Submitted:
07 April 2026
Posted:
09 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
2.1. The Analysis of Surface Morphology of the Prepared Catalyst
2.1.1. The Analysis of BET Data of Filtration vs Evaporation Prepared Catalyst
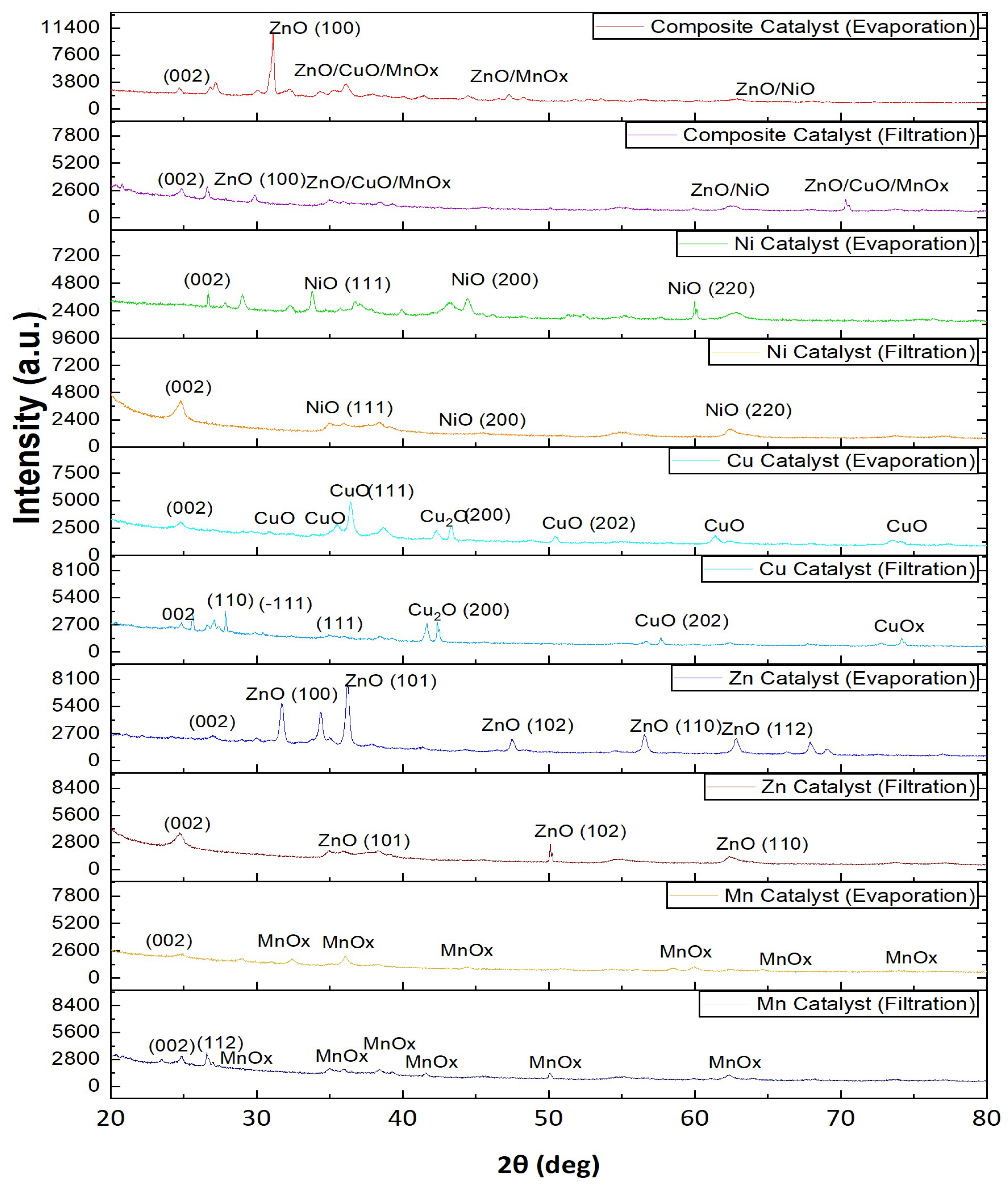
2.1.2. XRD Analysis of Catalysts Prepared by Filtration vs Evaporation
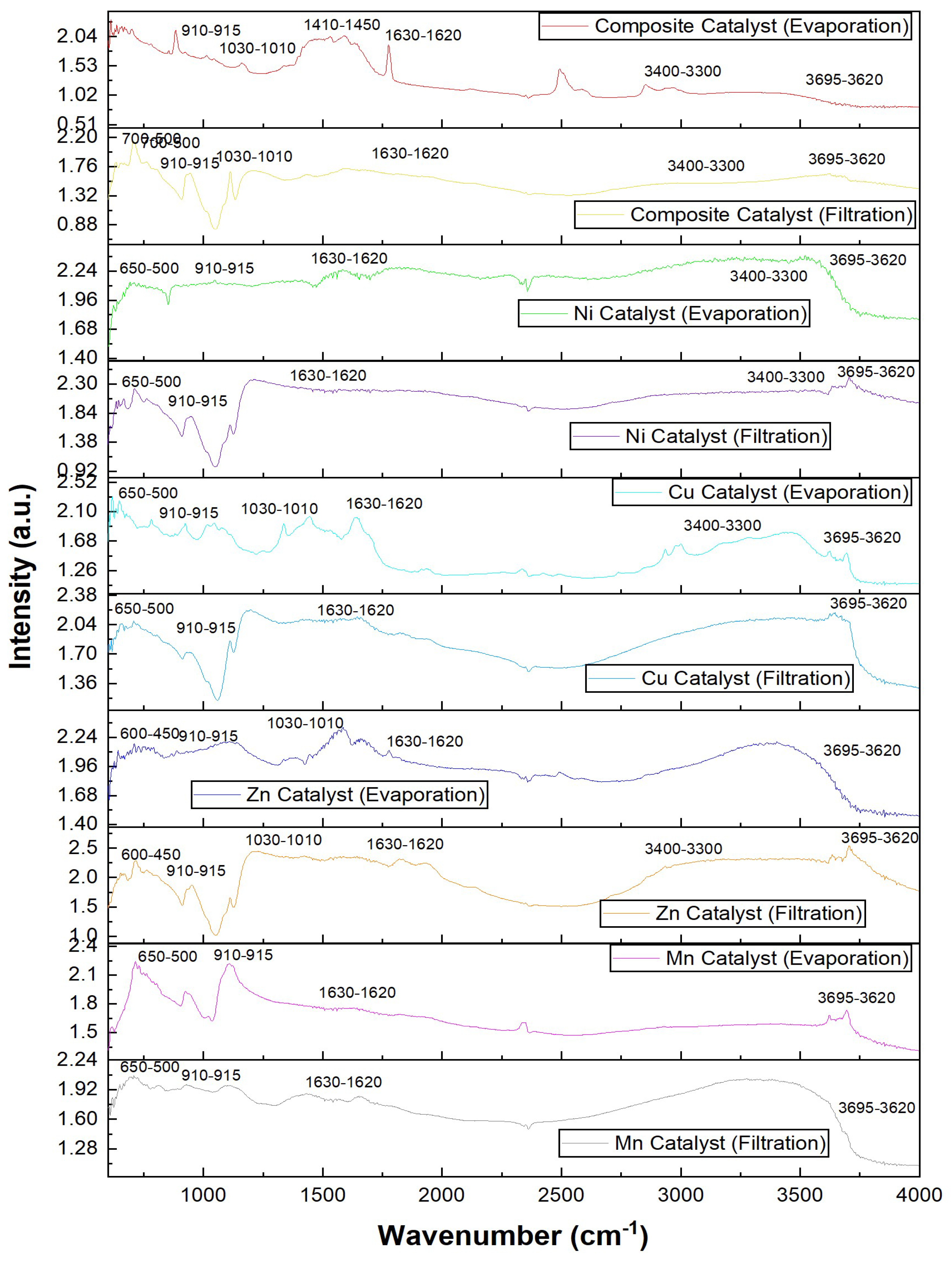
2.1.3. FTIR Analysis of Catalysts Prepared by Filtration vs Evaporation
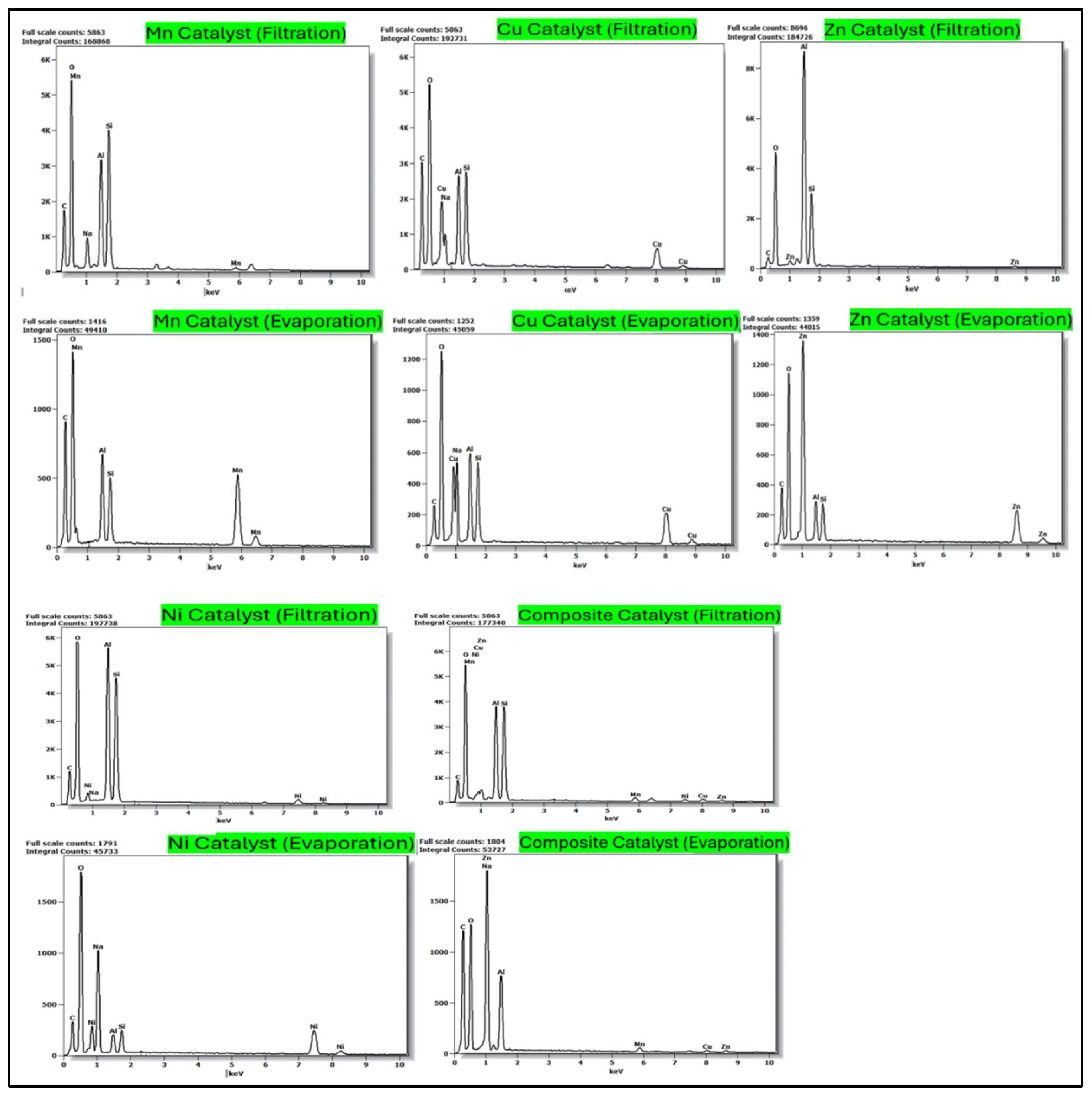
2.1.4. EDS Analysis of Catalysts Prepared by Filtration vs Evaporation
2.1.4. SEM Analysis of Catalysts Prepared by Filtration vs Evaporation
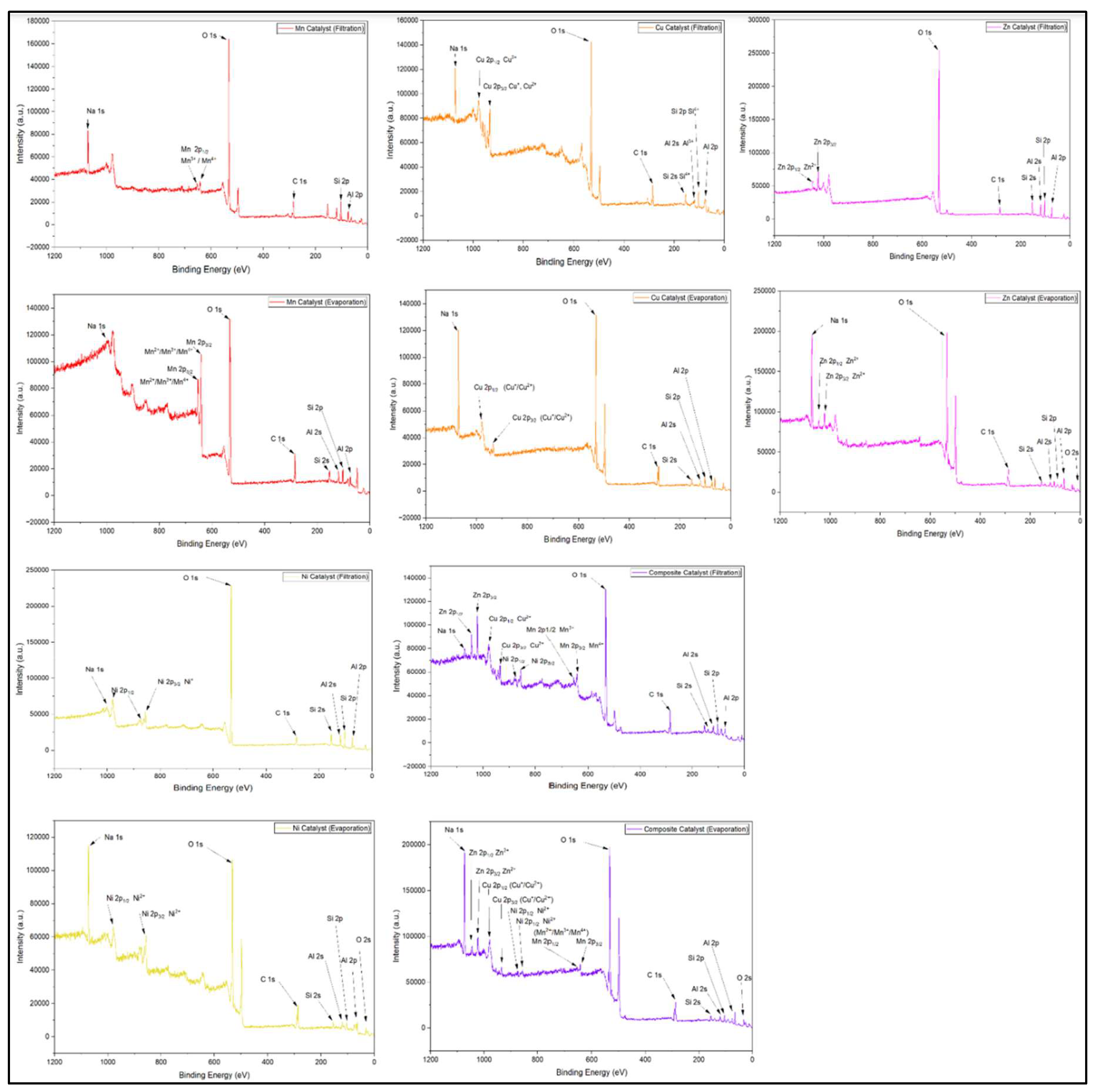
2.1.5. XPS Analysis of Catalysts Prepared by Filtration vs Evaporation
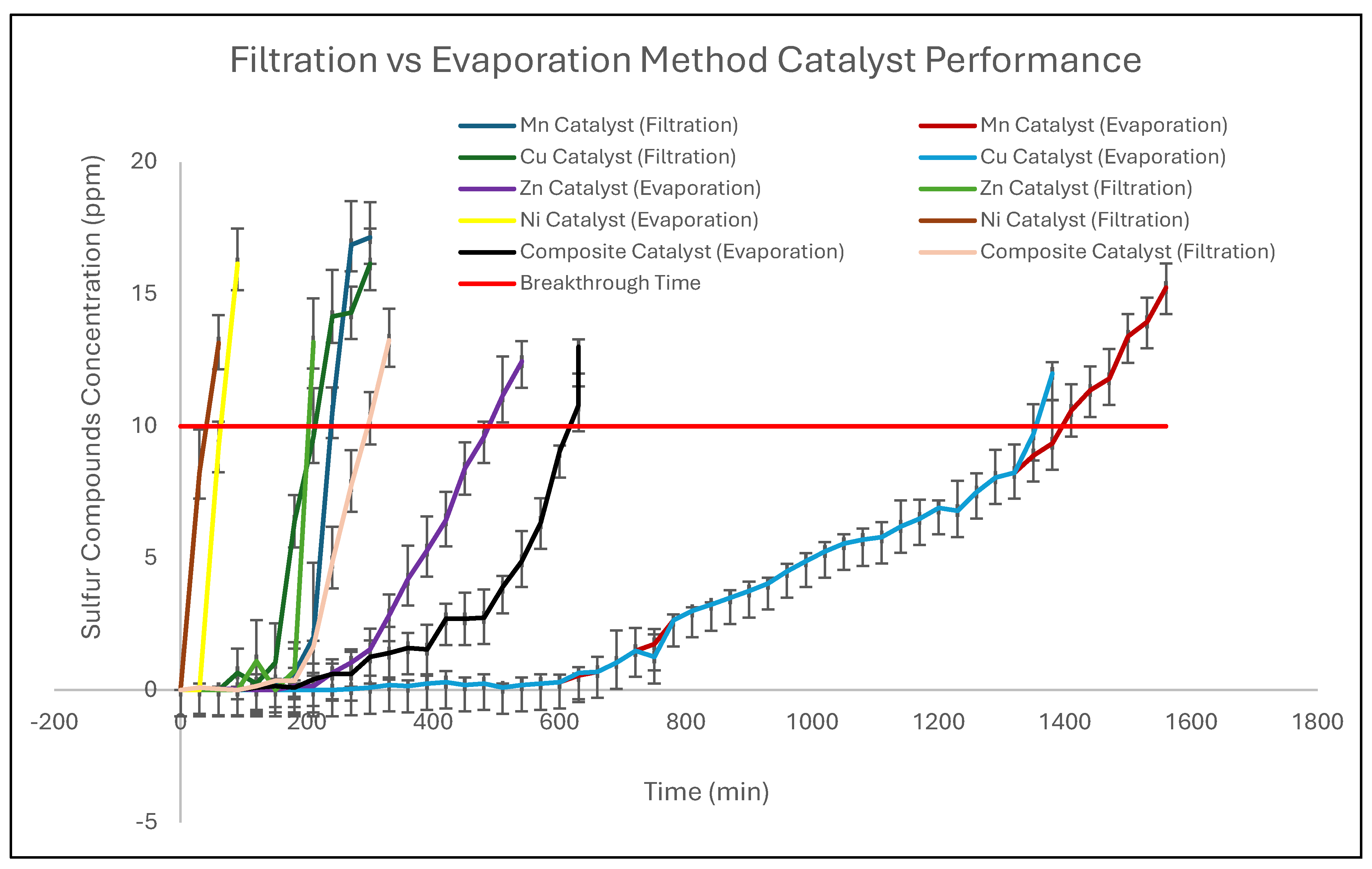
2.2. The Analysis of Catalysts Adsorption Performance
3. Materials and Methods
3.1. Materials
3.2. Catalyst Preparation
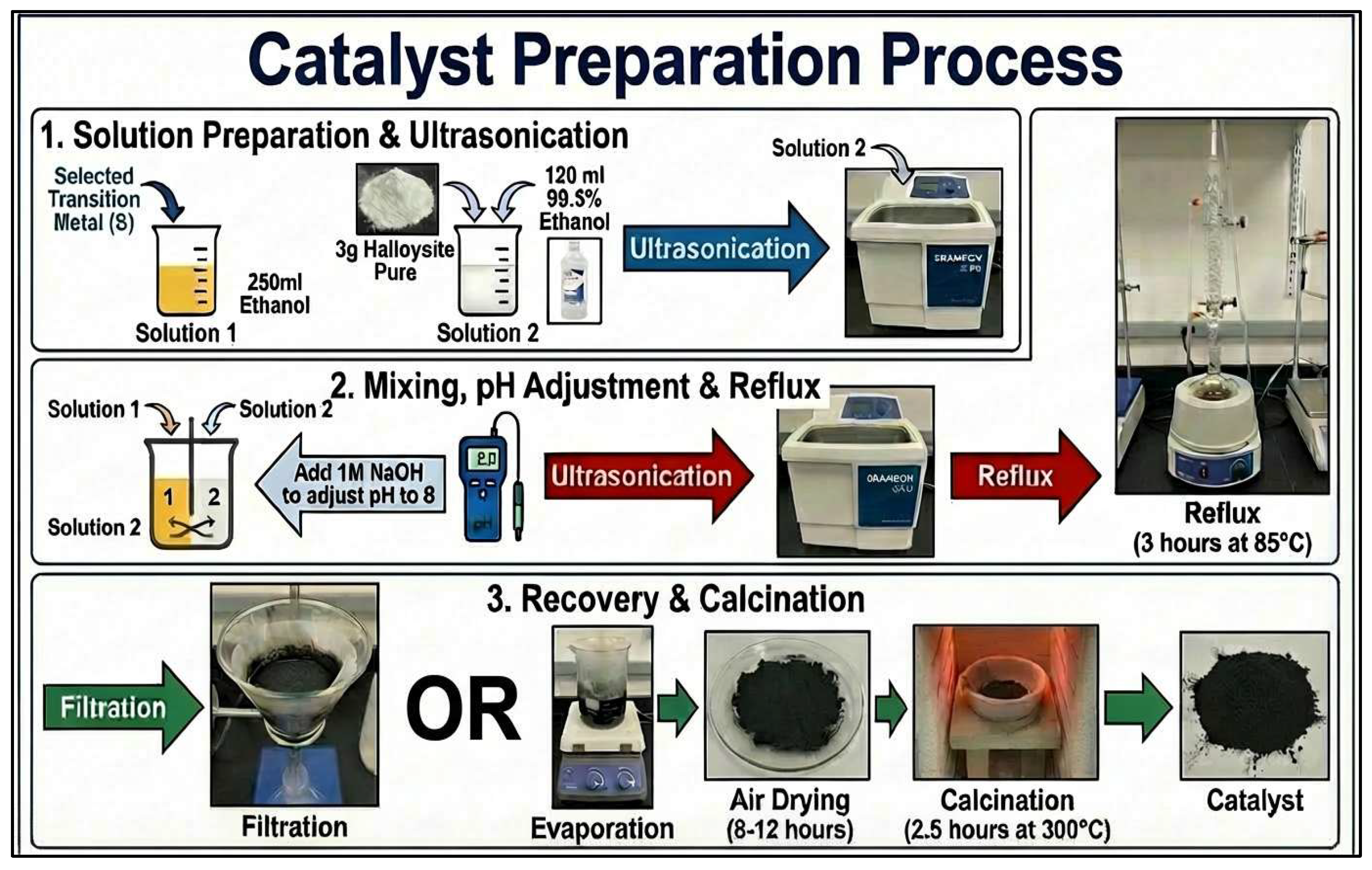
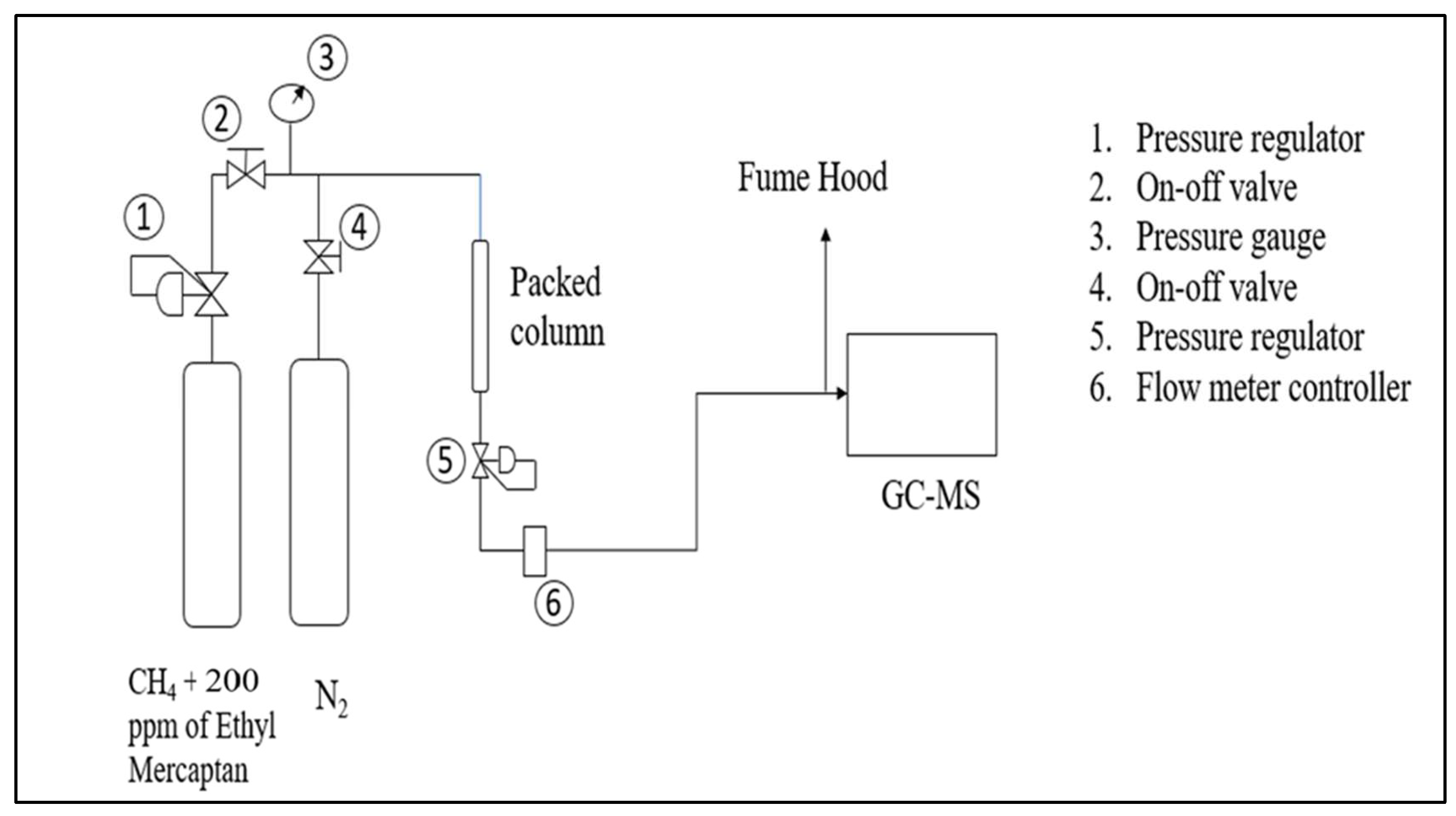
3.3. Catalyst Testing Process
4. Conclusions
- The catalyst preparation route was shown to be a critical factor controlling sulfur adsorption performance, with the evaporation method consistently outperforming filtration due to improved retention and utilization of active metal species on the support.
- Evaporation preserved a higher concentration of surface-accessible metal oxides, as confirmed by EDS and XPS, enabling stronger metal-oxygen active sites that enhanced interaction with sulfur-containing molecules.
- Structural characterization (SEM and BET) revealed that evaporation produced a more porous, rough, and accessible morphology, which improved gas diffusion and exposure of active sites, even in cases where total BET surface area was not maximized.
- Phase and dispersion analysis (XRD) indicated that evaporation promoted better developed and more finely distributed metal oxide phases, while filtration led to reduced oxide presence and less effective surface coverage due to precursor loss.
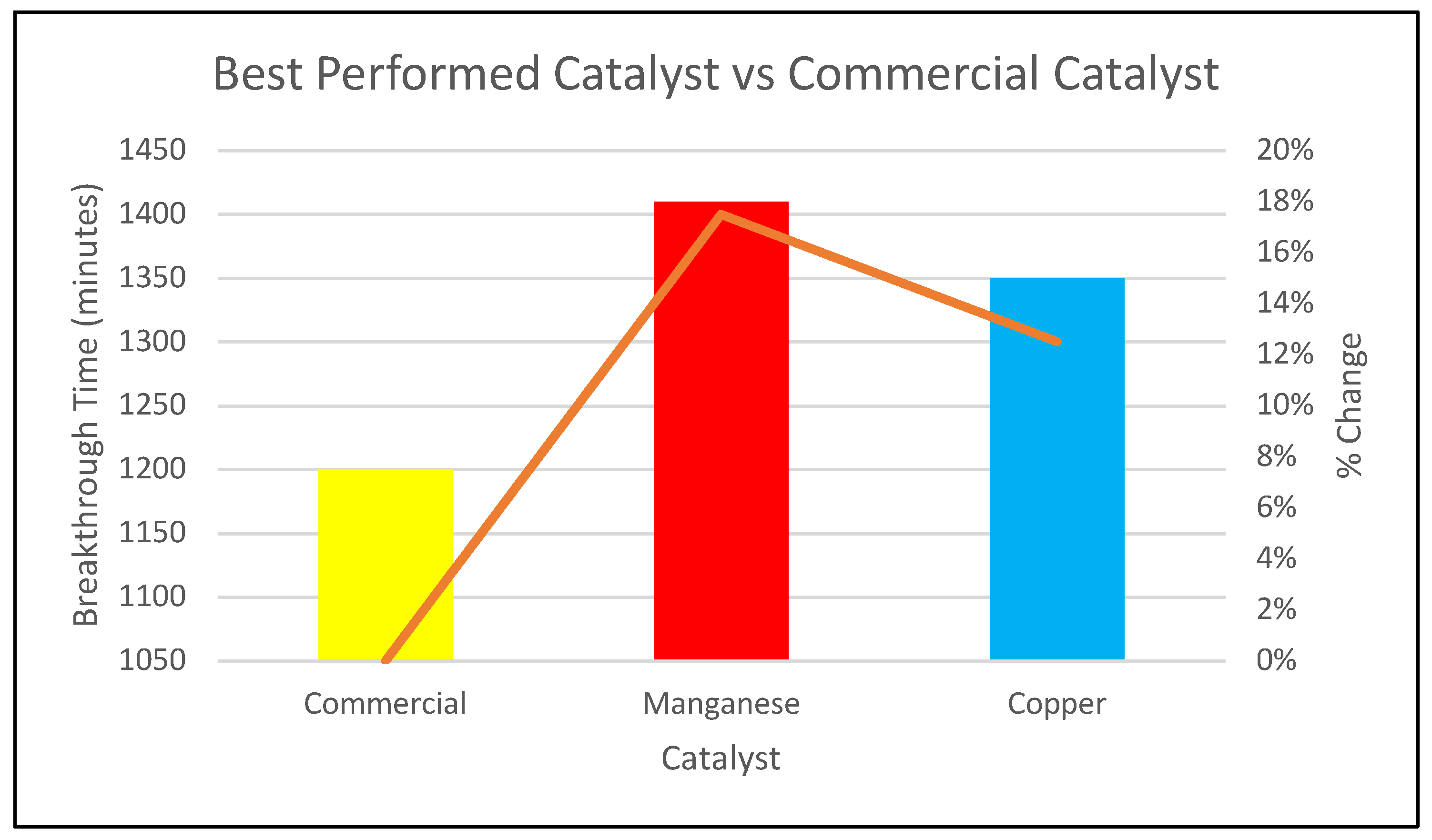
- The Mn and Cu catalysts prepared via evaporation exceeded the performance of the commercial catalyst, achieving longer breakthrough times and demonstrating that simple synthesis control can yield materials with commercially competitive or superior adsorption performance.
- Overall, the evaporation route provides a scalable, low-complexity, and effective strategy for producing high-performance halloysite-supported metal oxide catalysts, with strong potential for industrial natural gas desulfurization applications, particularly for the removal of trace sulfur compounds such as ethyl mercaptan.
- The evaporation-prepared Mn and Cu catalysts outperformed the commercial catalyst under identical test conditions, achieving longer breakthrough times (1410 min for Mn and 1350 min for Cu vs. 1200 min for the commercial catalyst) and higher calculated sulfur capacities, demonstrating that a simple and scalable preparation route can yield low-cost catalysts with strong potential for fixed-bed natural gas desulfurization applications.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Taheri, A., Babakhani, E.G. and Towfighi, J., 2017. Methyl mercaptan removal from natural gas using MIL-53 (Al). Journal of Natural Gas Science and Engineering, 38, pp.272-282. [CrossRef]
- Zhao, H., Hu, L., Zhang, X., Zhu, J. and He, J., 2022. Adsorption and separation of ethyl mercaptan from methane gas on Ni–Ti–LDH nanosheets. Applied Physics A, 128(8), p.687. [CrossRef]
- Castro, P.S., Zuniga, G.M., Holmes, W., Buchireddy, P.R., Gang, D.D., Revellame, E., Zappi, M. and Hernandez, R., 2023. Review of the adsorbents/catalysts for the removal of sulfur compounds from natural gas. Gas Science and Engineering, 115, p.205004. [CrossRef]
- Xie, Y., Bao, J., Song, X., Sun, X., Ning, P., Wang, C., Wang, F., Ma, Y., Fan, M. and Li, K., 2023. Catalysts for gaseous organic sulfur removal. Journal of hazardous materials, 442, p.130029. [CrossRef]
- Aminuddin, M.S., Bustam, M.A. and Johari, K., 2024. Latest technological advances and insights into capture and removal of hydrogen sulfide: a critical review. RSC sustainability, 2(4), pp.757-803. [CrossRef]
- Pinna, F., 1998. Supported metal catalysts preparation. Catalysis Today, 41(1-3), pp.129-137. [CrossRef]
- Lekhal, A., Glasser, B.J. and Khinast, J.G., 2001. Impact of drying on the catalyst profile in supported impregnation catalysts. Chemical engineering science, 56(15), pp.4473-4487. [CrossRef]
- Regalbuto, J. ed., 2016. Catalyst preparation: science and engineering. CRC press.
- Van Dillen, A.J., Terörde, R.J., Lensveld, D.J., Geus, J.W. and De Jong, K.P., 2003. Synthesis of supported catalysts by impregnation and drying using aqueous chelated metal complexes. Journal of Catalysis, 216(1-2), pp.257-264. [CrossRef]
- Joussein, E., Petit, S., Churchman, J., Theng, B., Righi, D. and Delvaux, B.J.C.M., 2005. Halloysite clay minerals—a review. Clay minerals, 40(4), pp.383-426. [CrossRef]
- Suslick, K.S., 1990. Sonochemistry. science, 247(4949), pp.1439-1445.
- Suslick, K.S. and Price, G.J., 1999. Applications of ultrasound to materials chemistry. Annual Review of Materials Science, 29(1), pp.295-326. [CrossRef]
- Busca, G., 2014. Heterogeneous catalytic materials.
- Du, M., Guo, B. and Jia, D., 2010. Newly emerging applications of halloysite nanotubes: a review. Polymer International, 59(5), pp.574-582. [CrossRef]
- Trovarelli, A. and Fornasiero, P., 2013. catalysis. by ceria and related materials London, (12), pp.1-888. [CrossRef]
- Winston, D. and Ariaratnam, S.T., 2022. Navigating Federal and State HDD Guidelines for Interstate Natural Gas Pipeline Crossings. Journal of Pipeline Systems Engineering and Practice, 13(2), p.04022008. [CrossRef]
- Babich, I.V. and Moulijn, J.A., 2003. Science and technology of novel processes for deep desulfurization of oil refinery streams: a review☆. Fuel, 82(6), pp.607-631. [CrossRef]
- Sing, K.S.W., 1967. Adsorption, surface area, and porosity. Academic press.
- Rouquerol, J., Rouquerol, F., Llewellyn, P., Maurin, G. and Sing, K., 2013. Adsorption by powders and porous solids: principles, methodology and applications. Academic press.
- Liu, X., Khinast, J.G. and Glasser, B.J., 2008. A parametric investigation of impregnation and drying of supported catalysts. Chemical Engineering Science, 63(18), pp.4517-4530. [CrossRef]
- Munnik, P., De Jongh, P.E. and De Jong, K.P., 2015. Recent developments in the synthesis of supported catalysts. Chemical reviews, 115(14), pp.6687-6718. [CrossRef]
- Yang, R.T., 2003. Adsorbents: fundamentals and applications. John Wiley & Sons.
- Zuniga, G.M., 2023. Developing Catalysts for the Removal of Methyl Mercaptan from Natural Gas. University of Louisiana at Lafayette.
- Yuan, P., Southon, P.D., Liu, Z., Green, M.E., Hook, J.M., Antill, S.J. and Kepert, C.J., 2008. Functionalization of halloysite clay nanotubes by grafting with γ-aminopropyltriethoxysilane. The Journal of Physical Chemistry C, 112(40), pp.15742-15751. [CrossRef]
- Frost, R.L. and Vassallo, A.M., 1996. The dehydroxylation of the kaolinite clay minerals using infrared emission spectroscopy. Clays and Clay minerals, 44(5), pp.635-651. [CrossRef]
- Vallarino, L.M., 1991. Macrocyclic complexes of the lanthanide (III), yttrium (III) and dioxouranium (VI) ions from metal-templated syntheses. Handbook on the physics and chemistry of rare earths, 15, pp.443-512.
- Farmer, V.C., 1974. The Infrared Spectra of Minerals. Mineralogical society monograph, 4, pp.331-363.
- Mehrabadi, B.A., Eskandari, S., Khan, U., White, R.D. and Regalbuto, J.R., 2017. A review of preparation methods for supported metal catalysts. Advances in catalysis, 61, pp.1-35.
- Arenas, L.T., Simm, C.W., Gushikem, Y., Dias, S.L., Moro, C.C., Costa, T.M. and Benvenutti, E.V., 2007. Synthesis of silica xerogels with high surface area using acetic acid as catalyst. Journal of the Brazilian Chemical Society, 18(5), pp.886-890. [CrossRef]
- Yuan, P., Tan, D. and Annabi-Bergaya, F., 2015. Properties and applications of halloysite nanotubes: recent research advances and future prospects. Applied Clay Science, 112, pp.75-93. [CrossRef]
- Theng, B.K., 2024. The chemistry of clay-organic reactions. CRC Press.
- Goldstein, J.I., Newbury, D.E., Michael, J.R., Ritchie, N.W., Scott, J.H.J. and Joy, D.C., 2017. Scanning electron microscopy and X-ray microanalysis. springer.
- Bartholomew, C.H. and Farrauto, R.J., 2011. Fundamentals of industrial catalytic processes. John Wiley & Sons.
- Cavani, F., Trifiro, F. and Vaccari, A.J.C.T., 1991. Hydrotalcite-type anionic clays: Preparation, properties and applications. Catalysis today, 11(2), pp.173-301. [CrossRef]
- Westmoreland, P.R. and Harrison, D.P., 1976. Evaluation of candidate solids for high-temperature desulfurization of low-Btu gases. Environmental Science & Technology, 10(7), pp.659-661. [CrossRef]
- Flytzani-Stephanopoulos, M., Gavalas, G.R., Tamhankar, S.S. and Sharma, P.K., 1984. Novel sorbents for high temperature regenerative H2S removal. United States Department of Energy.
- Biesinger, M.C., Payne, B.P., Grosvenor, A.P., Lau, L.W., Gerson, A.R. and Smart, R.S.C., 2011. Resolving surface chemical states in XPS analysis of first row transition metals, oxides and hydroxides: Cr, Mn, Fe, Co and Ni. Applied Surface Science, 257(7), pp.2717-2730. [CrossRef]
- Di Castro, V. and Polzonetti, G., 1989. XPS study of MnO oxidation. Journal of Electron Spectroscopy and Related Phenomena, 48(1), pp.117-123. [CrossRef]
- Ilton, E.S., Post, J.E., Heaney, P.J., Ling, F.T. and Kerisit, S.N., 2016. XPS determination of Mn oxidation states in Mn (hydr) oxides. Applied Surface Science, 366, pp.475-485. [CrossRef]
- Poulston, S., Parlett, P.M., Stone, P. and Bowker, M., 1996. Surface oxidation and reduction of CuO and Cu2O studied using XPS and XAES. Surface and Interface Analysis: An International Journal devoted to the development and application of techniques for the analysis of surfaces, interfaces and thin films, 24(12), pp.811-820.
- Van der Heide, P., 2011. X-ray photoelectron spectroscopy: an introduction to principles and practices. John Wiley & Sons.
- Grosvenor, A.P., Biesinger, M.C., Smart, R.S.C. and McIntyre, N.S., 2006. New interpretations of XPS spectra of nickel metal and oxides. Surface science, 600(9), pp.1771-1779. [CrossRef]
- Wang, P., Zhang, S., Wang, Z., Mo, Y., Luo, X., Yang, F., Lv, M., Li, Z. and Liu, X., 2023. Manganese-based oxide electrocatalysts for the oxygen evolution reaction: a review. Journal of Materials Chemistry A, 11(11), pp.5476-5494. [CrossRef]
- Li, S., Zhang, B., Yang, Y., Zhu, F., Zhao, D., Shi, S., Wang, S., Ding, S. and Chen, C., 2024. Insights into the Acidic Site in Manganese Oxide in Terms of the Sulfur and Water Tolerance of Low-Temperature NH3 Selective Catalytic Reduction. Langmuir, 40(28), pp.14504-14514. [CrossRef]

| Filtration Method | Evaporation Method | |||||
|---|---|---|---|---|---|---|
| Catalyst | Surface Area Cm2/g | Pore Volume cm3/g | Pore Size Å | Surface Area cm2/g | Pore Volume cm3/g | Pore Size Å |
| Manganese | 10.833 | 0.091 | 278.592 | 43.308 | 0.223 | 173.019 |
| Copper | 9.651 | 0.076 | 267.470 | 18.292 | 0.125 | 239.067 |
| Zinc | 38.769 | 0.213 | 187.902 | 14.034 | 0.096 | 274.758 |
| Nickel | 40.218 | 0.201 | 165.525 | 23.405 | 0.118 | 222.430 |
| Composite | 21.783 | 0.096 | 172.030 | 17.717 | 0.133 | 241.840 |
| Catalyst | Breakthrough Time (minutes) | % change | Sulfur Capacity, q (mg S/g) |
| Commercial | 1200 | 0% | 16848 |
| Mn | 1410 | 17.5% | 19796.4 |
| Cu | 1350 | 12.5% | 18954 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




