Submitted:
09 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Experimental Condition and Material
2.2. Experimental Design
2.3. Sample Collection and Processing
2.4. Measurements and Analytical Methods
2.4.1. Soil Bulk Weight
2.4.2. Soil Aggregate Analysis
2.4.3. Determination of Different Forms Nitrogen Content in Soil
2.4.4. Determination of Enzyme Activities in Soil
2.4.5. Determination of Maize Kernel Quality Indicators
2.5. Data Analysis
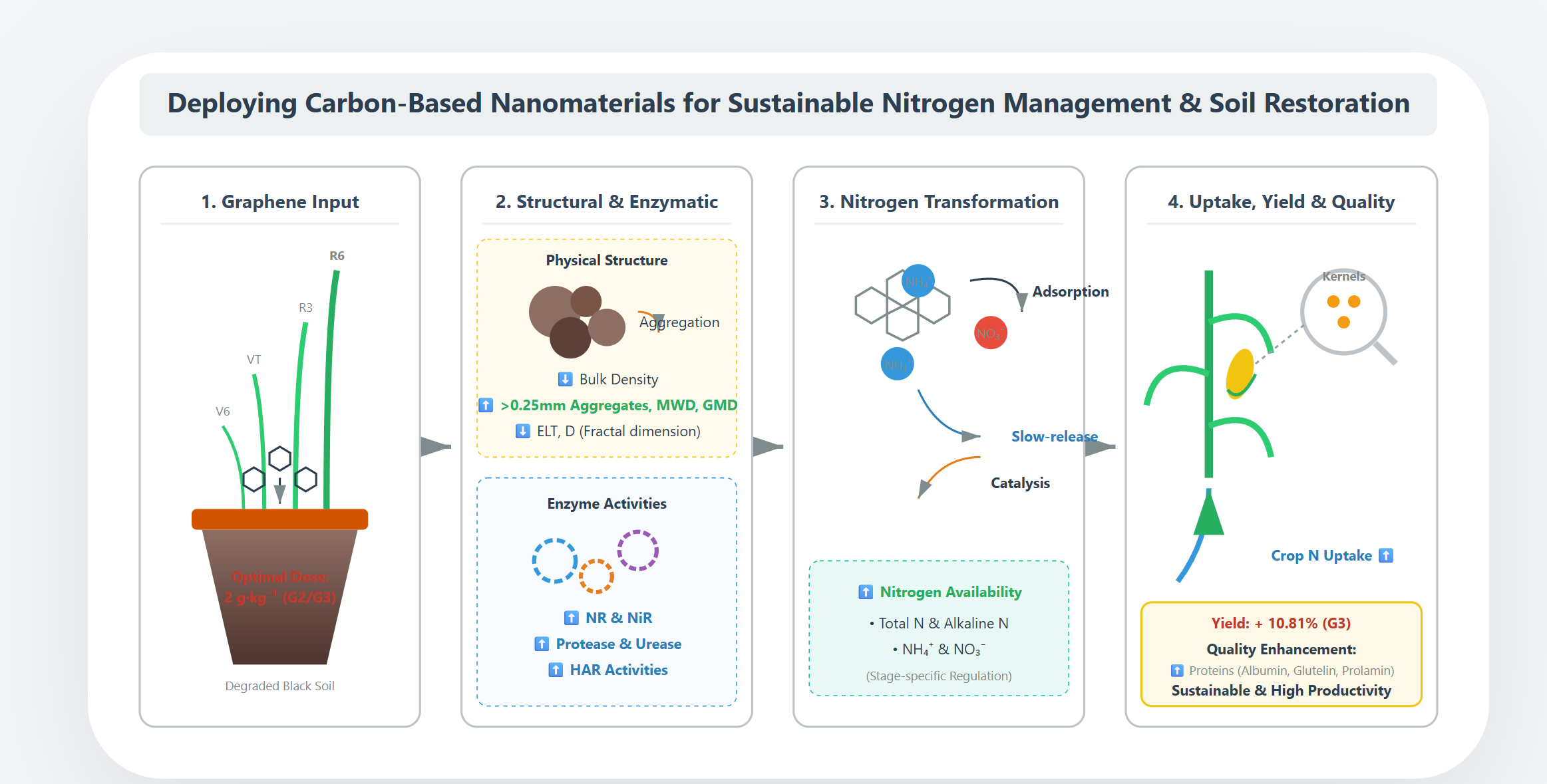
3. Results
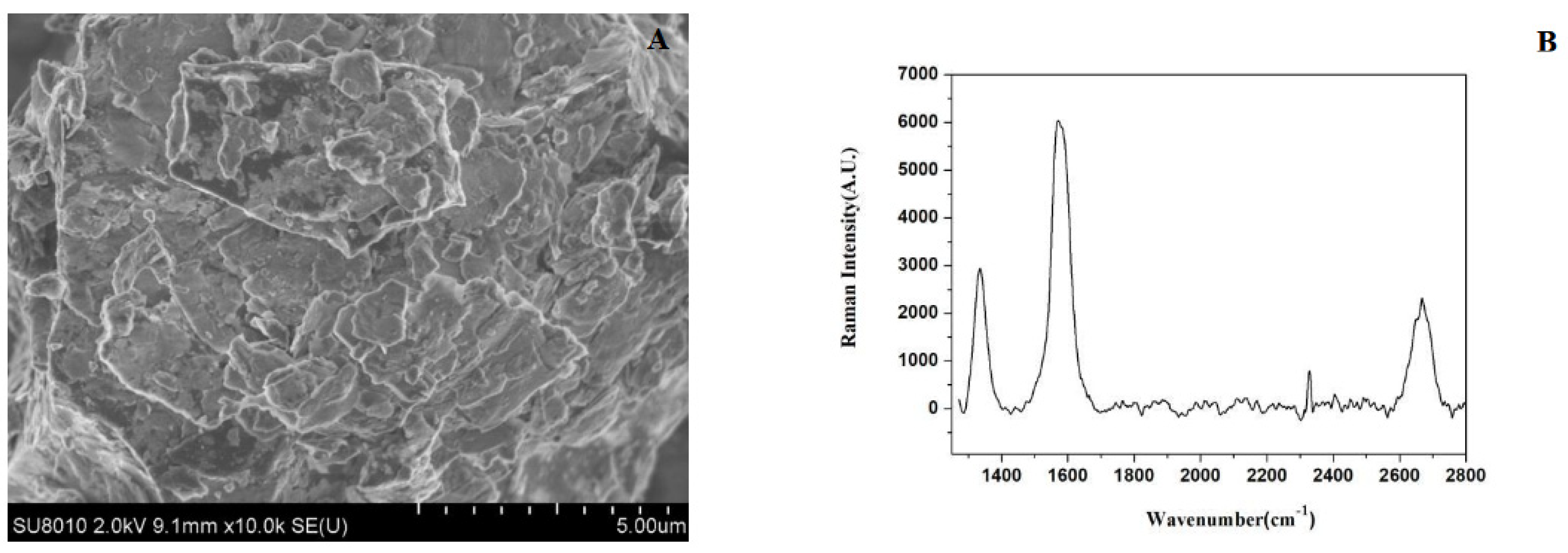
3.1. Characterization of Graphene
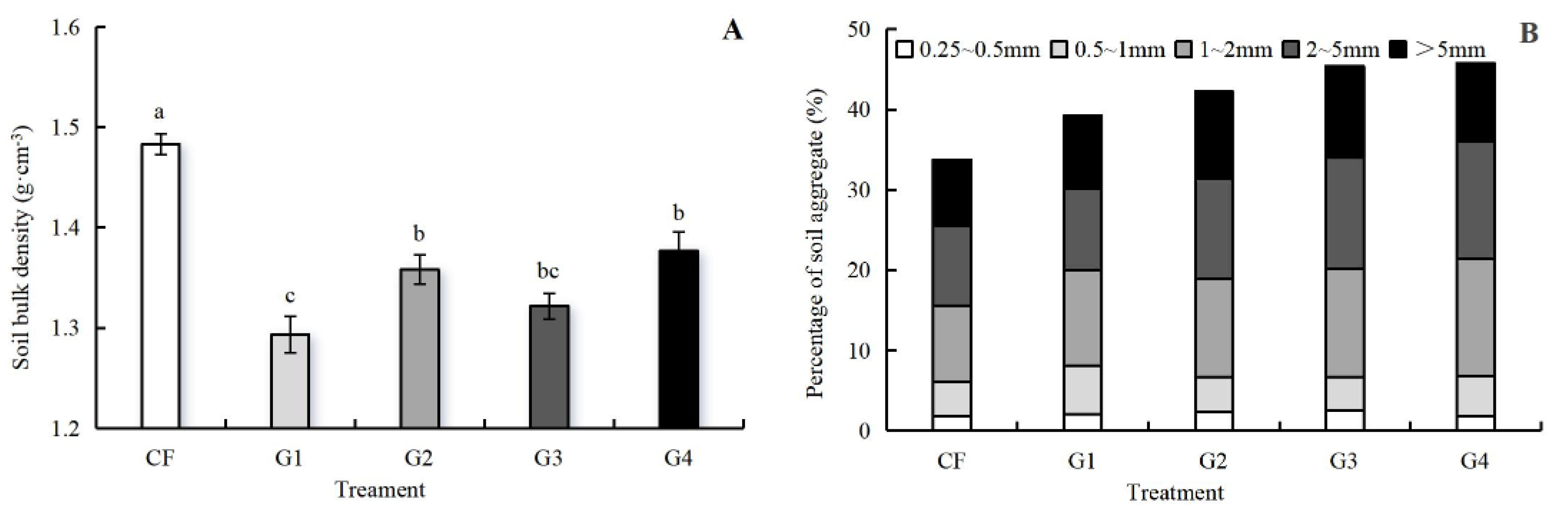
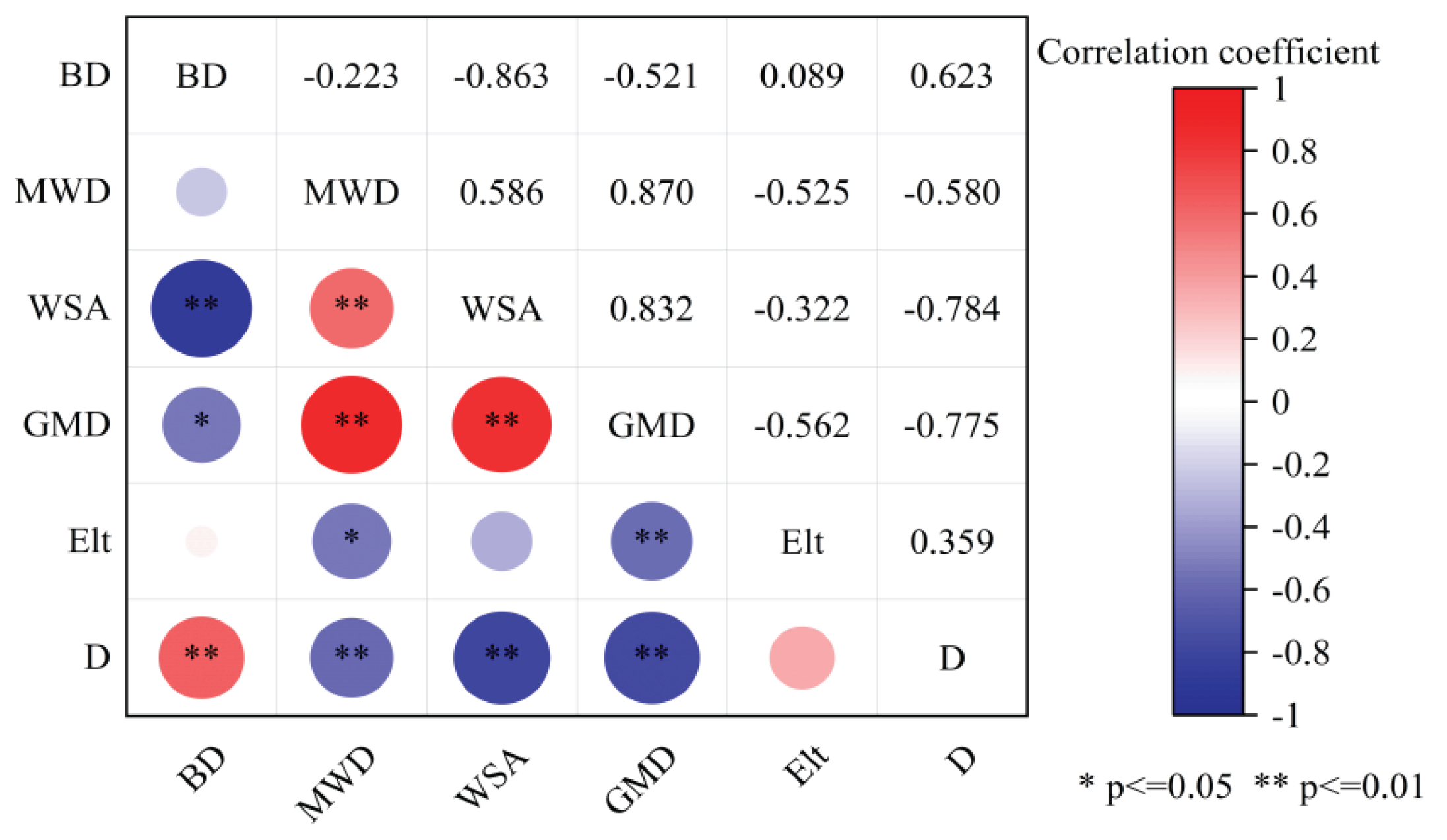
3.2. Effects of Graphene on Bulk Density and Aggregate Size Distribution of Soil
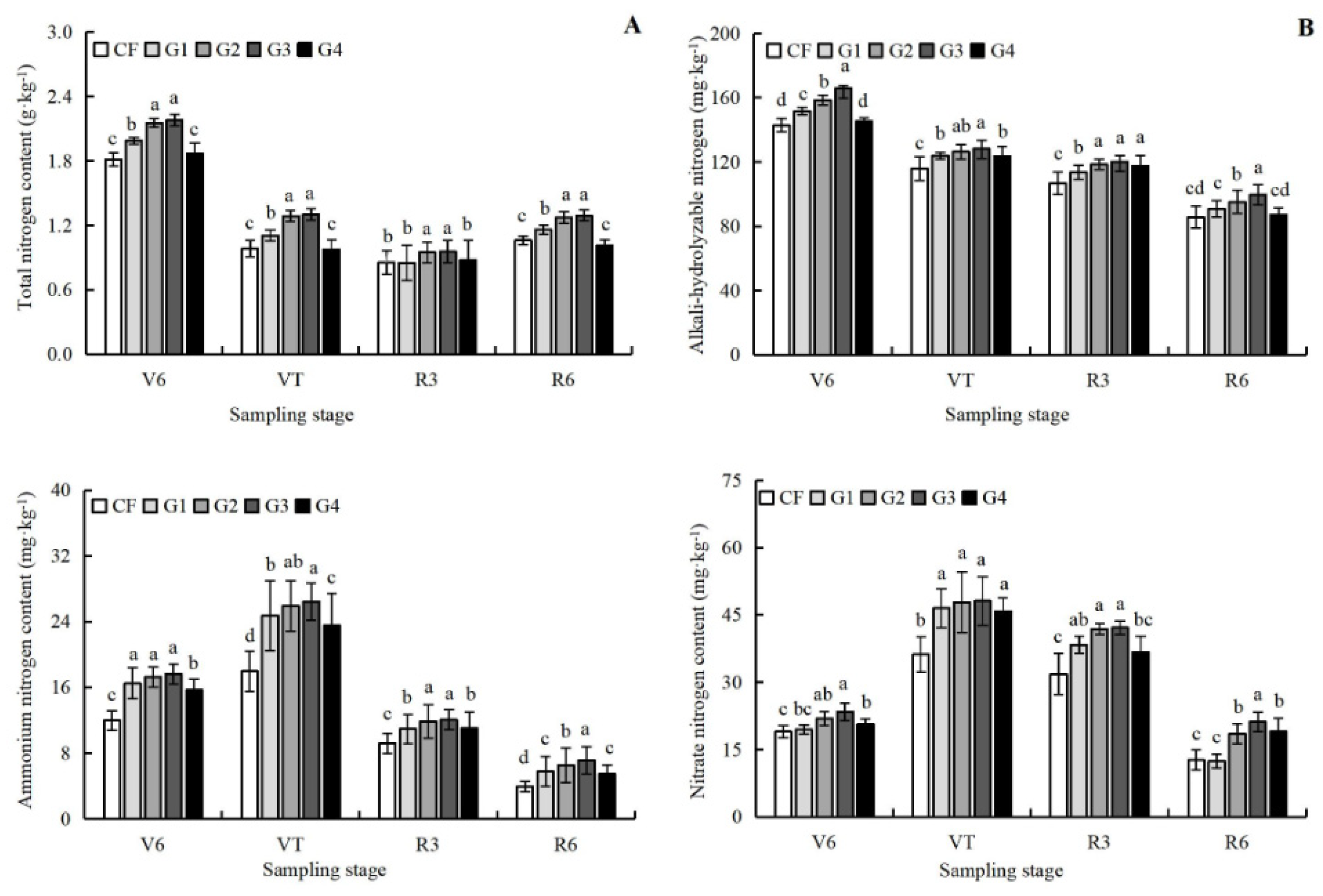
3.3. Effects of Graphene on Different Forms Soil Nitrogen
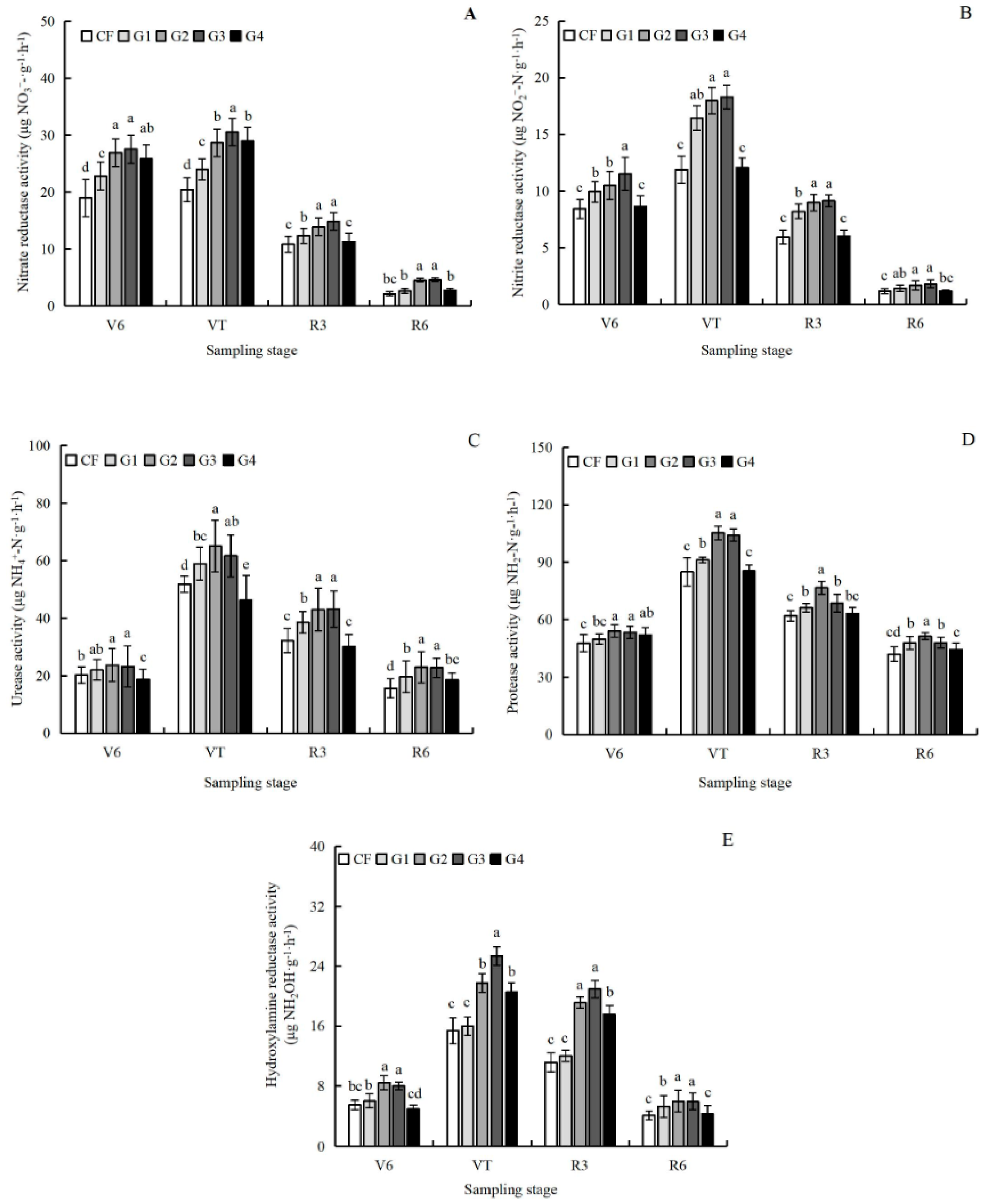
3.4. Effects of Graphene on Soil Enzyme Activity
3.5. Effects of Graphene on Ear Traits and Yield of Maize
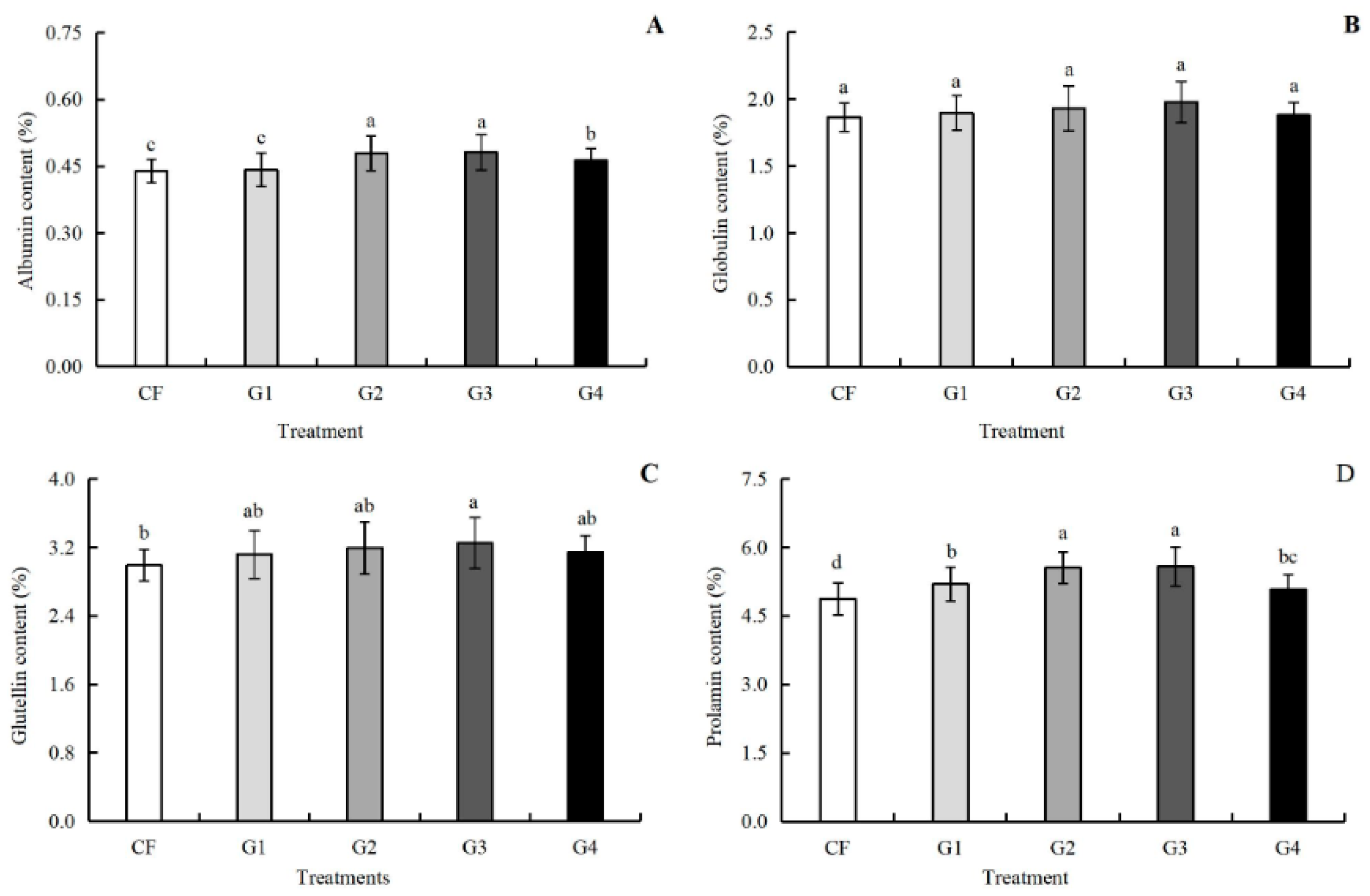
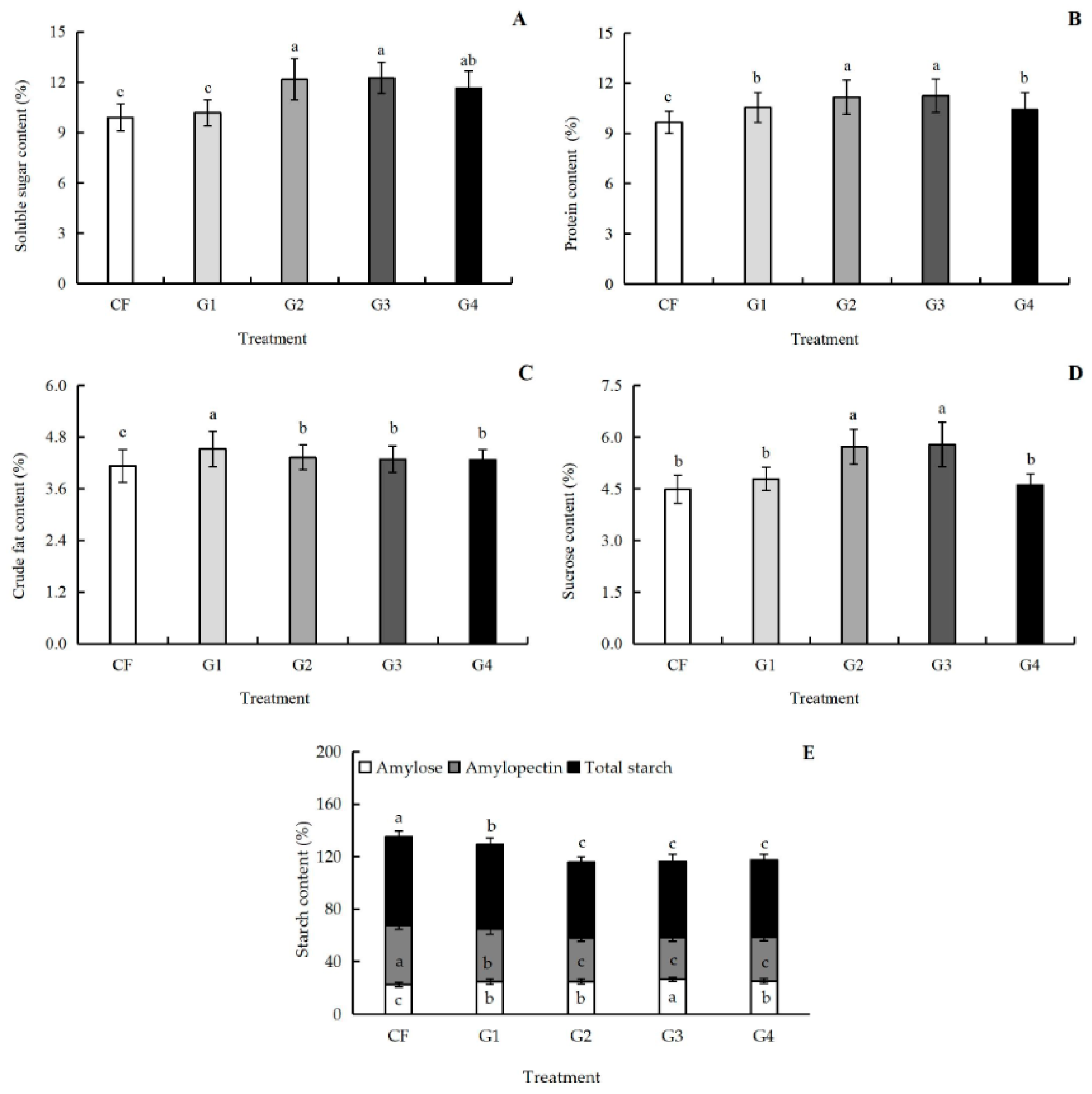
3.6. Effects of Graphene on Protein Components and Quality Index of Kernel
4. Discussion
4.1. Mechanisms of Graphene on Soil Physical Restructuring
4.2. Regulatory Dynamics of Graphene on the Soil Nitrogen Cycle
4.3. Graphene’s Scaffolding Effect on Nitrogen-Metabolizing Enzymes Activities
4.4. Linking Soil Micro-Environmental Optimization Into Maize Yield and Quality
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, R.; Hu, W.Y.; Jia, Z.J.; Liu, H.Q.; Zhang, C.; Huang, B.; Yang, S.H.; Zhao, Y.G.; Zhao, Y.C.; Shukla, M.K.; Taboada, M.N. Soil degradation: a global threat to sustainable use of black soils. Pedosphere 2024, 35, 264–279. [Google Scholar] [CrossRef]
- Wu, Z.X.; Jiang, J.Y.; Dong, W.C.; Cui, S. The Spatiotemporal characteristics and driving factors of soil degradation in the black soil region of Northeast China. Agronomy 2024, 14, 2870. [Google Scholar] [CrossRef]
- Ma, R.; Tian, Z.; Zhao, Y.; Wu, Y.; Liang, Y. Response of soil quality degradation to cultivation and soil erosion: A case study in a mollisol region of Northeast China. Soil and Tillage Res. 2024, 10, e106159. [Google Scholar] [CrossRef]
- Zhao, C.; Li, Y.; Huang, W.; He, N.; Borham, A.; Wang, J. Soil nutrient changes in black soil of Northeastern China under long-term cultivation with different fertilizer applications and tillage patterns: A meta-analysis. Commun. Soil Sci. Plant Anal. 2024, 56, 894–910. [Google Scholar] [CrossRef]
- Usman, M.; Farooq, M.; Wakeel, A.; Nawaz, A.; Cheema, S.; Rehman, H.; Ashraf, I.; Sanaullah, M. Nanotechnology in agriculture: Current status, challenges and future opportunities. Sci. Total Environ. 2020, 721, 137778. [Google Scholar] [CrossRef]
- Sharma, S.; Kundu, P.; Tyagi, D.; Shanmugam, V. Graphene-based nanomaterials applications for agricultural and food sector. Adv. Colloid Interface Sci. 2024, 336, 103377. [Google Scholar] [CrossRef]
- Bhattacharya, N.; Cahill, D.; Yang, W.; Kochar, M. Graphene as a nano-delivery vehicle in agriculture-current knowledge and future prospects. Crit.Revi. Biotechnol. 2022, 43, 851–869. [Google Scholar] [CrossRef]
- Li, L.J.; Tang, Y.M.; Bao, Z.J.; Tu, W.Y.; Peng, L.X.; Zou, L.; Zhao, C.S.; Li, Q. When graphene meets circular agriculture: Insights into agricultural sustainable development. Biosyst. Engin 2024, 237, 92–117. [Google Scholar] [CrossRef]
- Qiao, J.; Chen, Z.W.; Zhao, J.G.; Ren, J.; Wang, H.; Zhi, C.Y.; Li, J. W.; Xing, B.Y.; Nie, H. Graphene promotes the growth of Vigna angularis by regulating nitrogen metabolism and photosynthesis. Plos one 2024, 19, e0297892. [Google Scholar] [CrossRef] [PubMed]
- Luo, L.; Zhang, Y.; Xu, G. How does nitrogen shape plant architecture? J. Exp. Bot. 2020, 71, 4415–4427. [Google Scholar] [CrossRef] [PubMed]
- Anas, M.; Liao, F.; Verma, K.; Sarwar, M.; Mahmood, A.; Chen, Z.; Li, Q.; Zeng, X.; Liu, Y.; Li, Y. Fate of nitrogen in agriculture and environment: agronomic, eco-physiological and molecular approaches to improve nitrogen use efficiency. Biol. Res. 2020, 53, 47–66. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, R.; Kunal; Moulick, D.; Barek, V.; Brestic, M.; Gaber, A.; Skalicky, M.; Hossain, A. Sustainable strategies to limit nitrogen loss in agriculture through improving its use efficiency—aiming to reduce environmental pollution. J. Agric. Food Res. 2025, 22, 101957. [Google Scholar] [CrossRef]
- Geim, A. K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef]
- Avouris, P. Graphene: electronic and photonic properties and devices. Nano Lett. 2010, 11, 4285–4294. [Google Scholar] [CrossRef]
- Xiu, L.Q.; Zhang, W.M.; Wu, D.; Sun, Y.Y.; Zhang, H.H.; Gu, W.Q.; Wang, Y.N.; Meng, J.; Chen, W.F. Biochar can improve biological nitrogen fixation by altering the root growth strategy of soybean in Albic soil. Sci. Total Environ. 2021, 773, 144564. [Google Scholar] [CrossRef]
- Hu, X.G.; Zhou, Q.X. Novel hydrated graphene ribbon unexpectedly promotes aged seed germination and root differentiation. Sci. Rep. 2014, 4, 3782–3790. [Google Scholar] [CrossRef]
- Chen, Z.; Guo, Z.; Xu, N.; Cao, X.; Niu, J. Graphene nanoparticles improve alfalfa (Medicago sativa L.) growth through multiple metabolic pathways under salinity-stressed environment. J. Plant Physiol. 2023, 289, 154092. [Google Scholar] [CrossRef]
- Andelkovic, I.B.; Kabiri, S.; Tavakkoli, E. Graphene oxide-Fe(III) composite containing phosphate-a novel slow release fertilizer for improved agriculture management. J. Clean. Prod. 2018, 185, 977–1004. [Google Scholar] [CrossRef]
- Sun, Y.; Zhu, G.K.; Zhao, W.C. Engineered nanomaterials for improving the nutritional quality of agricultural products: A review. Nanomater. 2022, 12, 4219–4237. [Google Scholar] [CrossRef]
- Fang, J; Weng, Y.N.; Li, B.E.; Liu, H.J.; Liu, L.J.; Tian, Z.L.; Du, S.T. Graphene oxide decreases the abundance of nitrogen cycling microbes and slows nitrogen transformation in soils. Chemosphere 2022, 309, 136642. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Li, X.; Zhang, J. Effects of nitrogen application on soil pH, bacterial community, and available nitrogen in a wheat-maize rotation system. Eur. J. Soil Sci. 2022, 73, 1115–1126. [Google Scholar]
- Das, P.; Barker, C.; Park, Y.; Perreault, F.; Westerhoff, P.; Penton, C. Impact of graphite nano amendments on soil enzyme activities, functional genes and microbiome composition in a soil-plant system. Soil Biol. Biochem. 2025, 109714. [Google Scholar] [CrossRef]
- Yang, C.D.; Lu, S.G. Effects of five different biochars on aggregation, water retention and mechanical properties of paddy soil: A field experiment of three-season crops. Soil Tillage Res. 2021, 205, 104798. [Google Scholar] [CrossRef]
- Bremner, J.M.; Mulvaney, C.S. Methods of soil analysis. Part 2; American Society of Agronomy, Soil Science Society of America: Madison, 1996; pp. 1085–1121. [Google Scholar]
- Song, J.F.; Cao, K.; Duan, C.W.; Luo, N.; Cui, X.Y. Effects of graphene on Larix olgensis seedlings and soil properties of Haplic Cambisols in Northeast China. Forests 2020, 11, 258. [Google Scholar] [CrossRef]
- Cataldo, D.A.; Maroon, M.; Schrader, L.E. Phenol-disulfonic acid determination of nitrate-nitrogen in water extracts of plant tissue. Commun. Soil Sci. Plant Anal. 1973, 4, 717–778. [Google Scholar]
- Rong, Y.; Wang, Y.; Guan, Y.; Cai, Z.; Yang, G.; Zhao, X. Pyrosequencing reveals soil enzyme activities and bacterial communities impacted by graphene and its oxides. J. Agric. Food Chem. 2017, 65, 9191–9199. [Google Scholar] [CrossRef] [PubMed]
- Abdelmagid, H.M.; Tabatabai, M.A. Nitrate reductase activity of soils. Soil Biol. Biochem. 1987, 19, 421–427. [Google Scholar] [CrossRef]
- Cleemput, O.; Samater, A. Nitrite in soils: accumulation and role in the formation of gaseous N compounds. Fertil. Res. 1995, 45, 81–89. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, Y.; Sun, S.; Liu, W.; Zhu, L.; Yan, X. Different forms and proportions of exogenous nitrogen promote the growth of alfalfa by increasing soil enzyme activity. Plants 2022, 11, 1057. [Google Scholar] [CrossRef]
- Nayyar, H.; Bains, T.; Kumar, S. Low temperature induced floral abortion in chickpea: relationship to abscisic acid and cryoprotectants in reproductive organs. Environ. Exp. Bot. 2005, 53, 39–47. [Google Scholar] [CrossRef]
- Weegels, P.L.; Pijpekamp, A.M.; Graveland, A.; Hamer, L.J.; Schofield, J.D. Depolymerisation and repolymerisation of wheat glutenin during dough processing. I. Relationships between glutenin macropolymer content and quality parameters. J. Cereal Sci. 1996, 23, 103–111. [Google Scholar] [CrossRef]
- Liu, R.Q.; Lal, R. Potentials of engineered nanoparticles as fertilizers for increasing agronomic productions. Sci. Total Environ. 2015, 514, 131–139. [Google Scholar] [CrossRef]
- Liu, Z.Y.; Zhou, M.; Liao, W.F.; Liu, J.Y.; Luo, C.G.; Lu, C.Y.; Chen, Z.W.; Zhu, H.W. Fertilizer-holding performance of graphene on soil colloids based on double electric layer theory. Mater. 2023, 16, 2578. [Google Scholar] [CrossRef]
- Alessandrino, L.; Eusebi, A.L.; Aschonitis, V.; Mastrocicco, M.; Colombani, N. Variation of the hydraulic properties in sandy soils induced by the addition of graphene and classical soil improvers. J. Hydrol. 2022, 612, 128256. [Google Scholar] [CrossRef]
- Zhou, M.; Liu, C.Z.; Wang, J.; Meng, Q.F.; Yuan, Y.; Ma, X.F.; Liu, X.B.; Zhu, Y.X.; Ding, G.W.; Zhang, J.Z.; Zeng, X.N.; Du, W.L. Soil aggregates stability and storage of soil organic carbon respond to cropping systems on black soils of Northeast China. Sci. Rep. 2020, 10, 265–277. [Google Scholar] [CrossRef]
- Wang, Q.H.; Li, C.; Wang, Y.; Que, X.E. Phytotoxicity of graphene family nanomaterials and its mechanisms: A review. Front. Chem. 2019, 7, 292–298. [Google Scholar] [CrossRef]
- Costa, O.; Raaijmakers, J.; Kuramae, E. Microbial extracellular polymeric substances: ecological function and impact on soil aggregation. Front. Microbiol. 2018, 9, 1636–1649. [Google Scholar] [CrossRef]
- Wang, S.Y.; Liu, Y.; Wang, X.Y.; Xiang, H.T.; Kong, D.Y.; Wei, N.; Guo, W.; Sun, H.Y. Effects of graphene amendment on nutrient content, maize growth, and soil physicochemical parameters. Sci. Rep. 2023, 13, 2650–2662. [Google Scholar] [CrossRef] [PubMed]
- Verma, K.K.; Song, X.P.; Degu, D.H.; Guo, D. J.; Joshi, A.; Huang, H.R.; Xu, L.; Singh, M.; Huang, D.L.; Rajput, V.D.; Li, Y.R. Recent advances in nitrogen and nano-nitrogen fertilizers for sustainable crop production: a mini-review. Chem. Biol. Technol. Agric. 2023, 10, 111–124. [Google Scholar] [CrossRef]
- Haydar, M.; Ghosh, D.; Roy, S. Slow and controlled release nanofertilizers as an efficient tool for sustainable agriculture: Recent understanding and concerns. Plant NanoBiol. 2024, 7, 100058. [Google Scholar] [CrossRef]
- Zhao, F.L.; Xin, X.P.; Cao, Y.E.; Su, D.; Ji, P.H.; Zhu, Z.Q.; He, Z.L. Use of carbon nanoparticles to improve soil fertility, crop growth and nutrient uptake by corn (Zea mays L.). Nanomater. 2021, 11, 2717–2731. [Google Scholar] [CrossRef]
- Das, P.; Penton, C.R.; Bi, Y.Q.; Westerhoff, P. Unraveling mechanisms behind reduced nitrate leaching with graphite nanomaterials addition with fertilizers in soil column experiments. Chemosphere 2023, 337, 139417. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.Y.; White, J.C.; Wang, Z.Y.; Xing, B.S. Nano-enabled fertilizers to control the release and use efficiency of nutrients. Cur. Opin. Environ. Sci. Health 2018, 6, 77–83. [Google Scholar] [CrossRef]
- Das, P.; Davis, K.; Penton, C.R.; Westerhoff, P.; Bi, Y.Q. Impacts of graphitic nanofertilizers on nitrogen cycling in a sandy, agricultural soil. J. Nanopart. Res. 2022, 24, 120. [Google Scholar] [CrossRef]
- Saraiva, R.; Ferreira, Q.; Rodrigues, G.; Oliveira, M. Graphene oxide and its viability as a constituent in nanofertilizers. Front. Nanotechnol. 2025, 7, 1580066. [Google Scholar] [CrossRef]
- Fincheira, P.; Hoffmann, N.; Tortella, G.; Ruíz, A.; Cornejo, P.; Diez, M.C; Seabra, A.B.; Benavides-Mendoza, A.; Rubilar, O. Eco-efficient systems based on nanocarriers for the controlled release of fertilizers and pesticides: toward smart agriculture. Nanomater. 2023, 13, 1978–1989. [Google Scholar] [CrossRef] [PubMed]
- Datta, R.; Anand, S.; Moulick, A.; Baraniya, D.; Pathan, S.; Rejšek, K.; Vranová, V.; Sharma, M.; Sharma, D.; Kelkar, A.; Formánek, P. How enzymes are adsorbed on soil solid phase and factors limiting its activity: A Review. Int. Agrophys 2017, 31, 287–302. [Google Scholar] [CrossRef]
- Syshchykov, D.; Berezovskiy, A.; Agurova, I. The influence of degradation processes on enzymatic activity and the content of forms of mineral nitrogen in agricultural soils. Dokuchaev Soil Bull. 2024, 121, 28–46. [Google Scholar] [CrossRef]
- Adeel, M.; Bilal, M.; Rasheed, T.; Sharma, A.; Iqbal, H. Graphene and graphene oxide: Functionalization and nano-bio- catalytic system for enzyme immobilization and biotechnological perspective. Int. J. Biol. Macromol. 2018, 120, 1430–1440. [Google Scholar] [CrossRef]
- Jin, L.X.; Son, Y.; Yoon, T.K.; Kang, Y.J.; Kim, W.; Chung, H. High concentrations of single-walled carbon nanotubes lower soil enzyme activity and microbial biomass. Ecotoxicol..Environ. Saf. 2013, 88, 9–15. [Google Scholar] [CrossRef]
- Fang, J.; Weng, Y.N.; Li, B.E.; Liu, H.J.; Liu, L.J.; Tian, Z.L.; Du, S.T. Graphene oxide decreases the abundance of nitrogen cycling microbes and slows nitrogen transformation in soils. Chemosphere 2022, 309, 136642. [Google Scholar] [CrossRef]
- Chung, H.; Kim, M.J.; Ko, K.; Kim, J.H.; Kwon, H.A.; Hong, I.; Park, N.; Lee, S.W.; Kim, W. Effects of graphene oxides on soil enzyme activity and microbial biomass. Sci. Total Environ. 2015, 514, 307–313. [Google Scholar] [CrossRef]
- Fan, P.P.; Ming, B.; Evers, J.B.; Li, Y.Y; Li, S.K.; Xie, R.Z.; Anten, N.P.R. Nitrogen availability determines the vertical patterns of accumulation, partitioning, and reallocation of dry matter and nitrogen in maize. Field Crops Res. 2023, 297, 108927. [Google Scholar] [CrossRef]
- Hatakeyama, Y.; Araki, T.; Hatakeyama, K. Effects of prolonged graphene oxide treatment on biomass production and grain yield in hydroponically grown rice. ACS Agric. Sci. Technol. 2024, 11, 1184–1193. [Google Scholar] [CrossRef]
- Chen, Z.W.; Zhao, J.G.; Song, J.; Han, S.H.; Du, Y.Q.; Qiao, Y.Y.; Liu, Z.H.; Qiao, J.; Li, W.J.; Li, J.W.; Wang, H.Y.; Xing, B.Y.; Pan, Q.L. Influence of graphene on the multiple metabolic pathways of Zea mays roots based on transcriptome analysis. Plos One 2021, 16, e0244856. [Google Scholar] [CrossRef] [PubMed]
- Shewry, P.R.; Halford, N.G. Cereal seed storage proteins: structures, properties and role in grain utilization. J. Exp. Bot. 2002, 53, 947–958. [Google Scholar] [CrossRef]
| Test code | Graphene amount (g·kg−1) |
| CF | 0 |
| G1 | 1 |
| G2 | 2 |
| G3 | 5 |
| G4 | 10 |
| Treatment | GMD (mm) | MWD (mm) | WSA (%) | ELT (%) | D |
| CF | 0.235±0.008cd | 0.378±0.024b | 0.243±0.015d | 91.384±2.510b | 3.452±0.074a |
| G1 | 0.258±0.012c | 0.363±0.043b | 0.402±0.028b | 92.558±2.277a | 3.279±0.125bc |
| G2 | 0.305±0.023b | 0.447±0.023a | 0.428±0.014b | 89.172±2.630d | 3.203±0.065c |
| G3 | 0.343±0.028a | 0..458±0.023a | 0.466±0.012a | 89.114±2.148d | 3.181±0.103c |
| G4 | 0.250±0.015c | 0.340±0.028bc | 0.354±0.011c | 90.293±1.307c | 3.355±0.198b |
| Treatments | CF | G1 | G2 | G3 | G4 |
| Ear length (cm) | 20.00±1.00 a | 20.67±0.29 a | 20.78±0.29 a | 21.45±0.32a | 19.88±1.00 a |
| Ear diameter(mm) | 47.22±0.77 b | 46.78±1.58 b | 52.67±2.08 a | 52.58±1.84a | 47.47±2.25 b |
| Bald length (cm) | 3.50±0.50 a | 3.50±0.50 a | 2.83±0.76 b | 2.97±0.42b | 3.50±0.50 a |
| Kernels number of row | 35.67±0.58 b | 35.33±0.58 b | 38.00±0.24 a | 38.42±0.68a | 36.67±1.15 b |
| Row number of ear | 16.37±2.06 a | 15.33±1.15 b | 16.24±0.13 a | 16.78±0.82a | 16.67±3.16 a |
| Cob diameter (mm) | 24.30±1.57 b | 25.47±1.42 b | 29.07±0.83 a | 29.45±0.76a | 27.20±2.36 ab |
| Cob weight (g) | 38.94±0.68 a | 37.48±0.44 b | 37.78±0.61 b | 38.34±0.42a | 36.99±0.58 b |
| Yield (g·plant-1) | 179.35±6.31 b | 183.66±3.16 ab | 191.65±4.47 a | 198.74±5.41a | 184.57±3.38 ab |
| Index | Correlation coefficient | Direct path coefficient | Indirect path coefficient | ||
| X1-Y | X2-Y | X3-Y | |||
| Ear row number (X1) | 0.589* | 0.417 | - | 0.034 | 0.138 |
| Kernel number per row (X2) | 0.298 | 0.085 | 0.168 | - | 0.045 |
| 100-kernel weight (X3) | 0.572* | 0.428 | 0.135 | 0.009 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




