Submitted:
10 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
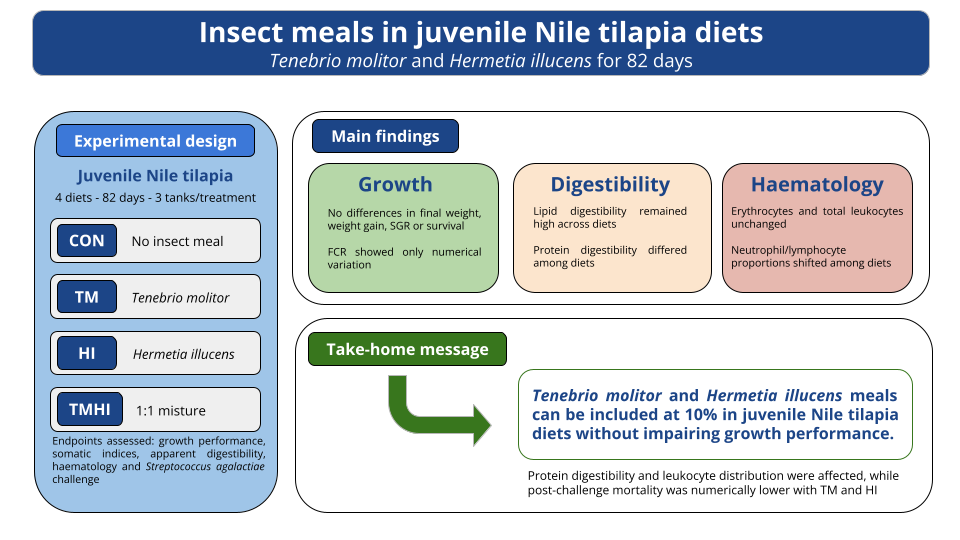
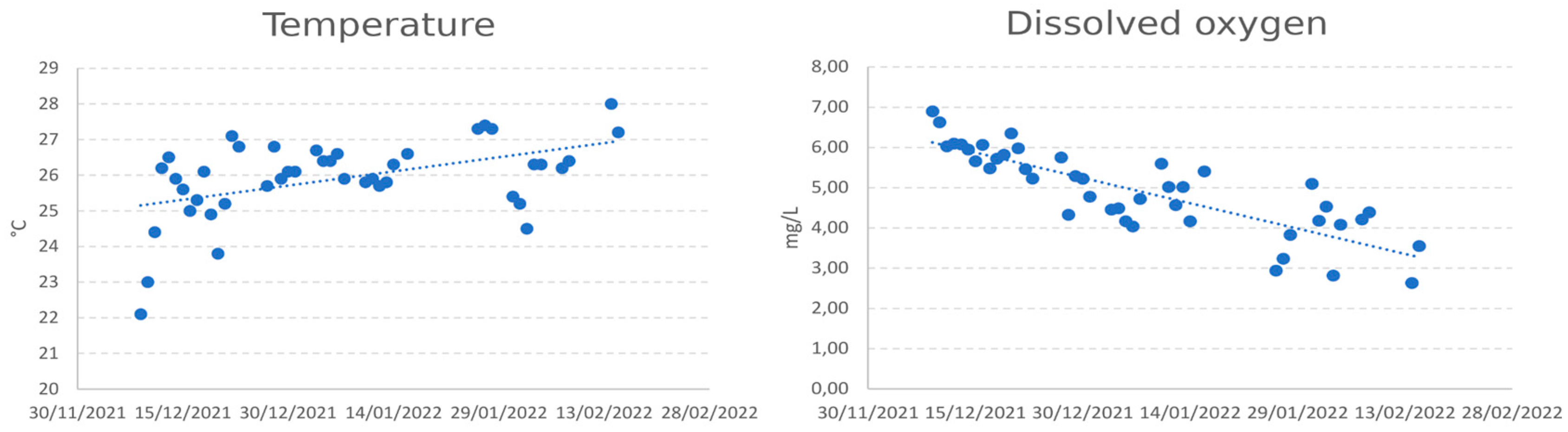
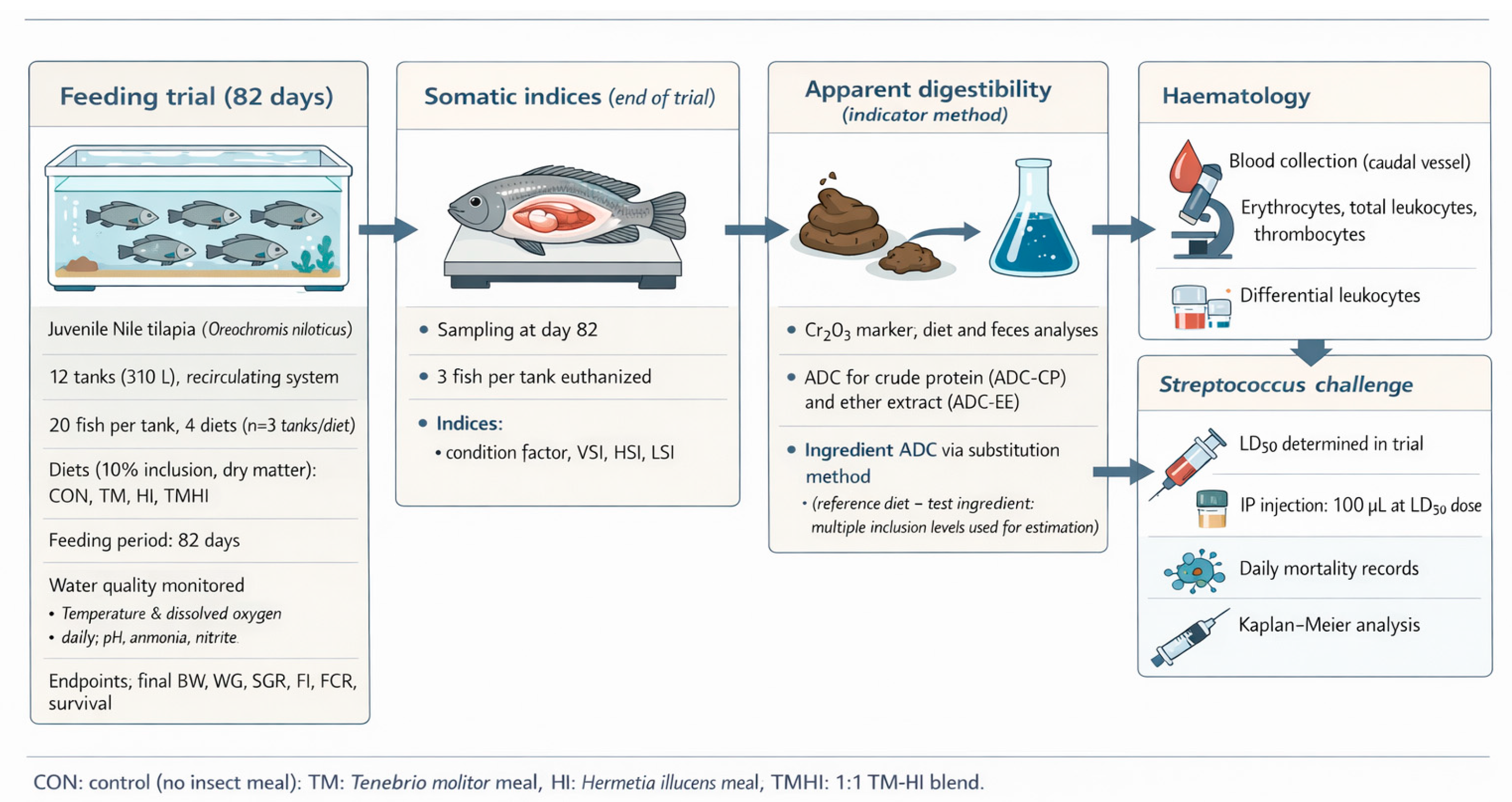
2.1. Experimental Facilities and Fish
2.2. Experimental Diets
2.2.1. Chemical Characterization of Insect Meals
| Identification* |
Crude protein (%) (mean ± SD**) |
p |
Total lipids (%) (mean ± SD**) |
p |
| TM | 49.9 ± 0.9 | NR | 35.5 ± 0.2 | NR |
| HI | 57.9 ± 0.2 | NR | 7.7 ± 0.2 | NR |
| Identification* | Saturated | Monounsaturated | ω-3 | ω-6 |
| TM | 7.97 | 16.63 | 0.26 | 8.49 |
| HI | 4.87 | 1.38 | 0.02 | 0.71 |
| Amino acid | TM* | HI* |
| Aspartic acid | 4.15 | 5.56 |
| Glutamic acid | 5.72 | 6.51 |
| Serine | 2.28 | 2.41 |
| Glycine | 3.13 | 3.86 |
| Histidine | 1.50 | 1.83 |
| Arginine | 2.81 | 3.11 |
| Threonine | 2.04 | 2.25 |
| Alanine | 3.59 | 3.50 |
| Proline | 2.85 | 3.03 |
| Tyrosine | 3.79 | 3.78 |
| Valine | 3.24 | 3.56 |
| Methionine | 0.80 | 1.12 |
| Cystine | 0.29 | 0.24 |
| Isoleucine | 2.20 | 2.40 |
| Leucine | 3.68 | 3.91 |
| Phenylalanine | 1.93 | 2.53 |
| Lysine | 2.71 | 3.51 |
| Tryptophan | 0.22 ± 0.00** | 0.42 ± 0.00** |
2.2.2. Diet Formulation and Processing
2.2.3. Growth Performance and Somatic Indices
2.2.4. Apparent Digestibility
2.2.5. Haematological Analyses
2.2.6. Streptococcus Agalactiae Challenge
2.2.7. Statistical Analysis
3. Results
3.1. Growth Performance
3.2. Survival and Somatic Indices
3.3. Apparent Digestibility Coefficients
3.4. Haematological Parameters
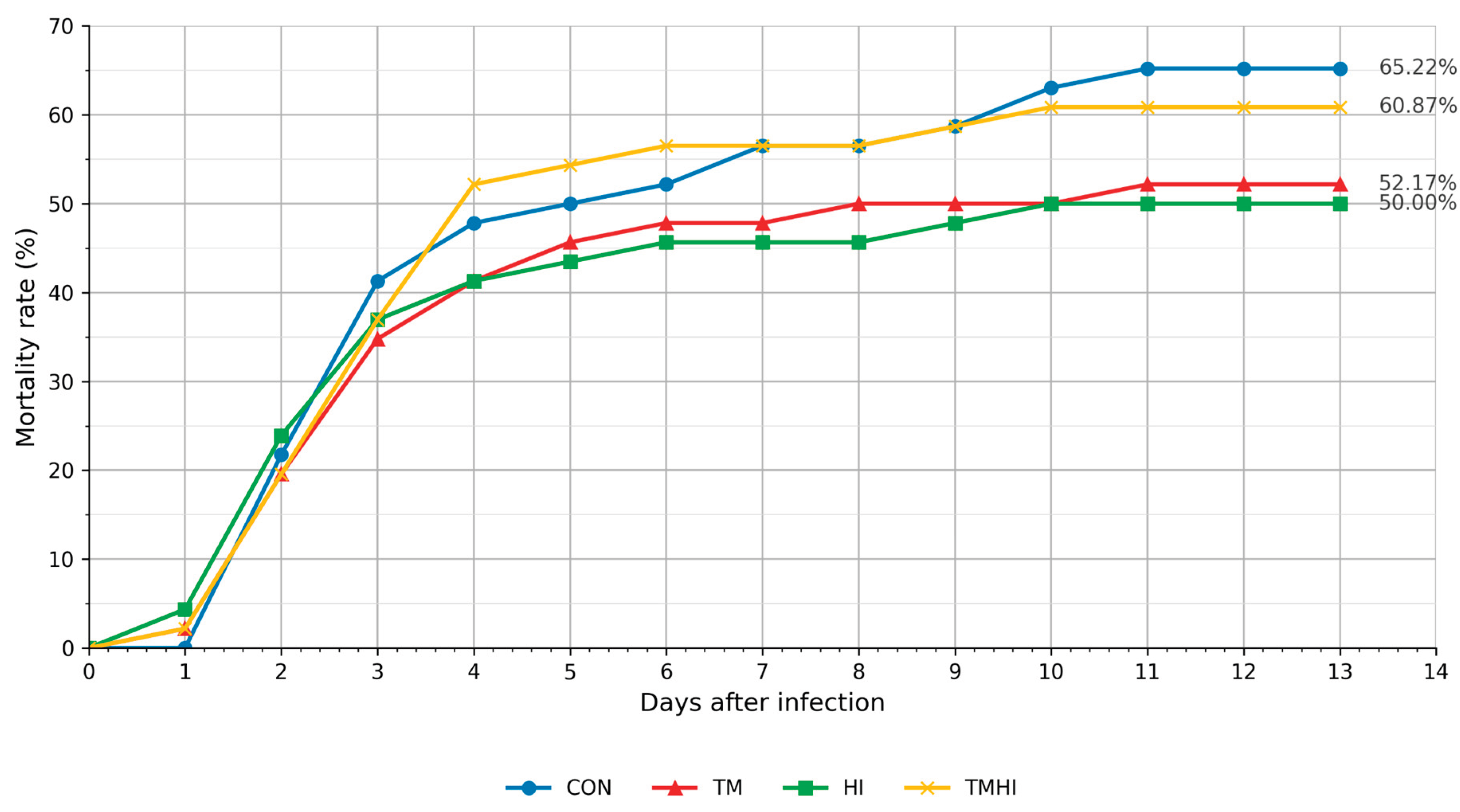
3.5. Streptococcus Agalactiae Challenge and Mortality
4. Discussion
4.1. Growth Performance and Somatic Responses
4.2. Nutrient Digestibility of Tenebrio Molitor and Hermetia Illucens Meals
4.3. Haematological Responses and Immunomodulation
4.4. Resistance to Streptococcus Agalactiae
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADC | Apparent Digestibility Coefficient |
| ADC-CP | Apparent Digestibility Coefficient of Crude Protein |
| ADC-EE | Apparent Digestibility Coefficient of Ether Extract |
| AOAC | Association of Official Analytical Chemists |
| BHI | Brain Heart Infusion |
| CFU | Colony-Forming Units |
| CON | Control Diet |
| Cr₂O₃ | Chromium(III) Oxide |
| EDTA | Ethylenediaminetetraacetic Acid |
| FAME | Fatty Acid Methyl Esters |
| FCR | Feed Conversion Ratio |
| HI | Hermetia illucens Larvae Meal Diet |
| HSI | Hepatosomatic Index |
| LD₅₀ | Median Lethal Dose |
| LSI | Liposomatic Index |
| PER | Protein Efficiency Ratio |
| SGR | Specific Growth Rate |
| TM | Tenebrio molitor Larvae Meal Diet |
| TMHI | Diet Containing a 1:1 Mixture of Tenebrio molitor and Hermetia illucens Larvae Meals |
| VSI | Viscerosomatic Index |
References
- Food and Agriculture Organization of the United Nations (FAO). The State of World Fisheries and Aquaculture 2024: Blue Transformation in Action; FAO: Rome, 2024; Available online: https://www.fao.org/publications/sofia-2024.
- Paredes-Trujillo, A.I.; Mendoza-Carranza, M. Streptococcosis in Nile tilapia farming: a severe yet overlooked health challenge in Mexico, a study case in Campeche, Mexico. Bull. Eur. Assoc. Fish. Pathol. Available from. 2024, 45, 1–17. [Google Scholar] [CrossRef]
- Hua, K. A meta-analysis of the effects of replacing fish meals with insect meals on growth performance of fish. Aquaculture Available from. 2021, 530, 735732. [Google Scholar] [CrossRef]
- Maulu, S.; Langi, S.; et al. Recent advances in the utilization of insects as an ingredient in aquafeeds: a review. 2022. Available online: https://pubmed.ncbi.nlm.nih.gov/36329686/.
- Islam, S.M.M.; Amrin, A.A.H.; Yousef, A.; Abol-Munafi, K.; Dayal, A.; Abdul-Rahim, A.M.K. Insect-based aquafeeds and fish mucosal immune responses: a systematic review. Rev. Aquac. 2024, 16, 1359–1390. [Google Scholar] [CrossRef]
- Hasan, M.M.; Alam, M.S.; Rayhan, N.; Rahman, M.M. Insects as alternative protein sources in fish nutrition: a review. Fishes 2023, 8, 291. [Google Scholar] [CrossRef]
- Ragozzino-Paulino, P.; Cocato, M.L.; de Souza Sarkis, J.E. Nutritional Potential of Edible Insects as Alternative Ingredients in Fish Feed: A Path to Modern Aquaculture. Aquac. Nutr. 2025, 7009004, 30. [Google Scholar] [CrossRef]
- Ihuț, A.; Răducu, C.; Uiuiu, P.; Munteanu, C. From gut to fillet: comprehensive effects of Tenebrio molitor in fish nutrition. Fishes Available from. 2025, 10, 468. [Google Scholar] [CrossRef]
- Liu, Q.; Li, J.; Liu, J.; Cai, Y.; Luo, Y.; Sangzhu, T.; et al. Enhanced Nile tilapia meat quality by the metabolomic effects of Tenebrio molitor larval meal dietary supplement. Appl. Food Res. 2025, 5, 101117. [Google Scholar] [CrossRef]
- Tippayadara, N.; Dawood, M.A.O.; Krutmuang, P.; Hoseinifar, S.H.; Doan, H.V.; Paolucci, M. Replacement of fish meal by black soldier fly (Hermetia illucens) larvae meal: effects on growth, haematology, and skin mucus immunity of Nile tilapia, Oreochromis niloticus. Animals 2021, 11, 193. [Google Scholar] [CrossRef]
- Shati, S.; Öz, M.; Erteken, A. Hermetia illucens in the feeding of Nile tilapia larvae. Aquat. Res. 2022, 5, 207–218. Available online: https://aquatres.scientificwebjournals.com/en/pub/issue/69220/1108366.
- Oliveira, C.G.; Araújo, R.D.; Prado, V.G.L.; Guilherme, H.O.; Paulino, R.R.; Costa, L.S. Impact of replacing fish meal with black soldier fly (Hermetia illucens) meal on diet acceptability in juvenile Nile tilapia: palatability and nutritional and health considerations for dietary preference. Aquac. Res. Available from. 2024, 2024, 3409955. [Google Scholar] [CrossRef]
- Wang, T.; Zhang, Y.Z.; Bao, M.Y.; Qin, X.P.; Guan, G.X. Defatted Hermetia illucens meal improves gut health and disease resistance in Nile tilapia under bacterial challenge. Fish Shellfish. Immunol. 2025, 110242. [Google Scholar] [CrossRef]
- Abd El-Gawad, E.A.; Zahran, E.; Youssuf, H.; Shehab, A.; Matter, A.F. Defatted black soldier fly (Hermetia illucens) diets improved hemato-immunological responses, biochemical parameters, and antioxidant activities in Streptococcus iniae-infected Nile tilapia (Oreochromis niloticus). BMC Vet. Res. 2025, 21, 104. [Google Scholar] [CrossRef]
- Haenen, O.L.M.; Dalsgaard, I.; Vendrell, D.; Peters, R.; et al. Bacterial diseases of tilapia, their zoonotic potential and risk management. Rev. Aquac. 2023, 15, e12743. Available online: https://onlinelibrary.wiley.com/doi/10.1111/raq.12743. [CrossRef]
- Abdallah, A.; Elbestawy, A.; Radwan, I.; Eltahan, A.; Soliman, W.S.; Nasr-Elsayed, S.; et al. Update on Streptococcus agalactiae infection in tilapia: epidemiology, pathogenesis, diagnosis and control. Biology. 2024. Available online: https://pubmed.ncbi.nlm.nih.gov/39472379/.
- Owatari, M.S.; Mouriño, J.L.P.; Martins, M.L. Antimicrobial resistance: a concern related to streptococcosis in tilapia farming. Mar Fish Sci (MAFIS) 2024, 37, 337–352. [Google Scholar] [CrossRef]
- Alves, A.P.C.; Paulino, R.R.; Pereira, R.T.; Costa, D.V.; Rosa, P.V. Nile tilapia fed insect meal: growth and innate immune response in different times under lipopolysaccharide challenge. Aquac. Res. Available from. 2021, 52, 529–540. [Google Scholar] [CrossRef]
- ABNT. NBR ISO 9001:2015. Sistemas de gestão da qualidade – requisitos. ABNT: Rio de Janeiro, 2015.
- AOAC. Official Methods of Analysis of AOAC International. In Method 2001.11, 17th edn; AOAC International: Gaithersburg, MD, 2002. [Google Scholar]
- AOAC. Official Methods of Analysis of AOAC International. In Method 920.39, 22nd edn; AOAC International: Rockville, MD, 2023. [Google Scholar]
- AOAC. Official Methods of Analysis of AOAC International. In Method 969.33, 22nd edn; AOAC International: Rockville, MD, 2023. [Google Scholar]
- AOAC. Official Methods of Analysis of AOAC International, Method 982.30, 22nd edn; AOAC International: Rockville, MD, 2023. [Google Scholar]
- Rockville. AOAC; 2023 Official Methods of Analysis of AOACInternational 22nd edn Method 99412 AOACInternational.
- AOAC. Official Methods of Analysis of AOAC International, Method 988.15, 22nd edn; AOAC International: Rockville, MD, 2023. [Google Scholar]
- National Research Council (NRC. Nutrient Requirements of Fish and Shrimp; National Academies Press: Washington, DC, USA, 2011; p. 376 pp. [Google Scholar]
- Bureau, D.P.; Harris, A.M.; Cho, C.Y. Apparent digestibility of rendered animal protein ingredients for rainbow trout (Oncorhynchus mykiss). Aquaculture 1999, 180, 345–358. [Google Scholar] [CrossRef]
- Abimorad, E.G.; Carneiro, D.J. Fecal collection methods and determination of crude protein and gross energy digestibility coefficients of feedstuffs for pacu, Piaractus mesopotamicus (Holmberg, 1887). Braz. J. Anim. Sci. 2004, 33, 1101–1109. [Google Scholar] [CrossRef]
- Nose, T. On the digestion of food protein by goldfish and rainbow trout. Bull. Freshw. Fish. Res. Lab. 1960, 10, 11–22. [Google Scholar]
- Goldenfarb, P. B.; Bowyer, F. P.; Hall, E.; Brosious, E. Reproducibility in the hematology laboratory: the microhematocrit determination. Am. J. Clin. Pathol. 1971, 56, 35–39. [Google Scholar] [CrossRef]
- Tran, H.Q.; Van Doan, H.; Stejskal, V. Environmental consequences of using insect meal as an ingredient in aquafeeds: A systematic view. Rev. Aquac. 2021, 14, 237–251. [Google Scholar] [CrossRef]
- Sánchez-Muros, M.J.; Barroso, F.G.; Manzano-Agugliaro, F. Insect meal as renewable source of food for animal feeding: a review. J. Clean. Prod. 2014, 65, 16–27. [Google Scholar] [CrossRef]
- Devic, E.; Leschen, W.; Murray, F.; Little, D.C. Growth performance, feed utilization and body composition of advanced nursing Nile tilapia (Oreochromis niloticus) fed diets containing Black Soldier Fly (Hermetia illucens) larvae meal. Aquacult Nutr. 2018, 24, 416–423. [Google Scholar] [CrossRef]
- Dietz, C.; Liebert, F. Does graded substitution of soy protein concentrate by an insect meal respond on growth and N-utilization in Nile tilapia (Oreochromis niloticus)? Aquac. Rep. 2018, 12, 43–48. [Google Scholar] [CrossRef]
- Goldenfarb, P.B.; Bowyer, F.P.; Hall, E.; Brosious, E. Reproducibility in the hematology laboratory: the microhematocrit determination. Am. J. Clin. Pathol. 1971, 56, 35–39. [Google Scholar] [CrossRef]
- Tubin, J.S.B.; Paiano, D.; Hashimoto, G.S.O.; Furtado, W.E.; Martins, M.L.; Durigon, E.; et al. Tenebrio molitor meal in diets for Nile tilapia juveniles reared in biofloc system. Aquaculture 2020, 519, 734763. [Google Scholar] [CrossRef]
- Fontes, T.V.; de Oliveira, K.R.B.; Gomes Almeida, I.L.; Maria Orlando, T.; Rodrigues, P.B.; Costa, D.V.d.; Rosa, P.V.e. Digestibility of insect meals for Nile tilapia fingerlings. Animals 2019, 9, 181. [Google Scholar] [CrossRef]
- Alfiko, Y.; Xie, D.; Astuti, R.T.; Wong, J.; Wang, L. Insects as a feed ingredient for fish culture: Status and trends. Aquac. Fish. 2022, 7, 166–178. [Google Scholar] [CrossRef]
- Li, S.; Ji, H.; Zhang, B.; Tian, J.; Zhou, J.; Yu, H. Influence of black soldier fly (Hermetia illucens) larvae oil on growth performance, body composition, tissue fatty acid composition and lipid deposition in juvenile Jian carp (Cyprinus carpio var. Jian). Aquaculture 2016, 465, 43–52. [Google Scholar] [CrossRef]
- Glencross, B.D. A feed is still only as good as its ingredients: An update on the nutritional research strategies for the optimal evaluation of ingredients for aquaculture feeds. Aquacult Nutr. 2020, 26, 1871–1883. [Google Scholar] [CrossRef]
- Santinha, P.J.M.; Gomes, E.F.S.; Coimbra, J.O. Effects of protein level of the diet on digestibility and growth of gilthead sea bream, Sparus auratus L. Aquacult Nutr. 1996, 2, 81–87. [Google Scholar] [CrossRef]
- Schrama, J.W.; Saravanan, S.; Geurden, I.; Heinsbroek, L.T.N.; Kaushik, S.J.; Verreth, J.A.J. Dietary nutrient composition affects digestible energy utilisation for growth: A study on Nile tilapia (Oreochromis niloticus) and a literature comparison across fish species. Br. J. Nutr. 2012, 108, 277–289. [Google Scholar] [CrossRef]
- Muin, H.; Taufek, N.M. Evaluation of growth performance, feed efficiency and nutrient digestibility of red hybrid tilapia fed dietary inclusion of black soldier fly larvae (Hermetia illucens). Aquac. Fish. 2024, 9, 46–51. [Google Scholar] [CrossRef]
- Hasan, I.; Gai, F.; Cirrincione, S.; Rimoldi, S.; Saroglia, G.; Terova, G. Chitinase and insect meal in aquaculture nutrition: A comprehensive overview of the latest achievements. Fishes 2023, 8, 607. [Google Scholar] [CrossRef]
- Eggink, K.M.; Pedersen, P.B.; Lund, I.; Dalsgaard, J. Chitin digestibility and intestinal exochitinase activity in Nile tilapia and rainbow trout fed different black soldier fly larvae meal size fractions. Aquac. Res. 2022, 53, 5536–5546. [Google Scholar] [CrossRef]
- Ebling, M.L.P.; Taschetto, E.A.; Pelizari, A.; Pietro, B.C.; Loureiro, B.B.; Lazzari, R.; et al. Tenebrio molitor larvae meal and evaluation of nutritional composition and apparent digestibility coefficient in silver catfish (Rhamdia quelen) feed. Cienc. Rural. 2025, 55, e20240408. [Google Scholar] [CrossRef]
- Quang Tran, H.; Woynarovich, A.; Thongvam, T.; Phuong, N.T. Environmental consequences of using insect meal as an alternative protein source in aquafeed: a review. Rev. Aquac. 2022, 14, 1474–1494. [Google Scholar] [CrossRef]
- Fazio, F. Fish hematology analysis as an important tool of aquaculture: A review. Aquaculture 2019, 500, 237–242. [Google Scholar] [CrossRef]
- Nogales-Mérida, S.; Gobbi, P.; Józefiak, D.; Mazurkiewicz, J.; Dudek, K.; Rawski, M.; Kierończyk, B.; Józefiak, A. Insect meals in fish nutrition. Rev. Aquac. 2019, 11, 1080–1103. [Google Scholar] [CrossRef]
- Hameed, A.; Majeed, A.; Maqsood, S.; Hamed, M.; Mahmood, A.; et al. Success of aquaculture industry with new insights of using insects as fish feed: a review. Fishes 2022, 7, 395. Available online: https://www.mdpi.com/2410-3888/7/6/395. [CrossRef]
- Patyra, E.; Kwiatek, K. Insect meals and insect antimicrobial peptides as an alternative for antibiotics and growth promoters in livestock production. Pathogens 2023, 12, 854. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, E.S.H.; Metwally, W.G.M.; Abdel-Rahman, M.A.M.; Albano, M.; Mahmoud, M.M. Streptococcus agalactiae infection in Nile tilapia (Oreochromis niloticus): A review. Biology 2024, 13, 914. [Google Scholar] [CrossRef] [PubMed]
- Delphino, M.K.V.C.; Leal, C.A.G.; Gardner, I.A.; Assis, G.C.; Jr, W.S.; Inoue, L.A.K.A.; et al. Economic appraisal of vaccination against Streptococcus agalactiae in Nile tilapia farms in Brazil. Aquaculture 2019, 518, 734881. Available online: https://pubmed.ncbi.nlm.nih.gov/30621892/.
- Fyrand, K.; Xu, C.; Evensen, Ø. Characterization of Streptococcus agalactiae 1a isolated from farmed Nile tilapia (Oreochromis niloticus) in North America, Central America, and Southeast Asia. Fish. Shellfish. Immunol. 2024, 154, 109919. [Google Scholar] [CrossRef]
- Owatari, M.S.; Mouriño, J.L.P.; Martins, M.L. Antimicrobial resistance: a concern related to streptococcosis in tilapia farming. Mar Fish Sci (MAFIS) 2024, 37, 337–352. [Google Scholar] [CrossRef]
- Delphino, M.; Joshi, R.; Alvarez, A.T. Economic appraisal of using genetics to control Streptococcus agalactiae in Nile tilapia under cage and pond farming system in Malaysia. Sci. Rep. 2022, 12, 8754. Available online: https://pmc.ncbi.nlm.nih.gov/articles/PMC9130118/. [CrossRef]
- Pau, E.J.J.N.; Yong, C.C. Input–output analysis of Streptococcus disease impact on Malaysian tilapia production and exports. Aquacult Int. 2025, 33, 494. [Google Scholar] [CrossRef]
- Henry, M.A.; Gasco, L.; Piccolo, G.; Fountoulaki, E. Effect of partial dietary replacement of fishmeal by yellow mealworm (Tenebrio molitor) larvae meal on the innate immune response and intestinal antioxidant enzymes of rainbow trout (Oncorhynchus mykiss). Fish. Shellfish. Immunol. 2018, 83, 308–313. [Google Scholar] [CrossRef]
- Su, J.; Gong, Y.; Cao, S.; Lu, F.; Han, D.; Liu, H.; et al. Effects of dietary Tenebrio molitor meal on the growth performance, immune response and disease resistance of yellow catfish (Pelteobagrus fulvidraco). Fish. Shellfish. Immunol. 2017, 69, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Sankian, Z.; Khosravi, S.; Kim, Y.O.; Lee, S.M. Effects of dietary inclusion of yellow mealworm (Tenebrio molitor) meal on growth performance, feed utilization, body composition, and immune responses of mandarin fish (Siniperca scherzeri). Aquaculture 2018, 496, 79–87. [Google Scholar] [CrossRef]
- Libanori, M.C.M.; Santos, G.G.; Pereira, S.A.; Lopes, G.R.; Owatari, M.S.; Soligo, T.A.; Yamashita, E.; Pereira, U.P.; Martins, M.L.; Mouriño, J.L.P. Dietary supplementation with benzoic organic acid improves the growth performance and survival of Nile tilapia (Oreochromis niloticus) after challenge with Streptococcus agalactiae (Group B). Aquaculture 2021, 545, 737204. [Google Scholar] [CrossRef]
- Rimoldi, S.; Di Rosa, A.R.; Oteri, M.; Chiofalo, B.; Hasan, I.; Saroglia, M.; Terova, G. The impact of diets containing Hermetia illucens meal on the growth, intestinal health, and microbiota of gilthead seabream (Sparus aurata). Fish. Physiol. Biochem. 2024, 50, 1003–1024. [Google Scholar] [CrossRef]
- Franco, A.; Scieuzo, C.; Salvia, R.; Pucciarelli, V.; Borrelli, L.; Addeo, N.F.; Bovera, F.; Laginestra, A.; Schmitt, E.; Falabella, P. Antimicrobial activity of lipids extracted from Hermetia illucens reared on different substrates. Appl. Microbiol. Biotechnol. 2024, 108, 167. [Google Scholar] [CrossRef]
- Costa, R.S.; Basto, A.; Monteiro, M.; Pinho, B.; Sá, T.; Santos, M.V.; Murta, D.; Schrama, J.W.; Valente, L.M.P. Combining Hermetia illucens and Tenebrio molitor meals in diets for European seabass: Effects on growth, nutrient utilisation, intestinal morphology and muscle quality. Aquaculture 2026, 610, 742899. [Google Scholar] [CrossRef]
| Diet | CON | TM | HI | TMHI |
| Ingredient composition (g kg⁻¹) | ||||
| Insect meal¹ | 0 | 100 | 100 | 100 |
| Poultry by-product meal | 109 | 38.6 | 40 | 42.5 |
| Meat and bone meal | 70 | 70 | 70 | 70 |
| Blood meal | 50 | 33.5 | 32.8 | 30 |
| Soybean meal | 310 | 320 | 303.7 | 314.1 |
| Wheat bran | 100 | 100 | 100 | 100 |
| Broken rice | 80 | 80 | 80 | 80 |
| Corn | 200 | 200 | 190 | 191 |
| Corn starch | 40 | 40 | 40 | 40 |
| Soybean oil | 26.5 | 0 | 28.6 | 17.3 |
| Dicalcium phosphate | 8.4 | 10 | 9.2 | 9.3 |
| DL-methionine | 1 | 2 | 1 | 0.7 |
| Vitamin–mineral premix² | 5 | 5 | 5 | 5 |
| Calculated composition | ||||
| Crude protein (%) | 31.5 | 31.5 | 31.5 | 31.5 |
| Digestible energy (kcal kg⁻¹) | 3176 | 3234 | 3244 | 3243 |
| Ether extract (%) | 6.7 | 6.7 | 6.7 | 6.7 |
| Ash (%) | 7.7 | 7.1 | 7.7 | 7.3 |
| Crude fibre (%) | 3.4 | 3.4 | 3.2 | 3.3 |
| Nitrogen-free extract (%) | 40.6 | 41 | 40.8 | 41.3 |
| Calcium (%) | 1.4 | 1.33 | 1.37 | 1.32 |
| Available phosphorus (%) | 0.81 | 0.83 | 0.84 | 0.82 |
| Lysine (%) | 1.72 | 1.76 | 1.79 | 1.79 |
| Methionine (%) | 0.54 | 0.65 | 0.54 | 0.56 |
| Diet | Initial body weight (g) | Final body weight (g) |
Weight gain (g) |
SGR (% day⁻¹) |
Feed intake (g) |
FCR |
| CON | 28.83 ± 0.64 | 248.31 ± 13.31 | 219.48 ± 13.23 | 2.76 ± 0.07 | 262.34 ± 15.74 | 1.20 ± 0.03 |
| TM | 28.28 ± 0.28 | 254.99 ± 10.77 | 226.71 ± 10.50 | 2.82 ± 0.04 | 260.58 ± 3.94 | 1.15 ± 0.05 |
| HI | 28.75 ± 0.75 | 252.97 ± 19.14 | 224.21 ± 19.05 | 2.79 ± 0.10 | 276.24 ± 13.38 | 1.25 ± 0.04 |
| TMHI | 29.22 ± 0.32 | 260.36 ± 6.35 | 231.14 ± 6.05 | 2.80 ± 0.02 | 270.63 ± 2.71 | 1.17 ± 0.02 |
| P-value | 0.2802 | 0.7388 | 0.7464 | 0.7110 | 0.3043 | 0.0525 |
| Diet | Survival (%) | Condition factor | VSI (%) | HSI (%) | LSI (%) |
| CON | 96.7 ± 5.8 | 2.05 ± 0.12 | 10.36 ± 0.24 | 2.38 ± 0.17 | 1.95 ± 0.34 |
| TM | 100.0 ± 0.0 | 2.19 ± 0.05 | 10.88 ± 0.51 | 1.99 ± 0.43 | 2.23 ± 0.32 |
| HI | 93.3 ± 5.8 | 2.09 ± 0.11 | 10.34 ± 2.01 | 2.14 ± 0.83 | 1.72 ± 0.49 |
| TMHI | 100.0 ± 0.0 | 2.10 ± 0.06 | 10.96 ± 0.74 | 1.81 ± 0.14 | 2.40 ± 0.06 |
| P-value | 0.7797 | 0.3832 | 0.8487 | 0.5478 | 0.0544 |
| Diet | Diets – test diets | Ingredients – insect meals | ||
| ADC-CP (%) | ADC-EE (%) | ADC-CP (%) | ADC-EE (%) | |
| CON | 86.95 ± 0.71ᵃ | 87.22 ± 1.57 | — | — |
| TM | 83.60 ± 1.87ᵇ | 87.66 ± 1.42 | 75.79 ± 6.22ᵇ | 88.70 ± 4.73 |
| HI | 87.31 ± 0.87ᵃ | 86.76 ± 1.66 | 88.16 ± 2.90ᵃ | 85.69 ± 5.52 |
| TMHI | 85.71 ± 0.42ᵃ | 88.95 ± 0.91 | 82.83 ± 1.41ᵃᵇ | 93.48 ± 2.93 |
| P-value | 0.0134** | 0.9132 | 0.0265* | 0.8343 |
| Diet | Erythrocytes (×10³ µL⁻¹) | Leukocytes (×10³ µL⁻¹) |
| CON | 2461.6 ± 216.5 | 26.58 ± 3.28 |
| TM | 2536.7 ± 270.3 | 25.71 ± 3.70 |
| HI | 2307.5 ± 284.8 | 26.37 ± 4.73 |
| TMHI | 2612.5 ± 369.3 | 31.18 ± 9.31 |
| P-value | 0.3332 | 0.3616 |
| Diet | Neutrophils (%) | Lymphocytes (%) | Monocytes (%) | Eosinophils (%) | Basophils (%) |
| CON | 32.33 ± 5.75ᵃ | 55.33 ± 3.62ᵇ | 12.00 ± 4.20 | 0.17 ± 0.41 | 0.17 ± 0.41 |
| TM | 26.33 ± 3.88ᵃᵇ | 61.83 ± 4.49ᵃᵇ | 11.67 ± 2.34 | 0.17 ± 0.41 | 0.00 ± 0.00 |
| HI | 22.50 ± 5.57ᵇ | 66.33 ± 6.41ᵃ | 10.33 ± 3.01 | 0.33 ± 0.52 | 0.50 ± 0.55 |
| TMHI | 21.83 ± 8.11ᵇ | 65.00 ± 7.77ᵃ | 13.00 ± 2.10 | 0.16 ± 0.41 | 0.00 ± 0.00 |
| P-value | 0.0247* | 0.0172* | 0.5101 | 0.8832 | 0.0628 |
| Diet | n (total) | Dead (n) | Survivors (n) | Cumulative mortality (%) [95% CI] | Survival (%) | Observation period (days) |
| CON | 46 | 30 | 16 | 65.2 [50.8–77.3] | 34.8 | 13 |
| TM | 46 | 24 | 22 | 52.2 [38.1–65.9] | 47.8 | 13 |
| HI | 46 | 23 | 23 | 50.0 [36.1–63.9] | 50.0 | 13 |
| TMHI | 46 | 28 | 18 | 60.9 [46.5–73.6] | 39.1 | 13 |
| Day post-challenge |
CON (dead n) |
CON (cum. mort. %) |
TM (dead n) |
TM (cum. mort. %) |
HI (dead n) |
HI (cum. mort. %) |
TMHI (dead n) |
TMHI (cum. mort. %) |
| 0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 |
| 1 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 |
| 2 | 0 | 0.0 | 1 | 2.2 | 2 | 4.3 | 1 | 2.2 |
| 3 | 10 | 21.7 | 8 | 19.6 | 9 | 23.9 | 8 | 19.6 |
| 4 | 9 | 41.3 | 7 | 34.8 | 6 | 37.0 | 8 | 37.0 |
| 5 | 3 | 47.8 | 3 | 41.3 | 2 | 41.3 | 7 | 52.2 |
| 6 | 1 | 50.0 | 2 | 45.7 | 1 | 43.5 | 1 | 54.3 |
| 7 | 1 | 52.2 | 1 | 47.8 | 1 | 45.7 | 1 | 56.5 |
| 8 | 2 | 56.5 | 0 | 47.8 | 0 | 45.7 | 0 | 56.5 |
| 9 | 0 | 56.5 | 1 | 50.0 | 0 | 45.7 | 0 | 56.5 |
| 10 | 1 | 58.7 | 0 | 50.0 | 1 | 47.8 | 1 | 58.7 |
| 11 | 2 | 63.0 | 0 | 50.0 | 1 | 50.0 | 1 | 60.9 |
| 12 | 1 | 65.2 | 1 | 52.2 | 0 | 50.0 | 0 | 60.9 |
| 13 | 0 | 65.2 | 0 | 52.2 | 0 | 50.0 | 0 | 60.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




