Submitted:
02 April 2026
Posted:
03 April 2026
You are already at the latest version
Abstract
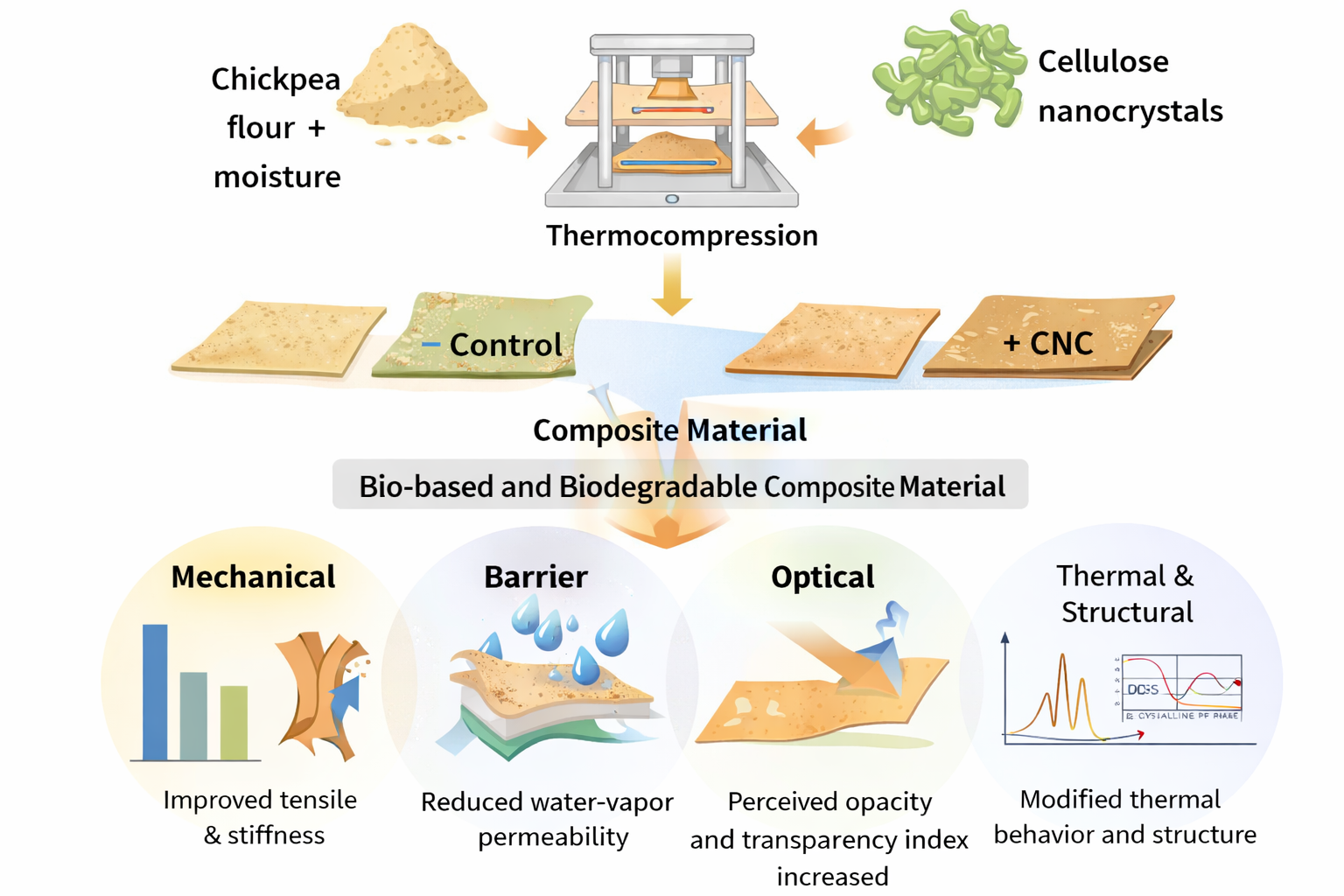
Chickpea (Cicer arietinum L.) flour is a promising raw material for the development of biodegradable packaging due to its protein and polyphenol content. In this study, thermocompressed chickpea flour sheets were reinforced with cellulose nanocrystals (CNC) to improve their barrier, mechanical, thermal, and structural properties. Preliminary trials identified 22% moisture as the most suitable condition for consistent sheet formation. CNC was incorporated at 0, 2.5, 5.0, and 7.5% (w/w), and the resulting sheets were evaluated for phenolic content, antioxidant activity, water vapor permeability (WVP), optical properties, thermal behavior, morphology, and structural characteristics. Thermocompression reduced the measurable phenolic fractions, although antioxidant activity was not significantly affected. CNC markedly reduced WVP, from 5.16x10-10 (control) to 5.93x10-12 g∙m-1∙s-1∙Pa-1 at 7.5% CNC. Tensile strength and Young's modulus increased with CNC loading, while elongation at break was highest at intermediate concentrations. SEM, DSC, XRD, and FTIR analyses indicated matrix reorganization and modified thermo-structural behavior. Overall, CNC improved the barrier and mechanical performance of thermocompressed chickpea flour sheets, supporting their potential for biodegradable packaging applications.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Experimental Design and Formulations
2.3. Preparation of Cellulose Nanocrystals
2.4. Transmission Electron Microscopy (TEM) and Nanoparticle Size Distribution Analysis
2.5. Preparation of Chickpea Flour Sheets by Thermocompression
2.6. Phenolic Compounds and Antioxidant Capacity
2.6.1. Extraction of Phenolic Compounds
2.6.2. Extraction of Bounds Phenolics (Condensed and Hydrolyzable Tannins)
2.6.3. Determination of Phenolic Compounds (Folin-Ciocalteu)
2.6.4. Antioxidant Capacity (DPPH Scavenging)
2.7. Thickness and Conditioning Prior to Testing
2.8. Mechanical Properties
2.9. Water Vapor Permeability
2.10. Color
2.11. Transparency, Opacity
2.12. Thermal Properties, TGA and DSC
2.13. X-ray Diffraction (XRD)
2.14. Scanning Electron Microscopy (SEM)
2.15. FTIR Spectroscopy
2.16. Statistical Analysis
3. Results
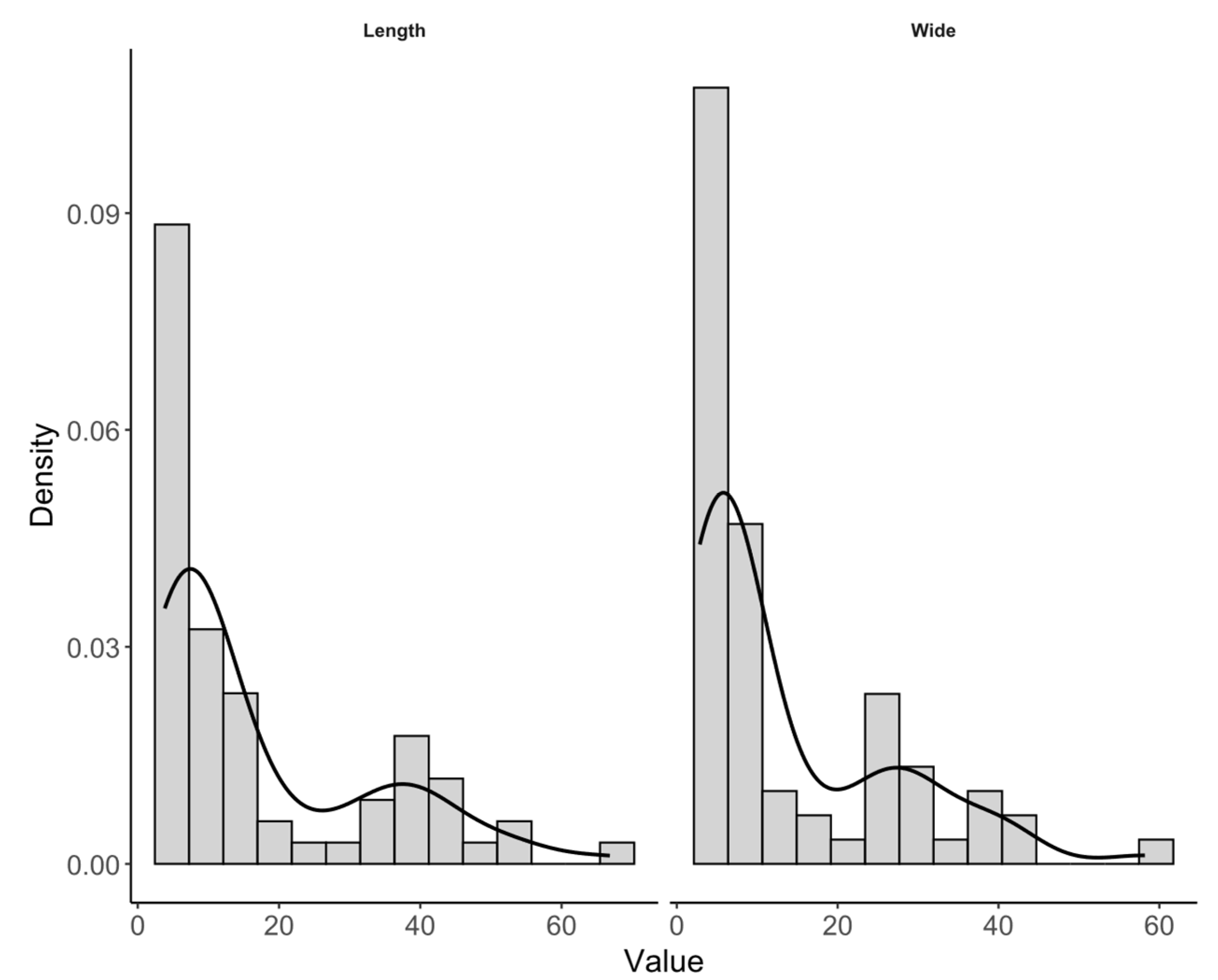
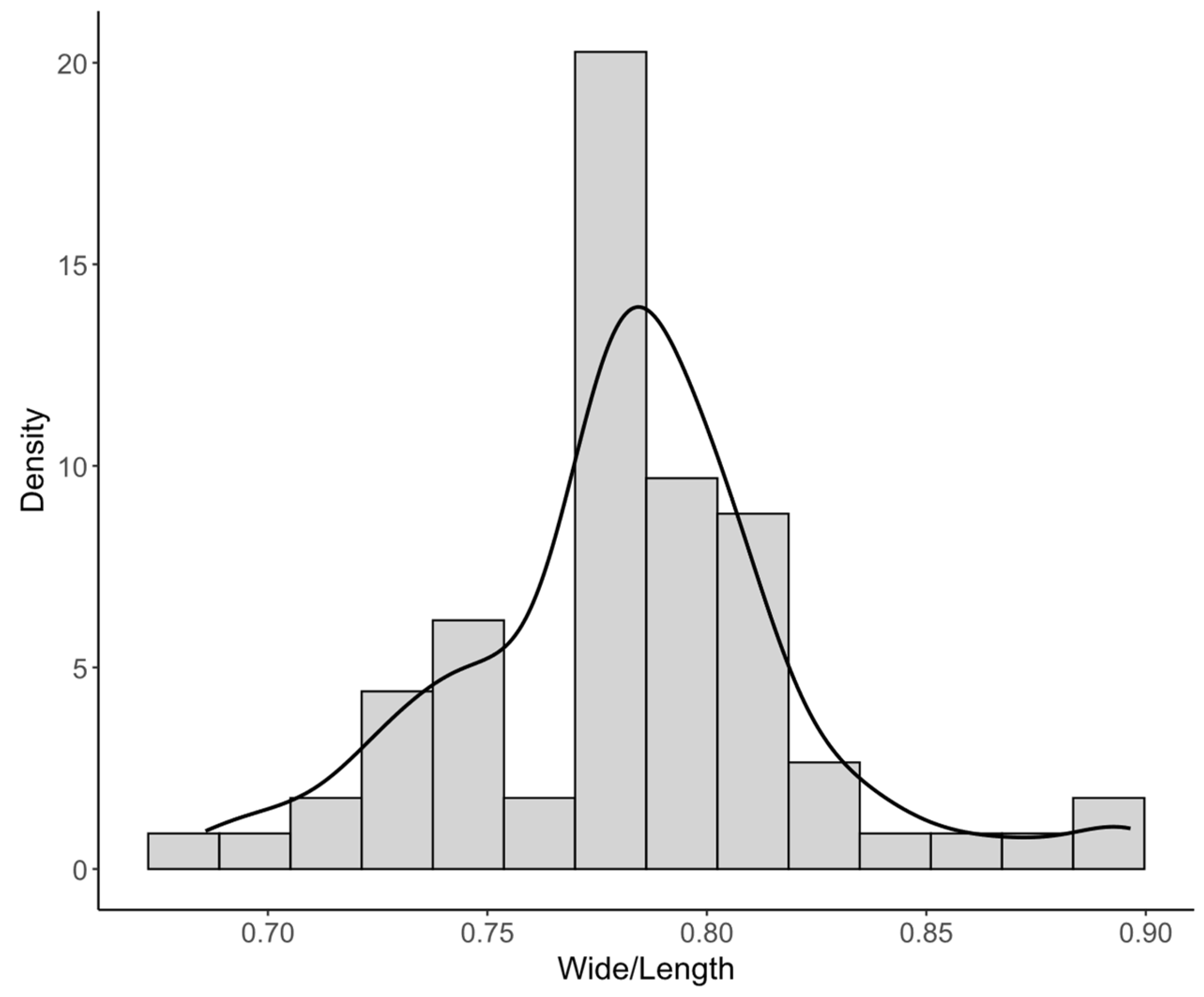
3.1. Characterization of Cellulose Nanocrystals (CNC)
| n | Median (Q1-Q3) (nm) |
Min-Max (nm) |
Mean ± SD (nm) |
CV (%) |
||
| Length | 70 | 8.94 (6.20-29.94) | 3.82-66.83 | 17.23 ±15.72 | 91.24 | |
| Width | 70 | 6.82 (4.92-23.53) | 2.86-58.21 | 13.58 ± 12.72 | 93.62 | |
| Width/Length ratio | 70 | 0.784 (0.763-0.803) | 0.686-0.897 | 0.782 ± 0.040 | 5.07 |
3.2. Phenolic Compounds
3.3. Antioxidant Capacity (DPPH Radical Scavenging)
3.4. Morphology of Thermocompressed Sheets (SEM)
3.5. Mechanical Properties
3.6. Water Vapor Permeability (WVP)
3.7. Optical Properties
3.7.1. Color
3.7.2. Transparency and Opacity
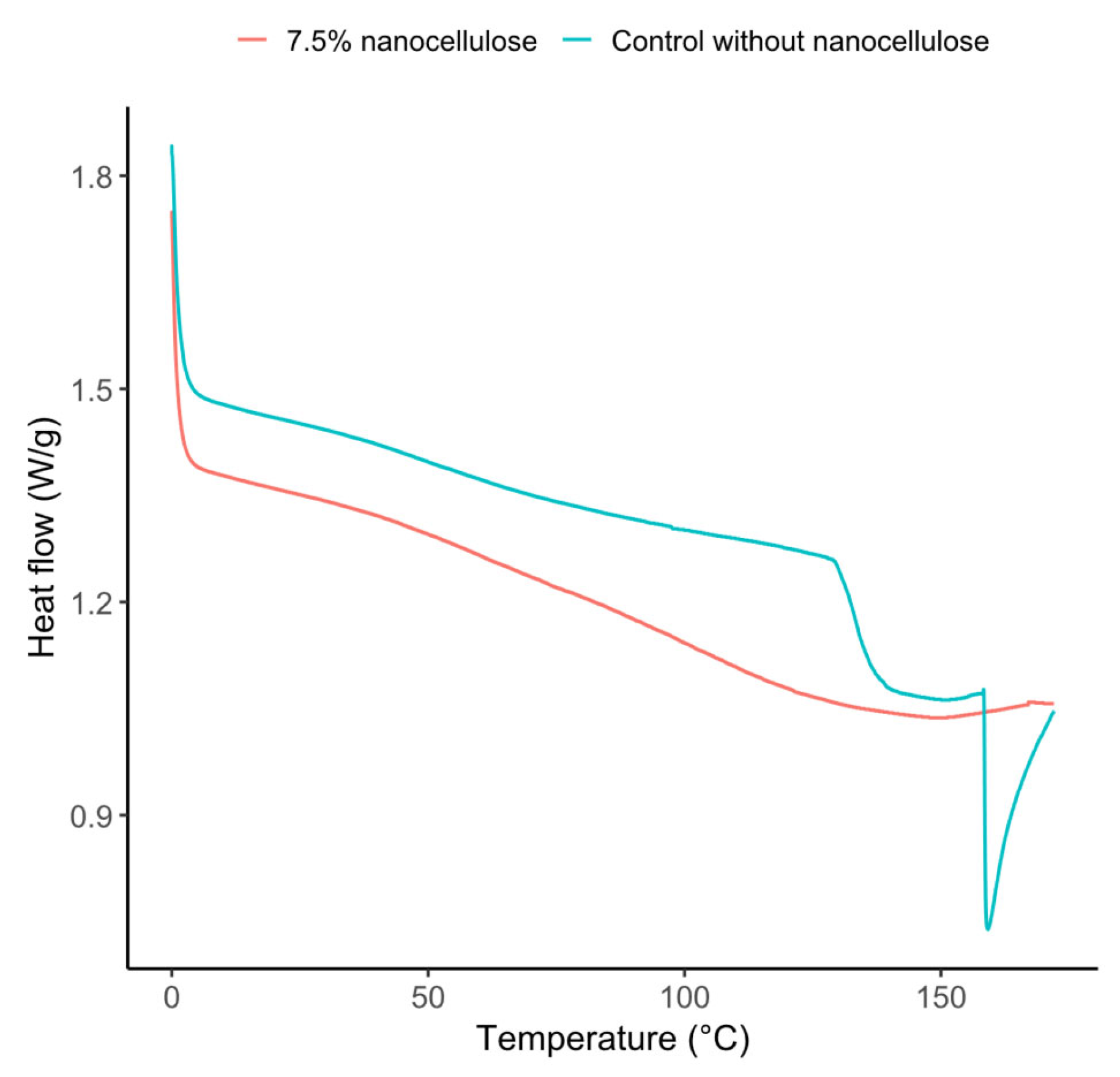
3.8. Thermal Properties
3.9. Structural Characterization
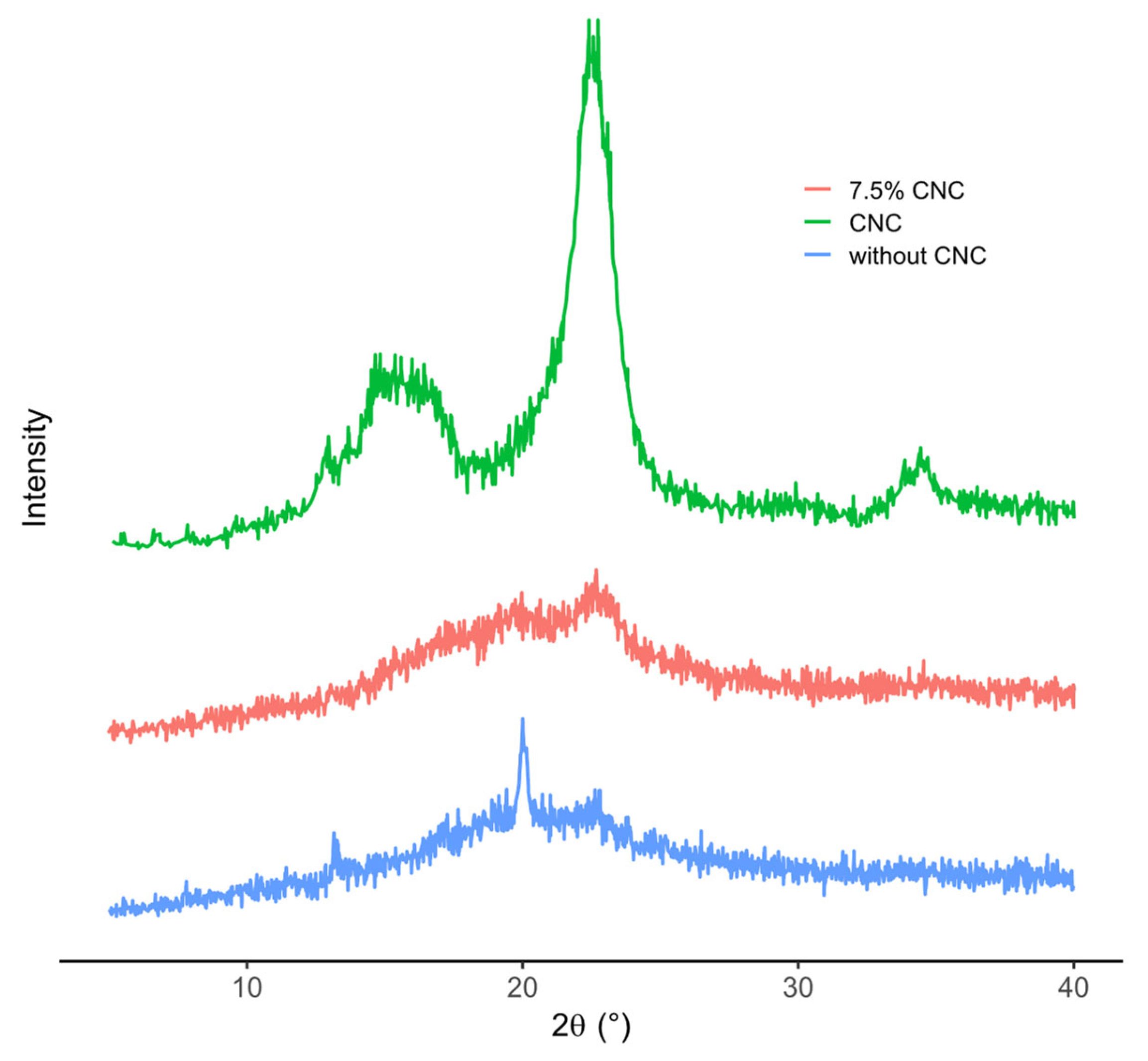
3.9.1. XRD
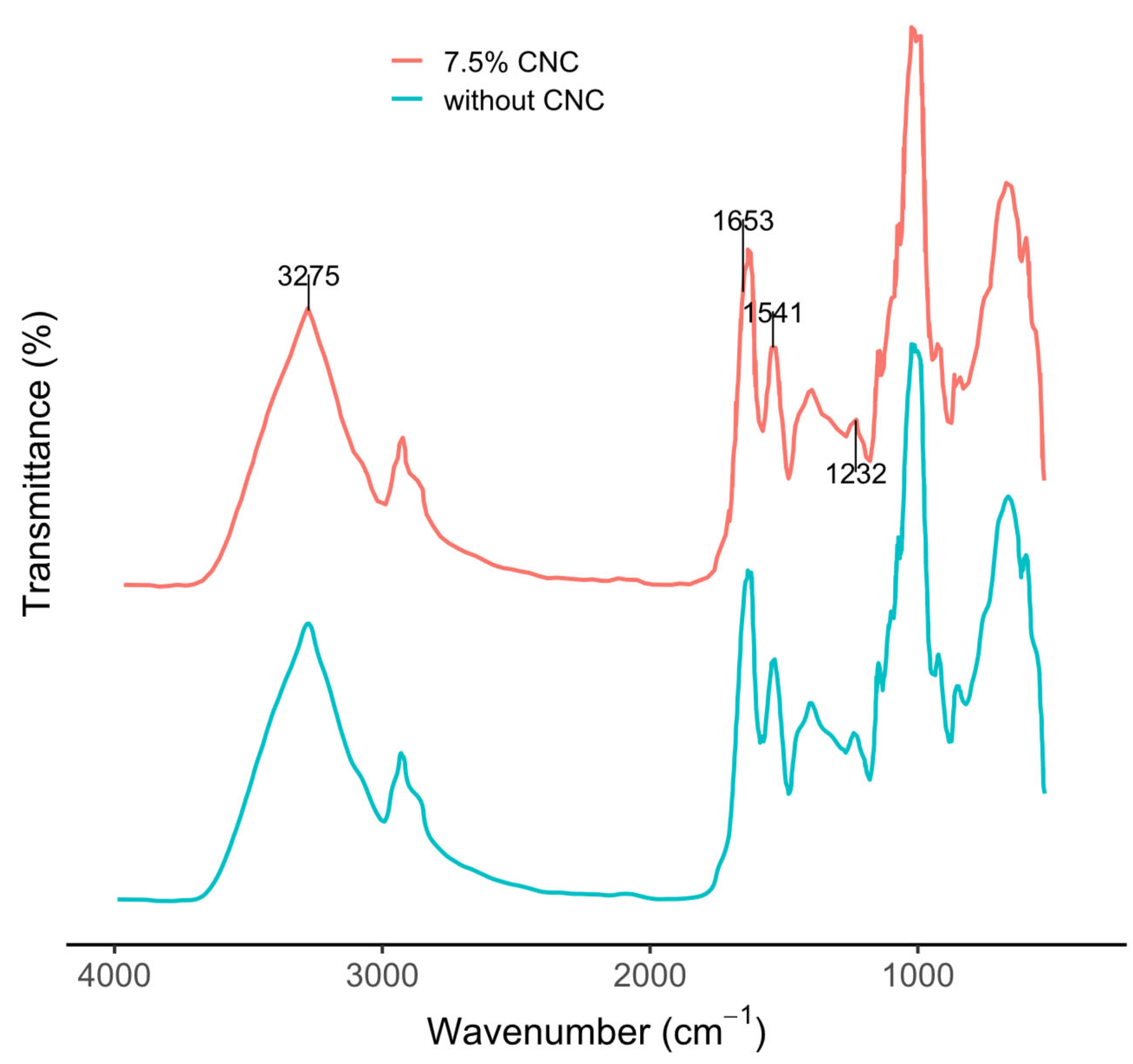
3.9.2. FTIR
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ASTM | American Society for Testing and Materials |
| ATR | Attenuated Total Reflectance |
| CIELAB | Commission International de l’Éclairage L*a*b* color space |
| CNC | Cellulose Nanocristals |
| CS | Control Sheet |
| CSN | CNC-reinforced Sheet |
| CV | Coefficient of Variation |
| DPPH | 2,2-Diphenyl-1-picrylhydrazyl |
| DSC | Differential Scanning Calorimetry |
| DTG | Derivative thermogravimetry |
| EAB | Elongation at Break |
| FTIR | Fourier Transform Infrared Spectroscopy |
| GAE | Galid Acid Equivalents |
| HSD | Honestly Significant Difference |
| MWCO | Molecular Weight Cut-off |
| Q1 | First Quartile |
| Q2 | Third Quartile |
| RH | Relative Humidity |
| RSA | Radical-Scavenging Activity |
| SD | Standard Deviation |
| T5% | Temperature at 5% Weigh Loss |
| T10% | Temperature at 10% Weigh Loss |
| Tmax | Temperature of Maximun Degradation Rate |
| Tonset | Onset Temperature |
| Tpeak | Peak temperature |
| TS | Tensile Strength |
| WVP | Water Vapor Permeability |
| XDR | X-ray Diffraction |
References
- Canpolat, M. Package-Borne Safety Issues in Food. In Food Safety; Nobel Tip Kitabevleri: Istanbul. Turkey, 2024; pp. 321–332.
- Priyanka, P.; Rani, R.; Yadav, R.; Bajal, N.; Mandal, B.S.; Kumar, A. Role of Active Packaging for Food Freshness and Quality Maintenance. International Journal of Environment, Agriculture and Biotechnology 2024, 9, 023–030. [CrossRef]
- Righetti, G.I.C.; Faedi, F.; Famulari, A. Embracing Sustainability: The World of Bio-Based Polymers in a Mini Review. Polymers (Basel). 2024, 16, 950. [CrossRef]
- Schäfer, L. Is There Hope to Switch Traditional Plastics into Sustainable? Science Insights 2025, 47, 1913–1917. [CrossRef]
- Long, J.; Zhang, W.; Zhao, M.; Ruan, C.-Q. The Reduce of Water Vapor Permeability of Polysaccharide-Based Films in Food Packaging: A Comprehensive Review. Carbohydr. Polym. 2023, 121267, 321. [CrossRef]
- Czajkowska–González, Y.A.; Alvarez–Parrilla, E.; del Rocío Martínez–Ruiz, N.; Vázquez–Flores, A.A.; Gaytán–Martínez, M.; de la Rosa, L.A. Addition of Phenolic Compounds to Bread: Antioxidant Benefits and Impact on Food Structure and Sensory Characteristics. Food Production, Processing and Nutrition 2021, 3, 25. [CrossRef]
- Bigne, F.; Romero, A.; Ferrero, C.; Puppo, M.C.; Guerrero, A. New Thermal and Rheological Approaches of Chickpea–Wheat Dough for Breadmaking. European Food Research and Technology 2021, 247, 1107–1115. [CrossRef]
- Soto-Madrid, D.; Pérez, N.; Gutiérrez-Cutiño, M.; Matiacevich, S.; Zúñiga, R.N. Structural and Physicochemical Characterization of Extracted Proteins Fractions from Chickpea (Cicer Arietinum L.) as a Potential Food Ingredient to Replace Ovalbumin in Foams and Emulsions. Polymers (Basel). 2022, 15, 110. [CrossRef]
- Jeya Jeevahan, J.; Chandrasekaran, M.; Venkatesan, S.P.; Sriram, V.; Britto Joseph, G.; Mageshwaran, G.; Durairaj, R.B. Scaling up Difficulties and Commercial Aspects of Edible Films for Food Packaging: A Review. Trends Food Sci. Technol. 2020, 100, 210–222. [CrossRef]
- Leya, B.; Franklin, R.S.; Pragalyaashree, M.M.; Monicka, A.A.; Tiroutchelvame, D.; Blessy, C.; Blessie, R.F. Biopolymer-Based Edible Packaging: A Critical Review on the Biomaterials, Formation, and Applications on Food Products. J. Appl. Biol. Biotechnol. 2024. [CrossRef]
- De Micheli, C.; Navarini, F.; Roncoroni, V. Process for the Manufacture of Totally Bio-Decomposable Films with High Mechanical Characteristics and Relevant Products and Applications 1995.
- Negrete-Bolagay, D.; Guerrero, V.H. Opportunities and Challenges in the Application of Bioplastics: Perspectives from Formulation, Processing, and Performance. Polymers (Basel). 2024, 16, 2561. [CrossRef]
- Saravacos, G.D.; Karathanos, V.T.; Marousis, S.N. Diffusion of Water in Starch Materials. In; 1992; pp. 329–340.
- Alibekov, R.S.; Urazbayeva, K.U.; Azimov, A.M.; Rozman, A.S.; Hashim, N.; Maringgal, B. Advances in Biodegradable Food Packaging Using Wheat-Based Materials: Fabrications and Innovations, Applications, Potentials, and Challenges. Foods 2024, 13, 2964. [CrossRef]
- Sapkota, J.; Jorfi, M.; Weder, C.; Foster, E.J. Reinforcing Poly(Ethylene) with Cellulose Nanocrystals. Macromol. Rapid Commun. 2014, 35, 1747–1753. [CrossRef]
- Hung, Y.-J.; Chiang, M.-Y.; Wang, E.-T.; Wu, T.-M. Synthesis, Characterization, and Physical Properties of Maleic Acid-Grafted Poly(Butylene Adipate-Co-Terephthalate)/Cellulose Nanocrystal Composites. Polymers (Basel). 2022, 14, 2742. [CrossRef]
- Duraccio, D.; Arrigo, R.; Strongone, V.; Capra, P.P.; Malucelli, G. Rheological, Mechanical, Thermal and Electrical Properties of UHMWPE/CNC Composites. Cellulose 2021, 28, 10953–10967. [CrossRef]
- Liu, G.-T.; Shi, S.-C.; Rahmadiawan, D. Revolutionizing Polymethyl Methacrylate Toughness: Achieving 190% Improvement with Nanocellulose Reinforcement While Maintaining Optical Clarity. Jurnal Pendidikan Teknologi Kejuruan 2024, 7, 229–237. [CrossRef]
- Le Duigou, A.; Deux, J.-M.; Davies, P.; Baley, C. PLLA/Flax Mat/Balsa Bio-Sandwich Manufacture and Mechanical Properties. Applied Composite Materials 2011, 18, 421–438. [CrossRef]
- Åkesson, D.; Skrifvars, M.; Hagström, B.; Walkenström, P.; Seppälä, J. Processing of Structural Composites from Biobased Thermoset Resins and Natural Fibres by Compression Moulding. J. Biobased Mater. Bioenergy 2009, 3, 215–225. [CrossRef]
- Lapuz, A.; Tsuchikawa, S.; Inagaki, T.; Ma, T.; Migo, V. Production of Nanocellulose Film from Abaca Fibers. Crystals (Basel). 2022, 12, 601. [CrossRef]
- R Core Team R: A Language and Enviromental for Statistical Computing 2024.
- Wilkinson, L. Ggplot2: Elegant Graphics for Data Analysis by WICKHAM, H. Biometrics 2011, 67. [CrossRef]
- Pérez-Jiménez, J.; Saura-Calixto, F. Literature Data May Underestimate the Actual Antioxidant Capacity of Cereals. J. Agric. Food Chem. 2005, 53, 5036–5040. [CrossRef]
- Makkar, H.P.S.; Gamble, G.; Becker, K. Limitation of the Butanol–Hydrochloric Acid–Iron Assay for Bound Condensed Tannins. Food Chem. 1999, 66, 129–133. [CrossRef]
- Hartzfeld, P.W.; Forkner, R.; Hunter, M.D.; Hagerman, A.E. Determination of Hydrolyzable Tannins (Gallotannins and Ellagitannins) after Reaction with Potassium Iodate. J. Agric. Food Chem. 2002, 50, 1785–1790. [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. [14] Analysis of Total Phenols and Other Oxidation Substrates and Antioxidants by Means of Folin-Ciocalteu Reagent. In; 1999; pp. 152–178.
- Singleton, V.L.; Rossi, J.A. Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents. Am. J. Enol. Vitic. 1965, 16, 144–158.
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a Free Radical Method to Evaluate Antioxidant Activity. LWT - Food Science and Technology 1995, 28, 25–30. [CrossRef]
- Molyneux, P. The Use of the Stable Free Radical Diphenylpicrylhydrazyl (Dpph) for Estimating Antioxidant Activity. Songklanakarin Journal of Science and Technology (SJST) 2004, 26, 211–219.
- Byun, Y.; Kim, Y.T.; Whiteside, S. Characterization of an Antioxidant Polylactic Acid (PLA) Film Prepared with α-Tocopherol, BHT and Polyethylene Glycol Using Film Cast Extruder. J. Food Eng. 2010, 100, 239–244. [CrossRef]
- ASTM International ASTM D882-02: Standard Test Method for Tensile Properties of Thin Plastic Sheeting. ASTM International 2002.
- ASTM International ASTM E96/E96M-22ae1: Standard Test Methods for Gravimetric Determination of Water Vapor Transmission Rate of Materials; West Conshohocken, PA, USA, 2022.
- Maftoonazad, N.; Badii, F.; Mohamed, A.; Ramaswamy, H. Evaluation of Physicochemical, Thermomechanical, and Structural Properties of Chickpea Flour Composite Films Reinforced with Crystalline Nanocellulose. J. Appl. Polym. Sci. 2020, 137. [CrossRef]
- Rojas-Lema, S.; Nilsson, K.; Trifol, J.; Langton, M.; Gomez-Caturla, J.; Balart, R.; Garcia-Garcia, D.; Moriana, R. “Faba Bean Protein Films Reinforced with Cellulose Nanocrystals as Edible Food Packaging Material.” Food Hydrocoll. 2021, 121, 107019. [CrossRef]


| Flour (mg GAE/ g sample) |
Sheets (mg GAE/ g sample) |
|
| Free Phenol | 0.813 ± 0.116a | 0.556 ± 0.023b |
| Condensed Tannins | 4.197 ± 0.609c | 2.259 ± 0.157d |
| Hydrolyzed Tannins | 1.121 ± 0.086e | 0.969 ± 0.022f |
| Flour (RSA %inhibition) |
Sheets (RSA %inhibition) |
|
| Free Phenol | 22.12 ± 1.85a | 20.80 ± 0.19a |
| Condensed Tannins | 84.48 ± 2.72b | 83.86 ± 1.04b |
| Hydrolyzed Tannins | 76.87 ± 2.43c | 66.31 ± 12.88c |
| CNC (%) | Tensile Strength (MPa) |
Elongation at Break(%) |
Young's Modulus (MPa) |
| 0 | 2.11 ± 0.56a | 6.18 ± 1.84a | 0.88 ± 0.07a |
| 2.5 | 2.67 ± 0.28b | 8.28 ± 1.35b | 0.97 ± 0.14a |
| 5.0 | 2.80 ± 0.27b,c | 9.22 ± 0.74b | 0.94 ± 0.11a |
| 7.5 | 3.10 ± 0.36c | 6.68 ± 1.25a | 1.35 ± 0.16b |
| CNC Concentration (%) |
WVP |
| 0.0 | a |
| 2.5 | b |
| 5.0 | c |
| 7.5 | d |
| CNC (%) | L* | Chroma | Hue |
| 0 | 40.00 ± 0.55a | 23.62 ± 0.12a | 41.81 ± 4.47a |
| 2.5 | 41.16 ± 0.94a | 25.29 ± 1.36a | 48.77 ± 1.59a |
| 5.0 | 52.40 ± 0.72b | 41.41 ± 1.00b | 59.27 ± 1.08b |
| 7.5 | 55.22 ± 1.65c | 44.86 ± 1.35c | 61.10 ± 2.63b |
| CNC (%) | Opacity (AU. mm-1) |
Transparency Index ( mm-1) |
| 0 | 2.21 ± 0.15a | 1.71 ± 0.07a |
| 2.5 | 2.26 ± 0.04a | 1.72 ± 0.02a |
| 5.0 | 3.04 ± 0.08b | 2.22 ± 0.06b |
| 7.5 | 3.15 ± 0.06b | 2.36 ± 0.11c |
| Sample | Event description |
Tonset (°C) |
Tpeak (°C) |
Tend_set (°C) |
Enthalpy (J/g) |
| Control without CNC |
Sharp endothermic | 127.4 | 133.7 | 143.0 | 64.0 |
| 7.5% CNC | Broad and less defined thermal event | 92.0 | 147.9 | 166.4 | 15.7 |
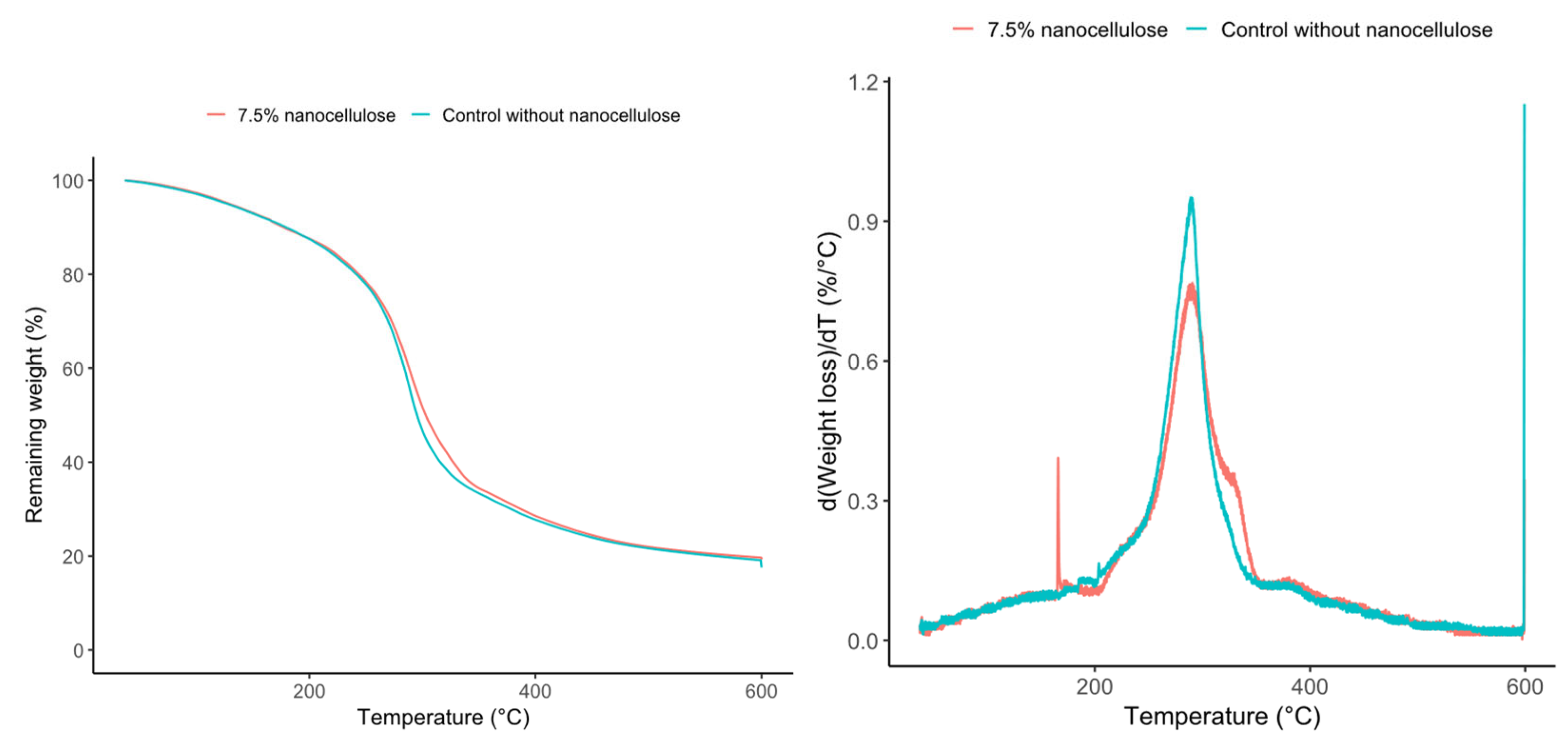
| Sample | T5%(°C) | T10%(°C) | Tonset(°C) | Tmax(°C) | Residual 600 °C (%) |
| Control without nanocellulose | 126 | 180 | 215.0 | 287.0 | 19.48 |
| 7.5% nanocellulose | 126 | 180 | 215.5 | 288.9 | 19.48 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




