Submitted:
13 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
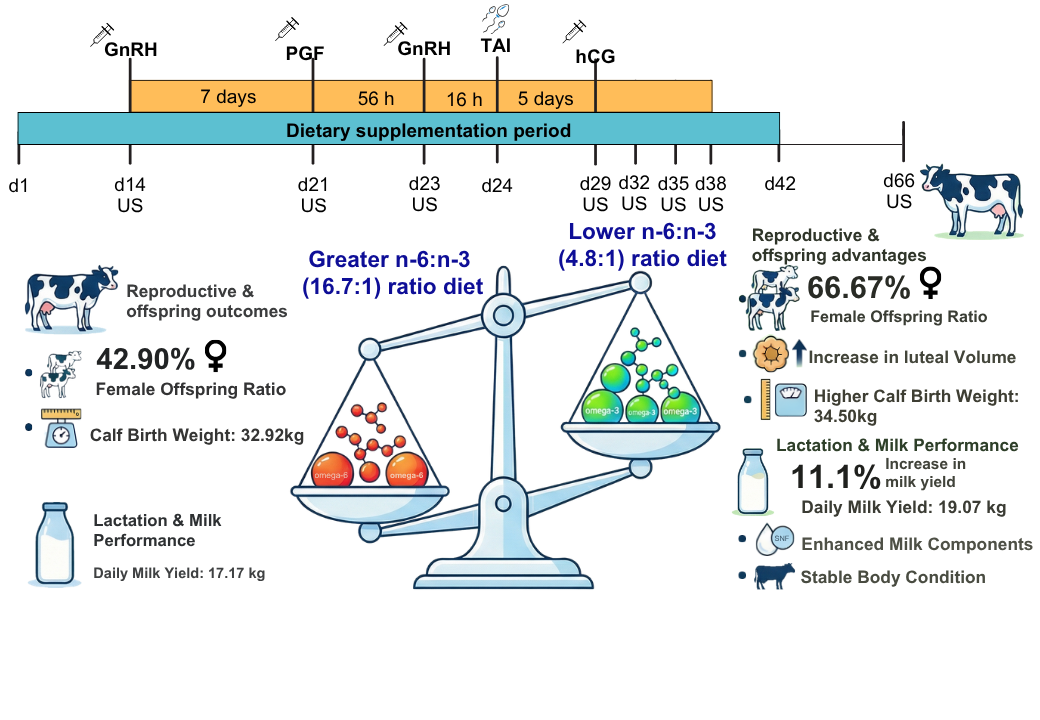
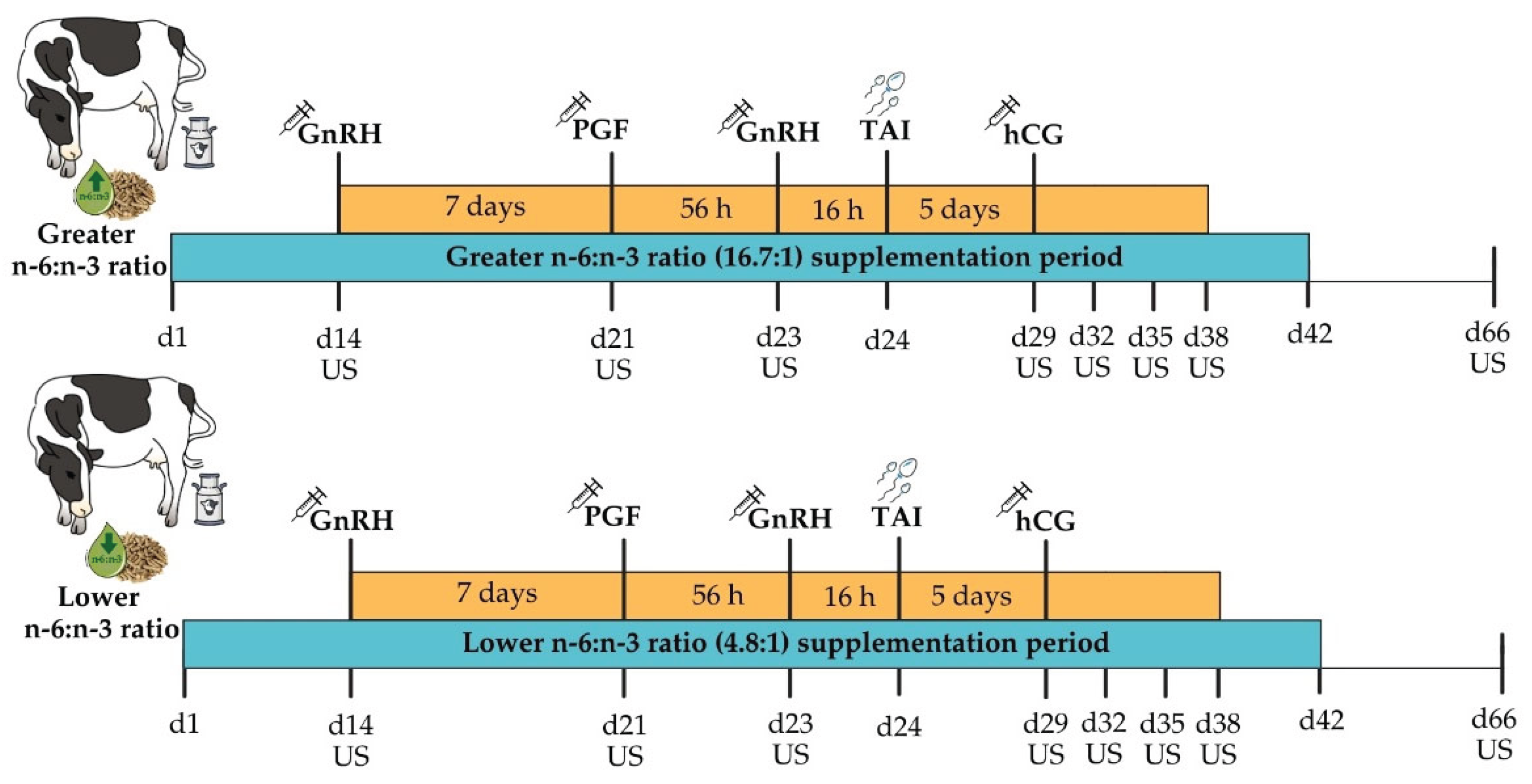
2.1. Animal Ethics and Experimental Design
2.2. Reproductive Management
2.3. Data and Sample Collection
2.4. Statistical Analysis
3. Results
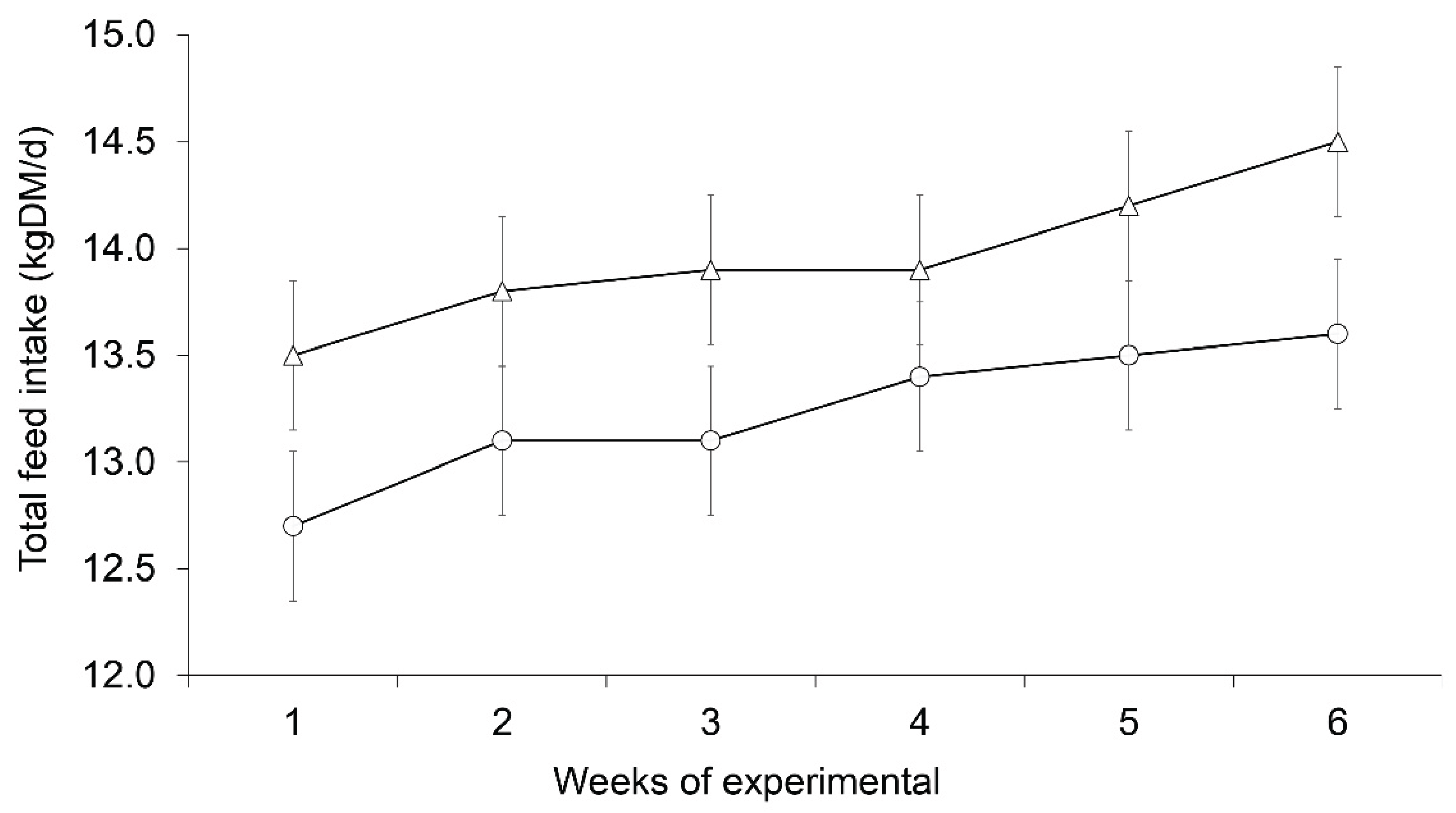
3.1. Feed intake, Body Weight, and Body Condition Score
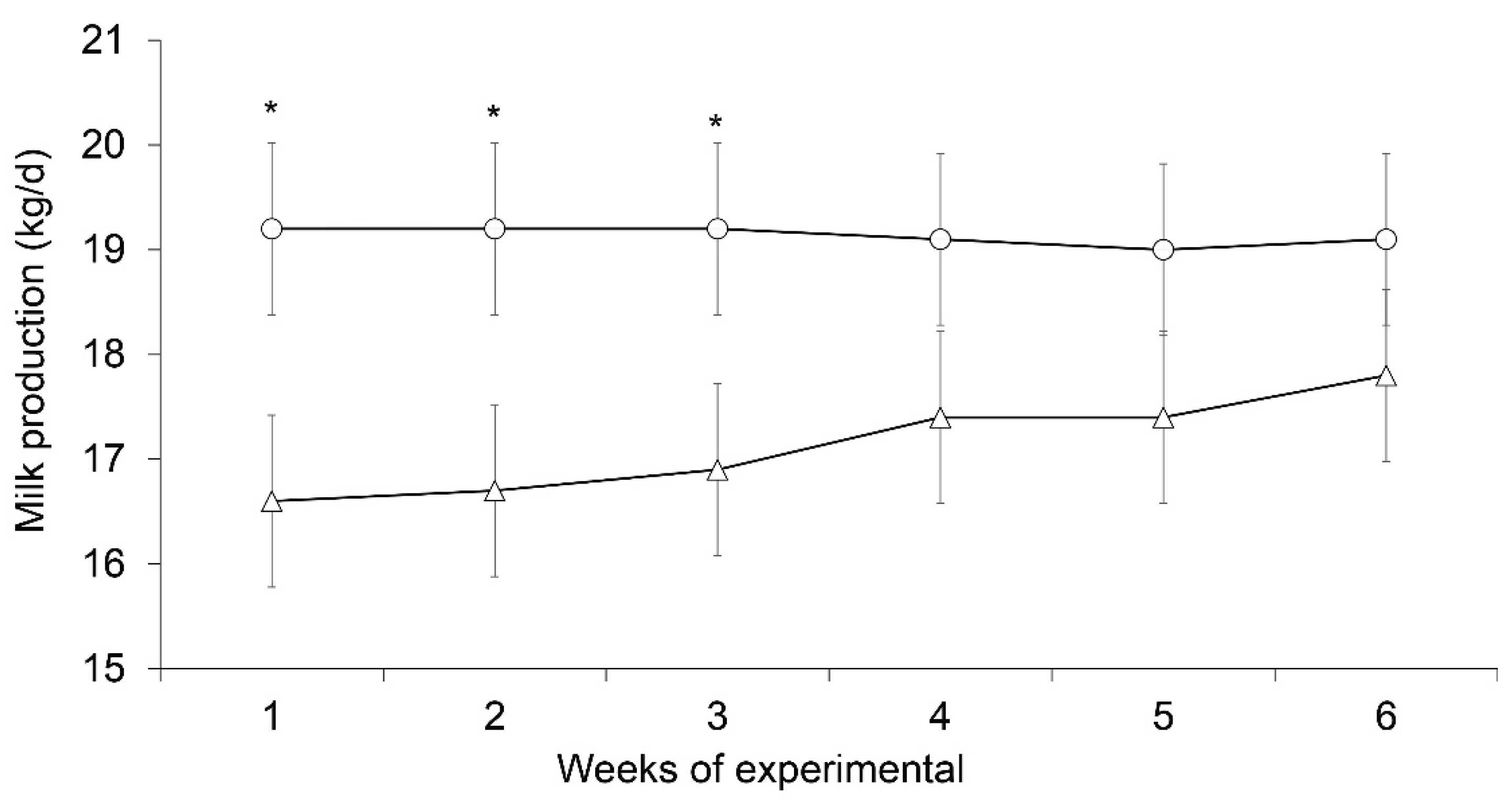
3.2. Milk Yield and Composition
3.3. Ovarian Dynamics, Reproductive Performance, and Offspring Characteristics
4. Discussion
4.1. Feed Intake and Lactation Performance
4.2. Ovarian Dynamics and Corpus Luteum Development
4.3. Maternal Nutrition and Offspring Sex Ratio
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Diniso, Y.S.; Jaja, I.F. A Retrospective Survey of the Factors Responsible for Culling and Mortality in Dairy Farms in the Eastern Cape Province, South Africa. Sci. Afr. 2021, 12, e00838. [Google Scholar] [CrossRef]
- Sheldon, I.M.; Cronin, J.G.; Bromfield, J.J. Tolerance and Innate Immunity Shape the Development of Postpartum Uterine Disease and the Impact of Endometritis in Dairy Cattle. Annu. Rev. Anim. Biosci. 2019, 7, 361–384. [Google Scholar] [CrossRef]
- Wiltbank, M.C.; Baez, G.M.; Garcia-Guerra, A.; Toledo, M.Z.; Monteiro, P.L.; Melo, L.F.; Ochoa, J.C.; Santos, J.E.; Sartori, R. Pivotal Periods for Pregnancy Loss during the First Trimester of Gestation in Lactating Dairy Cows. Theriogenology 2016, 86, 239–253. [Google Scholar] [CrossRef]
- Crouse, M.S.; Greseth, N.P.; McLean, K.J.; Crosswhite, M.R.; Pereira, N.N.; Ward, A.K.; Reynolds, L.P.; Dahlen, C.R.; Neville, B.W.; Borowicz, P.P. Maternal Nutrition and Stage of Early Pregnancy in Beef Heifers: Impacts on Hexose and AA Concentrations in Maternal and Fetal Fluids. J. Anim. Sci. 2019, 97, 1296–1316. [Google Scholar] [CrossRef]
- Staples, C.R.; Burke, J.M.; Thatcher, W.W. Influence of Supplemental Fats on Reproductive Tissues and Performance of Lactating Cows. J. Dairy Sci. 1998, 81, 856–871. [Google Scholar] [CrossRef] [PubMed]
- Butler, W.R. Nutritional Interactions with Reproductive Performance in Dairy Cattle. Anim. Reprod. Sci. 2000, 60–61, 449–457. [Google Scholar] [CrossRef]
- Roche, J.R.; Friggens, N.C.; Kay, J.K.; Fisher, M.W.; Stafford, K.J.; Berry, D.P. Invited Review: Body Condition Score and Its Association with Dairy Cow Productivity, Health, and Welfare. J. Dairy Sci. 2009, 92, 5769–5801. [Google Scholar] [CrossRef]
- Mekuriaw, Y. Negative Energy Balance and Its Implication on Productive and Reproductive Performance of Early Lactating Dairy Cows: Review Paper. J. Appl. Anim. Res. 2023, 51, 220–228. [Google Scholar] [CrossRef]
- Mattos, R.; Staples, C.R.; Thatcher, W.W. Effects of Dietary Fatty Acids on Reproduction in Ruminants. J. Reprod. Fertil. 2000, 5, 38–45. [Google Scholar] [CrossRef]
- Santos, J.; Bilby, T.; Thatcher, W.; Staples, C.; Silvestre, F. Long Chain Fatty Acids of Diet as Factors Influencing Reproduction in Cattle. Reprod. Domest. Anim. 2008, 43, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Thatcher, W.; Santos, J.E.P.; Staples, C.R. Dietary Manipulations to Improve Embryonic Survival in Cattle. Theriogenology 2011, 76, 1619–1631. [Google Scholar] [CrossRef] [PubMed]
- Cholewski, M.; Tomczykowa, M.; Tomczyk, M. A Comprehensive Review of Chemistry, Sources and Bioavailability of Omega-3 Fatty Acids. Nutrients 2018, 10, 1662. [Google Scholar] [CrossRef] [PubMed]
- Bionaz, M.; Vargas-Bello-Pérez, E.; Busato, S. Advances in Fatty Acids Nutrition in Dairy Cows: From Gut to Cells and Effects on Performance. J. Anim. Sci. Biotechnol. 2020, 11. [Google Scholar] [CrossRef]
- Sordillo, L.M. Nutritional Strategies to Optimize Dairy Cattle Immunity. J. Dairy Sci. 2016, 99, 4967–4982. [Google Scholar] [CrossRef]
- Hess, B.W.; Lake, S.L.; Scholljegerdes, E.J.; Weston, T.R.; Nayigihugu, V.; Molle, J.D.C.; Moss, G.E. Nutritional Controls of Beef Cow Reproduction1. J. Anim. Sci. 2005, 83, E90–E106. [Google Scholar] [CrossRef]
- Zeng, X.; Li, S.; Liu, L.; Cai, S.; Ye, Q.; Xue, B.; Wang, X.; Zhang, S.; Chen, F.; Cai, C.; et al. Role of Functional Fatty Acids in Modulation of Reproductive Potential in Livestock. J. Anim. Sci. Biotechnol. 2023, 14, 24. [Google Scholar] [CrossRef]
- Wathes, D.C.; Abayasekara, D.R.E.; Aitken, R.J. Polyunsaturated Fatty Acids in Male and Female Reproduction1. Biol. Reprod. 2007, 77, 190–201. [Google Scholar] [CrossRef]
- Rincón Cervera, M.Á.; Bravo Sagua, R.; Soares-Freitas, R.; Lopez Arana, S.L.; de Camargo, A. Monounsaturated and Polyunsaturated Fatty Acids: Structure, Food Sources, Biological Functions, and Their Preventive Role against Noncommunicable Diseases. In; 2022; pp. 185–210 ISBN 978-0-12-823569-0.
- Schmitz, G.; Ecker, J. The Opposing Effects of n−3 and n−6 Fatty Acids. Prog. Lipid Res. 2008, 47, 147–155. [Google Scholar] [CrossRef]
- Marei, W.F.A.; Khalil, W.A.; Pushpakumara, A.P.G.; El-Harairy, M.A.; Abo El-Atta, A.M.A.; Wathes, D.C.; Fouladi-Nashta, A. Polyunsaturated Fatty Acids Influence Offspring Sex Ratio in Cows. Int. J. Vet. Sci. Med. 2018, 6, S36–S40. [Google Scholar] [CrossRef] [PubMed]
- Moallem, U. Invited Review: Roles of Dietary n-3 Fatty Acids in Performance, Milk Fat Composition, and Reproductive and Immune Systems in Dairy Cattle. J. Dairy Sci. 2018, 101, 8641–8661. [Google Scholar] [CrossRef] [PubMed]
- Mattos, R.; Guzeloglu, A.; Badinga, L.; Staples, C.R.; Thatcher, W.W. Polyunsaturated Fatty Acids and Bovine Interferon-τ Modify Phorbol Ester-Induced Secretion of Prostaglandin F2α and Expression of Prostaglandin Endoperoxide Synthase-2 and Phospholipase-A2 in Bovine Endometrial Cells1. Biol. Reprod. 2003, 69, 780–787. [Google Scholar] [CrossRef] [PubMed]
- Robinson, R.; Hammond, A.; Wathes, D.; Hunter, M.; Mann, G. Corpus Luteum–Endometrium–Embryo Interactions in the Dairy Cow: Underlying Mechanisms and Clinical Relevance. Reprod. Domest. Anim. 2008, 43, 104–112. [Google Scholar] [CrossRef]
- Domingues, R.R.; Andrade, J.P.N.; Cunha, T.O.; Madureira, G.; Moallem, U.; Gomez-Leon, V.; Martins, J.P.N.; Wiltbank, M.C. Is Pregnancy Loss Initiated by Embryonic Death or Luteal Regression? Profiles of Pregnancy-Associated Glycoproteins during Elevated Progesterone and Pregnancy Loss. JDS Communications 2023, 4, 149–154. [Google Scholar] [CrossRef]
- Calder, P.C. Long-Chain Fatty Acids and Inflammation. Proc. Nutr. Soc. 2012, 71, 284–289. [Google Scholar] [CrossRef]
- Silvestre, F.T.; Carvalho, T.S.M.; Crawford, P.C.; Santos, J.E.P.; Staples, C.R.; Jenkins, T.; Thatcher, W.W. Effects of Differential Supplementation of Fatty Acids during the Peripartum and Breeding Periods of Holstein Cows: II. Neutrophil Fatty Acids and Function, and Acute Phase Proteins. J. Dairy Sci. 2011, 94, 2285–2301. [Google Scholar] [CrossRef]
- Thangavelu, G.; Colazo, M.G.; Ambrose, D.J.; Oba, M.; Okine, E.K.; Dyck, M.K. Diets Enriched in Unsaturated Fatty Acids Enhance Early Embryonic Development in Lactating Holstein Cows. Theriogenology 2007, 68, 949–957. [Google Scholar] [CrossRef]
- Giller, K.; Drews, B.; Berard, J.; Kienberger, H.; Schmicke, M.; Frank, J.; Spanier, B.; Daniel, H.; Geisslinger, G.; Ulbrich, S.E. Bovine Embryo Elongation Is Altered Due to Maternal Fatty Acid Supplementation. Biol. Reprod. 2018, 99, 600–610. [Google Scholar] [CrossRef]
- Sinedino, L.D.P.; Honda, P.; Souza, L.; Lock, A.; Boland, M.; Staples, C.; Thatcher, W.; Santos, J. Effects of Supplementation with Docosahexaenoic Acid on Reproduction of Dairy Cows. Reproduction 2017, 153, REP–16. [Google Scholar] [CrossRef]
- Ambrose, D.J.; Kastelic, J.P.; Corbett, R.; Pitney, P.A.; Petit, H.V.; Small, J.A.; Zalkovic, P. Lower Pregnancy Losses in Lactating Dairy Cows Fed a Diet Enriched in α-Linolenic Acid. J. Dairy Sci. 2006, 89, 3066–3074. [Google Scholar] [CrossRef] [PubMed]
- Fabjanowska, J.; Kowalczuk-Vasilev, E.; Klebaniuk, R.; Milewski, S.; Gümüş, H. N-3 Polyunsaturated Fatty Acids as a Nutritional Support of the Reproductive and Immune System of Cattle—A Review. Animals 2023, 13, 3589. [Google Scholar] [CrossRef]
- Mattos, R.; Staples, C.R.; Williams, J.; Amorocho, A.; McGuire, M.A.; Thatcher, W.W. Uterine, Ovarian, and Production Responses of Lactating Dairy Cows to Increasing Dietary Concentrations of Menhaden Fish Meal. J. Dairy Sci. 2002, 85, 755–764. [Google Scholar] [CrossRef]
- Iwata, H. Factors Affecting the Primary Sex Ratio in Cows. J. Mamm. Ova Res. 2012, 29, 96–102. [Google Scholar] [CrossRef]
- Seidel, G.E.; Schenk, J.L. Pregnancy Rates in Cattle with Cryopreserved Sexed Sperm: Effects of Sperm Numbers per Inseminate and Site of Sperm Deposition. Anim. Reprod. Sci. 2008, 105, 129–138. [Google Scholar] [CrossRef]
- Jones, C.; Heinrichs, J. Learn To Score Body Condition Available online: https://extension.psu.edu/learn-to-score-body-condition (accessed on 16 March 2022).
- Steel, R.G.D.; Torrie, J.H.; Dicky, D.A. Principles and Procedures of Statistics, A Biometrical Approach. 3rd Edition; McGraw Hill, Inc. Book Co., New York, 1997.
- Allred, S.L.; Dhiman, T.R.; Brennand, C.P.; Khanal, R.C.; McMahon, D.J.; Luchini, N.D. Milk and Cheese from Cows Fed Calcium Salts of Palm and Fish Oil Alone or in Combination with Soybean Products. J. Dairy Sci.e 2006, 89, 234–248. [Google Scholar] [CrossRef] [PubMed]
- NRC, N.R.C. Nutrient Requirements of Dairy Cattle, 7th Rev. Ed.; The National Academies Press: Washington, DC, USA, 2001.
- Huang, G.; Wang, J.; Liu, K.; Wang, F.; Zheng, N.; Zhao, S.; Qu, X.; Yu, J.; Zhang, Y.; Wang, J. Effect of Flaxseed Supplementation on Milk and Plasma Fatty Acid Composition and Plasma Parameters of Holstein Dairy Cows. Animals 2022, 12, 1898. [Google Scholar] [CrossRef]
- Leduc, M.; Létourneau-Montminy, M.-P.; Gervais, R.; Chouinard, P.Y. Effect of Dietary Flax Seed and Oil on Milk Yield, Gross Composition, and Fatty Acid Profile in Dairy Cows: A Meta-Analysis and Meta-Regression. J. Dairy Sci. 2017, 100, 8906–8927. [Google Scholar] [CrossRef]
- Do Prado, R.M.; Palin, M.F.; Do Prado, I.N.; Dos Santos, G.T.; Benchaar, C.; Petit, H.V. Milk Yield, Milk Composition, and Hepatic Lipid Metabolism in Transition Dairy Cows Fed Flaxseed or Linola. J. Dairy Sci. 2016, 99, 8831–8846. [Google Scholar] [CrossRef] [PubMed]
- Isenberg, B.J.; Soder, K.J.; Pereira, A.B.D.; Standish, R.; Brito, A.F. Production, Milk Fatty Acid Profile, and Nutrient Utilization in Grazing Dairy Cows Supplemented with Ground Flaxseed. J. Dairy Sci. 2019, 102, 1294–1311. [Google Scholar] [CrossRef]
- Sun, X.; Guo, C.; Wang, Q.; Zhang, Y.; Wang, Z.; Cao, Z.; Wang, W.; Li, S. Dietary N-3 Polyunsaturated Fatty Acids Enhance Dry Matter Intake in Transition Cows by Regulating Taste Transduction Gene Expression in the Liver Associated with Rumen Microbial Alteration. Anim. Nutr. 2025, 22, 502–513. [Google Scholar] [CrossRef]
- Petit, H.V.; Palin, M.F.; Doepel, L. Hepatic Lipid Metabolism in Transition Dairy Cows Fed Flaxseed. J. Dairy Sci. 2007, 90, 4780–4792. [Google Scholar] [CrossRef]
- Zachut, M.; Arieli, A.; Lehrer, H.; Livshitz, L.; Yakoby, S.; Moallem, U. Effects of Increased Supplementation of N-3 Fatty Acids to Transition Dairy Cows on Performance and Fatty Acid Profile in Plasma, Adipose Tissue, and Milk Fat. J. Dairy Sci. 2010, 93, 5877–5889. [Google Scholar] [CrossRef]
- Chilliard, Y.; Martin, C.; Rouel, J.; Doreau, M. Milk Fatty Acids in Dairy Cows Fed Whole Crude Linseed, Extruded Linseed, or Linseed Oil, and Their Relationship with Methane Output. J. Dairy Sci. 2009, 92, 5199–5211. [Google Scholar] [CrossRef]
- Moallem, U.; Lehrer, H.; Livshits, L.; Zachut, M. The Effects of Omega-3 α-Linolenic Acid from Flaxseed Oil Supplemented to High-Yielding Dairy Cows on Production, Health, and Fertility. Livest. Sci. 2020, 242, 104302. [Google Scholar] [CrossRef]
- Dirandeh, E.; Towhidi, A.; Ansari Pirsaraei, Z.; Adib Hashemi, F.; Ganjkhanlou, M.; Zeinoaldini, S.; Rezaei Roodbari, A.; Saberifar, T.; Petit, H.V. Plasma Concentrations of PGFM and Uterine and Ovarian Responses in Early Lactation Dairy Cows Fed Omega-3 and Omega-6 Fatty Acids. Theriogenology 2013, 80, 131–137. [Google Scholar] [CrossRef]
- Doyle, D.N.; Lonergan, P.; Diskin, M.G.; Pierce, K.M.; Kelly, A.K.; Stanton, C.; Waters, S.M.; Parr, M.H.; Kenny, D.A. Effect of Dietary N-3 Polyunsaturated Fatty Acid Supplementation and Post-Insemination Plane of Nutrition on Systemic Concentrations of Metabolic Analytes, Progesterone, Hepatic Gene Expression and Embryo Development and Survival in Beef Heifers. Theriogenology 2019, 127, 102–113. [Google Scholar] [CrossRef] [PubMed]
- Jaśkowski, B.M.; Opałka, A.; Gehrke, M.; Herudzińska, M.; Czeladko, J.; Baumgartner, W.; Jaśkowski, J.M. A Critical Overview on Prostaglandin Inhibitors and Their Influence on Pregnancy Results after Insemination and Embryo Transfer in Cows. Animals 2021, 11, 3368. [Google Scholar] [CrossRef] [PubMed]
- Trivers, R.L.; Willard, D.E. Natural Selection of Parental Ability to Vary the Sex Ratio of Offspring. Science 1973, 179, 90–92. [Google Scholar] [CrossRef]
- Wang, Y.; Fu, X.; Li, H. Mechanisms of Oxidative Stress-Induced Sperm Dysfunction. Front. Endocrinol. 2025, 16. [Google Scholar] [CrossRef]
- Rosenfeld, C.S. Sex-Specific Placental Responses in Fetal Development. Endocrinology 2015, 156, 3422–3434. [Google Scholar] [CrossRef] [PubMed]
- Roque-Jiménez, J.A.; Rosa-Velázquez, M.; Pinos-Rodríguez, J.M.; Vicente-Martínez, J.G.; Mendoza-Cervantes, G.; Flores-Primo, A.; Lee-Rangel, H.A.; Relling, A.E. Role of Long Chain Fatty Acids in Developmental Programming in Ruminants. Animals 2021, 11, 762. [Google Scholar] [CrossRef] [PubMed]
| Item | Treatments | |
|---|---|---|
| Greater n-6:n-3 (16.7:1) |
Lower n-6:n-3 (4.8:1) |
|
| Chemical composition (% of DM) | ||
| Moist | 9.90 | 10.10 |
| Dry matter | 90.10 | 89.90 |
| Crude protein | 24.00 | 24.48 |
| Fat | 4.05 | 4.13 |
| NDF | 28.26 | 30.70 |
| ADF | 15.27 | 16.59 |
| ADL | 5.23 | 5.34 |
| Ash | 9.19 | 8.43 |
| Fatty acids composition (g/100g) | ||
| Caprylic acid (C8:0) | 0.07 | 0.06 |
| Capric acid (C10:0) | 0.07 | 0.07 |
| Lauric acid (C12:0) | 0.93 | 0.99 |
| Myristic acid (C14:0) | 0.33 | 0.35 |
| Palmitic acid (C16:0) | 0.89 | 0.73 |
| Stearic acid (C18:0) | 0.15 | 0.35 |
| Arachidic acid (C20:0) | 0.02 | 0.02 |
| Lignoceric acid (C24:0) | 0.01 | 0.01 |
| Saturated Fat | 2.47 | 2.58 |
| Palmitoleic acid (C16:1n7) | 0.01 | 0.01 |
| Oleic acid (C18:1n9c; Omega-9) | 1.11 | 0.82 |
| Eicosenoic acid (C20:1n9; Omega-9) | 0.01 | - |
| Monounsaturated fatty acid | 1.13 | 0.83 |
| Linoleic acid (C18:2n6c; Omega-6) | 0.80 | 0.65 |
| Alpha-Linolenic acid (C18:3n3; ALA, Omega-3) | 0.05 | 0.13 |
| Polyunsaturated fatty acid | 0.85 | 0.78 |
| Unsaturated fat | 1.98 | 1.61 |
| Omega-3 (mg/100g) | 47.90 | 134.54 |
| Omega-6 (mg/100g) | 798.73 | 645.45 |
| Omega-9 (mg/100g) | 1115.76 | 824.82 |
| Omega-6 : Omega-3 | 16.70 | 4.80 |
| Item | Treatments | SEM | p-value | |
|---|---|---|---|---|
| Greater n-6:n-3 (16.7:1) |
Lower n-6:n-3 (4.8:1) |
|||
| Feed intake (DM) | ||||
| Concentrate | ||||
| kg/day | 7.82 | 8.30 | 0.25 | 0.35 |
| % BW | 1.45 | 1.55 | 0.04 | 0.25 |
| g/kg BW0.75 | 69.58 | 74.15 | 2.05 | 0.30 |
| Roughage | ||||
| kg/d | 5.52 | 5.72 | 0.12 | 0.16 |
| % BW | 1.02 | 1.06 | 0.02 | 0.17 |
| g/kg BW0.75 | 49.18 | 51.17 | 1.06 | 0.13 |
| Total | ||||
| kg/d | 13.34 | 14.02 | 0.32 | 0.20 |
| % BW | 2.46 | 2.60 | 0.05 | 0.17 |
| g/kg BW0.75 | 118.76 | 125.31 | 2.54 | 0.14 |
| BW (kg) | 539.62 | 541.13 | 6.85 | 0.99 |
| BCS | ||||
| Initial | 2.50 | 2.50 | 0.07 | 0.77 |
| Final | 2.65 | 2.71 | 0.07 | 0.51 |
| BCS Change | 0.16 | 0.22 | 0.03 | 0.23 |
| Item | Treatments | SEM | P-value | |
|---|---|---|---|---|
| Greater n-6:n-3 (16.7:1) |
Lower n-6:n-3 (4.8:1) |
|||
| Milk yield (Kg/d) | 17.17 | 19.07 | 0.76 | 0.08 |
| 3.5% FCM (Kg/d) | 17.17 | 19.28 | 1.02 | 0.15 |
| ECM (Kg/d) | 15.68 | 17.60 | 0.88 | 0.13 |
| Efficiency, ECM/DMI | 1.14 | 1.20 | 0.05 | 0.41 |
| Milk composition, % | ||||
| Fat | 3.28 | 3.12 | 0.19 | 0.52 |
| Protein | 2.99 | 2.92 | 0.07 | 0.48 |
| Lactose | 4.49 | 4.53 | 0.07 | 0.58 |
| Solid not fat | 8.08 | 8.06 | 0.11 | 0.98 |
| Total solid | 12.47 | 12.69 | 0.63 | 0.96 |
| SCC (x103 cells/mL) | 440.7 | 356.3 | 153.97 | 0.55 |
| Milk production, kg/d | ||||
| Fat yield | 0.59 | 0.64 | 0.05 | 0.43 |
| Protein yield | 0.53 | 0.60 | 0.03 | 0.10 |
| Lactose yield | 0.80 | 0.94 | 0.04 | 0.03 |
| Solid not fat yield | 1.45 | 1.67 | 0.08 | 0.05 |
| Total solid yield | 2.24 | 2.64 | 0.17 | 0.14 |
| Item | Treatments | SEM | p-value | |
|---|---|---|---|---|
| Greater n-6:n-3 (16.7:1) |
Lower n-6:n-3 (4.8:1) |
|||
| Synchronized ovulation rate | ||||
| first GnRH, % (no./no.) | 53.3 (16/30) | 58.6 (17/29) | 0.09 | 0.68 |
| second GnRH, % (no./no.) | 90.0 (27/30) | 96.6 (28/29) | 0.04 | 0.32 |
| Luteolysis (%) | 73.3 (22/30) | 72.4 (21/29) | 0.08 | 0.94 |
| pre-ovulatory follicle (mm) | 14.0 | 13.1 | 0.65 | 0.21 |
| Number of CL (n) | ||||
| d5 post TAI | 0.71 | 0.93 | 0.11 | 0.86 |
| d8 post TAI | 0.74 | 1.00 | 0.11 | 0.73 |
| d11 post TAI | 0.84 | 1.14 | 0.11 | 0.58 |
| d14 post TAI | 1.01 | 1.31 | 0.11 | 0.55 |
| CL diameter (mm) | ||||
| d5 post TAI | 16.97 | 15.35 | 0.84 | 0.17 |
| d8 post TAI | 19.51 | 21.21 | 0.84 | 0.15 |
| d11 post TAI | 21.66 | 24.04 | 0.86 | 0.05 |
| d14 post TAI | 24.61 | 26.58 | 0.89 | 0.10 |
| CL cross-sectional area (mm2) | ||||
| d5 post TAI | 246.38 | 196.04 | 26.49 | 0.18 |
| d8 post TAI | 313.71 | 366.87 | 30.52 | 0.16 |
| d11 post TAI | 380.10b | 461.94a | 30.90 | 0.04 |
| d14 post TAI | 474.14b | 560.32a | 32.41 | 0.03 |
| CL volume (mm3) | ||||
| d5 post TAI | 3,195.94 | 2,316.63 | 581.28 | 0.28 |
| d8 post TAI | 4,405.69 | 5,657.99 | 594.62 | 0.14 |
| d11 post TAI | 5,864.44 b | 7,774.80 a | 610.91 | 0.03 |
| d14 post TAI | 7,978.92 b | 10,395.00 a | 627.02 | 0.007 |
| Item | Treatments | SEM | p-value | |
|---|---|---|---|---|
| Greater n-6:n-3 (16.7:1) |
Lower n-6:n-3 (4.8:1) |
|||
| PR/AI, % (n/n) | ||||
| First AI | 23.33 (7/30) | 13.80 (4/29) | 0.08 | 0.37 |
| All AI | 96.67 (29/30) | 96.55 (28/29) | 0.01 | 1.00 |
| Number of services per conception, no. | 3.33 | 3.15 | 0.44 | 0.73 |
| Female offspring ratio, % (n/n) | 42.90 (12/28) | 66.67 (18/27) | 0.10 | 0.09 |
| Weight of offspring (Kg) | 32.92b | 34.50a | 0.54 | 0.02 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




