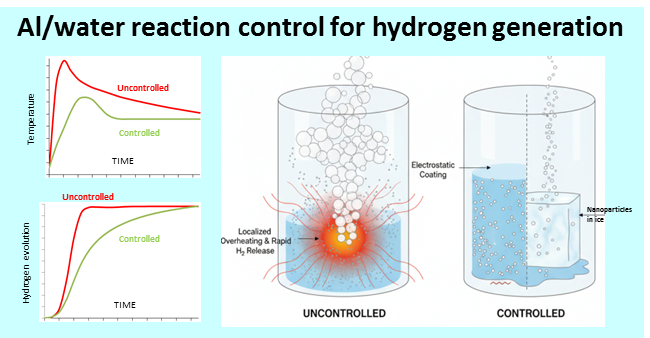
The reaction of aluminum with water is a promising method for producing hydrogen on-demand for autonomous energy systems. However, its practical implementation faces the challenge of process control due to high exothermicity, leading to particle sintering and thermal instability, especially when using highly reactive nanopowders. The goal of this study is to implement an integrated approach to controlling this reaction, aimed at minimizing these risks. The approach is based on the principle of spatial and temporal distribution of reactants to ensure uniform heat release. Two process management methods were investigated: electrostatic application of aluminum powder to the reactor walls with its gradual release and pre-treatment of a nanopowder-ice mixture. Using a macrokinetic mathematical model, calculations of the conversion kinetics and heat release were performed and compared with experimental data. The results showed that both methods prevent slurry self-heating and achieve uniform hydrogen generation at a constant rate. In particular, the use of a pre-frozen mixture ensured stable hydrogen production over a long period of time without additional heating or stirring. The proposed approaches can be used in the design of safe and efficient hydrogen generators for autonomous power plants.