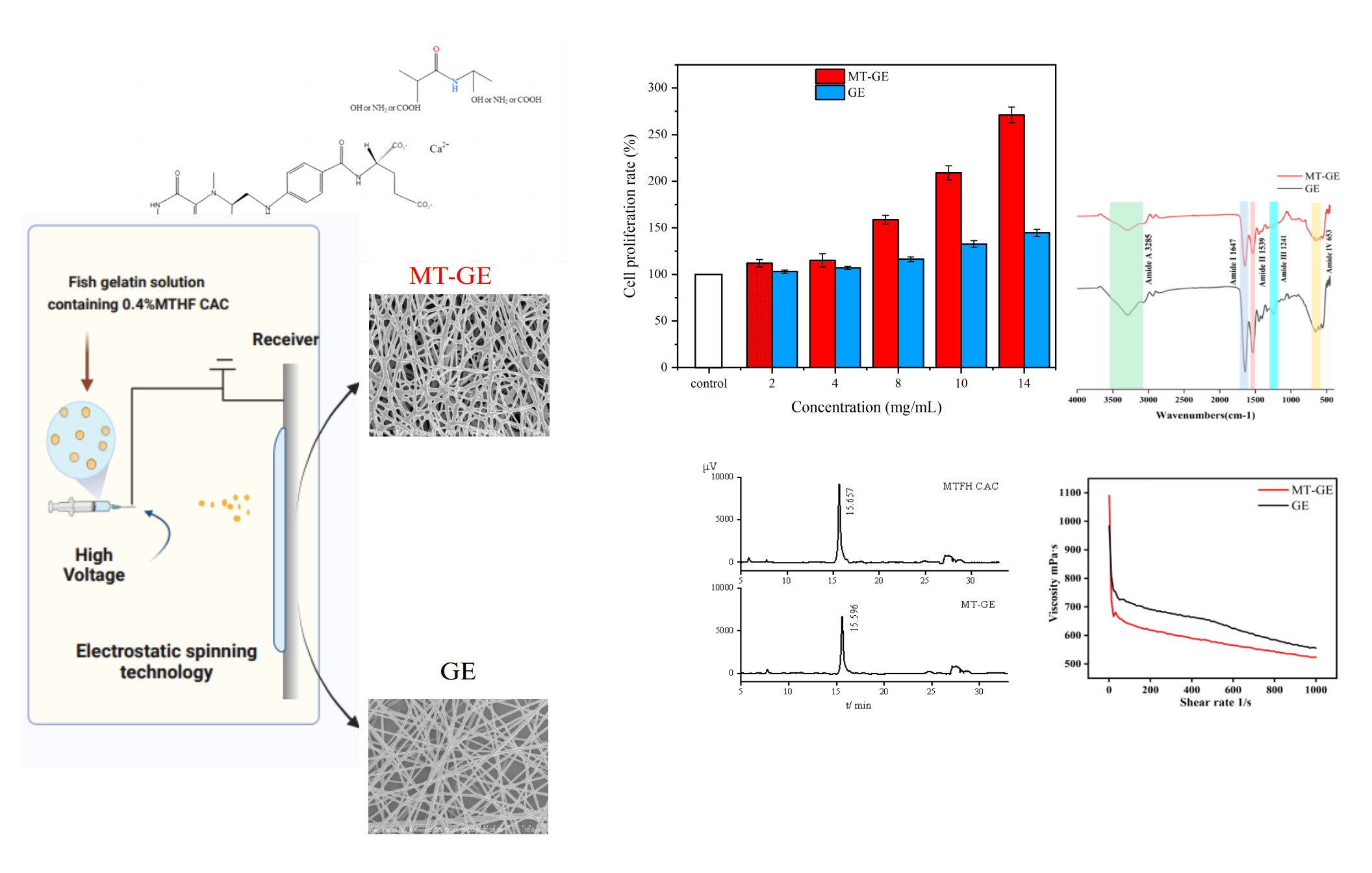
Folate is an essential vitamin associated with protein and DNA synthesis in the body. Compared with synthetic folic acid, 6S-5-methyltetrahydrofolate calcium salt crystal form C (MTHF CAC) is safer and has a higher bioavailability. In this study, a nanofiber membrane (MT-GE) was prepared from fish gelatin and MTHF CAC in the aqueous system via electrospinning. The differential scanning calorimetry results show the higher thermal stability of MT-GE than GE. The weight loss curve of MT-GE detected by thermogravimetric analysis was higher than that of GE. X-ray diffraction indicated the slightly higher crystalline strength of MT-GE than GE. Therefore, the inclusion of MTHF CAC improved the physical characteristics of GE nanofibers. High-performance liquid chromatography analysis revealed that the retention of MTHF CAC in MT-GE reached 85.57%, which suggests that electrospinning caused no effect on the properties of MTHF CAC. The MT-GE membrane supported cell proliferation, and the Cell Counting Kit-8 results indicate that the cell proliferation rate exceeded 100%, with the MT-GE solution demonstrating more than double the proliferation rate of the control group. Therefore, MT-GE has great potential for use as medical biomaterial.