Submitted:
09 April 2026
Posted:
10 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Species
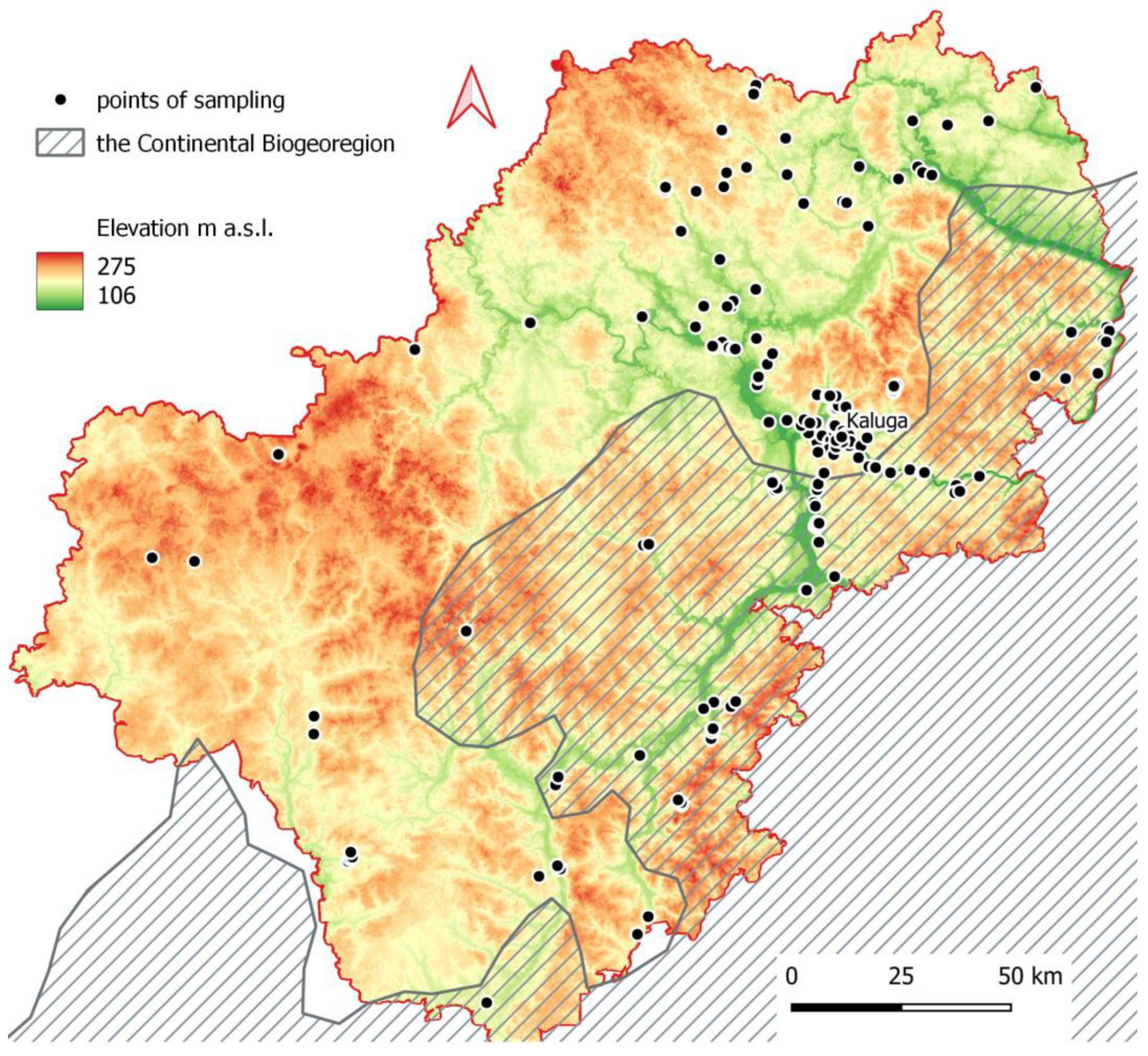
2.2. Study Area
2.3. Data Sampling
2.4. Data Analysis
3. Results
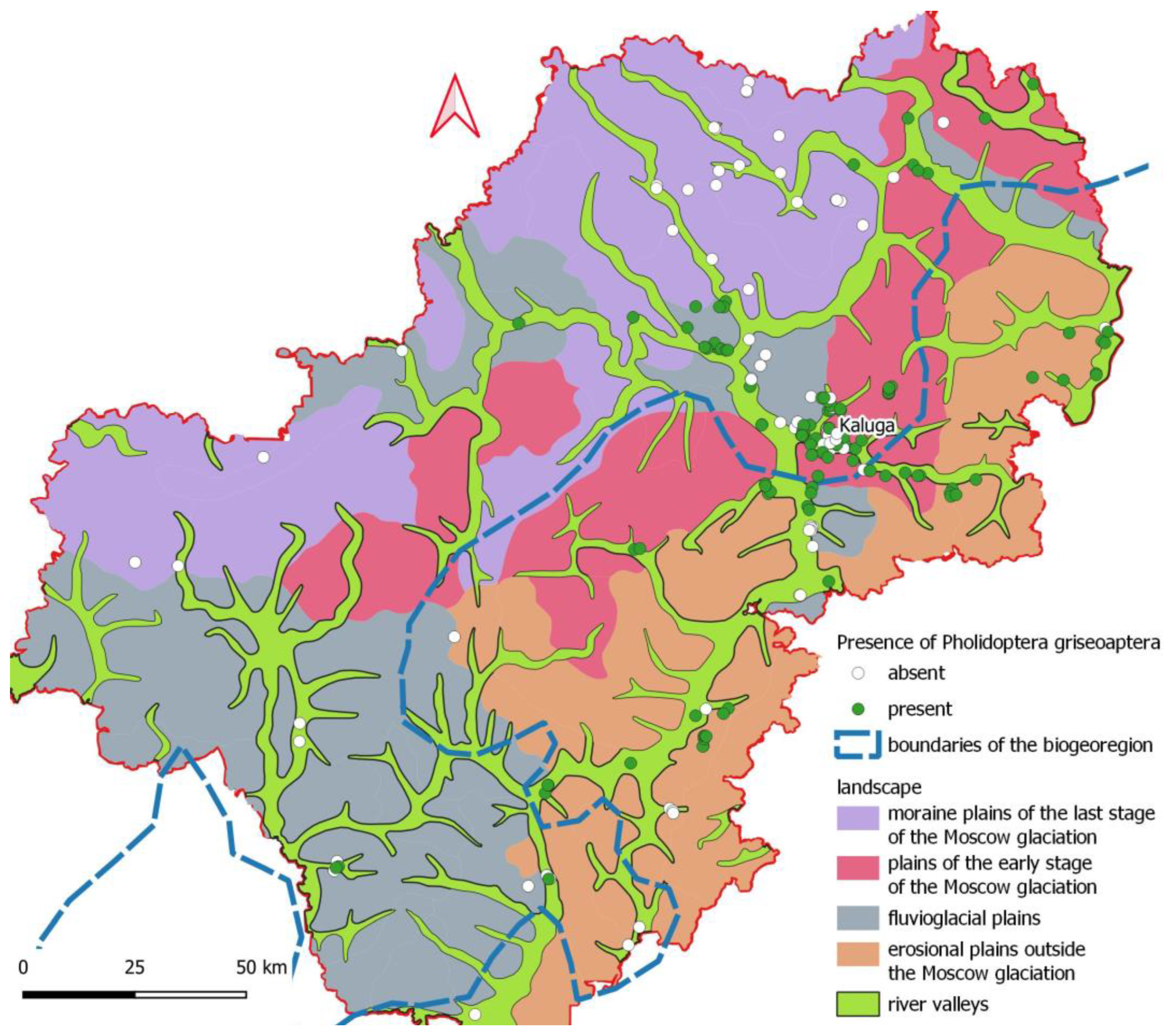
3.1. Distribution of Pholidoptera griseoaptera Across the Kaluga Region
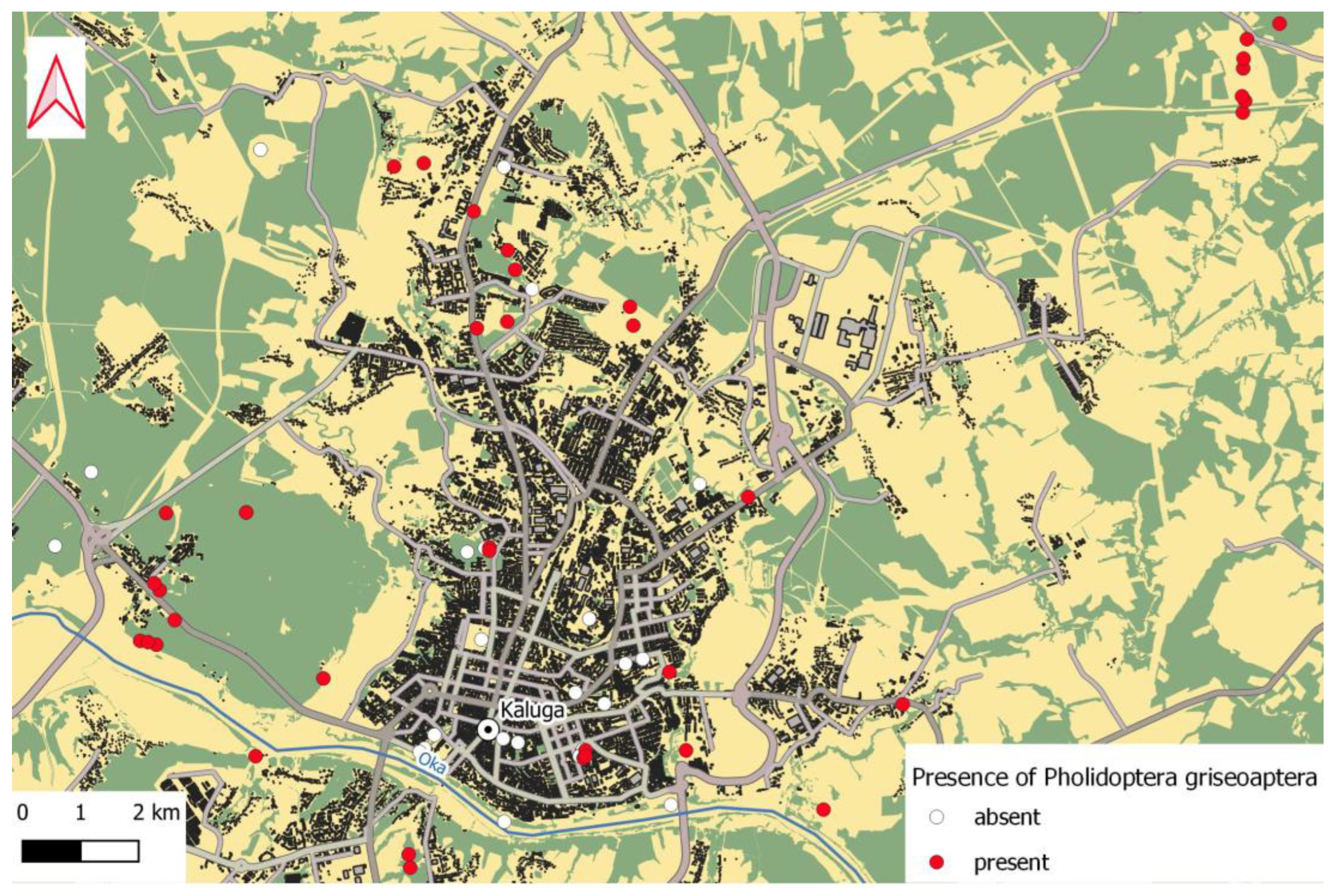
3.2. Distribution of Pholidoptera griseoaptera in Urban Areas
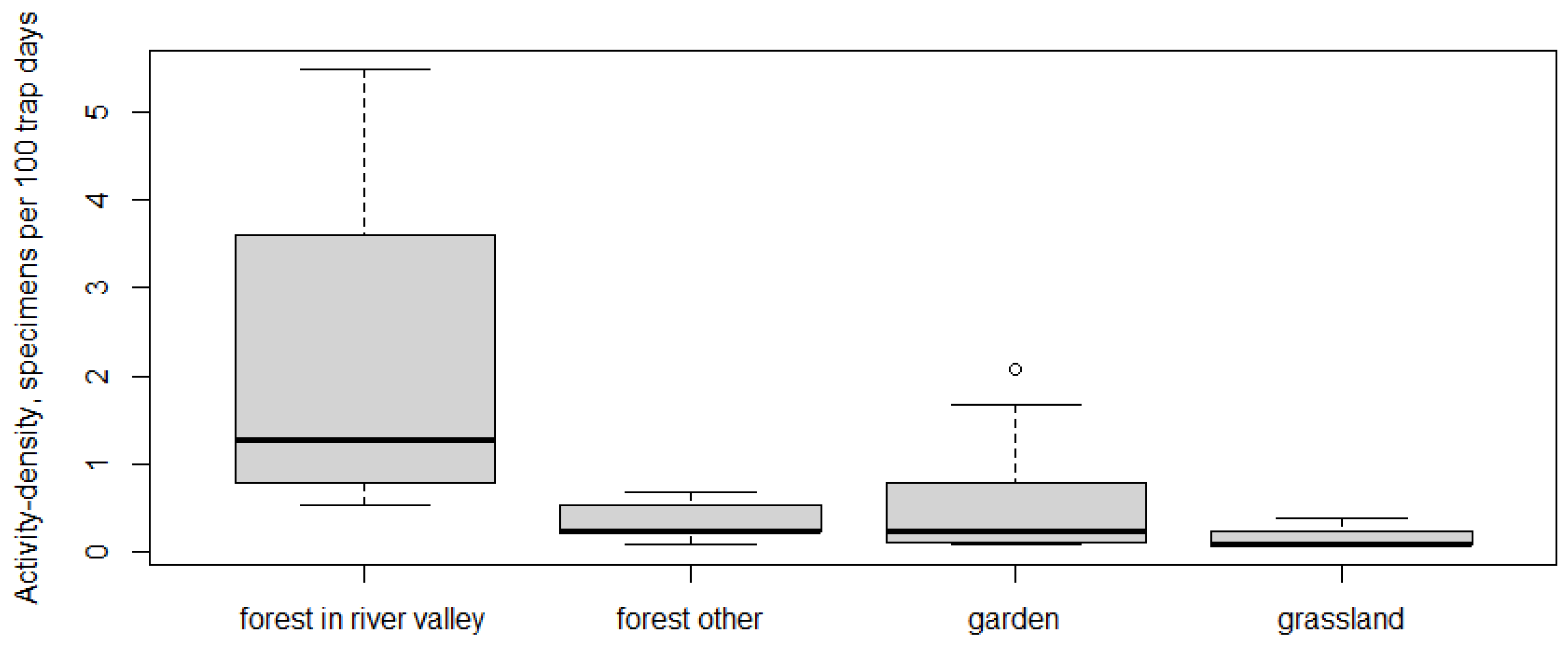
3.3. Abundance of Pholidoptera griseoaptera
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BP | before present |
| kyr | Thousand years |
References
- MacFadyen, A. Animal ecology. Aims and Methods, 2nd ed. Sir Isaac Pitman and Sons, Ltd.: London, 1963; XXIV+344 p.
- Molles, M.C.J. Ecology: concepts and applications. McGraw Hill: New York, USA, 2005; pp.
- Kindlmann, P.; Tsiftsis, S.; Buchbauerová, L.; Traxmandlová, I.; Štípková, Z. How Will Environmental Conditions Affect Species Distribution and Survival in the Coming Decades—A Review. Diversity, 2025, 17, 793. [CrossRef]
- Peterken, G.F.; Game, M. Historical factors affecting the number and distribution of vascular plant species in the woodlands of central Lincolnshire. The Journal of Ecology, 1984, 72, 155-182. [CrossRef]
- Ricketts, T.H. The matrix matters: effective isolation in fragmented landscapes. The American Naturalist, 2001, 158(1), 87-99. [CrossRef]
- Prevedello, J.A.; Vieira, M.V. Does the type of matrix matter? A quantitative review of the evidence. Biodiversity and Conservation, 2010, 19(5), 1205-1223. [CrossRef]
- European Environment Agency. EEA Report No 1/2002. Europe’s biodiversity – biogeographical regions and seas Biogeographical regions in Europe Published 31 May 2002. Available online: https://www.eea.europa.eu/publications/report_2002_0524_154909 (accessed on 12 November 2024).
- Smirnova, O.V.; Bobrovsky, M.V.; Khanina, L.G. (eds.). European Russian Forests. Their Current State and Features of Their History. Dordrecht: Springer Nature, 2017; 566 pp.
- Fartmann, T.; Krämer, B.; Stelzner, F.; Poniatowski, D. Orthoptera as ecological indicators for succession in steppe grassland. Ecological Indicators, 2012, 2, 337–344. [CrossRef]
- Samways, M.J.; Lockwood, J.A. Orthoptera conservation: pests and paradoxes. Journal of Insect Conservation, 1998, 2, 143–149. [CrossRef]
- Guido, M.; Gianelle, D. Distribution patterns of four Orthoptera species in relation to microhabitat heterogeneity in an ecotonal area. Acta Oecologica, 2001, 22(3), 175-185. [CrossRef]
- Kenyeres, Z.; Rácz, I.A.; Varga, Z. Endemism hot spots, core areas and disjunctions in European Orthoptera. Acta Zoologica Cracoviensia-Series B: Invertebrata, 2009, 52(1-2), 189-211. [CrossRef]
- Sergeev, M.G. Distribution patterns of grasshoppers and their kin over the Eurasian Steppes. Insects, 2021, 12(1), 77, 1–23. [CrossRef]
- Poniatowski, D.; Fartmann, T. What determines the distribution of a flightless bush-cricket (Metrioptera brachyptera) in a fragmented landscape? Journal of Insect Conservation, 2010, 14(6), 637-645. [CrossRef]
- Dvořák, T.; Hadrava, J.; Knapp, M. The ecological niche and conservation value of Central European grassland orthopterans: A quantitative approach. Biological Conservation, 2022, 265, 109406. [CrossRef]
- Bey-Bienko, G.Y. Orthoptera and Dermaptera. In Animal word of the USSR. Vol. IV. Forest belt; USSR Academy of Sciences Publ.: Moscow-Leningrad, Russia, 1953; pp. 527–552.
- Harz, K. Die Orthopteren Europas — The Orthoptera of Europe. Vol. I; Dr. W. Junk B.V.: The Hague, 1969, pp. i-xx, 1-749.
- Harz, K. Die Geradflügler Mitteleuropas; Jena: G. Fischer, 1957; 494 ss.
- Heller, K.G.; Korsunovskaya, O.; Ragge, D.R.; Vedenina, V.; Willemse, F.; Zhantiev, R.D.; Frantsevich, L. Check-list of European orthoptera. Articulata, 1998, 7, 1–61.
- Lucas, W.J. A Monograph of the British Orthoptera; The Ray Society: London, 1919; 264 pp.
- Chopard, L. Faune de France. 56, Orthoptéroïdes; Paul Lechevalier: Paris, 1951, 358 pp.
- Oschmann, M. Faunistisch-ökologische Untersuchungen an Orthopteren im Raum von Gotha. Hercynia-Ökologie und Umwelt in Mitteleuropa, 1969, 6(2), 115–168.
- Chernyakhovsky, M.E. Fauna of orthopteroid insects of the Moscow region. In Nauchnye osnovy okhrany zhivoy prirody Podmoscowia; Nauka: Moscow, Russia, 1988, pp. 72–78.
- Kaňuch, P.; Jarčuška, B.; Schlosserová, D.; Sliacka, A.; Paule, L.; Krištín, A. Landscape configuration determines gene flow and phenotype in a flightless forest-edge ground-dwelling bush-cricket, Pholidoptera griseoaptera. Evolutionary Ecology, 2012, 26(6), 1331-1343. [CrossRef]
- Diekötter, T.; Csencsics, D.; Rothenbühler, C.; Billeter, R.; & Edwards, P.J. Movement and dispersal patterns in the bush cricket Pholidoptera griseoaptera: the role of developmental stage and sex. Ecological Entomology, 2005, 30(4), 419-427. [CrossRef]
- Diekötter, T.; Speelmans, M.; Dusoulier, F.; Van Wingerden, W.K.; Malfait, J. P.; Crist, T. O.; .Edwards, P.J.; Dietz, H. Effects of landscape structure on movement patterns of the flightless bush cricket Pholidoptera griseoaptera. Environmental Entomology, 2007, 36(1), 90-98.
- Diekötter, T.; Baveco, H.; Arens, P.; Rothenbühler, C.; Billeter, R.; Csencsics, D.; De Philippi R.; Hendrickx F.; Speelmans, V.; Opdam, P.; Smulders, M. J. Patterns of habitat occupancy, genetic variation and predicted movement of a flightless bush cricket, Pholidoptera griseoaptera, in an agricultural mosaic landscape. Landscape Ecology, 2010, 25(3), 449-461. [CrossRef]
- Kaňuch, P.; Jarčuška, B.; Kovács, L.; Krištín, A. Environmentally driven variability in size-selective females’ mating frequency of bush-cricket Pholidoptera griseoaptera. Evolutionary Ecology, 2015, 29(5), 787-797. [CrossRef]
- Bey-Bienko G.Y. Changes of Habitats by Terrestrial Organisms as a Biological Principle. Zhurnal Obshchei Biologii, 1966, 20(1), 5–21.
- Sergeev, M.G. Patterns of Orthoptera distribution in North Asia; Nauka: Novosibirsk, Russia, 1986; 236 pp.
- Jacobson, G.G.; Bianki, V.L. Orthoptera and Pseudoneuroptera of the Russian Empire and bordering countries; Izdanie AF Devriena, St. Petersburg, Russia, 1905; 952 pp.
- Aleksanov, V.; Karmazina, I.; Shulaev, N.; Ruchin, A.; Lukiyanov, S.; Lobachev, E.; Nikolaeva, A.; Volodchenko, A.; Anikin, V.; Esin, M. Orthoptera and Mantodea in the Continental biogeographical region and adjacent areas of European Russia. Version 1.15. Joint Directorate of the Mordovia State Nature Reserve and National Park "Smolny". Occurrence dataset. (accessed on 09 July 2024). [CrossRef]
- Adakhovsky, D.A. Materials on the fauna, distribution and ecology of straight-winged insects (Orthoptera) Udmurtia. Bulletin of Udmurtia University Biology, 2006, 10: 120–121.
- Chernykhovsky, M.E.; Mironov, M.Yu.; Sobolev, N.A. Pholidoptera griseopatera. In Red Book of the City of Moscow, 3d edition; Department of Nature Conservation of the City of Moscow, Moscow, Russia, 2022; 311-312.
- Atlas of the Kaluga Region; Izd-vo N.Bochkarevoy: Kaluga, Russia, 2005; 48 pp.
- Lavrenko, E.M. The principles and units of geobotanical regionalization. In Geobot. Raionirovaniye SSSR. Nauka: Moscow-Leningrad, Russia, 1947; 9–13.
- Soloviyova M.P., Khomutova M.S. A tentative division of the Kaluga Region into geobotanical districts. Botanical Journal, 1969, 54, 721-728.
- Ogureeva G.N. (ed.). 2020. The Biodiversity of Russian Biomes. The Biomes of Plains; Institute of Global Climate and Ecology: Moscow, Russia, 623 pp.
- Barashkova Z.K., Lavrovich O.N., Shuleshkina E.N. The geological maps of the quaternary sediments in Kaluga region. Scale 1:500 000; Central geological center: Moscow, Russia, 1998, 1 p.
- Sheremet’eva E.V. Map of quaternary sediments. Sheet 1. State geological map of Russian Federation 1: 200 000; 2nd ed. Moscow series. Chart sheet N-37-XIII (Kaluga); Moscow, 2021, 1 p. Available from https://webftp.vsegei.ru/GGK200/N-37-XIII/N-37-XIII_KQO_1.pdf.
- GBIF.org .GBIF Occurrence Download (accessed on 04 April 2026). [CrossRef]
- Khanina L., Zaugolnova L., Smirnova O., Shovkun M., Glukhova E. Flora of vascular plants in the Central European Russia. Available from https://www.impb.ru/eco/ (accessed on 04 January 2026).
- "Shubert map". Fragment of topographic map of Kaluga Governorate years. Printed in 1860-1860. 3 versta in inch (1260 m in 1 cm). Available from http://www.etomesto.ru/shubert-map/13-14/ (accessed on 04 January 2026).
- Kaluga and its suburbs. Map of the Military Topographic Administration. Printed in 1919. http://www.etomesto.com/map-kaluga_1919/ (accessed on 01 March 2026).
- German map of the center of the Kaluga region and the environs of Vyazma, 1942. http://www.etomesto.com/map-kaluga_kozelsk-vyazma-medyn-1942/.
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing; Vienna, Austria. https://www.R-project.org/ (accessed on 01 March 2026).
- Jackman, S. pscl: Classes and Methods for R Developed in the Political Science Computational Laboratory. R package version 1.5.9; Sydney, Australia, 2024. URL https://github.com/atahk/pscl/ (accessed on 01 March 2026).
- Fox J, Weisberg S. An R Companion to Applied Regression, Third edition; Sage, Thousand Oaks CA, 2019. https://www.john-fox.ca/Companion/ (accessed on 01 March 2026).
- Bivand, R.; Wong, D. Comparing implementations of global and local indicators of spatial association. TEST, 2018, 27(3), 716-748. [CrossRef]
- Sliacka, A.; Krištìn, A.; Naďo, L. Response of Orthoptera to clear-cuts in beech forests. European Journal of Entomology, 2013, 110(2), 319-326. [CrossRef]
- Roesti, C.; Rutschmann, F. Orthoptera.ch. Der Heuschrecken-plattform für die Schweiz und Europa. Available from https://www.orthoptera.ch/. (accessed on 31.03.2026).
- Kleukers, R. M. J. C., van Nieukerken, E. J., Odé, B., Willemse, L. P. M., van Wingerden, W. K. R. E. De sprinkhanen en krekels van Nederland (Orthoptera). (Nederlandse fauna; No. 1); Nationaal Natuurhistorisch Museum: Leiden, 1997; pp. 1-413.
- Knuff, A.K.; Staab, M.; Frey, J.; Dormann, C.F.; Asbeck, T.; Klein, A.M. Insect abundance in managed forests benefits from multi-layered vegetation. Basic and Applied Ecology, 2020, 48, 124-135. [CrossRef]
- Hahn, S.; Kunert, G. Short-term consequences of nutritional depression on foraging behaviour of dark bush-crickets Pholidoptera griseoaptera (Orthoptera: Ensifera). European Journal of Entomology, 2006, 103(1), 249-253. [CrossRef]
- Chernov, Y.I. The zonation of nature and the terrestrial animals. Mysl: Moscow, Russia, 1975; pp. 1-222.
- Hartley, J.C.; Warne, A.C. The distribution of Pholidoptera griseoaptera (DeGeer) (Orthoptera: Tettigoniidae) in England and Wales related to accumulated temperatures. The Journal of Animal Ecology, 1973, 531-537. [CrossRef]
- Köhler, G.; Gossner, M.M.; Gruppe, A.; Gerstmeier, R. Orthopterans from flight-interception trap samples in spruce and larch crowns of two commercial forests in Upper Bavaria, Germany. Entomologische Zeitschrift mit Insekten-Börse, 2009, 119(6), 243-248.
- Mashkovtsev, V.P. The one and unique: Kaluga on old postcards. Faubourg: Vladimir, Russia, 2006; 287 pp.
- Sergeev, M.G. Ecogeographical distribution of Orthoptera. In: The Bionomics of Grasshoppers, Katydids and Their Kin. CAB International: Wallingford (UK) and New York, 1997, pp. 129–146.
- Milyakin, S.R. Motorization: History, Factors and Patterns. Studies on Russian Economic Development, 2023, 34(2), 254-262. [CrossRef]
- Novikov, V.P. (ed.). Oka Canyon Valley is a treasure of the Middle Russia; Biodiversity Conservation Center Publ.: Moscow – Kaluga, Russia, 2016, 96 pp.
- Varga, Z. Post-glacial dispersal strategies of Orthoptera and Lepidoptera in Europe and in the Carpathian basin. In Proceedings 13th international colloquium European Invertebrate Survey, Leiden, 2-5 September 2001. European Invertebrate Survey - the Netherlands, Leiden, 2001, pp. 93-105.
- Petrov, V.G. Geological structure and mineral resources of the Kaluga Region; Publishing house «Eydos», Kaluga, Russia, 2003; pp. 1-440.
- Penev, L.D. Large-scale variation in carabid assemblages, with special reference to the local fauna concept. Ann. Zool. Fennici, 1996, 33, 49-64.
- Hewitt, G.M. Post-glacial re-colonization of European biota. Biological Journal of the Linnean Society, 1999, 68(1-2), 87–112. [CrossRef]
- Lavrenko, E.M. History of the flora and the vegetation of the USSR according to data on the recent distribution of plants. In Vegetation of the USSR; Izd-vo AN SSSR, Moscow – Leningrad, Russian, 1938, pp. 235–396.
- Soloviyova, M.P.; Khomutova, M.S. Main results of the studying of the vegetation of the Kaluga Region. In Second conference of the regional studies of the Kaluga Region; Kaluga – Obninsk, Russia, 1970, pp. 52–56.
- Novenko, E.Y.; Tsyganov, A.N.; Mazei, N.G.; Kupriyanov, D.A.; Rudenko, O.V.; Bobrovsky, M.V., … Nizovtsev, V.A. Palaeoecological evidence for climatic and human impacts on vegetation in the temperate deciduous forest zone of European Russia during the last 4200 years: A case study from the Kaluzhskiye Zaseki Nature Reserve. Quaternary International, 2019, 516, 58-69. [CrossRef]
- Romanova, E.N.; Gobarova, E.O.; Zhil'cova, E.L. Methods for meso- and micro-climatic zoning for the purpose of optimizing the placement of crops with the use of technology for automated calculation; Gidrometeoizdat: St-Petersburg, Russia, 2003; 104 pp.
- Estevo, C.A.; Stralberg, D.; Nielsen, S.E.; Bayne, E. Topographic and vegetation drivers of thermal heterogeneity along the boreal–grassland transition zone in western Canada: Implications for climate change refugia. Ecology and Evolution, 2022, 12, e9008. [CrossRef]
- Nagy, A.; Sólymos, P. Relationship between microclimate and Orthoptera assemblages in different exposures of a dolina. Articulata, 2002, 17(1), 73-84.
- Decamps, H., Fortune, M., Gazelle, F., & Pautou, G. Historical influence of man on the riparian dynamics of a fluvial landscape. Landscape ecology, 1988, 1(3), 163-173. [CrossRef]


| Type of sample plots | Total | Presence | Absence |
|---|---|---|---|
| Gardens | 15 | 11 | 4 |
| Grasslands | 23 | 11 | 12 |
| Wetlands | 9 | 0 | 9 |
| Forests and edges (mesic), incl. | 189 | 117 | 75 |
| deciduous (non-riparian) | 101 | 72 | 29 |
| pine | 47 | 18 | 29 |
| spruce | 11 | 3 | 8 |
| riparian (willow or alder) | 30 | 17 | 13 |
| Label | Name | Levels | Description |
|---|---|---|---|
| Dec_Tree | Deciduous trees in tree layer | 0-1-2 | 0 – adult deciduous trees are absent 1 – sparse deciduous trees 2 – dense tree layer dominated by deciduous species |
| Dec_shrubs_un | Deciduous shrubs or undergrowth | 0-1 | 0 – deciduous shrubs (such as Corylus avellana L., Euonymus verrucosus Scop., Lonicera xylosteum L.) or undergrowth are not seen 1 – deciduous shrubs or undergrowth are conspicuous |
| Nem_herbs | Nemoral herbs | 0-1 | 0 – there are no nemoral species among dominants of the herb layer 1 – there are any nemoral species among the dominants of the herb layer |
| Nitr_herb | Nitrophilous herbs | 0-1 | 0 – there are no nitrophilous species among dominants of the herb layer 1 – there are any nitrophilous species among the dominants of the herb layer |
| Con_Tree | Coniferous trees | 0-1 | 0 – there are no coniferous trees in the tree stand. 1 – there are any coniferous trees (Pinus sylvestris or Picea abies) in the tree stand. |
| Con_un | Coniferous undergrowth | 0-1 | 0 – there is no coniferous undergrowth. 1 – there is coniferous undergrowth. |
| Hight_herb | Herbage height | 0-3 | 0 – there is no closed herb layer 1 – low herb layer (to 20 cm) 2 – moderate herb layer (20-60 cm) 3 – tall herb layer (above 60 cm) |
| Cover_herb | Herbage coverage | 0-2 | 0 – no closed herb layer 1 – sparse herb layer (coverage 10-70%) 2 – dense herb layer (70-100%) |
| Deadwood | Deadwood | 0-2 | 0 – deadwood was not seen when we sampled the insects 1 – moderate stock of deadwood (single stumps or trunks) 2 – large stock of deadwood |
| Isolation | Isolation | 0-1 | 0 – plot is not isolated by roads, buildings, bogs or other physical barriers from neighbouring forests 1 – plot is isolated by roads, buildings, bogs or other physical barriers from neighbouring forests |
| Old_forest | Presence of old forest | 0-2 | 0 – no forests within 5 km radius based on 1860 map 1 – forests within 500 m to 5 km distance 2 – forests on the same place or to 500 m |
| Landscape | Lanscapes | 5 levels | Mos_mor – moraine plains of the last stage of the Moscow glaciation Edg_mos – plains of the early stage (marginal zone) of the Moscow glaciation Ers – erosional plains outside the boundaries of the Moscow glaciation Flvg – fluvioglacial plains of both Moscow and Don glaciations Rip – landscapes of river valleys |
| Factor | Coeff. | Std. Error | z value | Pr(>|z|) | LR_Chisq | Partial_R2 |
|---|---|---|---|---|---|---|
| (Intercept) | -0.4570 | 1.3352 | -0.3422 | 0.7322 | ||
| Landscape: | 25.0711 | 0.134 | ||||
| Flvg | -0.7789 | 1.2306 | -0.6330 | 0.5268 | ||
| Edg_mos | 0.2994 | 1.2858 | 0.2329 | 0.8159 | ||
| Mos_mor | -3.2771 | 1.3855 | -2.3653 | 0.0180 | ||
| Rip | -0.2310 | 1.1601 | -0.1991 | 0.8422 | ||
| Ers | 15.7300 | 1446.1421 | 0.0109 | 0.9913 | ||
| Con_Tree | -1.3725 | 0.4366 | -3.1434 | 0.0017 | 10.5609 | 0.061 |
| Old_forest | 0.2022 | 0.2999 | 0.6742 | 0.5002 | 0.4556 | 0.003 |
| Nem_herbs | 2.1991 | 0.4553 | 4.8296 | <0.0001 | 27.2026 | 0.144 |
| Isolation | -3.4308 | 0.7535 | -4.5533 | <0.0001 | 26.5071 | 0.141 |
| Autocovariation | 121.1268 | 49.6305 | 2.4406 | 0.0147 | 6.5204 | 0.039 |
| Factor | Estimate | Std. Error | z value | Pr(>|z|) | LR_Chisq | Partial_R2 | Pr(>Chisq) |
|---|---|---|---|---|---|---|---|
| (Intercept) | -3.020 | 2.541 | -1.189 | 0.2346 | |||
| Con_Tree | -9.151 | 6.112 | -1.497 | 0.1343 | 6.106 | 0.308 | 0.0135 |
| Old_forest | 6.479 | 3.430 | 1.889 | 0.0589 | 22.408 | 0.620 | <0.0001 |
| Nem_herbs | 8.968 | 6.506 | 1.378 | 0.1681 | 6.716 | 0.329 | 0.0096 |
| Isolation | -7.504 | 3.792 | -1.979 | 0.0478 | 12.296 | 0.473 | 0.0005 |
| Autocovariation | -446.485 | 336.271 | -1.328 | 0.1843 | 4.344 | 0.241 | 0.0372 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




