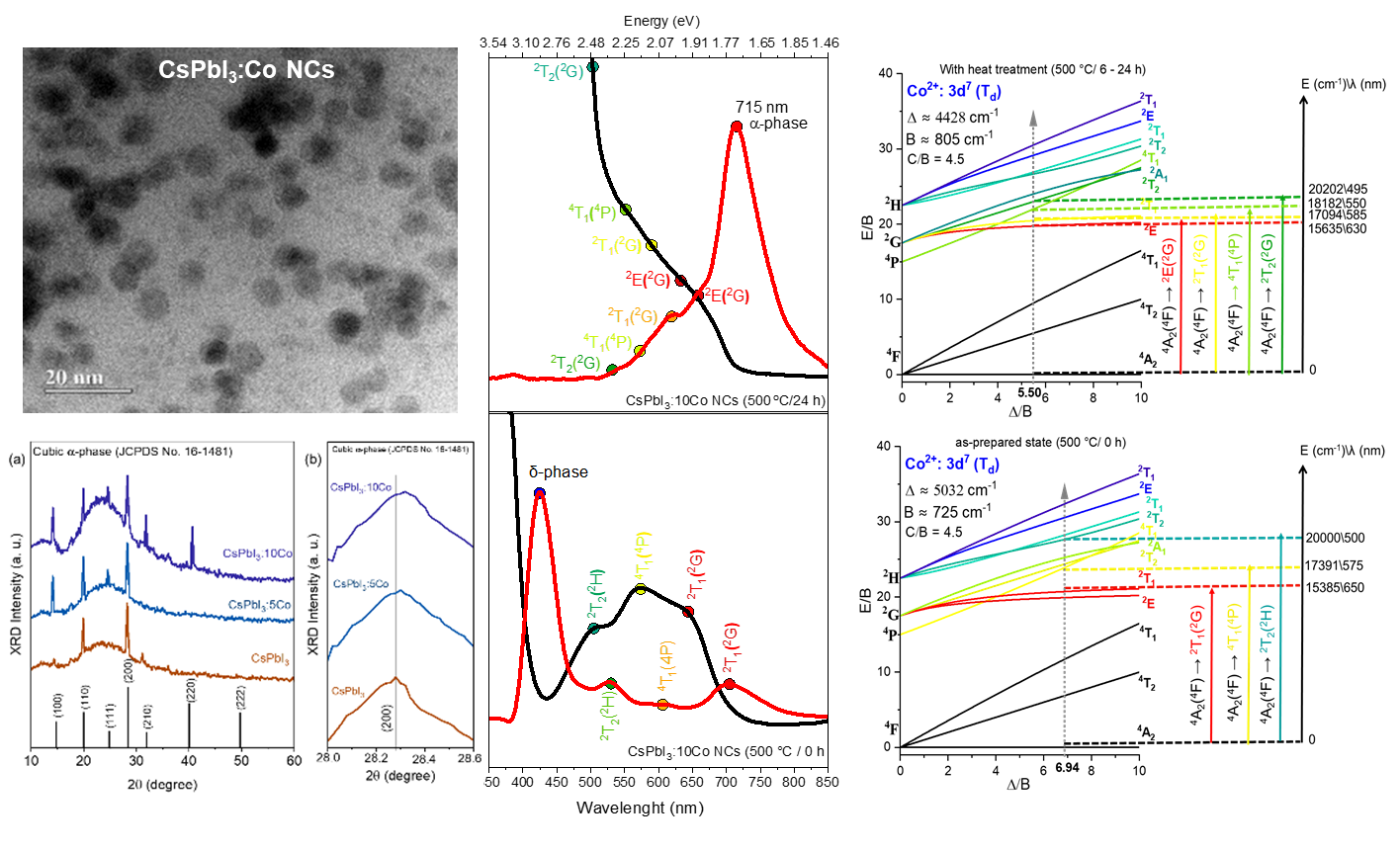
Co²⁺-doped CsPbI₃ nanocrystals (NCs) (CsPbI₃:xCo, x = 0, 0.05, and 0.10 mol%) were synthesized in situ within a borosilicate glass matrix by the fusion method followed by controlled thermal treatment at 500 °C for 6–24 h. Transmission electron microscopy images showed quasi-spherical NCs formed within the glass matrix with mean diameters ranging from 6.3 to 8.4 nm, consistent with diffusion-mediated growth under glass nanoconfinement. Energy-dispersive X-ray spectroscopy confirmed the presence of Co within the NCs regions. X-ray diffraction patterns confirm the exclusive stabilization of the cubic α-phase across all compositions, with a systematic shift of the (200) reflection to greater angles with increasing Co content, evidencing B-site lattice contraction due to the substitution of Pb²⁺ by Co²⁺. Optical absorption and photoluminescence spectra demonstrate that Co²⁺ incorporation enhances the δ-phase emission in the as-prepared condition, consistent with defect passivation, and that progressive thermal treatment stabilizes band-edge excitonic emission near ~1.74 eV without disrupting the fundamental optical response of the α-CsPbI₃ host. Crystal field theory and Tanabe–Sugano analysis for d⁷ ions in Td symmetry yielded Δ = 5032 cm⁻¹ and B = 725 cm⁻¹ in the as-prepared state, evolving to Δ = 4428 cm⁻¹ and B = 805 cm⁻¹ after thermal treatment, confirming the tetrahedral coordination of Co²⁺ and significant metal–iodide covalency. These results position CsPbI₃:xCo NCs embedded in glass as robust platforms for glass-integrated photonic applications.