Submitted:
14 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Myrtus communis L. (common myrtle) is an economically valuable Mediterranean shrub with diverse applications in food, pharmaceutical, and ornamental sectors. However, the biochemical diversity of drought- and salt-resistant genotypes remains insufficiently characterized, particularly regarding the relationship between primary and secondary metabolism and stress adaptation. This study investigated the biochemical and aroma profiles of six drought-resistant myrtle genotypes from natural populations in Antalya, Turkey, to identify chemotypic diversity and elucidate metabolic strategies underlying abiotic stress tolerance. Volatile compounds were analyzed using HS-SPME/GC-MS, while sugars and organic acids were quantified by HPLC. Multivariate statistical analyses (PCA, hierarchical clustering) were employed to evaluate metabolic relationships and genotype classification. Three chemotypes were identified: (i) Eucalyptol-type (G34, G36) with 35-40% 1,8-cineole; (ii) α-Pinene-type (G15, G37) with elevated terpenes (15.7-20.5%) and high sugar content (11.9-12.4 g/100 ml); and (iii) Ester-aldehyde type (G9) characterized by dominant esters (30.4%) and negligible eucalyptol. Significant genotypic variation was observed across metabolite classes (p < 0.001, η² > 0.90). Hierarchical clustering revealed three metabolic strategies: volatile-focused antioxidant defense (Cluster 1), osmotic adjustment with chemical defense (Cluster 2), and specialized stress signaling (Cluster 3). These findings highlight substantial metabolic plasticity and provide a basis for targeted breeding and diverse industrial applications.
Keywords:
1. Introduction
2. Results
2.1. Volatile Component Profile
2.2. Sugar Contents
2.3. Organic Acids Content
2.4. Hierarchical Clustering of Biochemical Profiles
3. Discussion
3.1. Chemotypic Diversity and Volatile Compound Profiles in Drought-Resistant Myrtle Genotypes
3.2. Primary Metabolite Accumulation and Drought Adaptation Strategies
3.3. Metabolic Trade-Offs and Integrated Drought Tolerance Strategies
3.4. Implications for Myrtle Domestication and Functional Food Development
4. Materials and Methods
4.1. Plant Material
4.2. Analysis of Volatile Compounds Using HS-SPME/GC-MS
4.3. Analysis of Sugar and Organic Acid Profile by HPLC
4.3.1. Sugar Analyses
2.3.2. Organic Acid Analyses
2.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HS-SPME | Headspace solid-phase microextraction |
| GC-MS | Gas Chromatography-Mass Spectrometry |
| HPLC | High Performance Liquid Chromatography |
| PCA | Principal Component Analysis |
| MVA | Mevalonate |
| MEP | Methylerythritol phosphate |
| ROS | Reactive Oxygen Species |
| TCA | Tricarboxylic Acid |
| ABA | Abscisic acid |
| SnRK2 | Sucrose nonfermenting 1–related protein kinase 2 |
| AREB/ABF | ABRE-Binding Proteins/ABRE Binding Factors |
| P5CS | Δ¹-pyrroline-5-carboxylate synthetase |
| SOS1 | SOS Ras/Rac Guanine Nucleotide Exchange Factor 1 |
| NHX | Na+/H+ antiporters |
| APX | Ascorbate peroxidase |
| GR | Glutathione reductase |
| HSD | Honestly Significant Difference |
| ANOVA | Analysis of Variance |
| LLE | Liquid-liquid extraction |
| LDL | Low-Density Lipoprotein |
| CAR/PDMS | Carboxen/Polydimethylsiloxane |
| RID | Refractive Index Detection |
| dw | Dry weight |
References
- Zilkah, S.; Goldschmidt, E.E. Myrtle (Myrtus communis L.)—A native Mediterranean and cultivated crop species. In Medicinal and Aromatic Plants of the Middle-East; Springer: Dordrecht, The Netherlands, 2014; pp. 253–267. [Google Scholar]
- Dafni, A. Myrtle (Myrtus communis) as a ritual plant in the Holy Land—A comparative study in relation to ancient traditions. Econ. Bot. 2016, 70, 222–234. [Google Scholar] [CrossRef]
- Asgarpanah, J.; Ariamanesh, A. Phytochemistry and pharmacological properties of Myrtus communis L. Indian J. Tradit. Knowl. 2015, 1, 82–87. [Google Scholar]
- Aksay, S.; Çınar, F.; Arslan, R.; Karatay, H.E. Development of myrtle (Myrtus communis L.) fruit-based herbal tea: Physicochemical and sensory characterization. Turk. J. Agric.-Food Sci. Technol. 2025, 13, 3285–3294. [Google Scholar] [CrossRef]
- Abate, L.; Bachheti, A.; Bachheti, R.K.; Husen, A.; Getachew, M.; Pandey, D.P. Potential role of forest-based plants in essential oil production: An approach to cosmetic and personal health care applications. In Non-Timber Forest Products: Food, Healthcare and Industrial Applications; Springer: Cham, Switzerland, 2021; pp. 1–18. [Google Scholar]
- Medda, S.; Fadda, A.; Mulas, M. Selection for ornamental purposes of ‘Angela’ myrtle (Myrtus communis L.) cultivar with unpigmented fruit. Sustainability 2022, 14, 13210. [Google Scholar] [CrossRef]
- Usai, M.; Marchetti, M.; Culeddu, N.; Mulas, M. Chemical composition of myrtle (Myrtus communis L.) berries essential oils as observed in a collection of genotypes. Molecules 2018, 23, 2502. [Google Scholar] [CrossRef]
- Külheim, C.; Yeoh, S.H.; Wallis, I.R.; Laffan, S.; Moran, G.F.; Foley, W.J. The molecular basis of quantitative variation in foliar secondary metabolites in Eucalyptus globulus. New Phytol. 2011, 191, 1041–1053. [Google Scholar] [CrossRef]
- Chen, M.; Qiao, Y.; Quan, X.; Shi, H.; Duan, Z. Physiological, biochemical and phytohormone responses of Elymus nutans to α-pinene-induced allelopathy. PeerJ 2022, 10, e14100. [Google Scholar] [CrossRef]
- Ilc, T. Role of cytochromes P450 in wine aroma. Ph.D. Thesis, Université de Strasbourg, Strasbourg, France, 2015. [Google Scholar]
- El Hadi, M.A.M.; Zhang, F.J.; Wu, F.F.; Zhou, C.H.; Tao, J. Advances in fruit aroma volatile research. Molecules 2013, 18, 8200–8229. [Google Scholar] [CrossRef]
- Gorjian, H.; Khaligh, N.G. Myrtle: A versatile medicinal plant. Nutrire 2023, 48, 10. [Google Scholar] [CrossRef]
- Falchi, R.; Bonghi, C.; Drincovich, M.F.; Famiani, F.; Lara, M.V.; Walker, R.P.; Vizzotto, G. Sugar metabolism in stone fruit: Source–sink relationships and environmental and agronomical effects. Front. Plant Sci. 2020, 11, 573982. [Google Scholar] [CrossRef]
- Afzal, S.; Chaudhary, N.; Singh, N.K. Role of soluble sugars in metabolism and sensing under abiotic stress. In Plant Growth Regulators: Signalling under Stress Conditions; Springer: Cham, Switzerland, 2021; pp. 305–334. [Google Scholar]
- Panchal, P.; Miller, A.J.; Giri, J. Organic acids: Versatile stress-response roles in plants. J. Exp. Bot. 2021, 72, 4038–4052. [Google Scholar] [CrossRef]
- Ali, S.; Hayat, K.; Iqbal, A.; Xie, L. Implications of abscisic acid in the drought stress tolerance of plants. Agronomy 2020, 10, 1323. [Google Scholar] [CrossRef]
- Prakash, P.; Saini, D.R.; Jangde, S.; Kamlapuri, S.S.; Kumar, K. Role of osmoprotectants on alleviation of elevated CO₂ and temperature stress. In Climate Change; CRC Press: Boca Raton, FL, USA, 2025; pp. 199–214. [Google Scholar]
- Almeida, D.M.; Oliveira, M.M.; Saibo, N.J. Regulation of Na⁺ and K⁺ homeostasis in plants: Towards improved salt stress tolerance in crop plants. Genet. Mol. Biol. 2017, 40, 326–345. [Google Scholar] [CrossRef] [PubMed]
- Wojtunik-Kulesza, K.A.; Kasprzak, K.; Oniszczuk, T.; Oniszczuk, A. Natural monoterpenes: Much more than only a scent. Chem. Biodivers. 2019, 16, e1900434. [Google Scholar] [CrossRef] [PubMed]
- Migliore, J.; Baumel, A.; Juin, M.; Médail, F. From Mediterranean shores to central Saharan mountains: Key phylogeographical insights from the genus Myrtus. J. Biogeogr. 2012, 39, 942–956. [Google Scholar] [CrossRef]
- Tüzün-Kis, B.; İkten, H. Assessment of genetic variation in wild myrtle (Myrtus communis L.) genotypes growing around the Mediterranean region of Turkey. Appl. Ecol. Environ. Res. 2022, 20. [Google Scholar] [CrossRef]
- Şan, B.; Yıldırım, F.; Yıldırım, A.N. Mersin (Myrtus communis L.) bitkisinin biyoaktif bileşenleri. Bahçe 2016, 45, 185–193. [Google Scholar]
- Yaşa, B.; Genç, M.; Angın, N.; Ertaş, M. Farklı bölgelerde yetişen mersin (Myrtus communis L.) meyvelerinin bazı fitokimyasal özelliklerinin karakterizasyonu. Tarim Doga Derg. 2023, 26, 1230. [Google Scholar] [CrossRef]
- Tuberoso, C.I.; Barra, A.; Angioni, A.; Sarritzu, E.; Pirisi, F.M. Chemical composition of volatiles in Sardinian myrtle (Myrtus communis L.) alcoholic extracts and essential oils. J. Agric. Food Chem. 2006, 54, 1420–1426. [Google Scholar] [CrossRef]
- Barboni, T.; Venturini, N.; Paolini, J.; Desjobert, J.M.; Chiaramonti, N.; Costa, J. Characterisation of volatiles and polyphenols for quality assessment of alcoholic beverages prepared from Corsican Myrtus communis berries. Food Chem. 2010, 122, 1304–1312. [Google Scholar] [CrossRef]
- Serreli, G.; Jerković, I.; Gil, K.A.; Marijanović, Z.; Pacini, V.; Tuberoso, C.I.G. Phenolic compounds, volatiles and antioxidant capacity of white myrtle berry liqueurs. Plant Foods Hum. Nutr. 2017, 72, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Vafadar Shoshtari, Z.; Rahimmalek, M.; Sabzalian, M.R.; Hosseini, H. Essential oil and bioactive compounds variation in myrtle (Myrtus communis L.) as affected by seasonal variation and salt stress. Chem. Biodivers. 2017, 14, e1600365. [Google Scholar] [CrossRef] [PubMed]
- Fadda, A.; Mulas, M. Chemical changes during myrtle (Myrtus communis L.) fruit development and ripening. Sci. Hortic. 2010, 125, 477–485. [Google Scholar] [CrossRef]
- Mulas, M.; Fadda, A.; Angioni, A. Effect of maturation and cold storage on the organic acid composition of myrtle fruits. J. Sci. Food Agric. 2013, 93, 37–44. [Google Scholar] [CrossRef]
- Yılmaz, S. Myrtus communis L. meyvesinin bazı kimyasal ve antioksidatif özelliklerinin belirlenmesi. Master’s Thesis, Bursa Uludag University, Bursa, Turkey, 2020. [Google Scholar]
- Chidouh, A.; Aouadi, S.; Heyraud, A. Extraction, fractionation and characterization of water-soluble polysaccharide fractions from myrtle (Myrtus communis L.) fruit. Food Hydrocoll. 2014, 35, 733–739. [Google Scholar] [CrossRef]
- Tuberoso, C.I.G.; Orrù, C.D. Myrtle (Myrtus communis L.) berries: Composition and properties. In Berries: Properties, Consumption and Nutrition; Nova Science Publishers: New York, NY, USA, 2011; pp. 145–155. [Google Scholar]
- Alim, E.; Çınar, O.; Uysal Bayar, F.; Çetinay, Ş.; Bayır Yeğin, A.; Polat, S.; Yüksel, K.; Şimşek, M.; Erçik, K. Determination of myrtle (Myrtus communis L.) genotypes suitable for different uses. Project No. TA-GEM/TBAD/B/19/A7/P6/970, 2013.
- Kraujalytė, V.; Leitner, E.; Venskutonis, P.R. Characterization of Aronia melanocarpa volatiles by HS-SPME, SDE and GC-O methods. J. Agric. Food Chem. 2013, 61, 4728–4736. [Google Scholar] [CrossRef]
- Miron, D.; Schaffer, A.A. Sucrose phosphate synthase, sucrose synthase, and invertase activities in developing fruit of Lycopersicon esculentum Mill. and the sucrose accumulating Lycopersicon hirsutum Humb. and Bonpl. Plant Physiol. 1991, 95, 623–627. [Google Scholar] [CrossRef]
- Bozan, B.; Başer, K.H.C.; Kara, S. Quantitative determination of naphthaquinones of Arnebia densiflora by an improved HPLC method. J. Chromatogr. A 1997, 782, 133–136. [Google Scholar] [CrossRef]
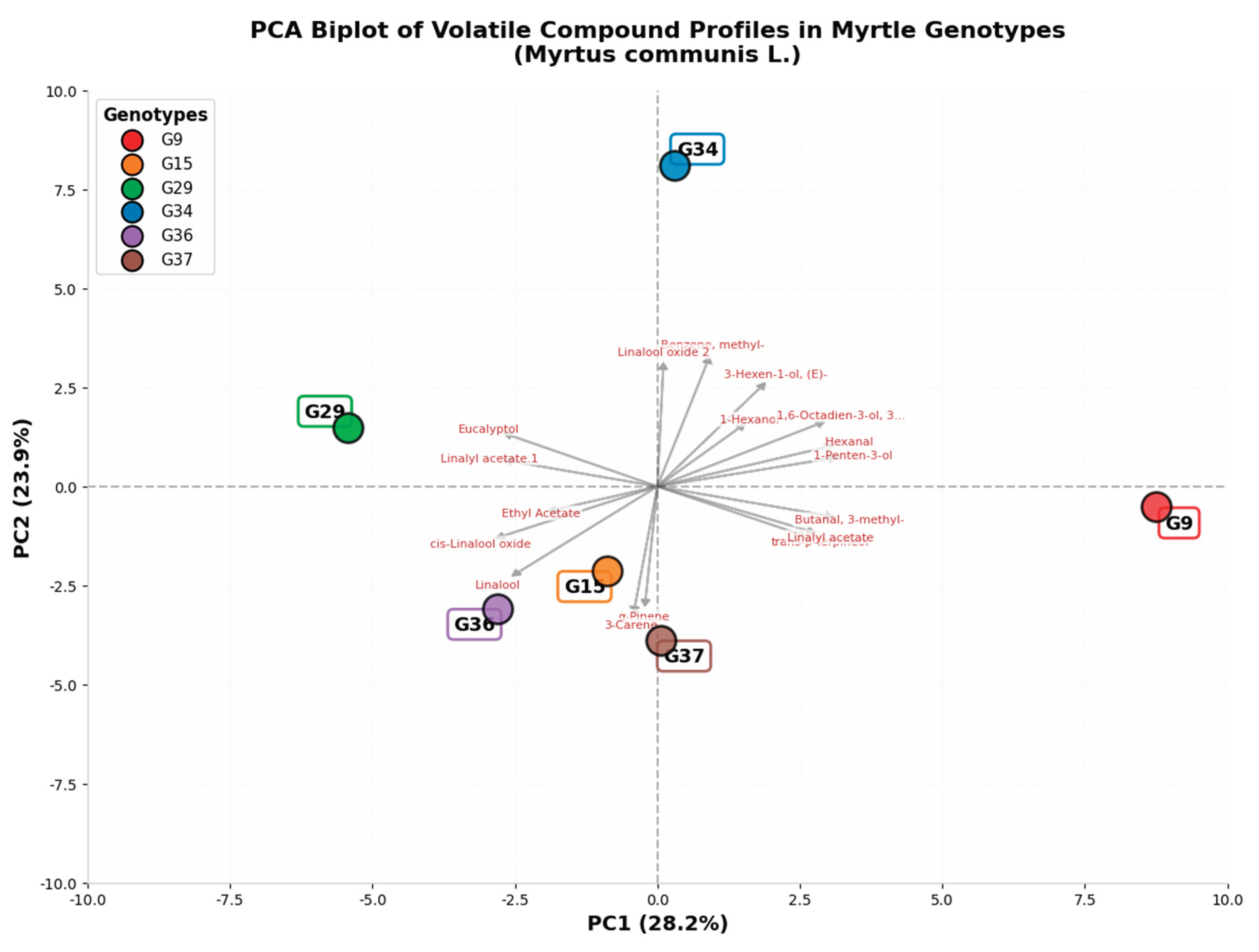
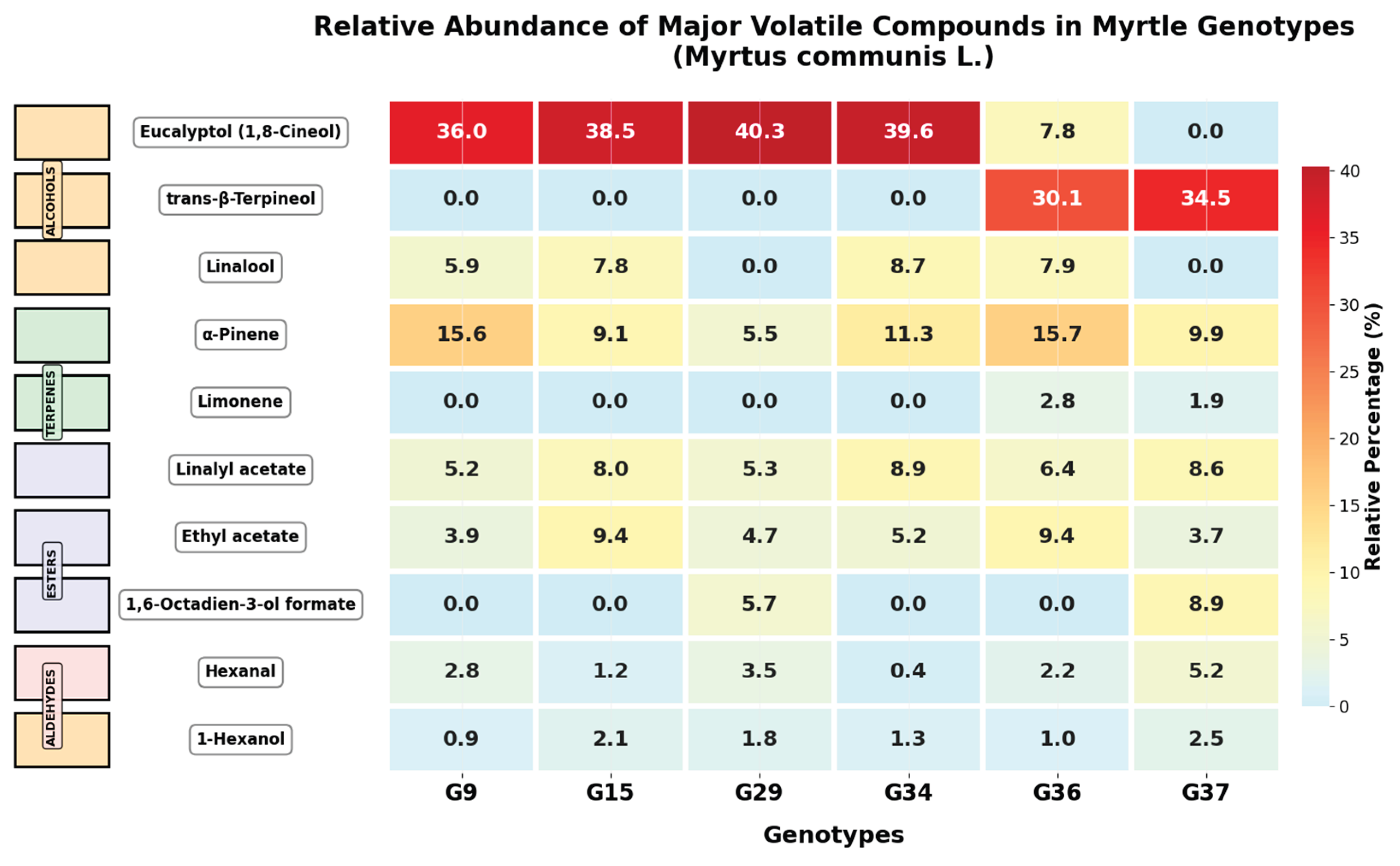
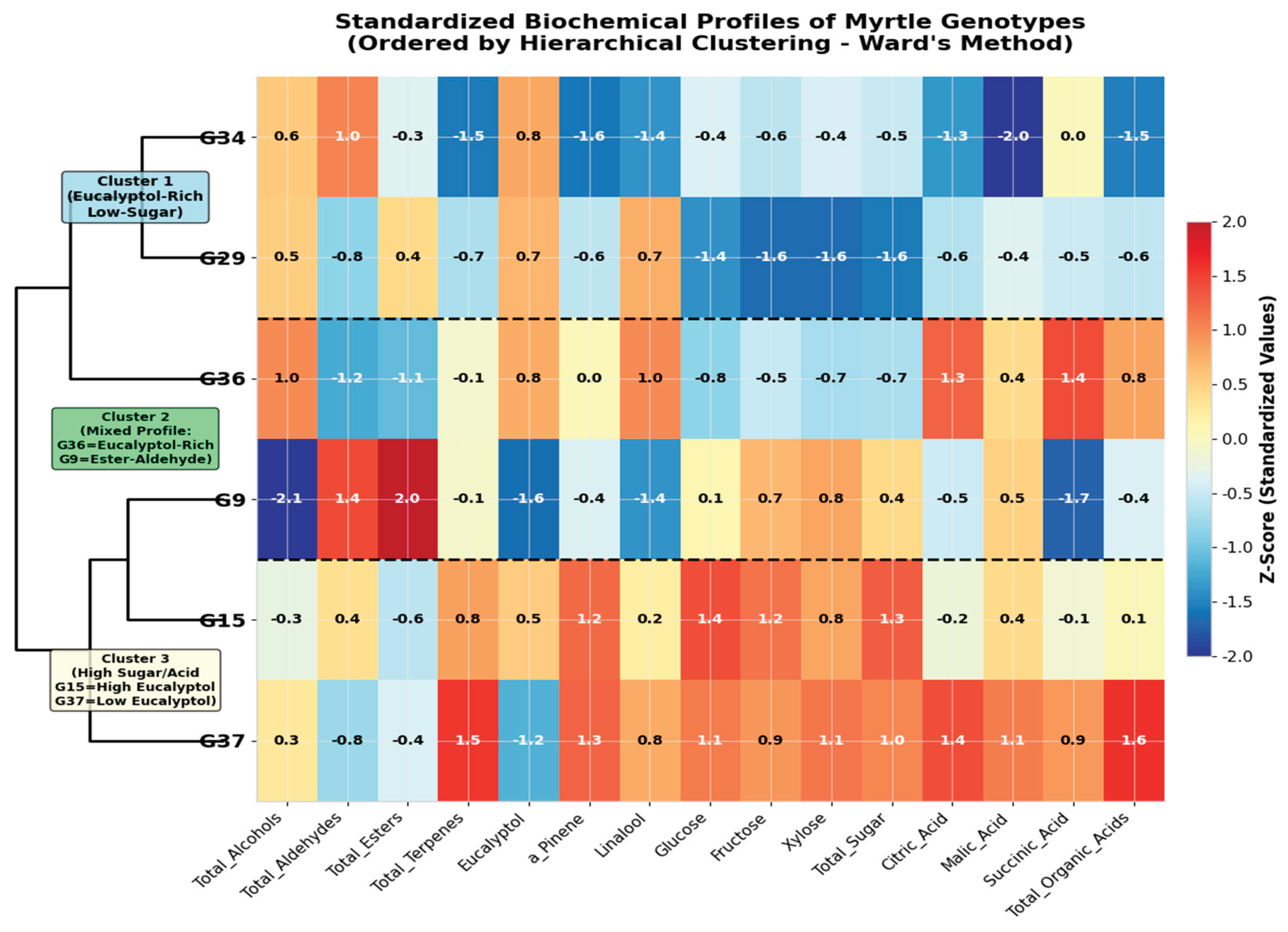
| Compound Group | Genotype 9 | Genotype 15 | Genotype 29 | Genotype 34 | Genotype 36 | Genotype 37 |
|---|---|---|---|---|---|---|
| Total Alcohols | 43.89 | 48.97 | 53.25 | 55.15 | 53.93 | 50.97 |
| Total Aldehydes | 7.04 | 4.38 | 2.08 | 5.67 | 1.38 | 2.45 |
| Total Esters | 30.40 | 23.13 | 26.98 | 24.43 | 24.05 | 25.34 |
| Total Terpenes | 13.74 | 19.10 | 11.64 | 7.74 | 15.24 | 20.50 |
| Total Ketones | 0.50 | 0.57 | 0.22 | 1.69 | 0.49 | 0.50 |
| Other Compounds | 4.43 | 3.85 | 4.98 | 5.32 | 4.55 | 0.24 |
| Parameter | Sum of Squares (Type III) | Mean Square | F-value | p-value | Partial η² |
|---|---|---|---|---|---|
| Glucose | 12857300.35 | 2571460.07 | 991.53 | < 0.001 | 0.998 |
| Fructose | 20407406.51 | 4081481.30 | 1638.36 | < 0.001 | 0.999 |
| Xylose | 179.85 | 35.97 | 304.76 | < 0.001 | 0.992 |
| Total Sugar | 64120238.62 | 12824047.73 | 1994.65 | < 0.001 | 0.999 |
| Sugar | Genotype 9 | Genotype 15 | Genotype 29 | Genotype 34 | Genotype 36 | Genotype 37 |
|---|---|---|---|---|---|---|
| Glucose | 4718.5 ± 28.5 c | 5819.1 ± 3.9 a | 3453.8 ± 37.3 f | 4314.9 ± 69.4 d | 3926.3 ± 86.2 e | 5571.3 ± 33.1 a |
| Xylose | 10.9 ± 0.2 b | 10.5 ± 0.3 b | 3.2 ± 0.3 e | 7.4 ± 0.4 c | 5.6 ± 0.2 d | 12.1 ± 0.6 a |
| Fructose | 6087.6 ± 72.3 c | 6588.7 ± 48.7 a | 3602.1 ± 4.0 e | 4728.6 ± 49.6 d | 4801.4 ± 40.5 d | 6341.0 ± 56.9 b |
| Total Sugar | 10817.0 ± 101.0 c | 12418.3 ± 52.7 a | 7059.1 ± 37.1 f | 9050.9 ± 60.5 d | 8733.3 ± 112.4 e | 11924.4 ± 89.1 b |
| Parameter | Sum of Squares (Type III) | Mean Square | F-value | p-value | Partial η² |
|---|---|---|---|---|---|
| Citric Acid | 83468.95 | 16693.79 | 51.61 | < 0.001 | 0.956 |
| Malic Acid | 257968.20 | 51593.64 | 35.29 | < 0.001 | 0.936 |
| Succinic Acid | 59123.68 | 11824.74 | 21.50 | < 0.001 | 0.900 |
| Total Organic Acids | 747187.56 | 149437.51 | 39.11 | < 0.001 | 0.942 |
| Organic Acid | Genotype 9 | Genotype 15 | Genotype 29 | Genotype 34 | Genotype36 | Genotype 37 |
|---|---|---|---|---|---|---|
| Citric Acid | 247.4 ± 14.0 b | 268.0 ± 20.6 b | 242.5 ± 19.8 b | 188.0 ± 4.2 c | 365.8 ± 4.5 a | 376.9 ± 29.8 a |
| Malic Acid | 804.4 ± 24.2 a | 791.9 ± 53.4 a | 681.5 ± 18.3 b | 500.2 ± 4.1 c | 785.9 ± 46.9 ab | 867.0 ± 52.7 a |
| Succinic Acid | 720.7 ± 24.4 d | 808.1 ± 33.7 bc | 794.7 ± 8.8 c | 820.9 ± 17.0 bc | 902.6 ± 12.9 a | 867.4 ± 32.2 ab |
| Total Organic Acids | 1772.5 ± 29.5 b | 1867.9 ± 78.2 b | 1718.7 ± 16.7 b | 1509.1 ± 24.2 c | 2054.3 ± 52.2 a | 2111.4 ± 111.2 a |
| Cluster | Genotypes | Key Characteristics (Z-scores) | Proposed Drought Strategy |
|---|---|---|---|
| Cluster 1 (Eucalyptol-Rich) | G29, G34, G36 | High Eucalyptol (+0.75), High Alcohols (+0.82), Low Sugars (-0.91) | Volatile-focused antioxidant defense |
| Cluster 2 (High Sugar/Terpene) | G15, G37 | High Glucose (+1.26), High Terpenes (+1.19), High Total Sugar (+1.15) | Osmotic adjustment with chemical defense |
| Cluster 3 (Ester-Dominant) | G9 | High Esters (+1.94), High Aldehydes (+1.57), Low Eucalyptol (-1.63) | Specialized stress signaling |
| Gen. No | Location Name | Altitude (m) | Fruit Color | Grafted/Natural | Irrigation | Vegetation | Plant Habitus |
|---|---|---|---|---|---|---|---|
| 9 | Antalya-Kalkan-İslamlar 3 | 278.0 | White | Grafted | No | Maquis, Red Pine | 4 |
| 15 | Antalya-Finike-Yeşilyurt 1 | 1.0 | White | Grafted | Yes | Small Garden | 3 |
| 29 | Antalya-Serik-Yumaklar | 403.6 | White | Grafted | Yes | Olive Grove | 4 |
| 34 | Antalya-Serik-Çetince 2 | 121.0 | White | Grafted | Yes | Garden | 5 |
| 36 | Muğla-Fethiye-Seydikemer-Kocaçınar | 120.0 | White | Grafted | Yes | Garden | 3 |
| 37 | Muğla-Fethiye-Seydikemer-Döver | 118.0 | White | Grafted | Yes | Garden | 2 |
| Gen. No | Fruit Weight (g) | Fruit Width (mm) | Fruit Length (mm) | Calyx Diameter (mm) | Fruit Stalk Length (mm) | Seed Number (count) | Seed Classification | Germinated Seed Count | Total Seed Weight (g) | Seed Ratio (%) |
|---|---|---|---|---|---|---|---|---|---|---|
| 9 | 1.32 | 12.49 | 14.93 | 5.34 | 23.46 | 16.60 | Multi-seeded | 0.00 | 0.11 | 8.33 |
| 15 | 1.46 | 13.18 | 16.69 | 5.47 | 20.70 | 26.50 | Multi-seeded | 0.70 | 0.16 | 10.96 |
| 29 | 1.28 | 12.85 | 15.84 | 4.46 | 16.20 | 21.20 | Multi-seeded | 0.10 | 0.14 | 10.94 |
| 34 | 1.36 | 12.85 | 17.14 | 5.12 | 20.15 | 24.10 | Multi-seeded | 0.20 | 0.14 | 10.29 |
| 36 | 1.18 | 12.19 | 15.19 | 4.93 | 13.56 | 18.60 | Multi-seeded | 0.20 | 0.11 | 9.32 |
| 37 | 1.48 | 13.56 | 16.55 | 5.39 | 16.83 | 18.60 | Multi-seeded | 0.00 | 0.14 | 9.46 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




