007, 356(11), 1140–51. [Google Scholar] [CrossRef]
- Petersen, K.F.; Dufour, S.; Befroy, D.; Garcia, R.; Shulman, G.I. Impaired mitochondrial activity in the insulin-resistant offspring of patients with type 2 diabetes. N Engl J Med 2004, 350(7), 664–71. [Google Scholar] [CrossRef]
- MacCannell, A.D.; Roberts, L.D. Metabokines in the regulation of systemic energy metabolism. Curr Opin Pharmacol 2022, 67, 102286. [Google Scholar] [CrossRef]
- Gwin, J.A.; Church, D.D.; Wolfe, R.R.; Ferrando, A.A.; Pasiakos, S.M. Muscle Protein Synthesis and Whole-Body Protein Turnover Responses to Ingesting Essential Amino Acids, Intact Protein, and Protein-Containing Mixed Meals with Considerations for Energy Deficit. Nutrients 2020, 12, 2457. [Google Scholar] [CrossRef]
- Park, S.; Church, D.D.; Azhar, G.; Schutzler, S.E.; Ferrando, A.A.; Wolfe, R.R. Anabolic response to essential amino acid plus whey protein composition is greater than whey protein alone in young healthy adults. J Int Soc Sports Nutr 2020, 17(1), 9. [Google Scholar] [CrossRef]
- Negro, M.; Perna, S.; Spadaccini, D.; Castelli, L.; Calanni, L.; Barbero, M.; Cescon, C.; Rondanelli, M.; D’Antona, G. Effects of 12 Weeks of Essential Amino Acids (EAA)-Based Multi-Ingredient Nutritional Supplementation on Muscle Mass, Muscle Strength, Muscle Power and Fatigue in Healthy Elderly Subjects: A Randomized Controlled Double-Blind Study. J Nutr Health Aging 2019, 23(5), 414–424. [Google Scholar] [CrossRef]
- Rondanelli, M.; Aquilani, R.; Verri, M.; Boschi, F.; Pasini, E.; Perna, S.; Faliva, A.; Condino, A.M. Plasma kinetics of essential amino acids following their ingestion as free formula or as dietary protein components. Aging Clin Exp Res 2017, 29(4), 801–805. [Google Scholar] [CrossRef] [PubMed]
- Ferrando, A.A.; Paddon-Jones, D.; Hays, N.P.; Kortebein, P.; Ronsen, O.; Williams, R.H.; McComb, S.; Symons, T.B.; Wolfe, R.R.; Evans, W. EAA supplementation to increase nitrogen intake improves muscle function during bed rest in the elderly. Clin Nutr 2010, 29, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Berrazaga, I.; Micard, V.; Gueugneau, M.; Walrand, S. The Role of the Anabolic Properties of Plant- versus Animal-Based Protein Sources in Supporting Muscle Mass Maintenance: A Critical Review. Nutrients 2019, 11(8), 1825. [Google Scholar] [CrossRef]
- Ispoglou, T.; Witard, O.C.; Duckworth, L.C.; Lees, M.J. The efficacy of essential amino acid supplementation for augmenting dietary protein intake in older adults: implications for skeletal muscle mass, strength and function. Proc Nutr Soc 2021, 80(2), 230–242. [Google Scholar] [CrossRef]
- Yoshihara, I.; Kondo, Y.; Okamoto, K.; Tanaka, H. Sepsis-Associated muscle wasting: a comprehensive review from bench to bedside. Int J Mol Sci 2023, 24, 5040. [Google Scholar] [CrossRef]
- Steiner, J.L.; Lang, C.H. Sepsis Attenuates the Anabolic Response to Skeletal Muscle Contraction. Shock 2015, 43(4), 344–351. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.C.; Wu, J.M.; Chen, K.Y.; Wu, M.H.; Yang, P.J.; Lee, P.C.; Chen, P.D.; Yeh, S.L.; Lin, M.T. Glutamine and leucine administration attenuates muscle atrophy in sepsis. Life Sci 2023, 314, 121327. [Google Scholar] [CrossRef]
- Xue, Q.; Zhang, D.; Zou, J.; Wang, H.; Shi, R.; Dong, L. Treatment advances of sepsis-induced myopathy (Review). Biomed Rep 2024, 22(2), 19. [Google Scholar] [CrossRef] [PubMed]
- Aquilani, R.; Zuccarelli, G.C.; Maestri, R.; Boselli, M.; Dossena, M.; Baldissarro, E.; Boschi, F.; Buonocore, D.; Verri, M. Essential amino acid supplementation is associated with reduced serum C-reactive protein levels and improved circulating lymphocytes in post-acute inflamed elderly patients. Int J Immunopathol Pharmacol 2021, 35, 20587384211036823. [Google Scholar] [CrossRef]
- Vadiveloo, M.K.; Gardner, C.D.; Bleich, S.N.; Khandpur, N.; Lichtenstein, A.H.; Otten, J.J.; Rebholz, C.M.; Singleton, C.R.; Vos, M.B.; Wang, S.; et al. Ultraprocessed Foods and Their Association With Cardiometabolic Health: Evidence, Gaps, and Opportunities: A Science Advisory From the American Heart Association. Circulation 2025, 152, e245–e263. [Google Scholar] [PubMed]
- Godsey, T.J.; Eden, T.; Emerson, S.R. Ultra-Processed Foods and Metabolic Dysfunction: A Narrative Review of Dietary Processing, Behavioral Drivers and Chronic Disease Risk. Metabolites 2025, 15, 784. [Google Scholar] [CrossRef]
- Horton, T.J.; Drougas, H.; Brachey, A.; Reed, G.W.; Peters, J.C.; Hill, J.O. Fat and carbohydrate overfeeding in humans: different effects on energy storage. Am J Clin Nutr 1995, 62(1), 19–29. [Google Scholar] [CrossRef]
- Leaf, A.; Antonio, J. The Effects of Overfeeding on Body Composition: The Role of Macronutrient Composition—A Narrative Review. Int J Exerc Sci 2017, 10(8), 1275–1296. [Google Scholar] [CrossRef] [PubMed]
- Webb, P.; Annis, J.F. Adaptation to overeating in lean and overweight men and women. Hum Nutr Clin Nutr 1983, 37(2), 117–131. [Google Scholar]
- Bray, G.A.; Smith, S.R.; de Jonge, L.; Xie, H.; Rood, J.; Martin, C.K.; Most, M.; Brock, C.; Mancuso, S.; Redman, L.M. Effect of dietary protein content on weight gain, energy expenditure, and body composition during overeating: a randomized controlled trial. JAMA 2012, 307(1), 47–55. [Google Scholar] [CrossRef]
- Peterson, C.M.; Zhang, B.; Johannsen, D.L.; Ravussin, E. Eight weeks of overfeeding alters substrate partitioning without affecting metabolic flexibility in men. Int J Obes (Lond) 2017, 41(6), 887–893. [Google Scholar] [CrossRef]
- Johannsen, D.L.; Marlatt, K.L.; Conley, K.E.; Smith, S.R.; Ravussin, E. Metabolic adaptation is not observed after 8 weeks of overfeeding but energy expenditure variability is associated with weight recovery. Am J Clin Nutr 2019, 110(4), 805–813. [Google Scholar] [CrossRef]
- Singh, P.; Beyl, R.A.; Marlatt, K.L.; Ravussin, E. Sleep Duration Alters Overfeeding-mediated Reduction in Insulin Sensitivity. J Clin Endocrinol Metab 2025, 110(5), e1625–e1630. [Google Scholar] [CrossRef]
- Klein, C.J.; Stanek, G.S.; Wiles, C.E., 3rd. Overfeeding macronutrients to critically ill adults: metabolic complications. J Am Diet Assoc 1998, 98(7), 795–806. [Google Scholar] [CrossRef] [PubMed]
- Tappy, L. Metabolic consequences of overfeeding in humans. Curr Opin Clin Nutr Metab Care 2004, 7(6), 623–8. [Google Scholar] [CrossRef]
- Yartsev, A. Complications of overfeeding the critically ill patient. 28/06/2015 Last updated 04/03/2025 – in. Available online: https://derangedphysiology.com/main/required-reading/gastrointestinal-intensive-care/Chapter-3121/consequences-malnutrition-critically-ill-patient (accessed on 27_02_2026).
- Cehan, V.D.; Cehan, A.R.; Pui, M.C.; Lazar, A. A New Perspective on Overfeeding in the Intensive Care Unit (ICU): Challenges, Dangers and Prevention Methods. Life (Basel) 2025, 15(5), 828. [Google Scholar] [CrossRef] [PubMed]
- Mehta, N.M.; Bechard, L.J.; Dolan, M.; Ariagno, K.; Jiang, H.; Duggan, C. Energy imbalance and the risk of overfeeding in critically ill children. Pediatr Crit Care Med 2011, 12(4), 398–405. [Google Scholar] [CrossRef] [PubMed]
- Mehanna, H.M.; Moledina, J.; Travis, J. Refeeding syndrome: what it is, and how to prevent and treat it. BMJ. 2008, 336(7659), 1495–8. [Google Scholar] [CrossRef] [PubMed]
- McGrath, K.; Culkin, A. Putting the PENG 2018 refeeding syndrome guidance into practice. Clinical Nutrition ESPEN 2023, 57, 844–845. [Google Scholar] [CrossRef]

| Study [Ref] | Title | Main Message |
|---|---|---|
| Cruz-Pierard, et al. 2026 [17] |
Synergistic effects of protein intake and resistance exercise. | Systematic review showing protein supplementation combined with resistance exercise enhances metabolic and anabolic biomarkers related to MPS, suggesting an interplay between nutrition and physical activity. |
| Prokopidis, et al. 2025 [18] |
Food matrix in the context of muscle and whole-body protein synthesis: a scoping review. | Highlights how the food matrix (nutrient interactions within whole foods) may influence MPS and whole-body protein synthesis beyond isolated protein dose. Limited data available, calling for future trials examining whole-food context. |
| Coker, & Coker, 2025 [19] |
Dietary proteins, AAs and insulin resistance: mini review. | Discusses how dietary protein and EAAs (especially leucine) influence insulin sensitivity and metabolic regulation, balancing anabolic stimulus with risks of chronic mTOR activation under nutrient overload. |
| Yimam, et al. 2025 [20] |
Postprandial aminoacidemia after alternative protein sources. | Focuses on postprandial AAs kinetics following ingestion of alternative proteins, underlining differences in AAs availability that can modulate metabolic responses and protein turnover. |
| Matthews, et al. 2025 [21] |
Understanding dietary protein quality. | Reviews methods to assess dietary protein quality emphasizing how quality metrics relate to digestibility, EAAs content, and metabolic impacts including protein synthesis stimulation. |
| Nutritional strategy |
Total caloric intake % E.E. |
Protein intake g/kg/day |
Effect on N.B. | Effect on LBM |
Predominant mechanisms |
[Ref.] |
|---|---|---|---|---|---|---|
| Very low calories + low protein |
<50% | <1.0 | Strongly negative | Rapid loss | ↑ gluconeogenesis from AA, ↑ proteolysis, ↓ protein synthesis |
[59,81] |
| Low calories + high protein |
<60–70% | ≥1.5–2.0 | Negative | Partial preservation |
AAs oxidation for energy purposes, incomplete protein-sparing effect | [56,57] |
| Adequate calories + adequate protein |
70–100% | 1.2–1.5 | Moderately negative or neutral | Better preservation |
↓ AAs oxidation, ↓ gluconeogenesis, insulin effect |
[79,81] |
| Adequate calories + high protein |
70–100% | 1.5–2.0 | Neutral or slightly positive | Maximum possible preservation (Stable phase) |
↓ proteolysis, ↑ protein synthesis (Limited by anabolic resistance) |
[56,82] |
| High calories + Very high protein |
>110–120% | ≥2.0 | Variable | No additional benefits |
Overfeeding, lipogenesis, ↑ metabolic stress |
[83,84] |
| Study [Ref] | Population | Study design |
Energy intake | Protein Intake g/kg/day |
Comparator | Outcome on LBM | Key findings |
|---|---|---|---|---|---|---|---|
| Stein, et al. 2024. [87] |
Obese | Additional protein intake in preservation of LBM | Hypo | 1.5 |
Lower intake (1 g/kg/day) |
significantly reduced in both groups | no differences in weight loss between the groups |
| Nunes, et al., 2022 [88] |
healthy adult (18 years or older) |
Randomized controlled trial | Normo | 1.2–1.59 and >1.6 |
Placebo or no intervention | increasing daily protein ingestion may enhance gains in LBM in studies enrolling subjects in RE | increasing protein ingestion results in small additional gains in LBM and lower body muscle strength gains. |
| Arends, et al. 2017 [89] |
Cancer patients | Clinical guidelines (ESPEN) | Normo/ Hyper |
1.2–1.5 | Inadequate intake | Partial preservation of LBM | Energy adequacy required to overcome anabolic resistance |
| Longland, et al., 2016 [90] |
Young man | Single-blind, randomized, parallel-group trial. RE training with high-intensity interval training. |
Hypo | 2.4 | Lower intake (1.2 g/kg/day) |
LBM increased in the higher protein group and loss of fat mass. | High protein diet was more effective in promoting increases in LBM and losses of fat mass when combined with high intensity RE and anaerobic exercise. |
| Weijs, et al. 2014 [81] |
ICU patients | Observational cohort | ≥80% measured energy expenditure | ≥1.2 | Lower intake | Reduced mortality and muscle loss | Best outcomes when energy and protein targets met together |
| Casaer, et al. 2011 [83] |
ICU patients | Randomized controlled trial | Early vs late parenteral nutrition | ~1.2 | Early high-calorie PN | Less muscle wasting with delayed calories | Early full calories blunt benefits of protein in acute phase |
| Villet, et al. 2005 [91] |
ICU patients | Prospective observational study | Hypo | ~1.0 | Adequate energy/protein | Progressive LBM loss | Energy deficit strongly associated with loss of fat-free mass |
| Paddon-Jones, et al. 2004 [92] |
Healthy adults (catabolic model) | Controlled feeding trial | Normo | High-quality protein + EAAs | Lower protein | Improved muscle protein synthesis | Adequate energy enhances anabolic response to protein |
| Wolfe, et al. 2000 [93] |
Critically ill patients | Narrative review of metabolic studies | Iso/ Hyper |
≥1.5 | Hypocaloric, low protein | Partial preservation of LBM | Energy–protein synergy limits endogenous protein oxidation |
| Wolfe, et al. 1983 [94] |
Severe burn patients | Metabolic balance study | Hyper | 2.0–2.5 | Lower protein intake | Improved nitrogen balance; reduced LBM loss | Adequate calories are required for protein to exert anabolic effects |
| Study [Ref] | Study design |
Study objective | Main findings |
|---|---|---|---|
| Hu, et al., 2025 [95] |
SR+MA | To evaluate the association between different levels of protein intake and renal adverse events and mortality in critically ill patients | Protein intake >1.3 g/kg/day was not associated with an increased risk of renal adverse events; no significant differences in 28-day, ICU, or hospital mortality were observed |
| Castro, et al. 2025 [96] |
SR+MA | To assess the impact of different protein intakes in patients with chronic critical illness | Higher protein intake (>1.3 g/kg/day) was associated with reduced early mortality, with no effect on late mortality or other clinical outcomes |
| Badpeyma, et al., 2025(1) [97] |
MA | Evaluating dose-response between high vs low protein in ICU patients | No significant effect on mortality or length of stay; reduction in muscle atrophy with high protein intake |
| Badpeyma, et al., 2025(2) [98] |
RCT | Comparing high protein (2.2 g/kg/day) vs conventional (1.0 g/kg/day) on mortality and clinical outcomes | High protein dose showed no clear improvement in mortality or major outcomes, highlighting uncertainties about efficacy and safety |
| Mohamed, et al., 2025 [82] |
SR+MA | To systematically determine the effect of protein dose (high >2.2 g/kg/day vs low <1.2 g/kg/day) on skeletal muscle strength in critically ill patients | Significant difference in skeletal muscle strength with higher vs lower protein intakes. Inconsistency was evident across the included studies. |
| O’Keefe, et al., 2025 [99] |
RCT | To test the hypothesis that supplementing enteral protein intake (2 g/kg/day vs standard nutritional care) would improve outcomes. | Protein supplementation did not improve outcomes but was associated with increased complications |
| Wang, et al., 2024 [100] |
RCT | To evaluate whether early high protein intake (1.5 g/kg/day) improves prognosis compared with low intake (0.8 g/kg/day) | No significant reduction in 28-day mortality; potential favorable effects on muscle preservation and duration of mechanical ventilation were reported |
| Qin, et al., 2024 [101] |
SR+MA | To compare higher (≥1.2 g/kg/day) vs lower (<1.2 g/kg/day) protein intake on clinical outcomes in adult ICU patients | No significant differences were found in mortality, ICU or hospital length of stay, duration of mechanical ventilation, or incidence of acute kidney injury |
| Blaauw, et al., 2024 [102] |
SR | To compare a protein intake group (≥1.2 g/kg/day) with a lower protein intake group (<1.2 g/kg/day) in critically ill adult patients on mortality, length of ICU and hospital stay. | Higher protein group results in little to no difference in mortality, with a probable slight increase in length of ICU stay and length of hospital stay. |
| Bels, et al., 2023 [57] | SR | Protein supplementation in ICU patients. | Protein supplementation has shown positive effects on recovery and mortality. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




