Submitted:
14 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Sample
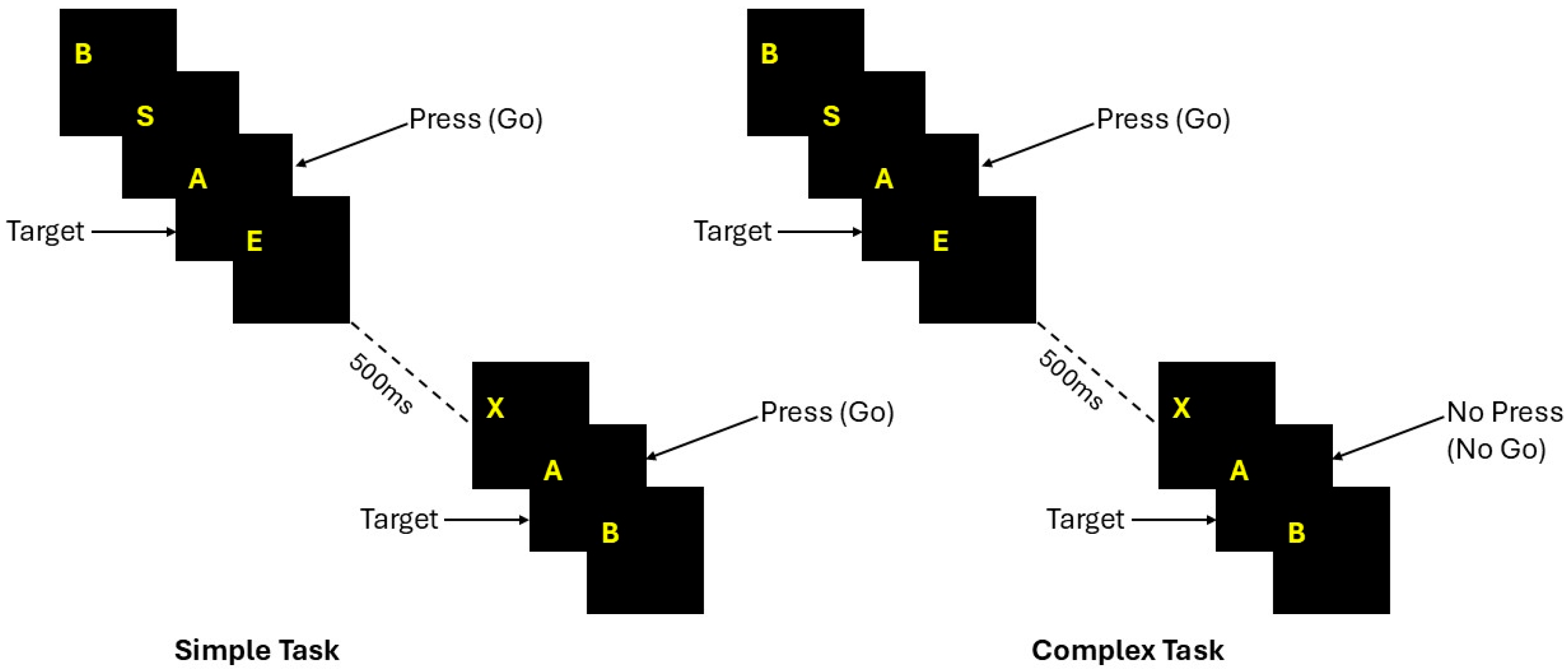
Stimuli and Paradigm
Acquisition and Preprocessing of Neuroimaging Data
Estimation of the Diffusion Tensor and Fiber Tracking
EEG Data Recording and Preprocessing
Research Design and Statistical Analysis
3. Results
Behavioral Performance Characteristics
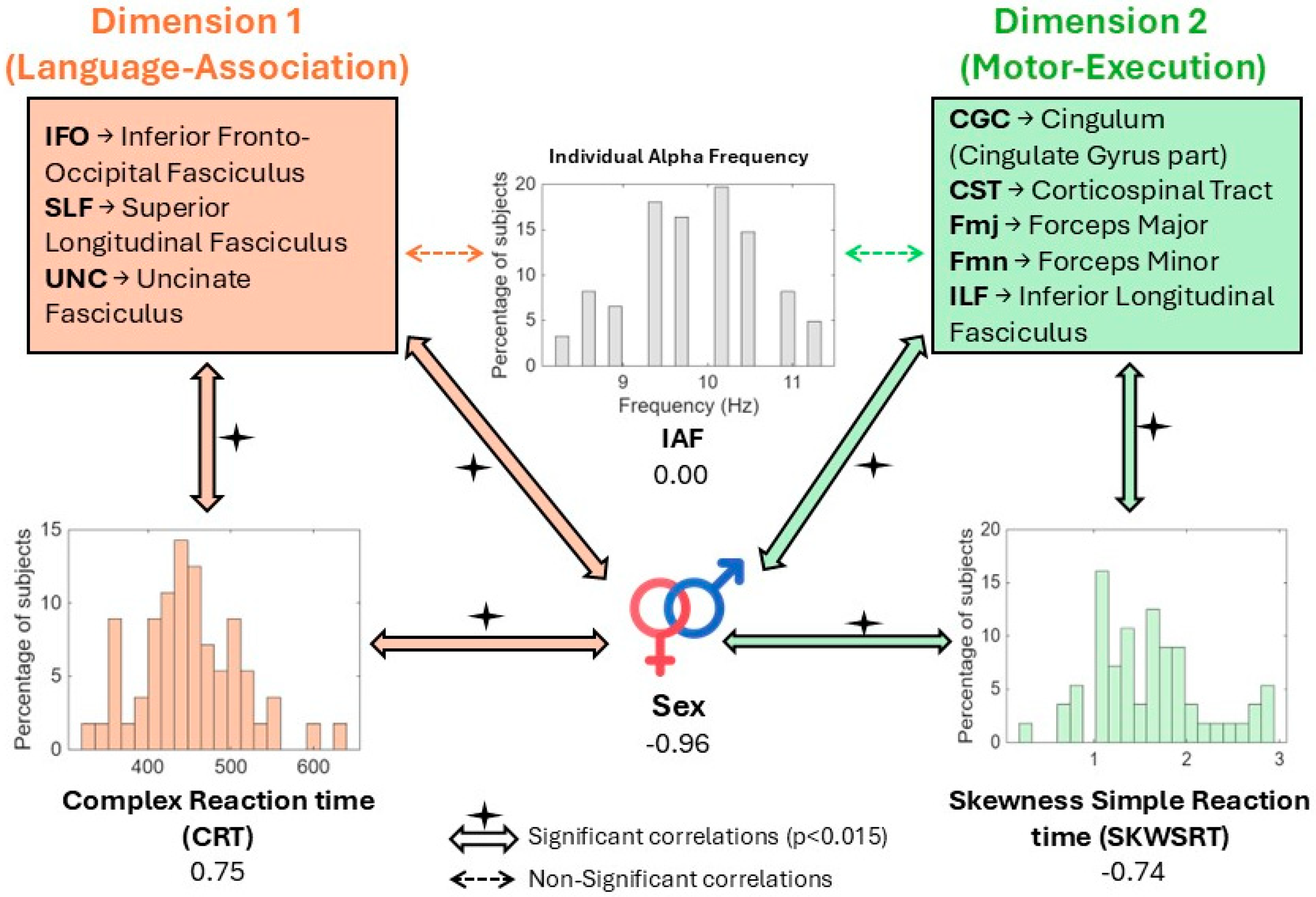
Sparse Multiple CCA Identifies Two Significant Dimensions
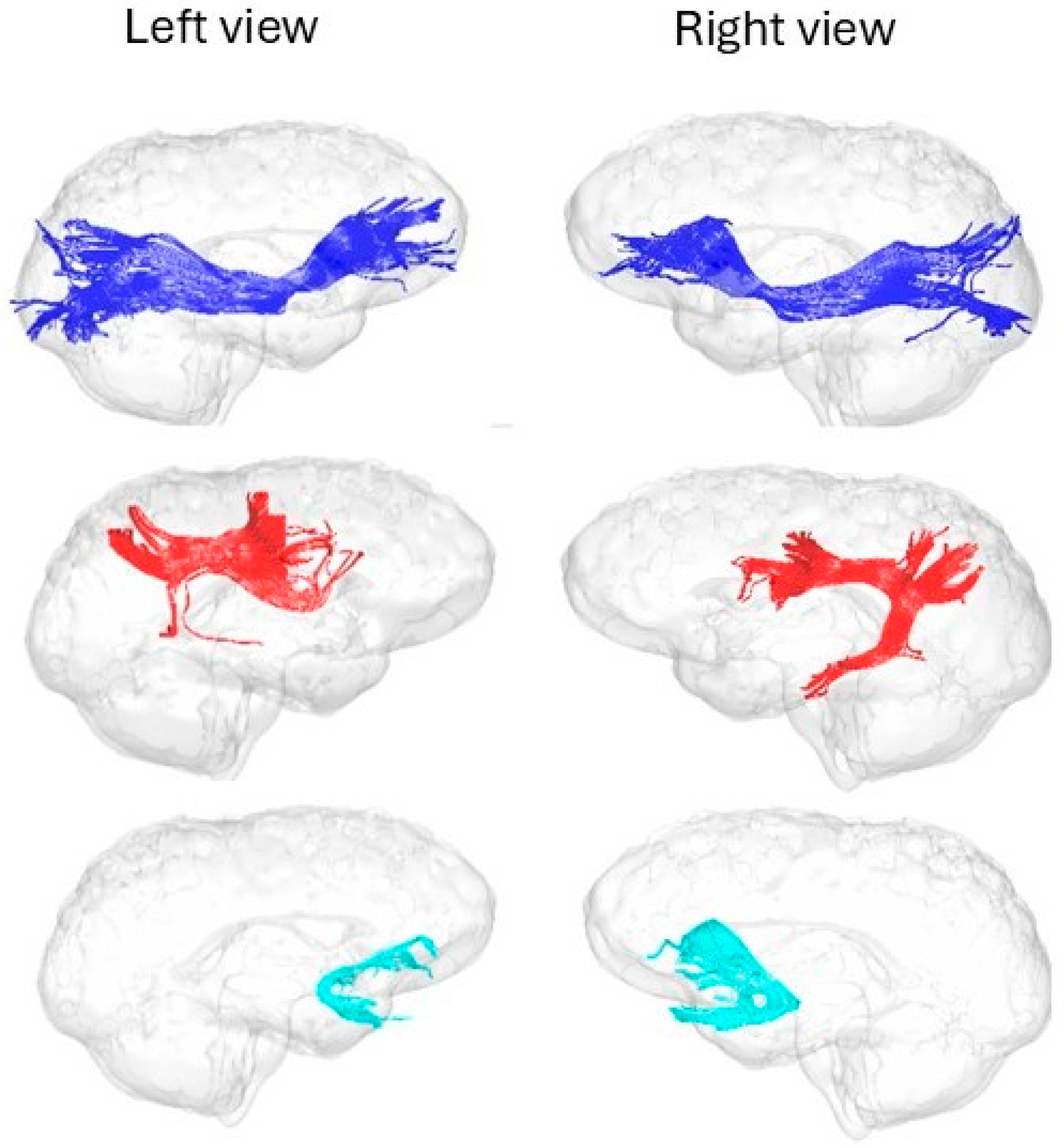
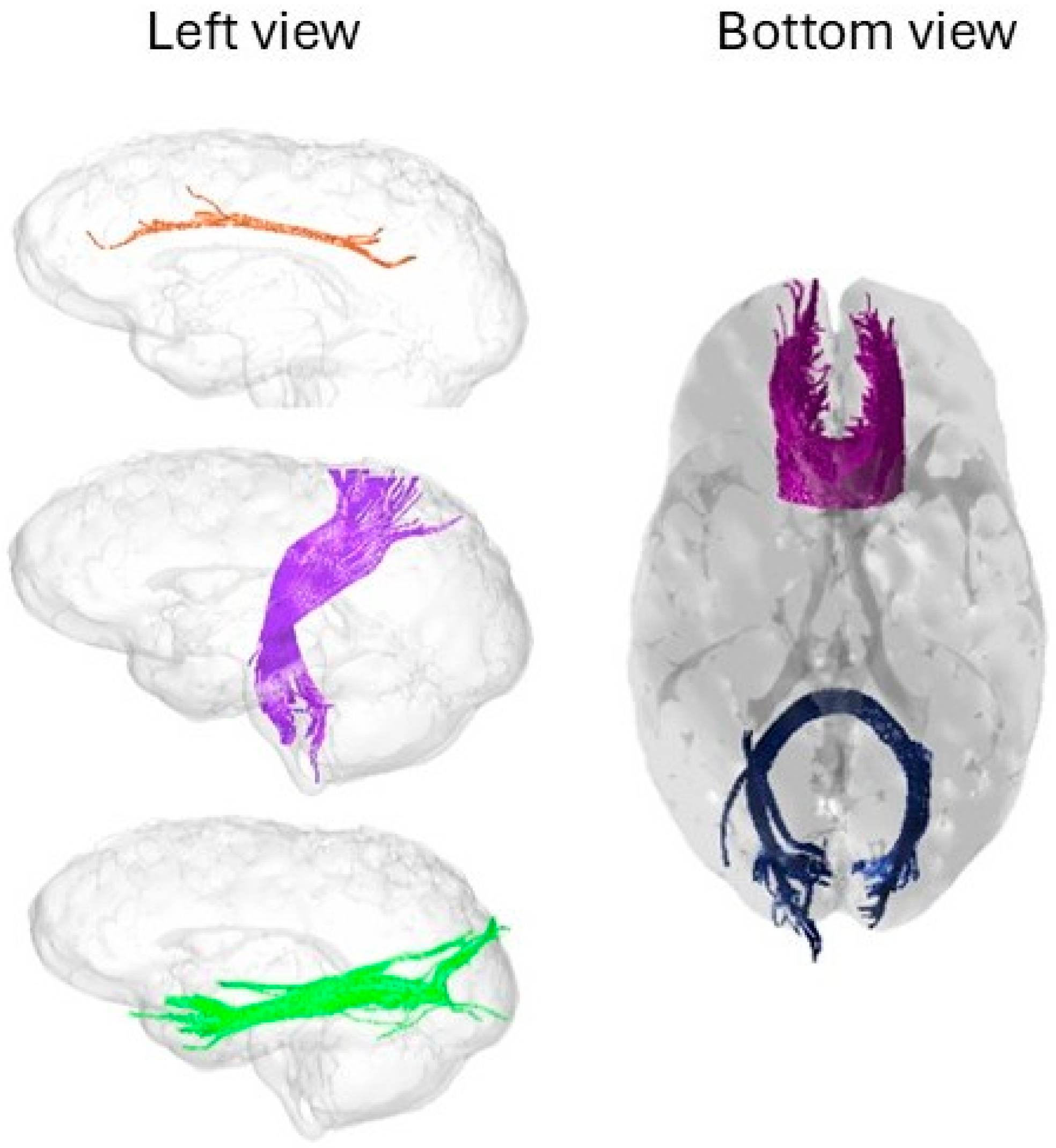
Dimension 1: Language-Association Tracts and Complex Task Performance
Dimension 2: Motor-Interhemispheric Tracts and Intra-Individual Variability
Absence of Significant Associations for EEG Alpha Peak and Visual Pathways
4. Discussion
Distinct White Matter Systems for Cognitive Complexity and Motor Consistency
The Absence of EEG Alpha Peak Contributions: Structural Constraints on Oscillatory Activity
Sex Differences in Brain-Behavior Relationships
Comparison with Existing Literature and Methodological Considerations
Implications, Limitations, and Future Direction
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Donders, F.C. On the Speed of Mental Processes. Acta Psychologica 1969, 30, 412–431. [Google Scholar] [CrossRef]
- Stufflebeam, S.M.; Witzel, T.; Mikulski, S.; Hämäläinen, M.S.; Temereanca, S.; Barton, J.J.S.; Tuch, D.S.; Manoach, D.S. A Non-Invasive Method to Relate the Timing of Neural Activity to White Matter Microstructural Integrity. Neuroimage 2008, 42, 710–716. [Google Scholar] [CrossRef]
- Penke, L.; Maniega, S.M.; Murray, C.; Gow, A.J.; Valdés Hernández, M.C.; Clayden, J.D.; Starr, J.M.; Wardlaw, J.M.; Bastin, M.E.; Deary, I.J. A General Factor of Brain White Matter Integrity Predicts Information Processing Speed in Healthy Older People. J Neurosci 2010, 30, 7569–7574. [Google Scholar] [CrossRef]
- Kuznetsova, K.A.; Maniega, S.M.; Ritchie, S.J.; Cox, S.R.; Storkey, A.J.; Starr, J.M.; Wardlaw, J.M.; Deary, I.J.; Bastin, M.E. Brain White Matter Structure and Information Processing Speed in Healthy Older Age. Brain Struct Funct 2016, 221, 3223–3235. [Google Scholar] [CrossRef]
- Tamnes, C.K.; Fjell, A.M.; Westlye, L.T.; Østby, Y.; Walhovd, K.B. Becoming Consistent: Developmental Reductions in Intraindividual Variability in Reaction Time Are Related to White Matter Integrity. J. Neurosci. 2012, 32, 972–982. [Google Scholar] [CrossRef] [PubMed]
- Klimesch, W. Alpha-Band Oscillations, Attention, and Controlled Access to Stored Information. Trends in Cognitive Sciences 2012, 16, 606–617. [Google Scholar] [CrossRef]
- Nunez, P. EEG Coherency II: Experimental Comparisons of Multiple Measures. Clinical Neurophysiology 1999, 110, 469–486. [Google Scholar] [CrossRef]
- Srinivasan, R.; Nunez, P.L.; Silberstein, R.B. Spatial Filtering and Neocortical Dynamics : Estimates of EEG Coherence. 1998, 45, 814–826. [Google Scholar] [CrossRef]
- Valdés-Hernández, P.A.; Ojeda-González, A.; Martínez-Montes, E.; Lage-Castellanos, A.; Virués-Alba, T.; Valdés-Urrutia, L.; Valdes-Sosa, P.A. White Matter Architecture Rather than Cortical Surface Area Correlates with the EEG Alpha Rhythm. NeuroImage 2010, 49, 2328–2339. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Gonzalez, G.; Bringas-Vega, M.L.; Galán-Garcia, L.; Bosch-Bayard, J.; Lorenzo-Ceballos, Y.; Melie-Garcia, L.; Valdes-Urrutia, L.; Cobas-Ruiz, M.; Valdes-Sosa, P.A.; Galan-Garcia, L.; et al. Multimodal Quantitative Neuroimaging Databases and Methods: The Cuban Human Brain Mapping Project. Clinical EEG and neuroscience : official journal of the EEG and Clinical Neuroscience Society (ENCS) 2011, 42, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Góngora, D.; Domínguez, M.; Bobes, M.A. Characterization of Ten White Matter Tracts in a Representative Sample of Cuban Population. BMC Med Imaging 2016, 16, 59. [Google Scholar] [CrossRef] [PubMed]
- Valdes-Sosa, P.A.P.A.; Galan-Garcia, L.; Bosch-Bayard, J.; Bringas-Vega, M.L.M.L.; Aubert-Vazquez, E.; Rodriguez-Gil, I.; Das, S.; Madjar, C.; Virues-Alba, T.; Mohades, Z.; et al. The Cuban Human Brain Mapping Project, a Young and Middle Age Population-Based EEG, MRI, and Cognition Dataset. Scientific Data 2021, 8, 45. [Google Scholar] [CrossRef] [PubMed]
- Gongora, D.; Vega-Hernandez, M.; Valdes-Sosa, P.; Jahanshahi, M.; Bringas-Vega, M. Tract-Based Fractional Anisotropy Predicts WAIS Intelligence Quotient Indices and Subtest Performance. IBRO Reports 2019, 6, S443--S444. [Google Scholar] [CrossRef]
- EIWA-III - Escala de Inteligencia de Wechsler Para Adultos | Tercera Edicion | Pearson Assessments US. Available online: https://www.pearsonassessments.com/en-us/Store/Professional-Assessments/Cognition-%26-Neuro/Escala-de-Inteligencia-de-Wechsler-para-Adultos-%7C-Tercera-Edicion/p/100000192?srsltid=AfmBOorXuVeWbka6mzmHAouga5_zr8oFemdlOhfgtD5dec93nrpZJnrD (accessed on 10 April 2026).
- Borrego, M.; Trujillo-Barreto, N.; Rodr\’\iguez-Puentes, Y.; Bosch-Bayard, J.; Mart\’\inez-Montes, E.; Melie-Garc\’\ia, L.; Aubert, E.; Valdés-Sosa, P. “Neuronic Localizador de Fuentes”: Sistema Para El Cálculo de La Tomograf{\’\i}a Eléctrica/Magnética Cerebral.; 2013; pp. 1110–1113.
- Petersen, S.E.; Posner, M.I. The Attention System of the Human Brain: 20 Years After. Annual Review of Neuroscience 2012, 35, 73–89. [Google Scholar] [CrossRef]
- SPM2 Software - Statistical Parametric Mapping. Available online: https://www.fil.ion.ucl.ac.uk/spm/software/spm2/ (accessed on 10 April 2026).
- Le Bihan, D.; Mangin, J.-F.; Poupon, C.; Clark, C.A.; Pappata, S.; Molko, N.; Chabriat, H. Diffusion Tensor Imaging: Concepts and Applications. Journal of Magnetic Resonance Imaging 2001, 13, 534–546. [Google Scholar] [CrossRef]
- Le Bihan, D.; van Zijl, P. From the Diffusion Coefficient to the Diffusion Tensor. NMR in Biomedicine 2002, 15, 431–434. [Google Scholar] [CrossRef]
- SPM5 Software - Statistical Parametric Mapping. Available online: https://www.fil.ion.ucl.ac.uk/spm/software/spm5/ (accessed on 10 April 2026).
- Kim, H.Y. AN ANISOTROPIC DIFFUSION WITH MEANINGFUL SCALE PARAMETER.
- Kreher, B.W.; Hennig, J.; Il’yasov, K.A. DTI&FiberTools: A Complete Toolbox for DTI Calculation, Fiber Tracking, and Combined Evaluation.
- Hernández-González, G. de los Á.; Álvarez-Sánchez, M.; Jordán-González, J.; Valdés-Urrutia, L.; Melie-García, L.; Valdés-Sosa, P.A. Prevalencia de Hallazgos Incidentales En Las Im{á}genes de Resonancia Magn{é}tica: Proyecto Cubano de Mapeo Cerebral Humano. Revista CENIC Ciencias Biol{ó}gicas 2012, 43. [Google Scholar]
- Klimesch, W. The Frequency Architecture of Brain and Brain Body Oscillations: An Analysis. European Journal of Neuroscience 2018, 48, 2431–2453. [Google Scholar] [CrossRef]
- HOTELLING, H. RELATIONS BETWEEN TWO SETS OF VARIATES*. Biometrika 1936, 28, 321–377. [Google Scholar] [CrossRef]
- Wilms, I.; Croux, C. Robust Sparse Canonical Correlation Analysis. BMC Syst Biol 2016, 10, 72. [Google Scholar] [CrossRef]
- Witten, D.M.; Tibshirani, R.; Hastie, T. A Penalized Matrix Decomposition, with Applications to Sparse Principal Components and Canonical Correlation Analysis. Biostatistics 2009, 10, 515–534. [Google Scholar] [CrossRef] [PubMed]
- Witten, D.; Tibshirani, R.; Gross, S.; Narasimhan, B. PMA: Penalized Multivariate Analysis 2024.
- Witten, D.M.; Tibshirani, R.J. Extensions of Sparse Canonical Correlation Analysis with Applications to Genomic Data. Stat Appl Genet Mol Biol 2009, 8, 28. [Google Scholar] [CrossRef]
- Kamali, A.; Sair, H.I.; Radmanesh, A.; Hasan, K.M. Decoding the Superior Parietal Lobule Connections of the Superior Longitudinal Fasciculus/Arcuate Fasciculus in the Human Brain. Neuroscience 2014, 277, 577–583. [Google Scholar] [CrossRef]
- Caverzasi, E.; Papinutto, N.; Amirbekian, B.; Berger, M.S.; Henry, R.G. Q-Ball of Inferior Fronto-Occipital Fasciculus and Beyond. PLOS ONE 2014, 9, e100274. [Google Scholar] [CrossRef]
- Casto, C.; Poliak, M.; Tuckute, G.; Small, H.; Sherlock, P.; Wolna, A.; Lipkin, B.; D’Mello, A.M.; Fedorenko, E. The Cerebellar Components of the Human Language Network. Neuron 2026, S0896627325009869. [Google Scholar] [CrossRef]
- Walhovd, K.B.; Fjell, A.M. White Matter Volume Predicts Reaction Time Instability. Neuropsychologia 2007, 45, 2277–2284. [Google Scholar] [CrossRef]
- Glasser, M.F.; Coalson, T.S.; Robinson, E.C.; Hacker, C.D.; Harwell, J.; Yacoub, E.; Ugurbil, K.; Andersson, J.; Beckmann, C.F.; Jenkinson, M.; et al. A Multi-Modal Parcellation of Human Cerebral Cortex. Nature 2016, 536, 171–178. [Google Scholar] [CrossRef]
- Rushworth, M.F.S.; Behrens, T.E.J. Choice, Uncertainty and Value in Prefrontal and Cingulate Cortex. Nat Neurosci 2008, 11, 389–397. [Google Scholar] [CrossRef]
- Bastos, A.M.; Vezoli, J.; Bosman, C.A.; Schoffelen, J.-M.; Oostenveld, R.; Dowdall, J.R.; De Weerd, P.; Kennedy, H.; Fries, P. Visual Areas Exert Feedforward and Feedback Influences through Distinct Frequency Channels. Neuron 2015, 85, 390–401. [Google Scholar] [CrossRef] [PubMed]
- Hennessee, J.P.; Reggente, N.; Cohen, M.S.; Rissman, J.; Castel, A.D.; Knowlton, B.J. White Matter Integrity in Brain Structures Supporting Semantic Processing Is Associated with Value-Directed Remembering in Older Adults. Neuropsychologia 2019, 129, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Der, G.; Deary, I.J. Age and Sex Differences in Reaction Time in Adulthood: Results from the United Kingdom Health and Lifestyle Survey. Psychology and Aging 2006, 21, 62–73. [Google Scholar] [CrossRef] [PubMed]
- Ingalhalikar, M.; Smith, A.; Parker, D.; Satterthwaite, T.D.; Elliott, M.A.; Ruparel, K.; Hakonarson, H.; Gur, R.E.; Gur, R.C.; Verma, R. Sex Differences in the Structural Connectome of the Human Brain. Proceedings of the National Academy of Sciences 2014, 111, 823–828. [Google Scholar] [CrossRef] [PubMed]
- Del Mauro, G.; Zeng, X.; Wang, Z. Normative Brain Entropy Across the Lifespan. bioRxiv 2025, 2025.05.08.652915. [Google Scholar] [CrossRef]
- Kochunov, P.; Thompson, P.M.; Winkler, A.; Morrissey, M.; Fu, M.; Coyle, T.R.; Du, X.; Muellerklein, F.; Savransky, A.; Gaudiot, C.; et al. The Common Genetic Influence over Processing Speed and White Matter Microstructure: Evidence from the Old Order Amish and Human Connectome Projects. NeuroImage 2016, 125, 189–197. [Google Scholar] [CrossRef]
- Tuch, D.S.; Salat, D.H.; Wisco, J.J.; Zaleta, A.K.; Hevelone, N.D.; Rosas, H.D. Choice Reaction Time Performance Correlates with Diffusion Anisotropy in White Matter Pathways Supporting Visuospatial Attention. Proc Natl Acad Sci U S A 2005, 102, 12212–12217. [Google Scholar] [CrossRef]
- Durham, E.L.; Ghanem, K.; Stier, A.J.; Cardenas-Iniguez, C.; Reimann, G.E.; Jeong, H.J.; Dupont, R.M.; Dong, X.; Moore, T.M.; Berman, M.G.; et al. Multivariate Analytical Approaches for Investigating Brain-Behavior Relationships. Front Neurosci 2023, 17, 1175690. [Google Scholar] [CrossRef] [PubMed]
| Neuroimaging measures | Mean | Minimum | Maximum | Std. Dev. |
|---|---|---|---|---|
| ATR-L mean FA | 0.445 | 0.331 | 0.601 | 0.087 |
| ATR-R mean FA | 0.480 | 0.365 | 0.654 | 0.087 |
| CGC-L mean FA | 0.417 | 0.305 | 0.517 | 0.067 |
| CGC-R mean FA | 0.404 | 0.319 | 0.530 | 0.060 |
| CGH- L mean FA | 0.355 | 0.228 | 0.456 | 0.082 |
| CGH-R mean FA | 0.335 | 0.219 | 0.491 | 0.081 |
| CST-L mean FA | 0.553 | 0.431 | 0.674 | 0.081 |
| CST-R mean FA | 0.586 | 0.386 | 0.747 | 0.107 |
| Fmj mean FA | 0.522 | 0.422 | 0.621 | 0.067 |
| Fmn mean FA | 0.457 | 0.367 | 0.575 | 0.074 |
| IFOF-L mean FA | 0.479 | 0.360 | 0.593 | 0.091 |
| IFOF-R mean FA | 0.447 | 0.352 | 0.531 | 0.069 |
| ILF-L mean FA | 0.467 | 0.346 | 0.607 | 0.090 |
| ILF-R mean FA | 0.464 | 0.351 | 0.792 | 0.105 |
| SLF-L mean FA | 0.465 | 0.336 | 0.596 | 0.091 |
| SLF-R mean FA | 0.463 | 0.338 | 0.621 | 0.089 |
| UNC- L mean FA | 0.379 | 0.257 | 0.487 | 0.080 |
| UNC-R mean FA | 0.369 | 0.275 | 0.481 | 0.073 |
| Alpha Peak Frequency | 10.303 | 8.594 | 11.328 | 0.718 |
| Variable | Mean | Minimum | Maximum | Std. Dev |
| SRT | 463.54 | 408.00 | 518.00 | 35.49 |
| CRT | 433.37 | 357.00 | 555.00 | 47.14 |
| SDSTR | 78.58 | 44.04 | 114.78 | 17.59 |
| SDCRT | 85.81 | 45.24 | 138.71 | 22.75 |
| SKWSRT | 1.08 | -0.36 | 2.10 | 0.59 |
| Variable | Mean | Minimum | Maximum | Std. Dev |
| SKWCRT | 1.41 | -0.41 | 2.41 | 0.55 |
| Commission errors SRT | 1.8 | 0 | 9 | 2.10 |
| Omission errors SRT | 2.08 | 0 | 15 | 1.92 |
| Commission errors CRT | 2.71 | 0 | 12 | 2.56 |
| Omission errors CRT | 4.33 | 0 | 18 | 4.21 |
| Variables | Dimension 1 Canonical variate weights (p=0.015) |
Dimension 2 Canonical variate weights (p=0.013) |
|---|---|---|
| ATR-L mean FA | 0 | 0 |
| ATR-R mean FA | 0 | 0 |
| CGC-L mean FA | 0 | 0.363 |
| CGC-R mean FA | 0 | 0.303 |
| CGH-L mean FA | 0 | 0 |
| CGH- R mean FA | 0 | 0 |
| CST-L mean FA | 0 | 0.357 |
| CST-R mean FA | 0 | 0.346 |
| Fmj mean FA | 0 | 0.373 |
| Fmn mean FA | 0 | 0.307 |
| IFOF- L mean FA | 0.333 | 0 |
| IFOF-L mean FA | 0.337 | 0 |
| ILF- L mean FA | 0 | 0.301 |
| ILF-R mean FA | 0 | 0.409 |
| SLF-L mean FA | 0.423 | 0 |
| SLF-R mean FA | 0.439 | 0 |
| UNC-L mean FA | 0.379 | 0 |
| UNC-R mean FA | 0.345 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




