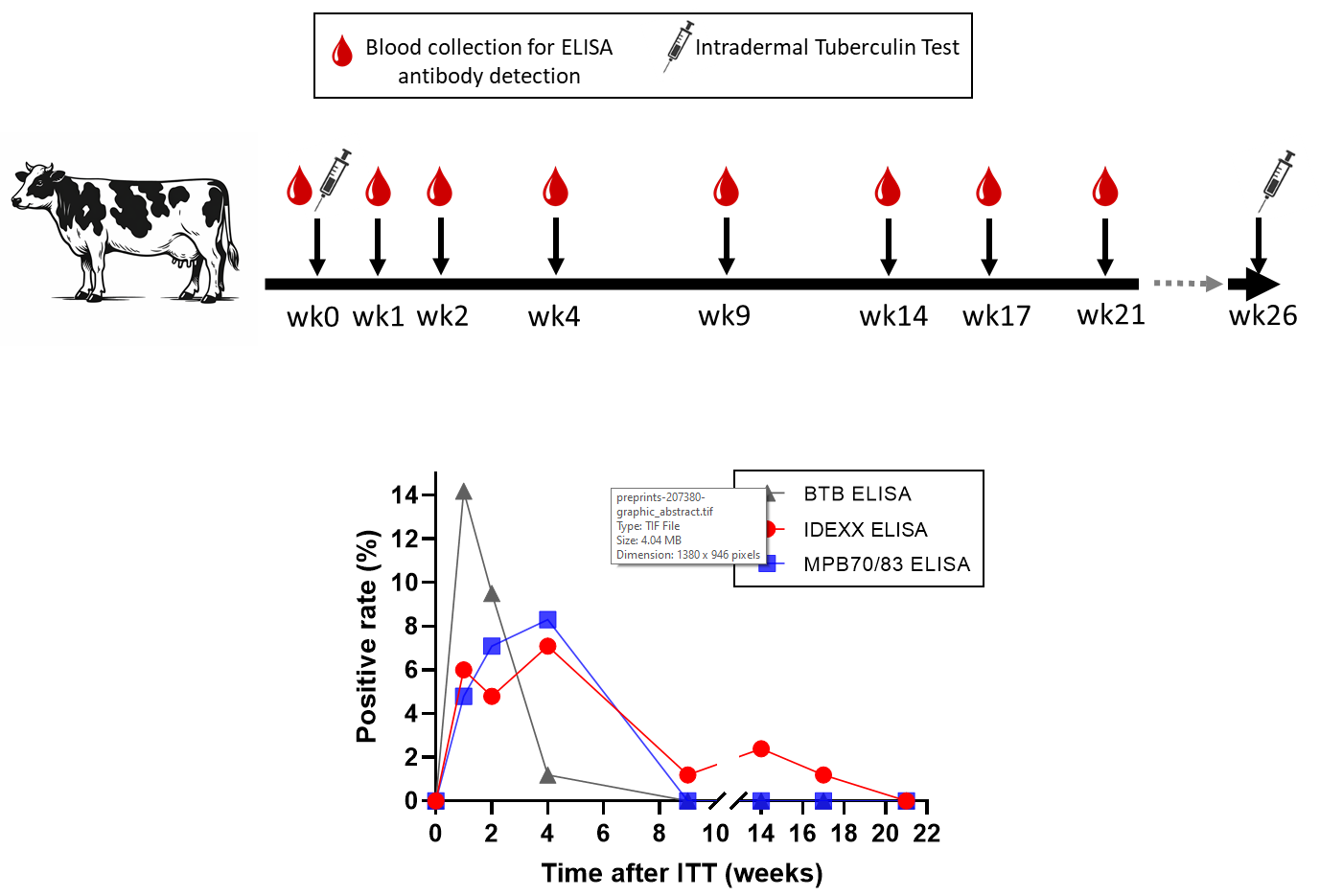
A zoonotic disease caused primarily by Mycobacterium bovis (M. bovis), bovine tuberculosis (bTB), remains a considerable global concern. The intradermal tuberculin test (ITT) is a primary global screening tool for infected animals through their cellular immune response. However, ITT fails to identify all bTB-infected animals. Serological enzyme-linked immunosorbent assays (ELISAs), which detect humoral immune responses are a potential complementary approach for bTB diagnosis. Herein, 86 serum samples collected from a bTB-free herd were analyzed using three bTB serological ELISA kits: the IDEXX M. bovis antibody test (IDEXX), the BIONOTE BTB antibody ELISA 2.0 kit (BTB), and an in-house ELISA using MPB70 and MPB83 as antigens (termed homemade). Antibody responses were monitored before and after ITT administration for 21 weeks. All serum samples collected before ITT administration tested negative with all three ELISA kits. However, 1 week after ITT administration, samples tested positive using the IDEXX, BTB, and homemade ELISA kits. Week 9, all samples tested negative with the BTB and homemade ELISA kits, whereas for IDEXX they remained negative until week 21. ITT-induced a serological response against M. bovis, engendering false-positive results. Therefore, collecting serum samples for bTB antibody testing should be avoided for at least 21 weeks following ITT.