Submitted:
02 April 2026
Posted:
07 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Description of the Model
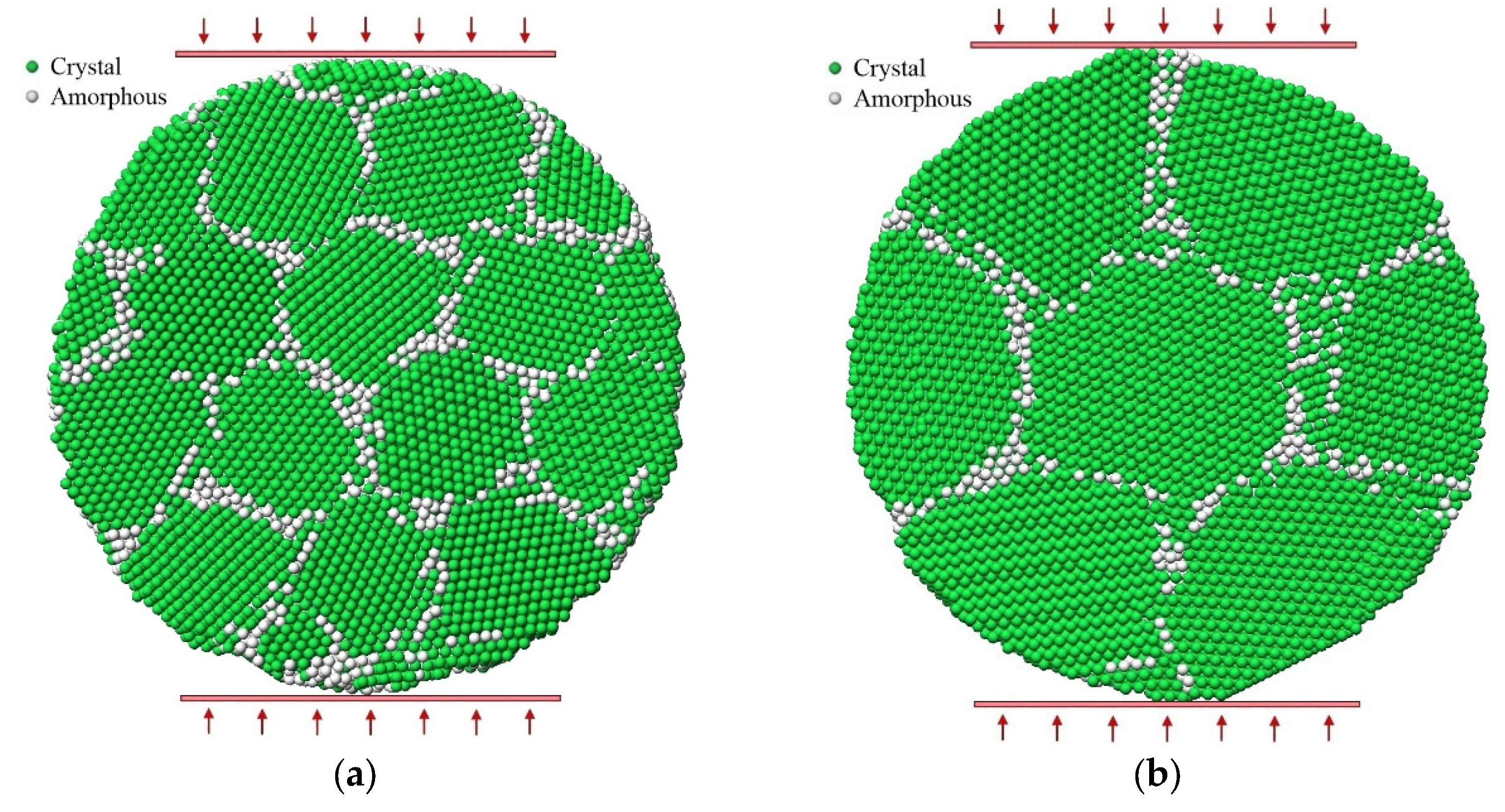
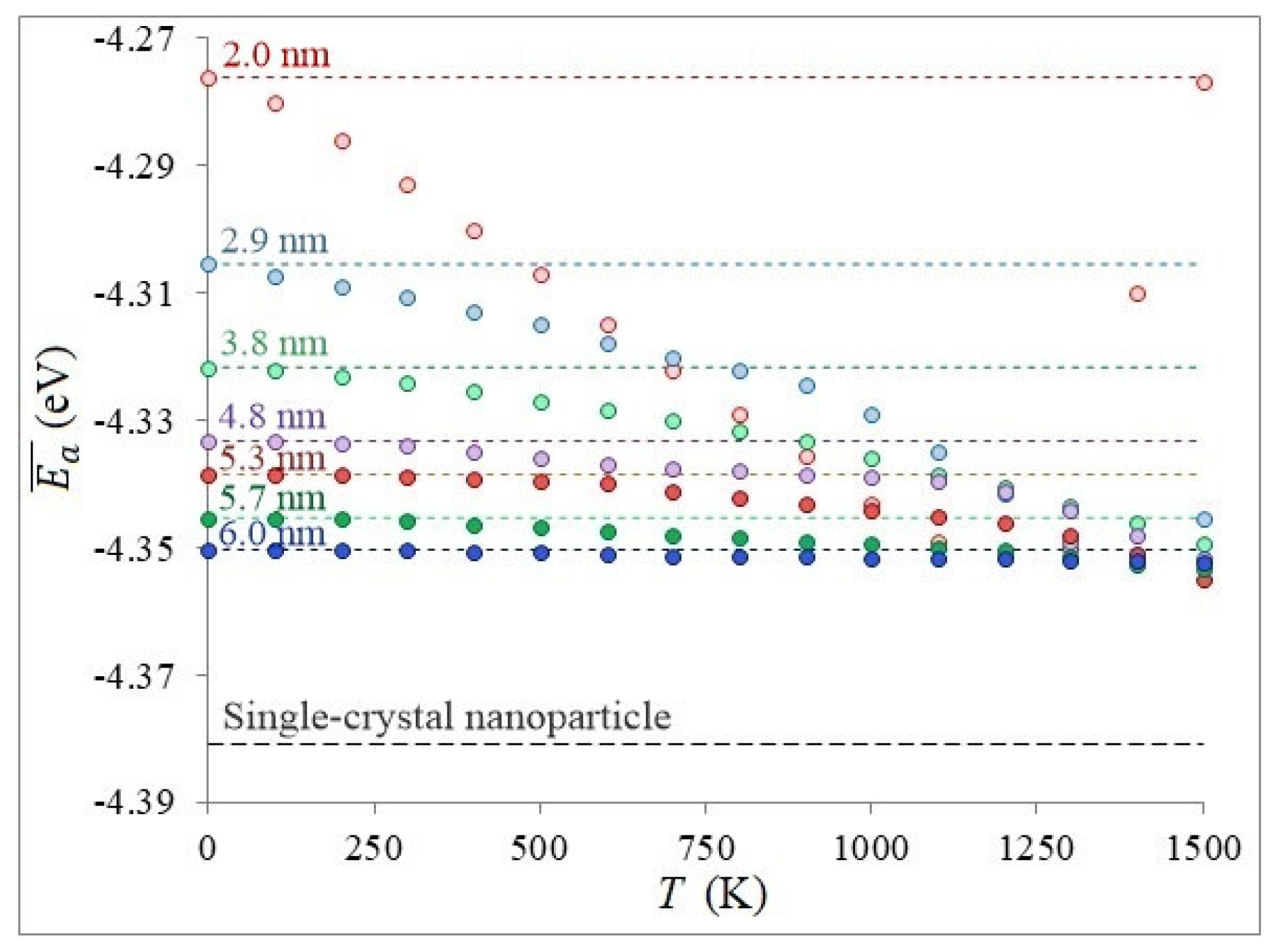
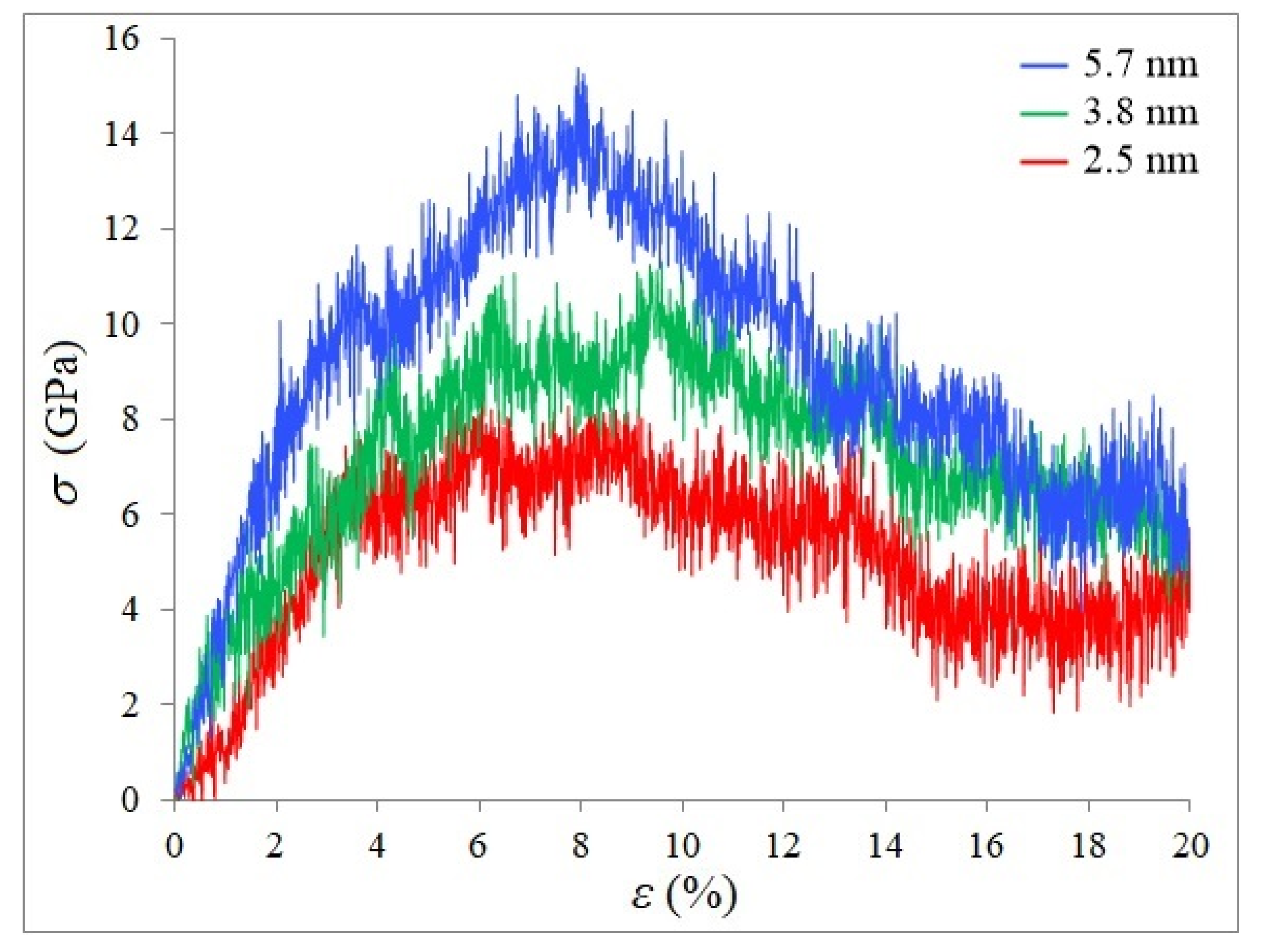
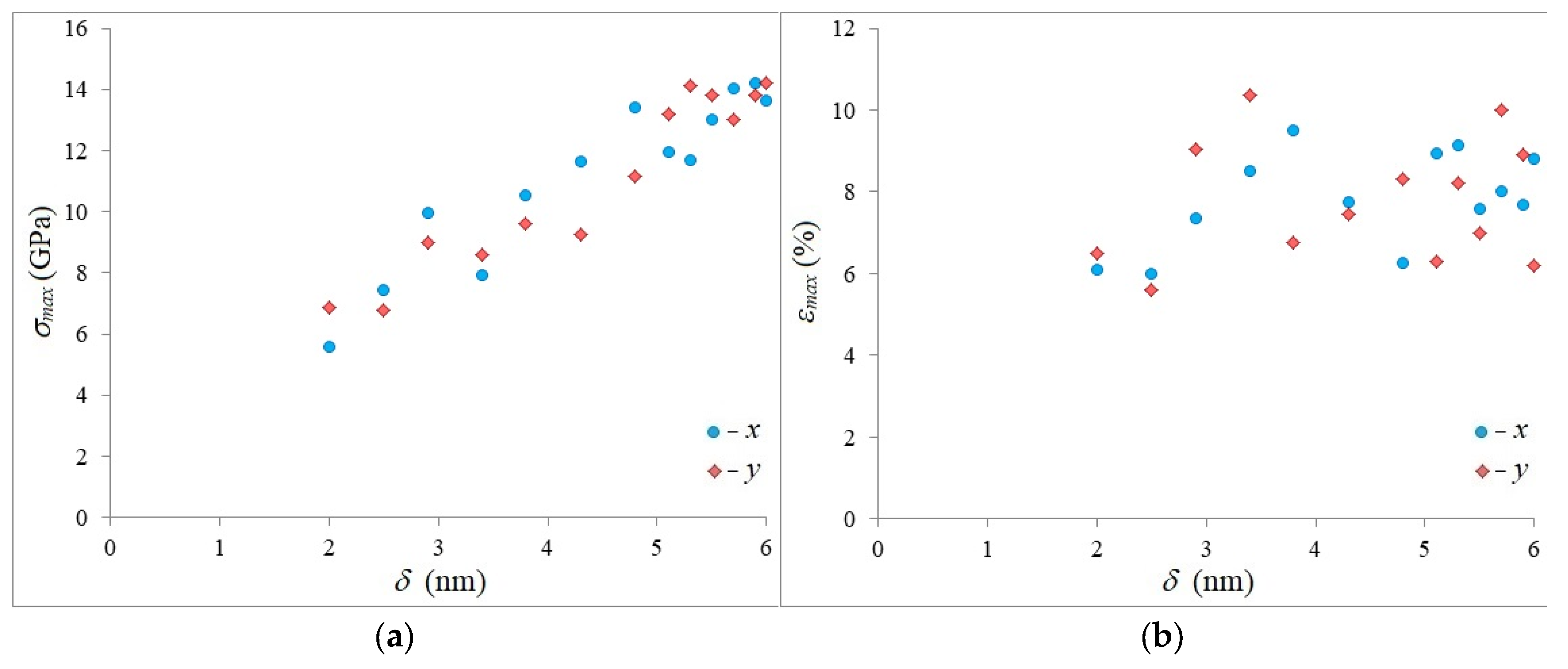
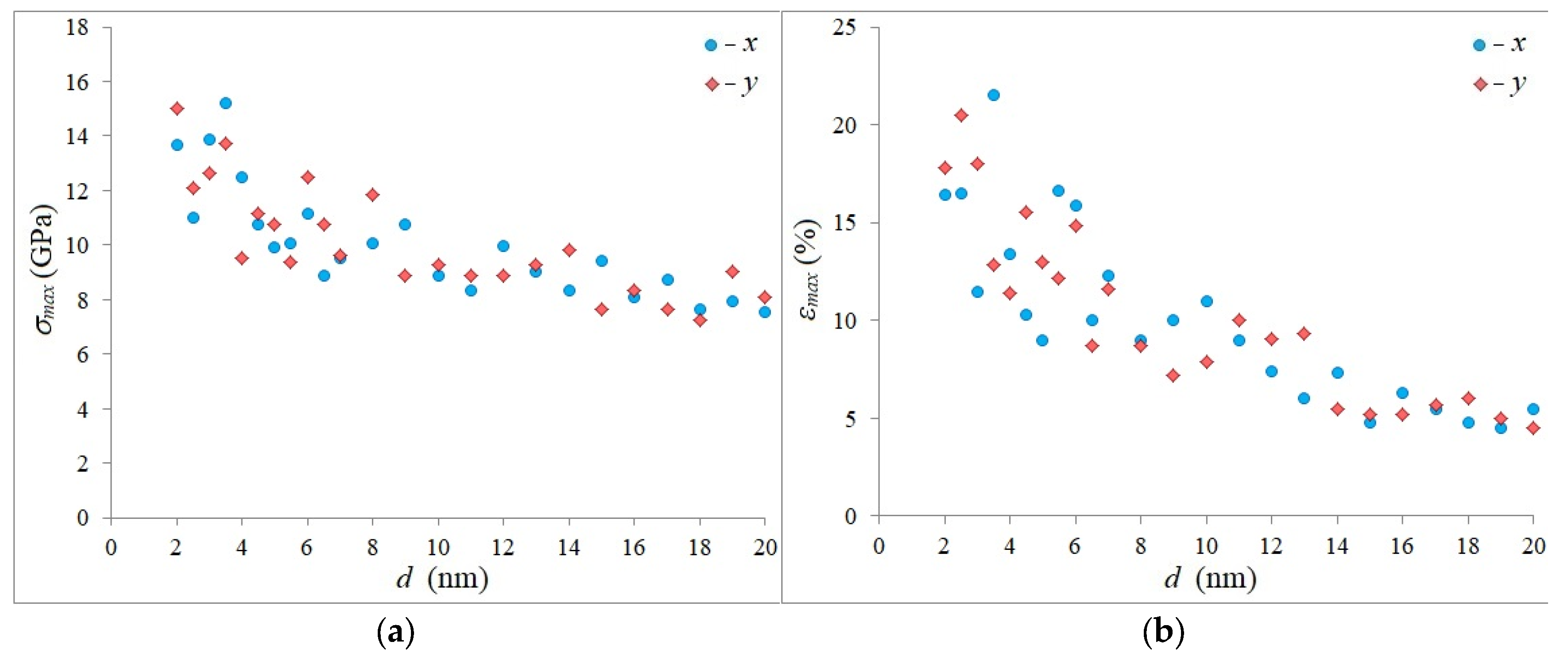
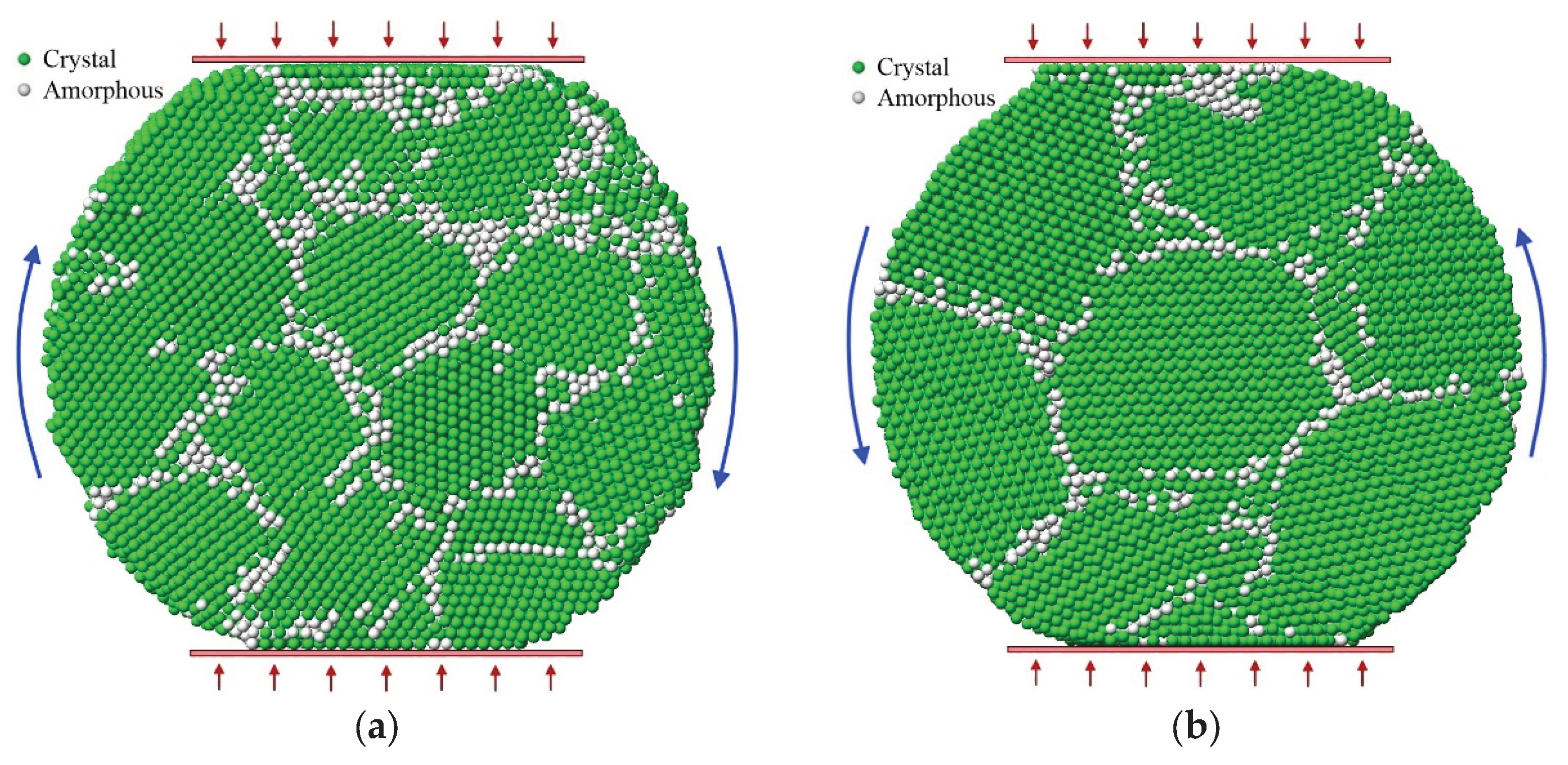
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Dekker Encyclopedia of Nanoscience and Nanotechnology, 3rd ed; Schwarz, J.A., Lyshevski, S.E., Contescu, C.I., Eds.; CRC Press: Boca Raton, USA, 2014; 4200p. [Google Scholar]
- Humbert, C.; Noblet, T.; Dalstein, L.; Busson, B.; Barbillon, G. Sum-frequency generation spectroscopy of plasmonic nanomaterials: a review. Materials 2019, 12, 836. [Google Scholar] [CrossRef] [PubMed]
- Mantri, Y.; Jokerst, J.V. Engineering plasmonic nanoparticles for enhanced photoacoustic imaging. ACS Nano 2020, 14, 9408‒9422. [Google Scholar] [CrossRef]
- Kodama, K.; Nagai, T.; Kuwaki, A.; Jinnouchi, R.; Morimoto, Y. Challenges in applying highly active Pt-based nanostructured catalysts for oxygen reduction reactions to fuel cell vehicles. Nat. Nanotechnol. 2021, 16, 140‒147. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, S.; Qin, R.; Zheng, N.; Perez-Ramirez, J. Nanoscale engineering of catalytic materials for sustainable technologies. Nat. Nanotechnol. 2021, 16, 129‒139. [Google Scholar] [CrossRef]
- Jain, T.K.; Morales, M.A.; Sahoo, S.K.; Leslie-Pelecky, D.L.; Labhasetwar, V. Iron oxide nanoparticles for sustained delivery of anticancer agents. Mol. Pharm. 2005, 2, 194‒205. [Google Scholar] [CrossRef]
- Shim, S.Y.; Lim, D.K.; Nam, J.M. Ultrasensitive optical biodiagnostic methods using metallic nanoparticles. Nanomedicine 2008, 3, 215‒232. [Google Scholar] [CrossRef]
- Carlton, C.E.; Ferreira, P.J. In situ TEM nanoindentation of nanoparticles. Micron 2012, 43, 1134‒1139. [Google Scholar] [CrossRef]
- Deneen, J.; Mook, W.M.; Minor, A.; Gerberich, W.W.; Carter, C.B. In situ deformation of silicon nanospheres. J. Mater. Sci. 2006, 41, 4477‒4483. [Google Scholar] [CrossRef]
- Sharma, A.; Hickman, J.; Gazit, N.; Rabkin, E.; Mishin, Y. Nickel nanoparticles set a new record of strength. Nat. Commun. 2018, 9, 4102. [Google Scholar] [CrossRef]
- Guo, D.; Xie, G.; Luo, J. Mechanical properties of nanoparticles: basics and applications. J. Phys. D Appl. Phys. 2014, 47, 013001. [Google Scholar] [CrossRef]
- Ramos, M.; Ortiz-Jordan, L.; Hurtado-Macias, A.; Flores, S.; Elizalde-Galindo, J.; Rocha, C.; Torres, B.; Zarei-Chaleshtori, M.; Chianelli, R. Hardness and elastic modulus on six-fold symmetry gold nanoparticles. Materials 2013, 6, 198‒205. [Google Scholar] [CrossRef]
- Mordehai, D.; Lee, S.-W.; Backes, B.; Srolovitz, D.J.; Nix, W.D.; Rabkin, E. Size effect in compression of single-crystal gold microparticles. Acta Mater. 2011, 59, 5202‒5215. [Google Scholar] [CrossRef]
- Han, W.-Z.; Huang, L.; Ogata, S.; Kimizuka, H.; Yang, Z.-C.; Weinberger, C.; Li, Q.-J.; Liu, B.-Y.; Zhang, X.-X.; Li, J.; Ma, E.; Shan, Z.-W. From “smaller is stronger” to “size-independent strength plateau”: towards measuring the ideal strength of iron. Adv. Mater. 2015, 27, 3385‒3390. [Google Scholar] [CrossRef]
- Hong, Y.; Zhang, N.; Zaeem, M.A. Metastable phase transformation and deformation twinning induced hardening-stiffening mechanism in compression of silicon nanoparticles. Acta Mater. 2018, 145, 8‒18. [Google Scholar] [CrossRef]
- Amodeo, J.; Pizzagalli, L. Modeling the mechanical properties of nanoparticles: a review. Comptes Rendus. Physique, Plasticity and Solid State Physics 2021, 22, 35‒66. [Google Scholar] [CrossRef]
- Feruz, Y.; Mordehai, D. Towards a universal size-dependent strength of face-centered cubic nanoparticles. Acta Mater. 2016, 103, 433‒441. [Google Scholar] [CrossRef]
- Poletaev, G.; Sannikov, A.; Gafner, Y.; Gafner, S.; Zorya, I. Mechanical properties of crystalline and amorphous nickel nanoparticles: molecular dynamics simulation. J. Nanoparticle Res. 2025, 27, 153. [Google Scholar] [CrossRef]
- Kiener, D.; Minor, A.M. Source truncation and exhaustion: insights from quantitative in situ TEM tensile testing. Nano Lett. 2011, 11, 3816‒3820. [Google Scholar] [CrossRef]
- Mompiou, F.; Legros, M.; Sedlmayr, A.; Gianola, D.S.; Caillard, D.; Kraft, O. Source-based strengthening of sub-micrometer Al fibers. Acta Mater. 2012, 60, 977‒983. [Google Scholar] [CrossRef]
- Dunstan, D.J.; Bushby, A.J. The scaling exponent in the size effect of small scale plastic deformation. Int. J. Plast. 2013, 40, 152‒162. [Google Scholar] [CrossRef]
- Phani, P.S.; Johanns, K.E.; George, E.P.; Pharr, G.M. A simple stochastic model for yielding in specimens with limited number of dislocations. Acta Mater. 2013, 61, 2489‒2499. [Google Scholar] [CrossRef]
- Kilymis, D.; Gerard, C.; Pizzagalli, L. Mechanical properties of amorphous silicon nanoparticles. In TMS 2019 148th Annual Meeting & Exhibition Supplemental Proceedings. The Minerals, Metals & Materials Series; Springer: Cham, 2019; p. pp. 1347‒1354. [Google Scholar] [CrossRef]
- Issa, H.K.; Taherizadeh, A.; Maleki, A. Atomistic-level study of the mechanical behavior of amorphous and crystalline silica nanoparticles. Ceram. Int. 2020, 46, 21647‒21656. [Google Scholar] [CrossRef]
- Zhao, J.; Nagao, S.; Odegard, G.M.; Zhang, Z.; Kristiansen, H.; He, J. Size-dependent mechanical behavior of nanoscale polymer particles through coarse-grained molecular dynamics simulation. Nanoscale Res. Lett. 2013, 8, 541. [Google Scholar] [CrossRef] [PubMed]
- Beaber, A.; Nowak, J.; Ugurlu, O.; Mook, W.; Girshick, S.; Ballarini, R.; Gerberich, W. Smaller is tougher. Phil. Mag. 2011, 91, 1179‒1189. [Google Scholar] [CrossRef]
- Sun, J.; He, L.; Lo, Y.C.; Xu, T.; Bi, H.; Sun, L.; Zhang, Z.; Mao, S.X.; Li, J. Liquid-like pseudoelasticity of sub-10-nm crystalline silver particles. Nat. Mater. 2014, 13, 1007‒1012. [Google Scholar] [CrossRef]
- Issa, I.; Amodeo, J.; Rethore, J.; Joly-Pottuz, L.; Esnouf, C.; Morthomas, J.; Perez, M.; Chevalier, J.; Masenelli-Varlot, K. In situ investigation of MgO nanocube deformation at room temperature. Acta Mater. 2015, 86, 295‒304. [Google Scholar] [CrossRef]
- Mook, W.; Nowak, J.; Perrey, C.; Carter, C.; Mukherjee, R.; Girshick, S.; McMurry, P.; Gerberich, W. Compressive stress effects on nanoparticle modulus and fracture. Phys. Rev. B 2007, 75, 214112. [Google Scholar] [CrossRef]
- Gerberich, W.W.; Stauffer, D.D.; Beaber, A.R.; Tymiak, N.I. A brittleness transition in silicon due to scale. J. Mater. Res. 2012, 27, 552‒561. [Google Scholar] [CrossRef]
- Liang, S.-X.; Zhang, L.-C.; Reichenberger, S.; Barcikowski, S. Design and perspective of amorphous metal nanoparticles from laser synthesis and processing. Phys. Chem. Chem. Phys. 2021, 23, 11121‒11154. [Google Scholar] [CrossRef]
- Sun, J.; Sinha, S.K.; Khammari, A.; Picher, M.; Terrones, M.; Banhart, F. The amorphization of metal nanoparticles in graphitic shells under laser pulses. Carbon 2020, 161, 495‒501. [Google Scholar] [CrossRef]
- He, D.S.; Huang, Y.; Myers, B.D.; Isheim, D.; Fan, X.; Xia, G.-J.; Deng, Y.; Xie, L.; Han, S.; Qiu, Y.; Wang, Y.-G.; Luan, J.; Jiao, Z.; Huang, L.; Dravid, V.P.; He, J. Single-element amorphous palladium nanoparticles formed via phase separation. Nano Res. 2022, 15, 5575–5580. [Google Scholar] [CrossRef]
- Qian, Y.; Silva, A.; Yu, E.; Anderson, C.L.; Liu, Y.; Theis, W.; Ercius, P.; Xu, T. Crystallization of nanoparticles induced by precipitation of trace polymeric additives. Nat. Commun. 2021, 12, 2767. [Google Scholar] [CrossRef]
- Poletaev, G.M.; Bebikhov, Y.V.; Semenov, A.S. Molecular dynamics study of the formation of the nanocrystalline structure in nickel nanoparticles during rapid cooling from the melt. Mater. Chem. Phys. 2023, 309, 128358. [Google Scholar] [CrossRef]
- Kumar, K.S.; Van Swygenhoven, H.; Suresh, S. Mechanical behavior of nanocrystalline metals and alloys. Acta Mater. 2003, 51, 5743–5774. [Google Scholar] [CrossRef]
- Meyers, M.A.; Mishra, A.; Benson, D.J. Mechanical properties of nanocrystalline materials. Prog. Mater. Sci. 2006, 51, 427–556. [Google Scholar] [CrossRef]
- Quek, S.S.; Chooi, Zh.H.; Wu, Zh.; Zhang, Y.W.; Srolovitz, D.J. The inverse Hall-Petch relation in nanocrystalline metals: A discrete dislocation dynamics analysis. J. Mech. Phys. Solids. 2016, 88, 252–266. [Google Scholar] [CrossRef]
- Naik, S.N.; Walley, S.M. The Hall-Petch and inverse Hall-Petch relations and the hardness of nanocrystalline metals. J. Mater. Sci. 2020, 55, 2661–2681. [Google Scholar] [CrossRef]
- Purja Pun, G.P.; Mishin, Y. Development of an interatomic potential for the Ni-Al system. Phil. Mag. 2009, 89, 3245‒3267. [Google Scholar] [CrossRef]
- Levchenko, E.V.; Ahmed, T.; Evteev, A.V. Composition dependence of diffusion and thermotransport in Ni-Al melts: a step towards molecular dynamics assisted databases. Acta Mater. 2017, 136, 74‒89. [Google Scholar] [CrossRef]
- Poletaev, G.M.; Bebikhov, Yu.V.; Semenov, A.S.; Starostenkov, M.D. Self-diffusion in melts of Ni-Al and Ti-Al systems: molecular dynamics study. Letters on Materials 2021, 11, 438‒441. [Google Scholar] [CrossRef]
- Poletaev, G.M.; Gafner, Y.Y.; Gafner, S.L. Molecular dynamics study of melting, crystallization and devitrification of nickel nanoparticles. Letters on Materials 2023, 13, 298‒303. [Google Scholar] [CrossRef]
- Bian, J.; Zhang, H.; Niu, X.; Wang, G. Anisotropic deformation in the compressions of single crystalline copper nanoparticles. Crystals 2018, 8, 116. [Google Scholar] [CrossRef]
- Bian, J.J.; Yang, L.; Niu, X.R.; Wang, G.F. Orientation-dependent deformation mechanisms of bcc niobium nanoparticles. Phil. Mag. 2018, 98, 1848‒1864. [Google Scholar] [CrossRef]
- Xiao, S.; Hu, W.; Yang, J. Melting temperature: from nanocrystalline to amorphous phase. J. Chem. Phys. 2006, 125, 184504. [Google Scholar] [CrossRef]
- Noori, Z.; Panjepour, M.; Ahmadian, M. Study of the effect of grain size on melting temperature of Al nanocrystals by molecular dynamics simulation. J. Mater. Res. 2015, 30, 1648‒1660. [Google Scholar] [CrossRef]
- Poletaev, G.M.; Sitnikov, A.A.; Bebikhov, Yu.V.; Semenov, A.S. Influence of mean grain size and excess energy on the melting temperature of nanocrystalline aluminum. Letters on Materials 2025, 15, 77‒83. [Google Scholar] [CrossRef]
- Poletaev, G.; Rakitin, R.; Bebikhov, Y.; Semenov, A. Molecular dynamics study of melting initiation at tilt grain boundaries depending on their misorientation angle in aluminum. Phys. Scr. 2025, 100, 015988. [Google Scholar] [CrossRef]
- Goryaeva, A.M.; Fusco, C.; Bugnet, M.; Amodeo, J. Influence of an amorphous surface layer on the mechanical properties of metallic nanoparticles under compression. Phys. Rev. Mater. 2019, 3, 033606. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



