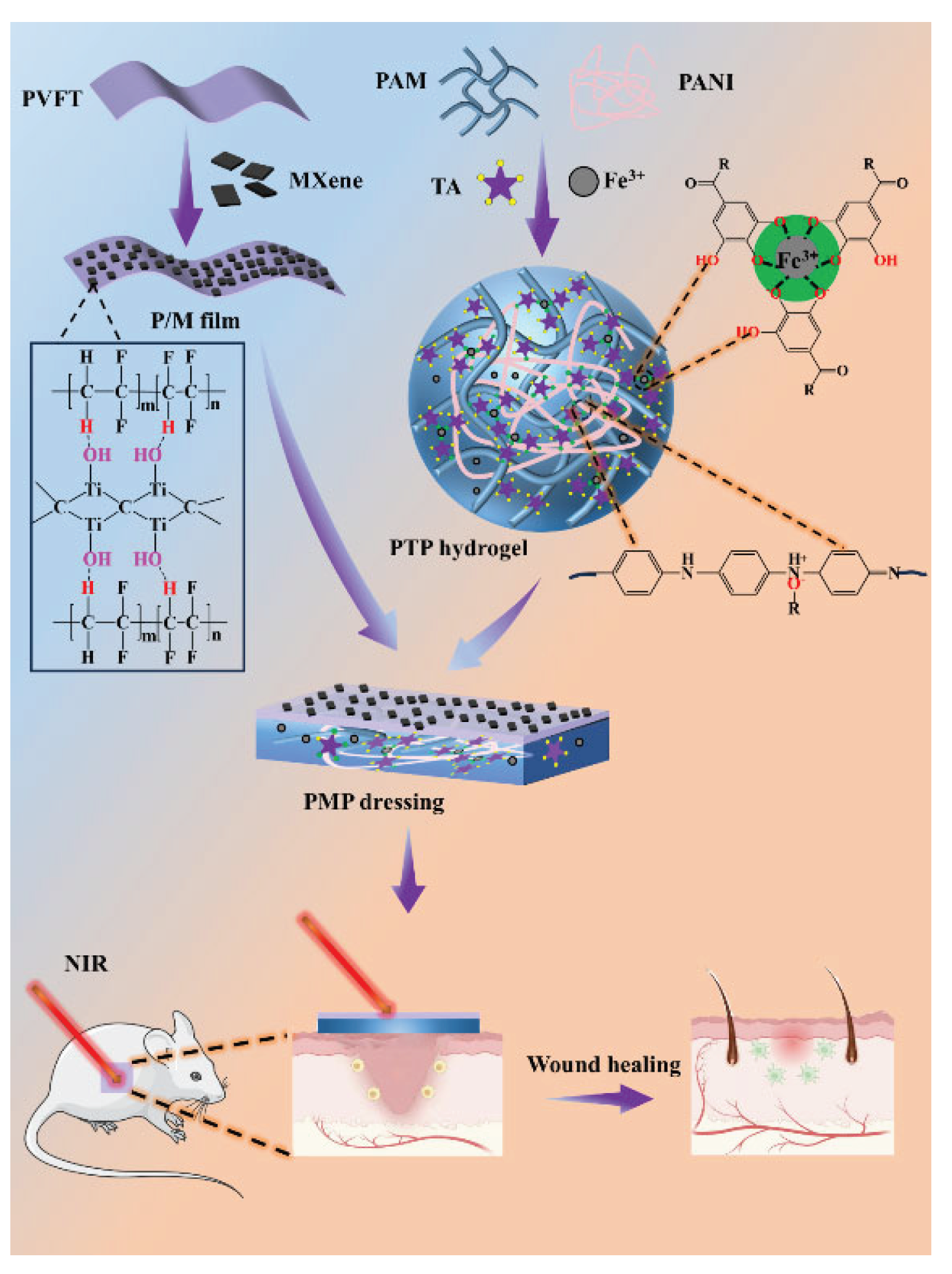
2. Materials and Methods
2.1. Materials
Poly(vinylidene fluoride-trifluoroethylene) (PVFT, FC-30) was purchased from Acoma, France. Ti3C2TX MXene was purchased from Suzhou BKNANAO Materials Co., Ltd. N,N-dimethylformamide (DMF, AR), ferric chloride (FeCl3, AR), acrylamide (AM, CP), ammonium persulfate (APS, AR), tetramethylethylenediamine (TEMED, 99%), and aniline (ANI, AR) were purchased from Chemical Reagents Co., Ltd. Tannic acid (TA, AR) and N,N-methylenebisacrylamide (MBA, AR) were purchased from Shanghai McLean Biochemical Technology Co., Ltd. Escherichia coli (E. coli, ATCC25922) and Staphylococcus aureus (S. aureus, CMCC (B) 26003) strains were purchased from the Beijing Biological Treasure Center. L929 cells were cultured in MEM medium (Gibco, USA) obtained from Shanghai Zhongqiao Xinzhou Biotechnology Co., Ltd., China. The CCK-8 kit and Live/Dead Assay kit were purchased from Beyotime Biotechnology Co., Ltd. All aqueous solutions were prepared using ultrapure water generated by a water purification system (MST-11-10, Shanghai Mosu Scientific Equipment Co., Ltd.).
2.2. Cell Lines and Animals
L929 cells were obtained from ATCC (NCTC clone 929-CCL-1). The cells were cultured in RPMI 1640 medium supplied with 10% v/v of FBS, 100 U/mL of penicillin, and 100 μg/mL of streptomycin. The cells were incubated in a 37 °C humidified environment with 5% CO2 supply. Six-week-old BALB/c mice (18-20 g, female) were obtained from Hubei Provincial Center for Disease Control and Prevention, Wuhan, China. All animal experiments were performed under the guidelines approved by the regulations of Chinese law and the local Ethical Committee. The animal experiments were approved by the Institutional Animal Care and Use Committee (IACUC Number: 4441).
2.3. Preparation of the P/M Composite
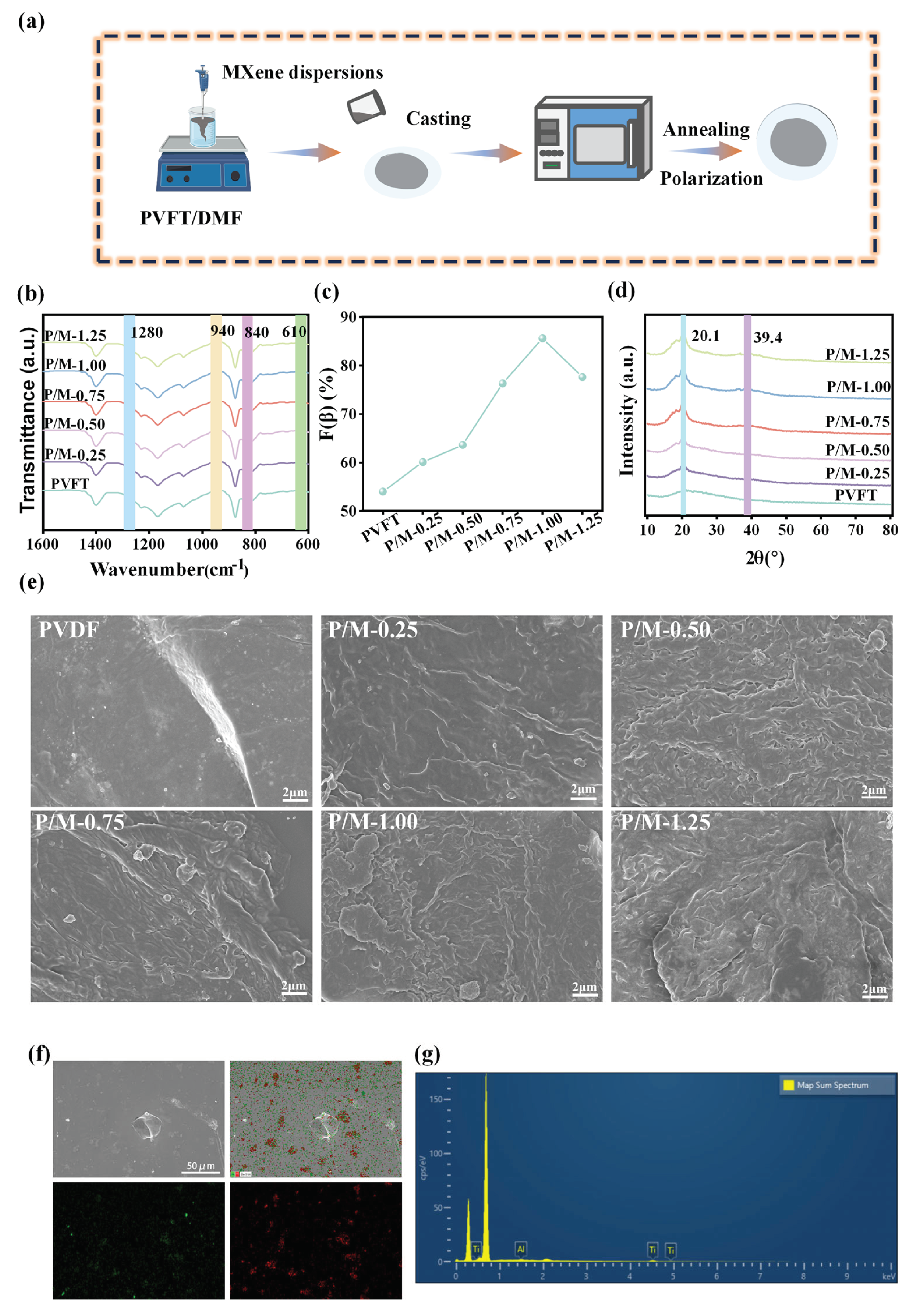
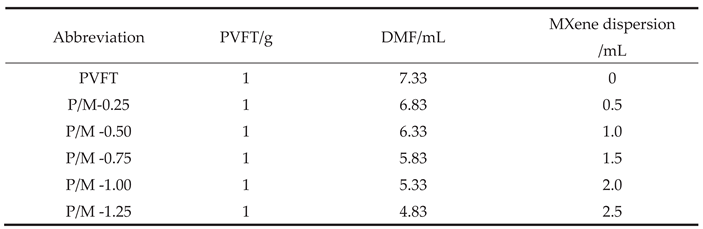
The P/M composite membrane was prepared by tape casting method, firstly, 0.25 g of MXene was dissolved in 50 mL of DMF solution, stirred at room temperature for30 min and then sonicated for 10 min to obtain a uniformly dispersed 5 mg / mL MXene dispersion. Then, 1 g PVFT powder was added into DMF, stirred at room temperature (RT) for 3 h, and then ultrasonically treated to obtain mixed PVFT solution. Then MXene dispersion was added, and the ultrasonic stirring was continued to make the solution uniform. The solution was then dumped on a clean glass plate and placed in a vacuum drying oven for 12 h at the temperature of 50 °C. Finally, the temperature of the vacuum drying oven was adjusted to 120 °C for 2 h, and then the glass plate was immersed in deionized water. The film was washed with deionized water after falling off from the surface of the glass plate, and then dried.
The specific parameters were shown in
Table 1 below. The film was washed with deionized water after falling off from the surface of the glass plate, and then dried. The prepared composite film was cut into 60x60 mm
2 sample specifications for piezoelectric polarization. The specific parameters were set as follows: the polarization voltage was 24 kV, the polarization time was 30 min, the distance between the sample and the polarization needle was set to 20 cm, and the polarization was carried out under dry conditions at RT (The P/M composite membrane is the P/M-1.00 composite membrane by default).
2.4. Fabrication of PTP Conductive Hydrogels
The preparation of PTP hydrogel was completed by one-pot method. A certain amount of AM was uniformly dissolved in ionic water (32wt %, 7.6mL), and then 0.27 g of TA, 0.1 g of ferric chloride and 0.2 g of MBA were dissolved in 10 mL of deionized water. Then, a certain amount of TA, Fe3+, MBA solution and aniline (ANI) were added to the AM solution to continue stirring, and then an appropriate amount of TEMED and APS were immediately added. Then, the PTP hydrogel was obtained by uniformly mixing and standing for a period of time and placing it in an oven at 60 °C. Named according to different ANI content, for example, ANI with the content of 0.02 mL was named PTP-0.02 (PTP hydrogel defaults to PTP-0.06).
2.5. Characterization of P/M Composite
FT-IR can be used to study the chemical structure and composition of the samples. The specific operation steps were as follows: the P/M composite film was cut into a suitable shape and placed in the drying box for 30 min to remove moisture. Then, the film was pressed with a clamp and vacuumized for scanning, with the scanning range of 4000-400 cm-1.
The crystal structure of P/M composite membrane was detected by XRD. The P/M composite film was cut into a square of 20x20 mm2, and the crystal phase was analyzed by X-ray diffractometer. The target was Cu-Kα target, the scanning speed was 2 °/s, and the detection range was 10-80°.
The surface morphology of the P/M composite membrane was observed. Firstly, the prepared composite membrane was washed with ethanol-water solution, and then rinsed repeatedly with deionized water and dried. The P/M composite film was cut into a suitable shape and adhered to the test bench. Then vacuum treatment and surface gold spraying were carried out to observe the surface morphology and obtain pictures.
2.6. Characterization of P/M Composite
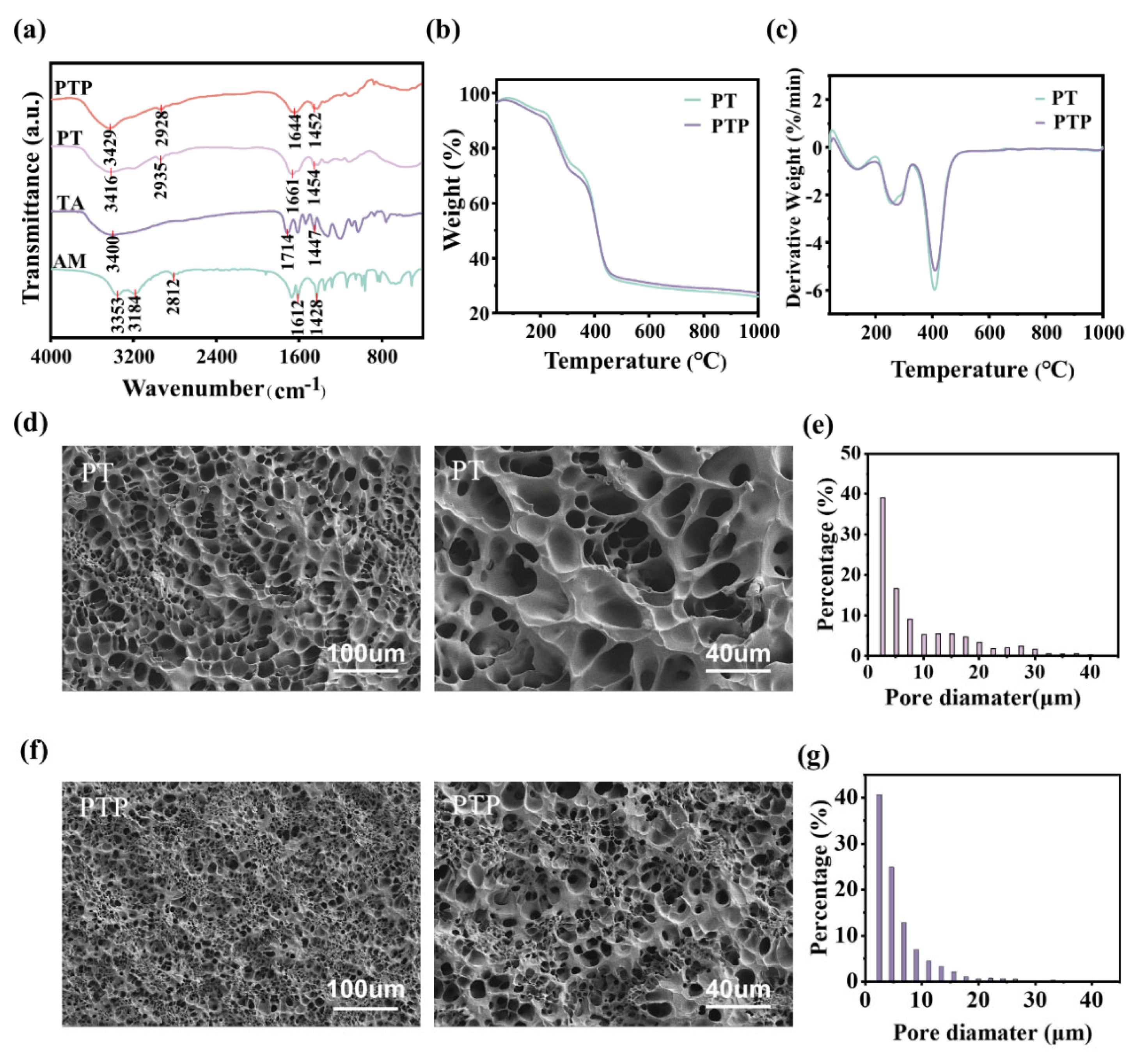
The functional groups of the prepared hydrogel samples were characterized by Fourier transform infrared spectroscopy. The freeze-dried hydrogel samples were ground into powder and dried at 50 °C for 6 h in a blast drying oven. The samples were mixed with potassium bromide (KBr) powder in certain proportion, ground and pressed, and then tested. The scanning test range was 400-4000 cm-1.
The crystal structure and properties of the samples were analyzed by X-ray diffractometer. The processed freeze-dried hydrogel sample powder was placed in the sample scaffold, and the test diffraction angle range was set to 5°-90°.
Each hydrogel sample was frozen and crisped in liquid nitrogen, and then freeze- dried for 48 h. The sample was fixed on the sample holder using conductive adhesive, and then gold was sprayed on the cross section of the sample. The internal microstructure of the hydrogel was observed under a scanning electron microscope.
The hydrogel sample after freeze-drying treatment was cut into very fine particles, and then the vacuum drying oven was used to remove water. Then a certain amount of sample was weighed, put into the instrument and connected to N2, and the weight change of the sample from RT to 1000 °C was tested.
2.7. Water Contact Angle Assay
The hydrophilicity of P/M composite piezoelectric film was evaluated by static contact angle test at RT. The specific test steps were as follows: Control the knob to spin a droplet of the appropriate size at the tip of the needle, and then increased the height of the plane where the composite film was located until the droplet drops on the surface of the film. After the shape of the water droplets was stable, a clear image was taken, and then the image data was processed to calculate the contact angle of the composite film. In order to ensure the authenticity and reliability of the data, each group of composite membranes was subjected to three parallel tests, and the results were averaged.
2.8. Swelling Performance of PTP Conductive Hydrogels
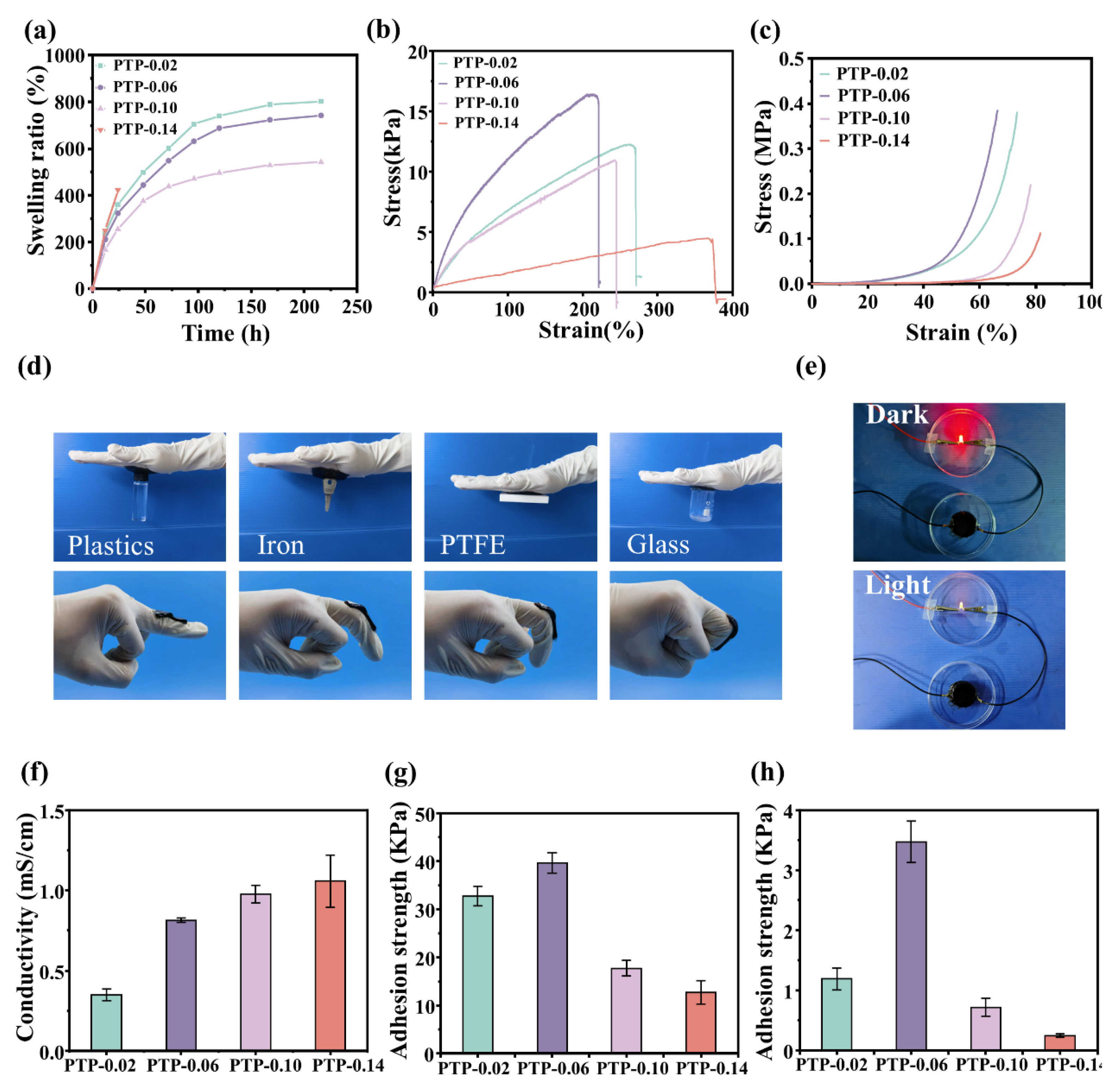
In order to evaluate the water absorption capacity and swelling stability of PTP hydrogels, the swelling ratio needed to be tested. The hydrogel samples were prepared to ensure the uniform shape and size of the samples. The hydrogel samples were freeze-dried for 48 h to constant weight (Mt). The freeze-dried hydrogels were immersed in PBS buffer at pH 7.4 at 37 °C. During the soaking process, the hydrogel sample fully absorbed water and reached an equilibrium state. At regular intervals, the soaked hydrogel samples were taken out and the excess water on the surface was gently sucked with filter paper, and the weight was weighed (recorded as M
0). Continuous measurement until the hydrogel reached stable swelling equilibrium state. In order to ensure the accuracy of the data, each group of hydrogel samples were tested three times in parallel. The swelling ratio of the hydrogel was calculated according to Formula (1) [
28,
29] :
2.9. Mechanical Properties
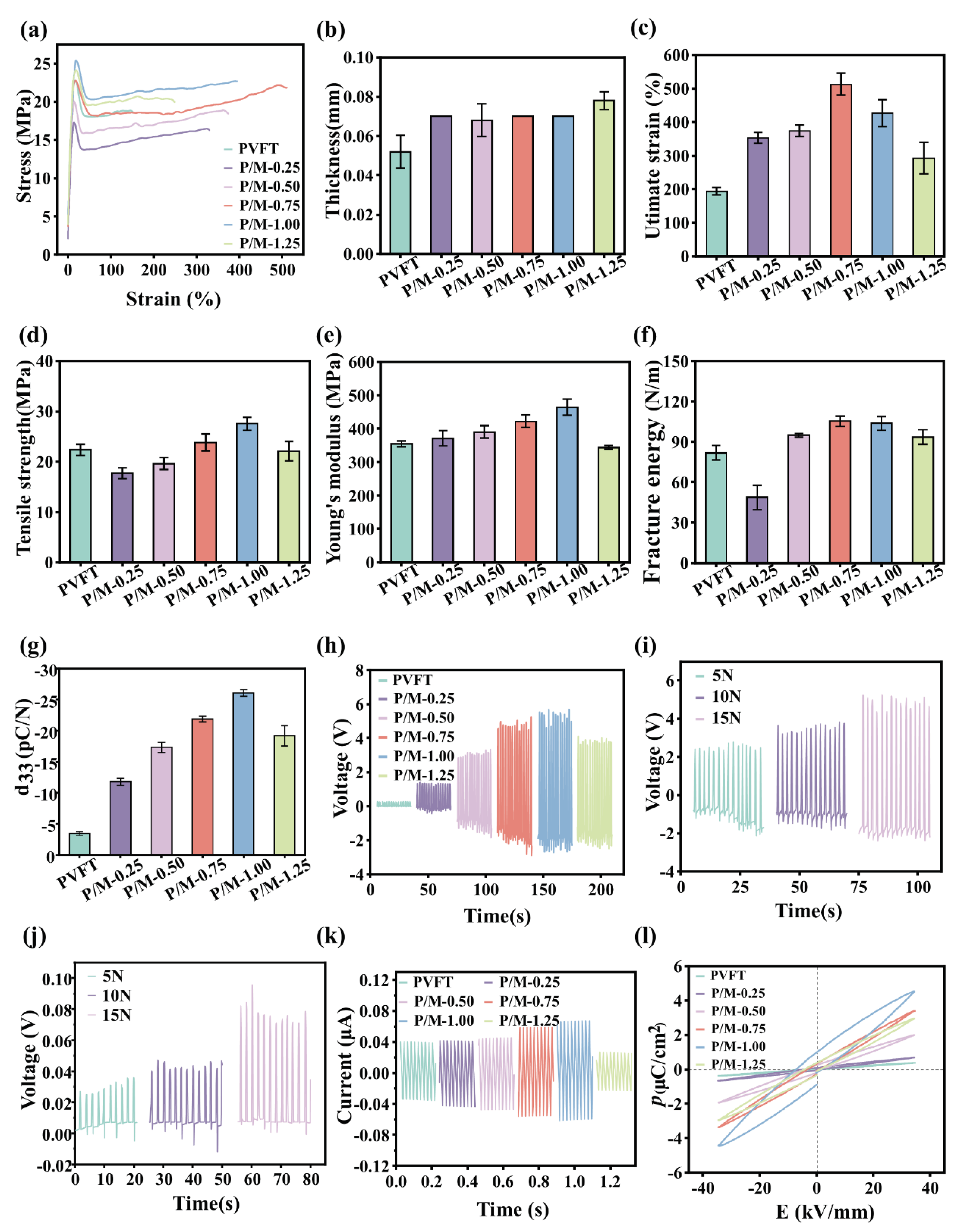
The composite piezoelectric film was cut into a rectangular size of 60x10 mm2, and the upper and lower ends of the film were pasted with cotton tape to facilitate fixing and increasing the friction force. The thickness and width of the composite film were measured using a vernier caliper. At RT, the tensile test of the composite film was carried out at the speed of 8 mm/min by using an electronic universal material testing machine. According to the measured stress-strain data curve, three parallel tests were performed on each film using the same method, and the results were averaged.
The hydrogel was prepared into a dumbbell shape with size of 20 mm long, 3 mm wide and 1 mm thick. The electronic universal material testing machine was used to test the hydrogel at the speed of 5 mm/min, and the stress-strain data curve was measured. The hydrogel was subjected to three parallel tests using the same method, and the results were averaged.
The hydrogel was prepared into a cylinder with a diameter of 25 mm and a height of 20 mm, and equilibrated for 6 h after gelation. The hydrogel sample was placed on the machine to apply the compression force, and the sample was compressed at a speed of 20 mm/min at RT with a universal testing machine. The instrument was compressed to the critical point of the hydrogel sample to obtain the stress-strain curve of the hydrogel sample. In order to ensure the accuracy of the data, each group of hydrogel samples were tested three times in parallel.
Rheological measurements were performed on a rheometer ( TA DHR-2, USA ) to prepare disc-like hydrogel samples ( 20mm×2mm ). The gel-sol transition point of the hydrogel was determined by strain scanning in the strain range of 0.1% -400% at a constant frequency of 1 Hz at 37 °C. Under the condition of constant strain amplitude γ = 5%, the frequency sweep was carried out in the frequency range of 0.1 ~ 10 Hz.
2.10. Tissue Adhesion of Hydrogels
The adhesion ability of the hydrogel was tested by the lap shear method, and the pig skin was used to simulate the human surface to test the adhesion of the hydrogel. Firstly, the fresh pig skin was treated cleanly, cut into a long strip shape of 40x10 mm
2, and the cut pig skin was soaked in PBS solution for 2 h. The prepared hydrogel was adhered between two pigskins. The contact area between the pig skin and the hydrogel was 10x10 mm
2, and then stand for 1h to ensure that the hydrogel was fully adhered to the pig skin. Subsequently, the clamp was clamped and the tensile test was carried out using a universal material testing machine. When the two pigskins were separated, it had the maximum fracture strength. The magnitude of the force at this time was recorded and calculated according to the following formula. Each group of hydrogels was tested three times, and the experimental results were averaged. The hydrogel adhesion to pig skin and P/M film were also tested according to the above steps, and pig skin was replaced with composite film. The bonding strength is obtained from Formula (2):
Among them, F was the maximum fracture force; S was the contact area (cm2) between the hydrogel and the pig skin.
2.11. Electrical Conductivity of PTP Conductive Hydrogels
In order to explore the effect of adding PANI on the conductivity of hydrogels, the resistance of hydrogels was tested by electrochemical workstation. The prepared hydrogel was cut into a cuboid of appropriate size, and the resistance of the hydrogel was measured by an electrochemical workstation with electrodes connected at both ends. The conductivity σ of the hydrogel was calculated according to Formula (3):
L was the length of the test hydrogel, R and S represented the resistance and cross- sectional area of the hydrogel sample, respectively.
2.12. Electrical Conductivity of PTP Conductive Hydrogels
In the piezoelectric coefficient test, the P/M composite film was first smoothed to ensure that the surface had no obvious defects. The sensor was connected to the surface of the sample. The instrument automatically processed the piezoelectric coefficient d33 according to the pressure applied to the sample surface and the corresponding charge signal. The same method was used to test three positions randomly selected for each membrane, and the results were averaged.
The ferroelectric performance test ensured that the surface of the P/M composite film wass flat and defect-free, and cut into an appropriate size. The electrode was coated on the surface of the sample to ensure that the electrode was in good contact with the surface of the sample, and the area size of the electrode was appropriate. The sample was placed in a test device and an applied electric field was applied with an electric field intensity of 40 kV/mm and the frequency of 25 Hz. The measurement process was stable and then record the measurement results. For the accuracy of the data, each group of composite membranes was tested in parallel for 3 times, and the results were averaged.
Open circuit voltage and conductive current test, the P/M composite film was cut to a suitable size, and its front and back sides were tightly connected with the conductive copper foil respectively. The surface of the copper foil was covered with a layer of PET plastic to protect the internal device. Then the copper foil was completely in contact with the measuring electrode of the electrochemical workstation. A mechanical arm was used to apply pressure perpendicular to the membrane surface to ensure that the force applied was consistent in size and frequency. The open circuit voltage measured by the measuring instrument was recorded. Each group of composite membranes was tested three times in parallel, and the results were averaged. The P/M composite film was cut to suitable size, and the prepared hydrogel was cut into a shape of suitable size. The piezoelectric film was bonded with the hydrogel to form composite dressing. A mechanical arm was used to apply pressure perpendicular to the membrane surface to ensure that the force applied was consistent in size and frequency. The open circuit voltage measured by the measuring instrument was recorded. Each group of composite membranes was tested three times in parallel, and the results were averaged.
Surface potential test and surface potential stability test, surface potential was an important method to evaluate the piezoelectric properties and surface charge distribution of composite films. The P/M composite film was cut to an appropriate size, and the surface potential measuring instrument was connected to the electrode of the sample to measure the potential distribution on the surface of the sample. Ensure that the grounding connection of the measuring instrument was good to avoid interference and error. Each group of composite membranes was tested in parallel for 3 times, and the results were averaged. In order to explore the stability of the surface potential of the P/M composite membrane, the composite membrane was placed in Eagle medium for 28 days. After the composite membrane sample was taken out every 1 week to rinse its surface, the surface potential test was performed, and the above steps were repeated for testing.
2.13. Photothermal Performance Evaluation
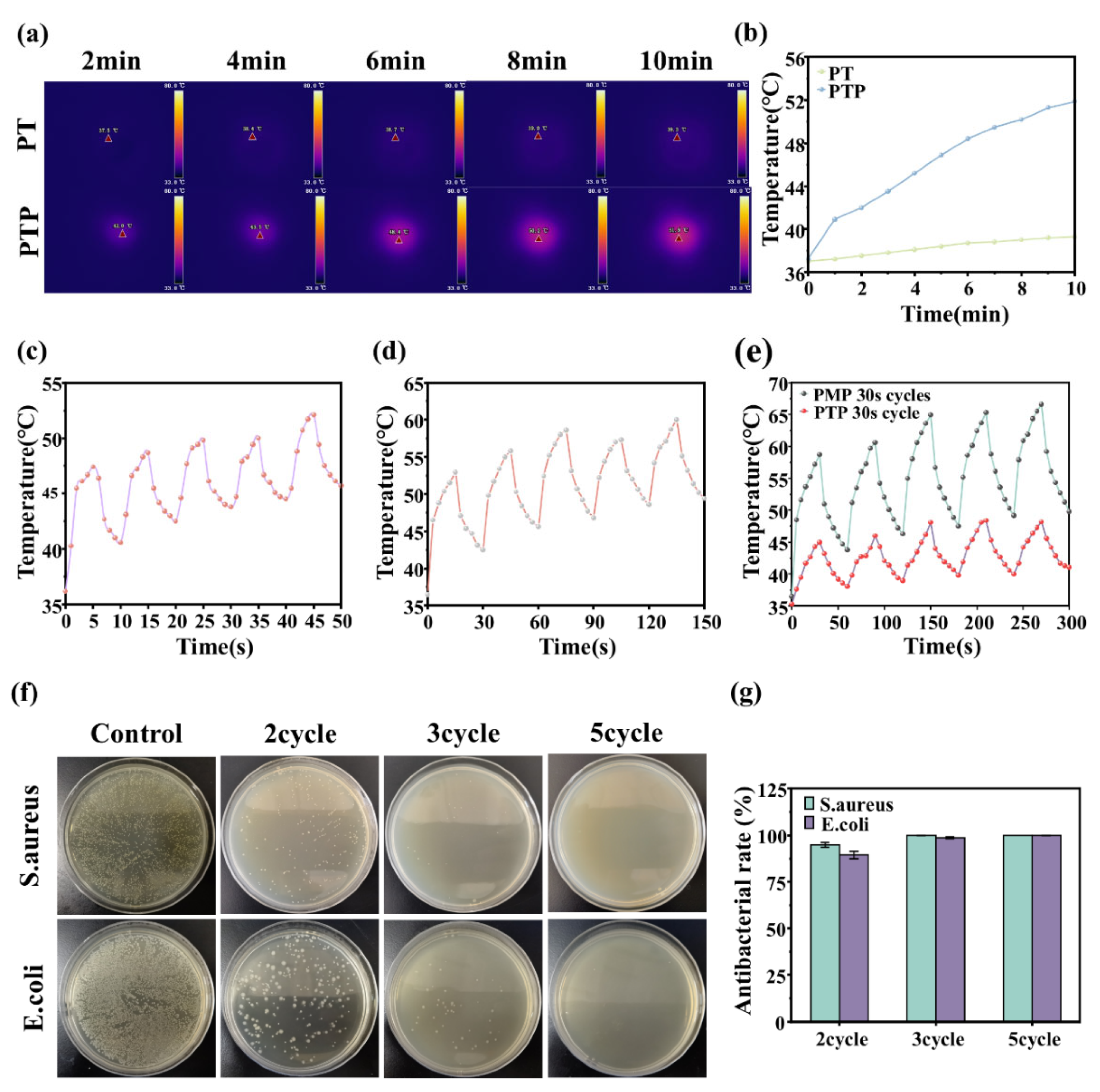
The 808 nm near-infrared laser was used to irradiate the piezoelectric film, hydrogel and composite dressing at a distance of 15 cm from the near-infrared laser, and the power was 1.0 W/cm2. Three cycles of 5 s, 15 s and 30 s were used to irradiate the composite dressing. The irradiation time of the hydrogel sample was 10 min, and the PTP hydrogel was also irradiated for 30 s as a control of the composite dressing. The near-infrared thermal imager was used to record the temperature change and take pictures.
2.14. Antibacterial Assay In Vitro
The antibacterial activity of hydrogels and composite dressings against Staphylococcus aureus and Escherichia coli was tested and evaluated. After the two strains were activated, they were cultured for 24 h. A certain amount of the prepared hydrogel was added to the 24-well culture plate, and then moved into the ultra-clean bench for ultraviolet sterilization. A 100 μL bacterial suspension (PBS, 10
6 CFU / mL) was added to the surface of the hydrogel disc, and then the 24-well culture plate was cultured at 37 °C for 2h. The hydrogel group was irradiated with 808 nm NIR laser for different times (3 min, 5 min, 10 min) and without irradiation. The composite dressing group was irradiated with 808 nm NIR laser for 30 s cycles (2, 3, 5 cycles), and continued to be cultured at 37 °C for 2 h. PBS was added to each well to re-suspend the bacteria. As a negative control group, 100 μL sterile PBS (10
6 CFU / mL) bacterial suspension was added to 1 mL PBS. Then 50μL was evenly applied to the solid agar plate. After incubation at 37 °C for 18-24 h, the number of colonies on the plate was observed and recorded. In order to ensure the accuracy of the data, each group of samples were tested at least 3 times, and the results were averaged. The antibacterial Formula (4):
Among them, M0 and M1 represent the average number of colonies on blank plates and sample plates, respectively.
2.15. In Vitro Biocompatibility and Cell Scratch Assay
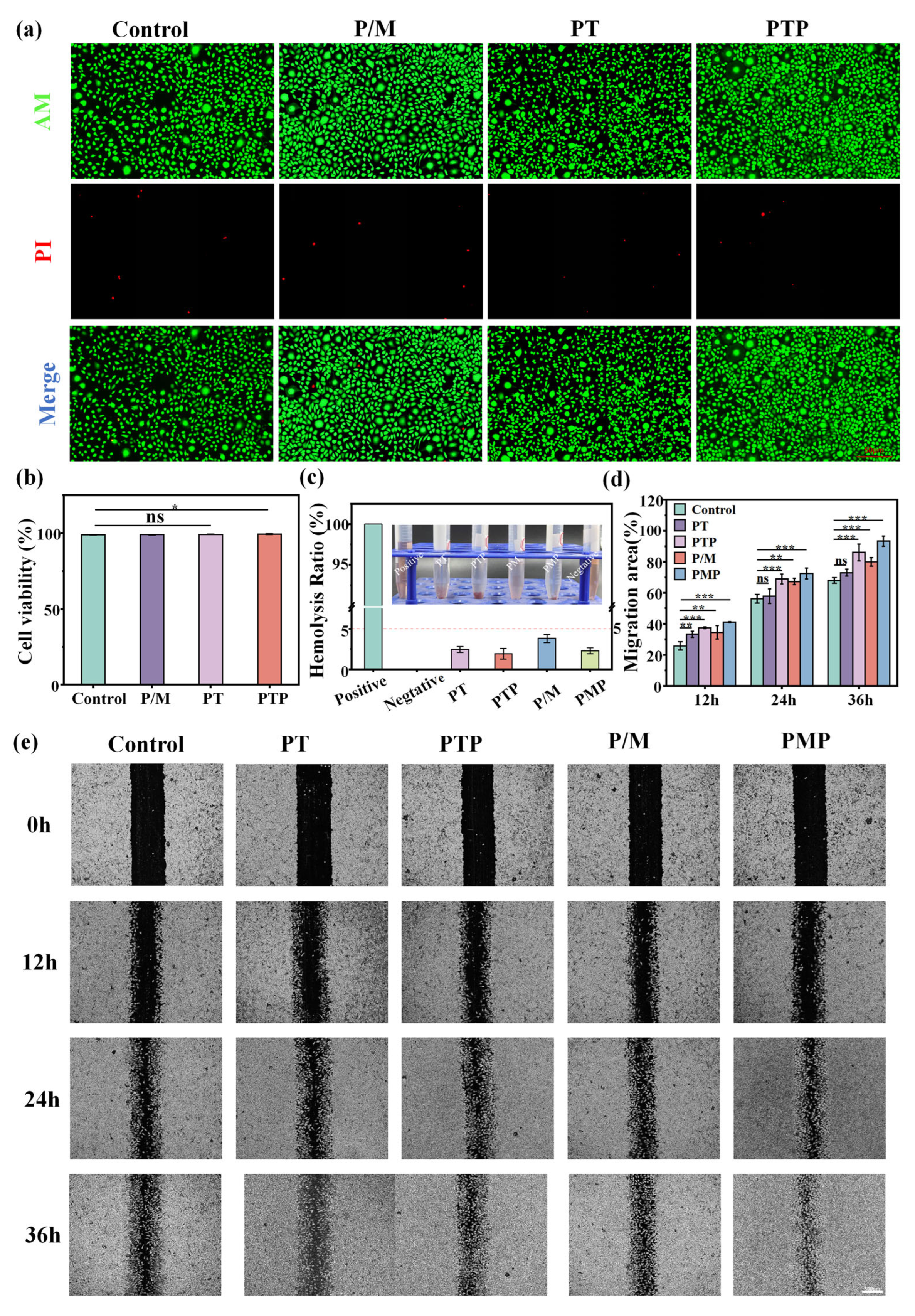
L929 cells were cultured in MEM medium supplemented with 10% fetal bovine serum and 1% penicillin-streptomycin. The culture was maintained in a controlled environment of 37 °C, 5% CO2 and humidified air, and the medium was changed every other day. L929 cells were seeded in 96-well plates and co-cultured with various biomaterials. CCK-8 kit was used for viability assessment. The evaluation procedure consisted of dispensing the CCK-8 solution into each well and incubating the plate at 37 °C for 2 hours. The absorbance was then measured at 450 nm using the Varioskan TM LUX Microplate Reader (Thermo Fisher Scientific) to determine the number of living cells based on the OD450 value. Live / Dead Assay kit (Beyotime Biotechnology, China) was used to evaluate the cytotoxicity 3 days after seeding. The experimental protocol included staining of living cells with calcein-AM (emitting green fluorescence), staining of non-living cells with propidium iodide (emitting red fluorescence), and observation of the results with a fluorescence microscope.
The blood obtained from SD rats was diluted to obtain 2% red blood cell (RBC) suspension. The material was ground into powder, 25 mg was weighed and placed in a test tube, and 10 mL normal saline was slowly dripped into the test tube. After it was completely dissolved/dispersed, the test tube was placed in water bath for 0.5 h at 37 °C. The diluted anticoagulant whole blood 0.1 mL was taken to the test tube, shaken well, placed in a water bath, and continued for 1 h at 37 °C. The test tube was taken out, and the blood cells were centrifuged at a speed of 1500 rpm/min for 10 min. The supernatant was taken out and placed in an ultraviolet spectrophotometer for absorbance determination. The wavelength was set to 545 nm. The hemolysis rate (HR) of the sample was calculated according to Formula (5):
In the equation, HR meant hemolysis rate, the absorbance of the A1 was sample group, the absorbance of the A2 was positive control group (10 mL deionized water, 0.1 mL anticoagulant whole blood, no sample material), and the absorbance of the A3 was negative control group (10 mL normal saline, 0.1 mL anticoagulant whole blood, no sample material), the experiment was repeated for five times.
The migration of L929 cells was studied by a special wound model of culture-insertion in 2 wells (ibidi GmbH, Martinsried, Germany). Approximately 70 mL ( 4 × 10
5 cells/mL ) of the cell suspension was placed into a hole in a culture plate on a 12-well plate. After incubation in complete medium for 24 hours, the cells converge around the insert, forming a 500 mm wide gap when the insert was removed. Then the cells were co-cultured with PT hydrogel extract (1mg/mL) and PTP hydrogel extract for 12, 24 or 36 hours, respectively. In the P/M group, the positive and negative sides of the membrane were closely connected with the conductive copper foil, respectively, and inserted into the wire. The surface of the copper foil was covered with a layer of PET plastic to protect the internal device. The wire was placed in the cell culture medium, and then the force generation device was knocked to make the membrane produce electrical stimulation (knocking for 15 min every 12 h). In the PMP group, P/M was replaced with PMP, and PTP hydrogel extract was added to the cell culture medium. Cell migration was continuously monitored and quantitatively evaluated at each interval using an inverted microscope and ImageJ software. The cell migration rate was as follows:
A0 and At were the initial scratch area and the scratch area after a period of culture, respectively.
2.16. In Vivo Wound Healing Analysis
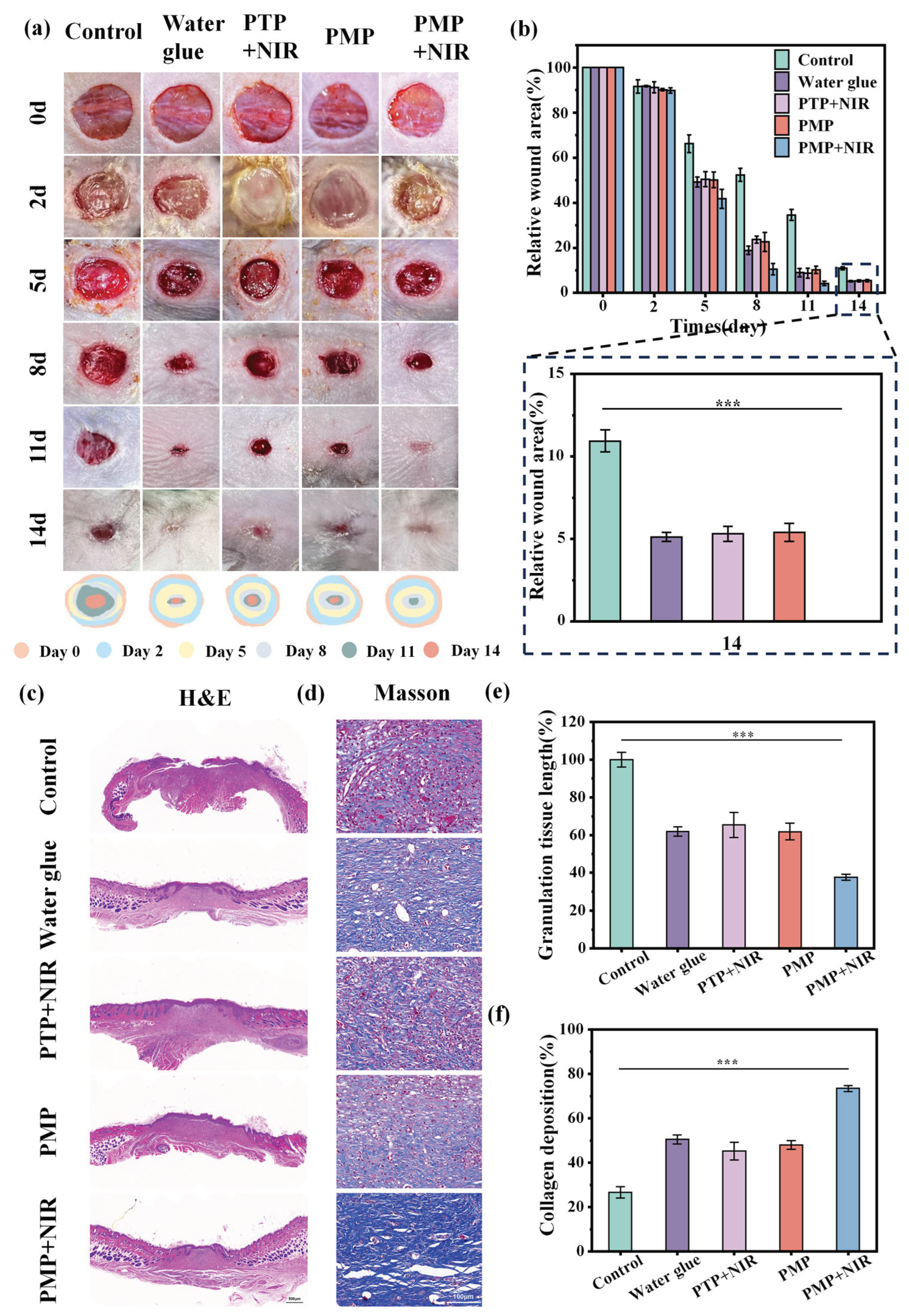
All animal experiments were approved by the Animal Protection Committee of Tongji Medical College. Six-week-old male BALB/c mice were anesthetized with 1% pentobarbital sodium (Sigma, USA), depilated and disinfected on the back, and then underwent full-thickness wound resection with a diameter of 10 mm. Each wound was injected with 10 μL (1×107 CFU/mL) Staphylococcus aureus suspension. The wound was then covered with a film dressing for two days to simulate the infected wound surface. The mice were divided into five experimental groups: control group, medical dressing water glue group, PTP hydrogel+NIR group (808 nm, 1W/cm2 for 10 minutes), PMP, PMP+NIR group (808 nm, 1 W/cm2 for 5 cycle). In all composite dressing groups, the piezoelectric effect was generated through deformation of the composite membrane induced by the natural movements and joint bending of the mice.
Wound tissues were collected on day 2, 5, 8 and 11 after injury. Wound tissues were collected on day 14, fixed with 4% paraformaldehyde, stained with hematoxylin and eosin (H&E), and stained with Masson ‘s trichrome to evaluate granulation tissue width and collagen deposition, respectively. ImageJ software was used to collect wound images and analyze them. The relative wound area calculation formula (7) is as follows:
Area0 was the initial wound area, and Arean was the wound area on the 2nd, 5th, 8th and 11th day, respectively.
2.17. Statistical Analysis
All numerical data were presented as mean ± standard deviation (SD) and statistically analyzed using GraphPad Prism 9.0 (GraphPad Software Inc., USA) with three or more replicate values. Significant differences were determined using Student’s t-test for comparison of two groups and one-way ANOVA with post-hoc Tukey’s test for multiple comparisons. The statistical significance was declared when *p < 0.05, **p < 0.01, ***p < 0.001 and ****p < 0.0001.