Submitted:
14 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Experimental Methods
2.1. Preparation of Phosphors
2.2. WLEDs Packaging and Aging
2.3. Characterizations
3. Results and Discussion
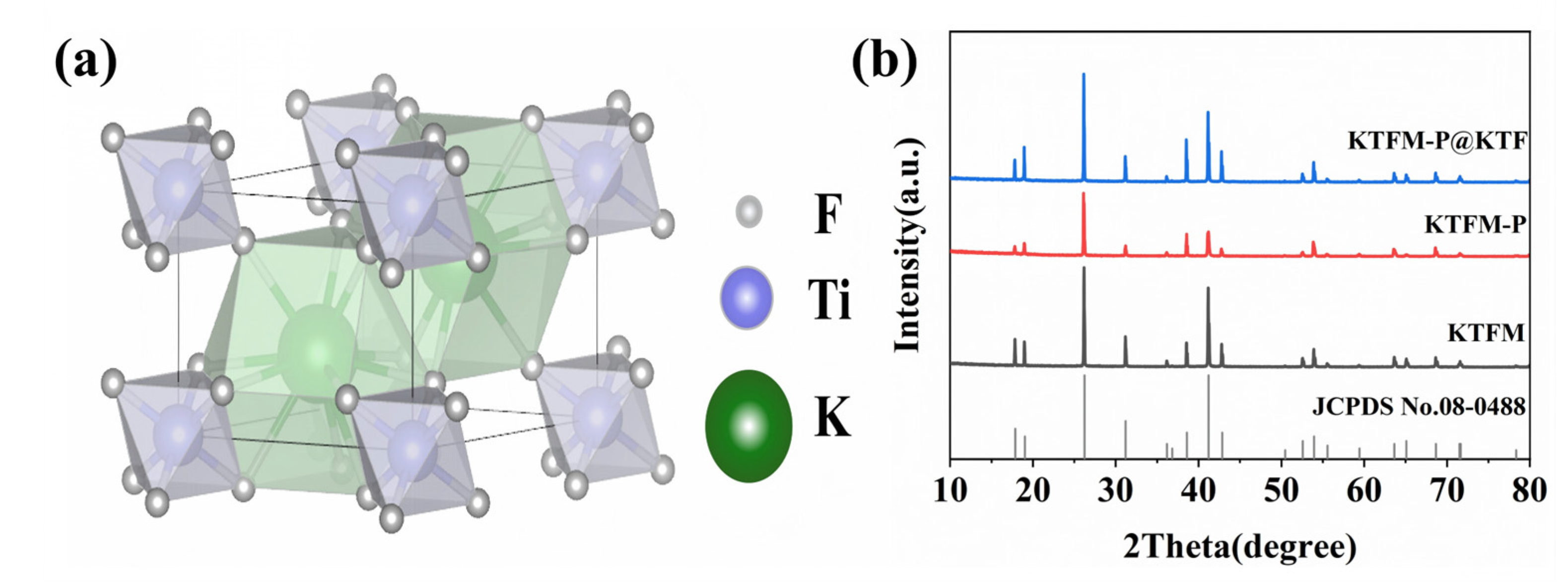
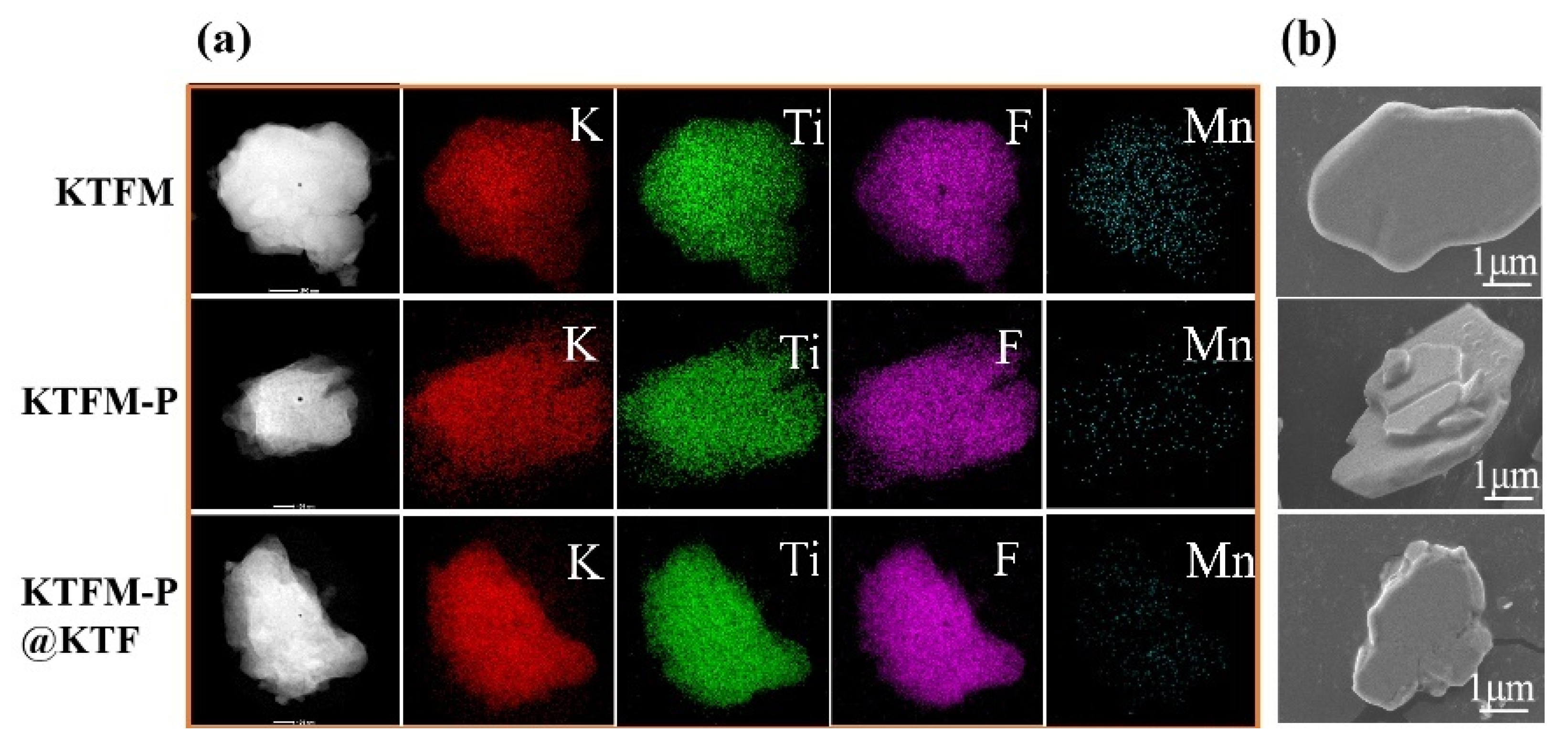
3.1. Structure, Morphology and Composition Analysis
3.2. Structure, Morphology and Composition Analysis
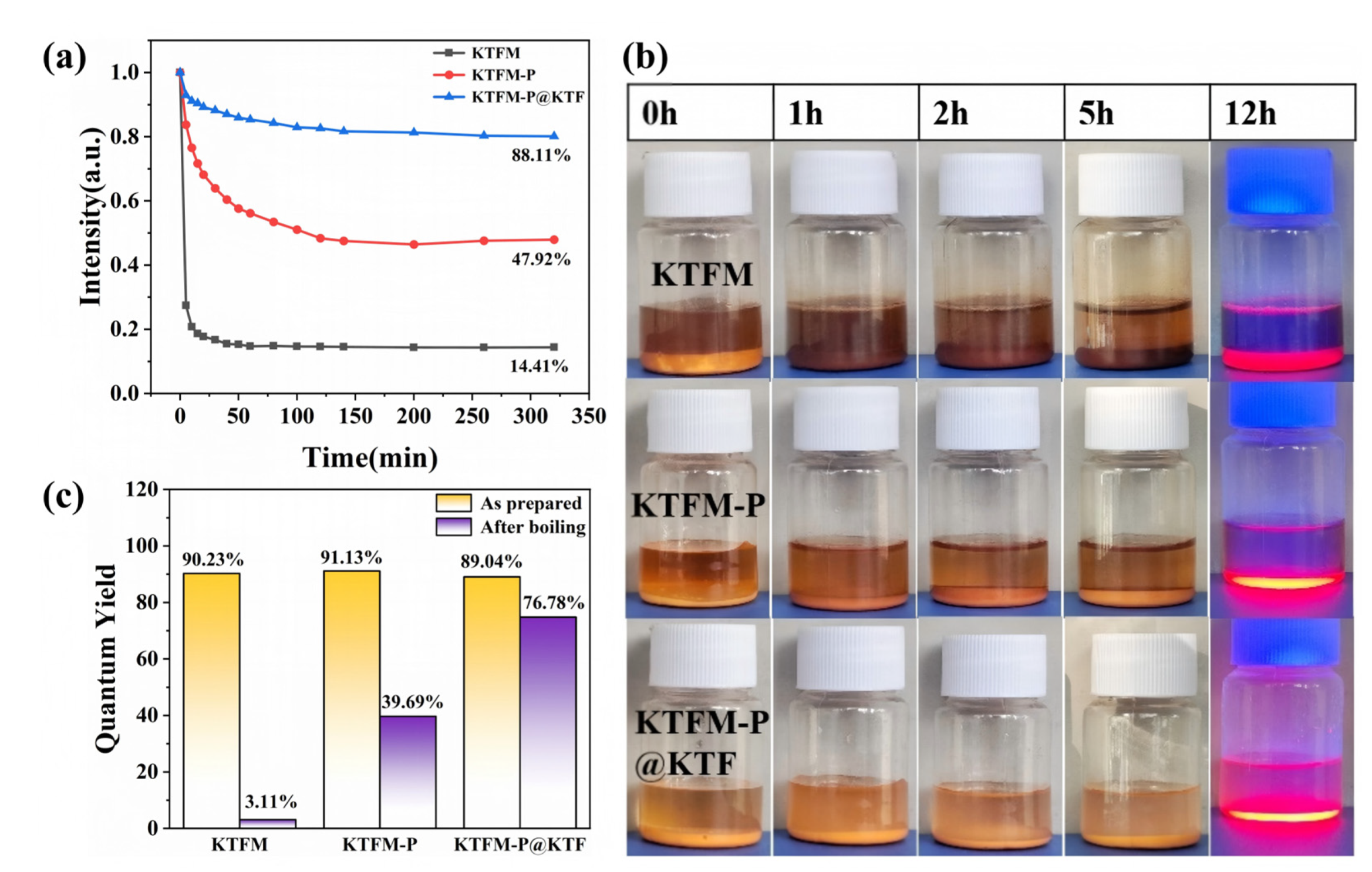
3.3. Moisture Resistance Mechanism
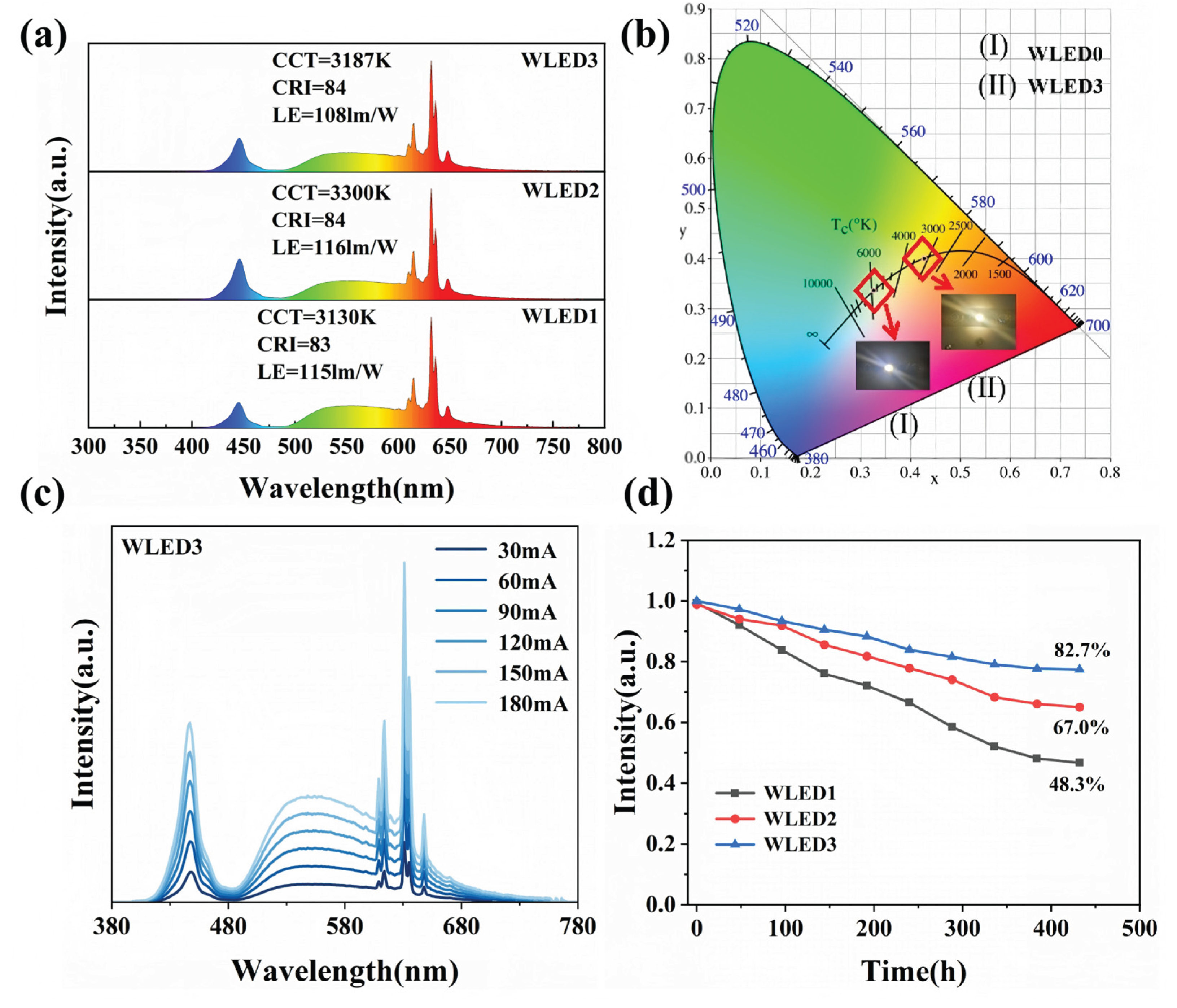
3.4. WLEDs Aging Test
4. Conclusion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| WLEDs | white light-emitting diodes |
| IQY | internal quantum yield |
| OP | alkyl phosphate |
| ODTMS | octadecyltrimethoxysilane |
| OA | oleic acid |
| CA | citric acid |
| EL | electroluminescence |
| YAG | Y3Al5O12:Ce3+ |
| XRD | X-ray diffraction |
| SEM | scanning electron microscopy |
| EDS | energy dispersive X-ray spectroscopy |
| TEM | transmission electron microscope |
| XPS | X-ray photoelectric spectrum |
| PL | photoluminescence |
| PLE | photoluminescence excitation |
| LE | luminous efficacy |
| CIE | International Commission on Illumination |
| CCT | correlated color temperature |
| CRI | color-rendering index |
| CQS | color quality scale |
References
- Zhang, H.; Zhang, H.; Pan, A.; et al. Rare earth-free luminescent materials for WLEDs: Recent Progress and Perspectives. Adv. Mater. Technol. 2021, 6, 2000648. [Google Scholar] [CrossRef]
- Yu, Y.; Wang, T.; Zhong, X.; et al. High Luminescent Thermal Stability and Water Resistance of K2SiF6:Mn4+@CaF2 Red Emitting Phosphor. Ceram. Int. 2021, 47, 33172–33179. [Google Scholar] [CrossRef]
- Yang, J.; Luo, P.; Wan, P.; et al. Surface engineered environment-stable red-emitting fluorides for white light emitting diodes. Ceram. Int. 2022, 48, 5009–5016. [Google Scholar] [CrossRef]
- Qiang, J.; Wang, L.; Wang, T.; et al. Improvement of the luminescent thermal stability and water resistance of K2SiF6:Mn4+ by surface passivation. Ceram. Int. 2022, 48, 17253–17260. [Google Scholar] [CrossRef]
- Zhou, Q.; Tan, H.; Zhou, Y.; et al. Optical performance of Mn4+ in a new hexa-coordinated fluorozirconate complex of Cs2ZrF6. J. Mater. Chem. C 2016, 4, 7443–7448. [Google Scholar] [CrossRef]
- Song, E.H.; Wang, J.Q.; Ye, S.; et al. Room-temperature synthesis and warm-white LED applications of Mn4+ ion doped fluoroa-luminate red phosphor Na3AlF6: Mn4+. J. Mater. Chem. C 2016, 4, 2480–2487. [Google Scholar] [CrossRef]
- Lian, H.; Huang, Q.; Chen, Y.; et al. Resonance emission enhancement (REE) for narrow band red-emitting A2GeF6: Mn4+ (A= Na, K, Rb, Cs) phosphors synthesized via a precipitation-cation exchange route. Inorg. Chem. 2017, 56, 11900–11910. [Google Scholar] [CrossRef] [PubMed]
- Ming, H.; Liu, L.; He, S.; et al. An ultra-high yield of spherical K2NaScF6: Mn4+ red phosphor and its application in ultra-wide color gamut liquid crystal displays. J. Mater. Chem. C 2019, 7, 7237–7248. [Google Scholar] [CrossRef]
- Zhou, Q.; Zhou, Y.; Liu, Y.; et al. A new and efficient red phosphor for solid-state lighting: Cs2TiF6: Mn4+. J. Mater. Chem. C 2015, 3, 9615–9619. [Google Scholar] [CrossRef]
- Arai, Y.; Adachi, S. Optical properties of Mn4+-activated Na2SnF6 and Cs2SnF6 red phosphors. J. Lumin. 2011, 131, 2652–2660. [Google Scholar] [CrossRef]
- Suehiro, T.; Xie, R.-J.; Hirosaki, N. Gas-reduction- nitridation synthesis of CaAlSiN3: Eu2+ fine powder phosphors for solid-state lighting. Ind. Eng. Chem. Res. 2014, 53, 2713–2717. [Google Scholar] [CrossRef]
- Liu, Y.-X.; Hu, J.-X.; Ju, L.-C.; et al. Hydrophobic surface modification toward highly stable K2SiF6: Mn4+ phosphor for white light-emitting diodes. Ceram. Int. 2020, 46(7), 8811–8818. [Google Scholar] [CrossRef]
- Arunkumar, P.; Kim, Y.H.; Kim, H.J.; et al. Hydrophobic organic skin as a protective shield for moisture-sensitive phosphor-based optoelectronic devices. ACS Appl. Mater. Interfaces 2017, 9, 7232–7240. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.-Y.; Song, E.-H.; Deng, T.-T.; et al. Waterproof narrow-band fluoride red phosphor K2TiF6: Mn4+ via facile superhydrophobic surface modification. ACS Appl. Mater. Interfaces 2018, 10, 880–889. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.; Lin, C.; Liu, R.-S. Waterproof alkyl phosphate coated fluoride phosphors for optoelectronic materials. Angew. Chem. Int. Ed. 2015, 127, 11012–11016. [Google Scholar] [CrossRef]
- Huang, L.; Liu, Y.; Si, S.; et al. A new reductive dl-mandelic acid loading approach for moisture-stable Mn4+ doped fluorides. Chem. Commun. 2018, 54, 11857–11860. [Google Scholar] [CrossRef]
- Yu, Y.; Jiang, C.; Chang, G.; et al. Preparation of red phosphor K2SiF6:Mn4+ with high water-resistant and negative thermal quenching effect induced by citric acid-passivation. J. Lumin. 2024, 270, 0022–2313. [Google Scholar] [CrossRef]
- Huang, D.; Zhu, H.; Deng, Z.; et al. Moisture-resistant Mn4+-doped core-shell-structured fluoride red phosphor exhibiting high luminous efficacy for warm white light-emitting diodes. Angew. Chem. Int. Ed. 2019, 58, 3843–3847. [Google Scholar] [CrossRef]
- Zhu, H.; Lin, C.; Luo, W.; et al. Highly efficient non-rare-earth red emitting phosphor for warm white light-emitting diodes. Nat. Commun. 2014, 5, 4312. [Google Scholar] [CrossRef]
- Zhang, Y.; Ran, Q.; Wang, Q.; et al. High-efficiency red organic light-emitting diodes with external quantum efficiency close to 30% based on a novel thermally activated delayed fluorescence emitter. Adv. Mater. 2019, 31, 1902368. [Google Scholar] [CrossRef]
- Huang, L.; Zhu, Y.; Zhang, X.; et al. HF-free hydrothermal route for synthesis of highly efficient narrow-band red emitting phosphor K2Si1-xF6:xMn4+ for warm white light-emitting diodes. Chem. Mater. 2016, 28, 1495–1502. [Google Scholar] [CrossRef]
- Liu, L.; Wu, D.; He, S.; et al. A Reverse Strategy to Restore the Moisture-deteriorated Luminescence Properties and Improve the Humidity Resistance of Mn4+-doped Fluoride Phosphors. Chem. Asian J. 2020, 15, 3326–3337. [Google Scholar] [CrossRef]
- Jiang, C.; Li, L.; Brik, M.G.; et al. Epitaxial growth via anti-solvent-induced deposition towards a highly efficient and stable Mn4+ doped fluoride red phosphor for application in warm WLEDs. J. Mater. Chem. C 2019, 7, 6077–6084. [Google Scholar] [CrossRef]
- Qiao, J.; Ning, L.; Molokeev, M.S.; et al. Eu2+ site preferences in the mixed cation K2BaCa(PO4)2 and thermally stable luminescence. J. Am. Chem. Soc. 2018, 140, 9730–9736. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Wang, L.; Zhou, T.; et al. Moisture-induced degradation and its mechanism of (Sr, Ca)AlSiN3: Eu2+, a red-color- converter for solid state lighting. J. Mater. Chem. C 2015, 3, 3181–3188. [Google Scholar] [CrossRef]

| Sample | C1s | O1s | F1s | K2p | Ti2p |
| KTFM | 33.44 | 13.87 | 38.42 | 9.84 | 4.43 |
| KTFM-P | 32.84 | 11.05 | 40.80 | 10.47 | 4.84 |
| KTFM-P@KTF | 30.36 | 6.57 | 45.22 | 12.53 | 5.32 |
| WLEDs | LE(lm/W) | CCT(K) | CRI | CQS |
| WLED0 | 145.5 | 5771.97 | 56.23 | 64.6 |
| WLED1 | 115.3 | 3130.12 | 83.11 | 83.1 |
| WLED2 | 116.1 | 3300.05 | 84.11 | 80.4 |
| WLED3 | 108.8 | 3187.91 | 84.86 | 81.7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




