Submitted:
01 April 2026
Posted:
03 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
- analyze the chemical composition of cold pressed aluminum briquettes produced from recycled machining chips,
- evaluate their chemical homogeneity after remelting,
- identify the presence of trace elements and potential contaminants,
- assess their suitability for use as deoxidizers in steelmaking.
1.1. State of the Art
2. Materials and Methods
- filling the pressing chamber with loose chips,
- pre compression at a low pressure of approximately 10–20 MPa,
- the main pressing step at 100–160 MPa,
- maintaining the pressure for 3–5 s to stabilize the shape and reduce elastic springback,
- pressure release and ejection of the briquette from the chamber.
- uniform material compaction,
- minimization of internal voids,
- stable briquette geometry,
- reproducibility of production.
- reduced oxidation during heating,
- limited metal loss in the form of fine particulate matter,
- improved stability of chemical composition,
- higher aluminum yield (80–90%, depending on the melting method).
2.1. Sample Preparation
- Sample A – the cold pressed briquette in its original state
- Sample B – the remelted and resolidified block
2.2. Chemical Composition Analysis (XRF)
- FP corrections (Fundamental Parameters),
- automatic deconvolution of overlapping peaks,
- internal calibration models for aluminum alloys.
2.3. Microstructural and Local Chemical Analysis (SEM–EDS)
2.4. Surface Topography and 3D Profilometry
2.5. Density, Porosity and Compaction Analysis
- C < 0.70 – low compaction, high porosity
- 0.70 ≤ C ≤ 0.90 – standard industrial compaction
- C > 0.90 – highly compact briquettes with minimal porosity and high metal yield.
3. Results
3.1. Analysis of Sample A
3.1.1. Chemical Composition Analysis (XRF)
3.1.2. Density, Porosity, and Degree of Compaction of Sample A
3.2. Analysis of Sample B
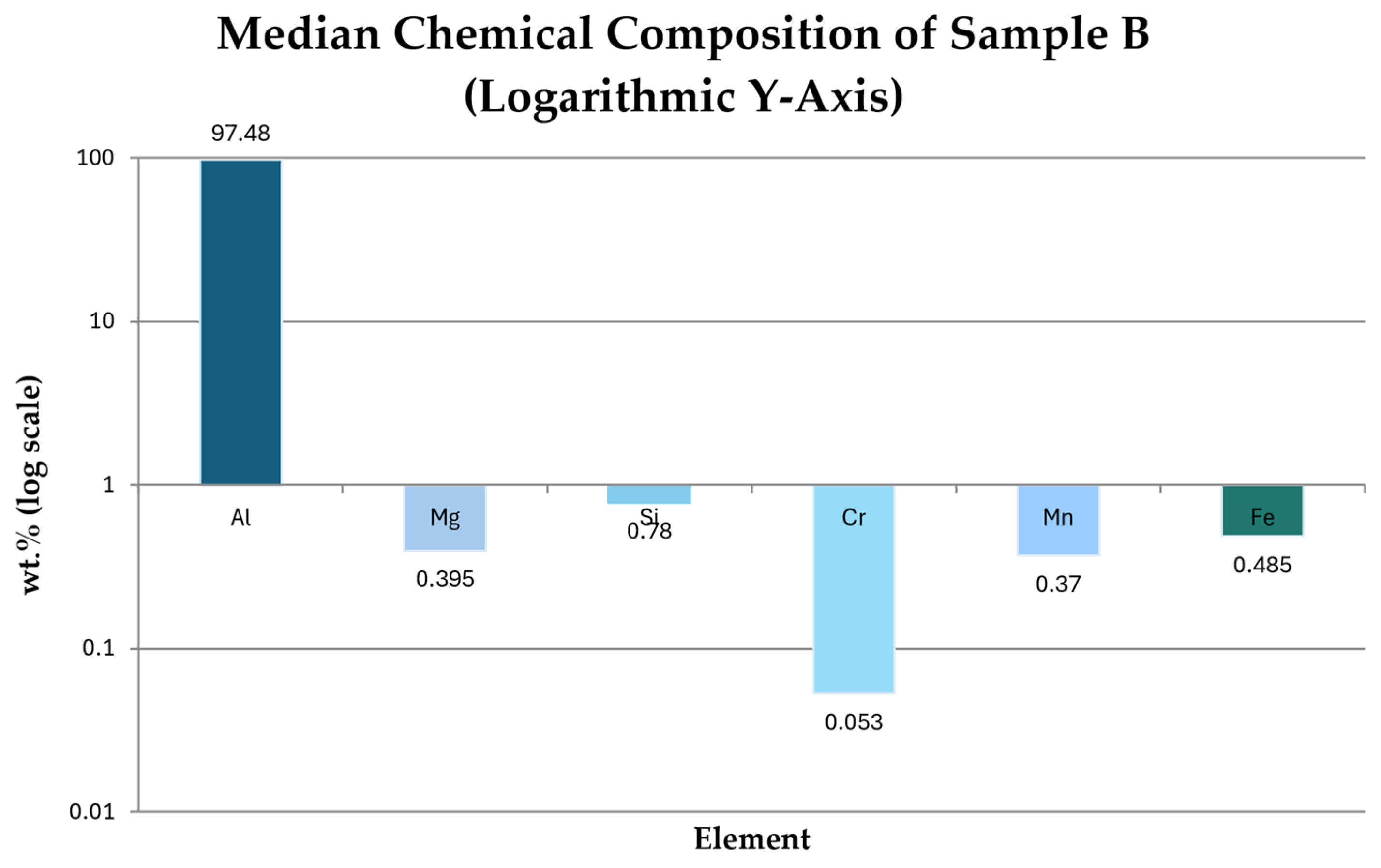
3.2.1. Chemical Composition of the Remelted Briquette (Sample B)
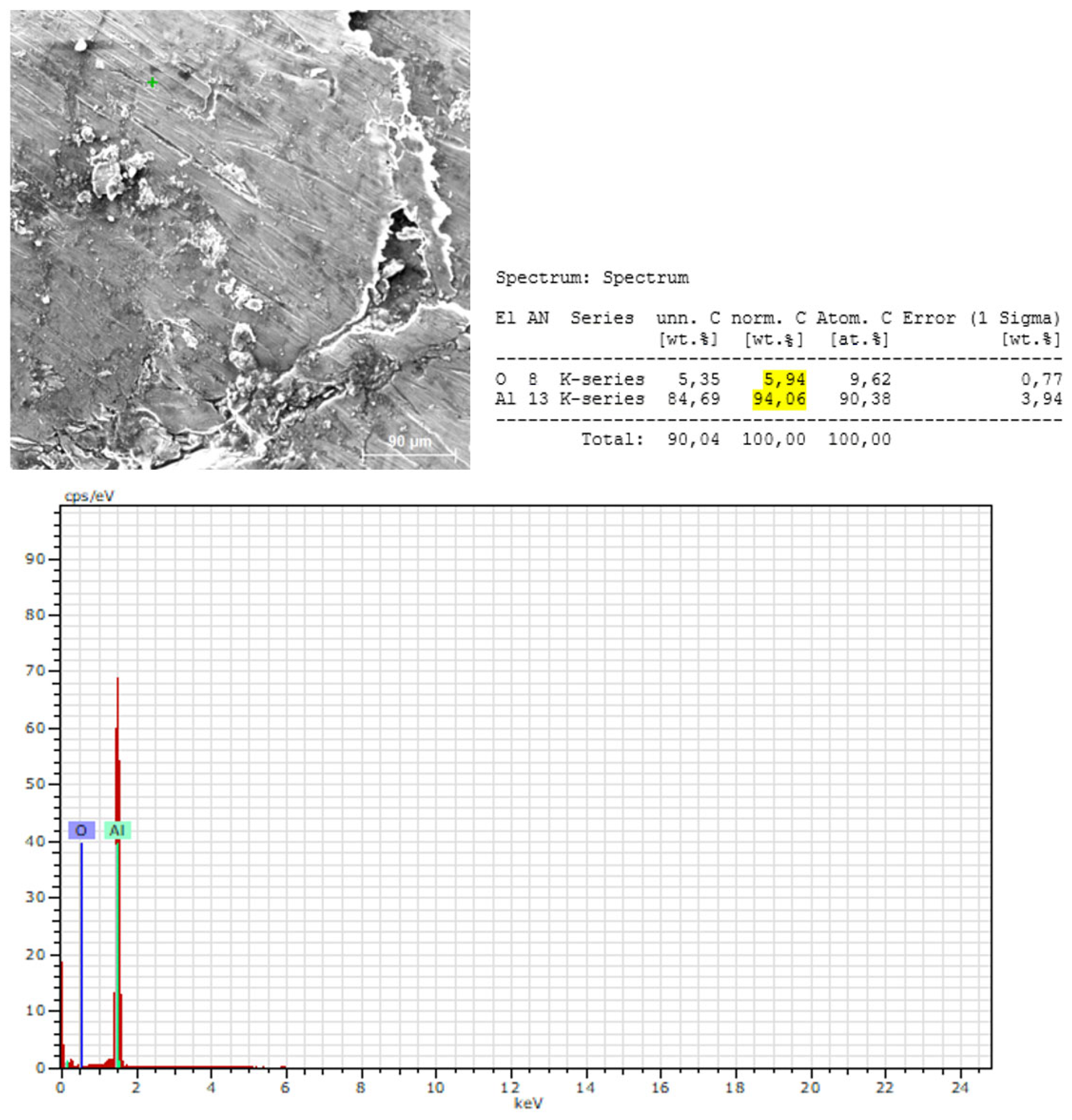
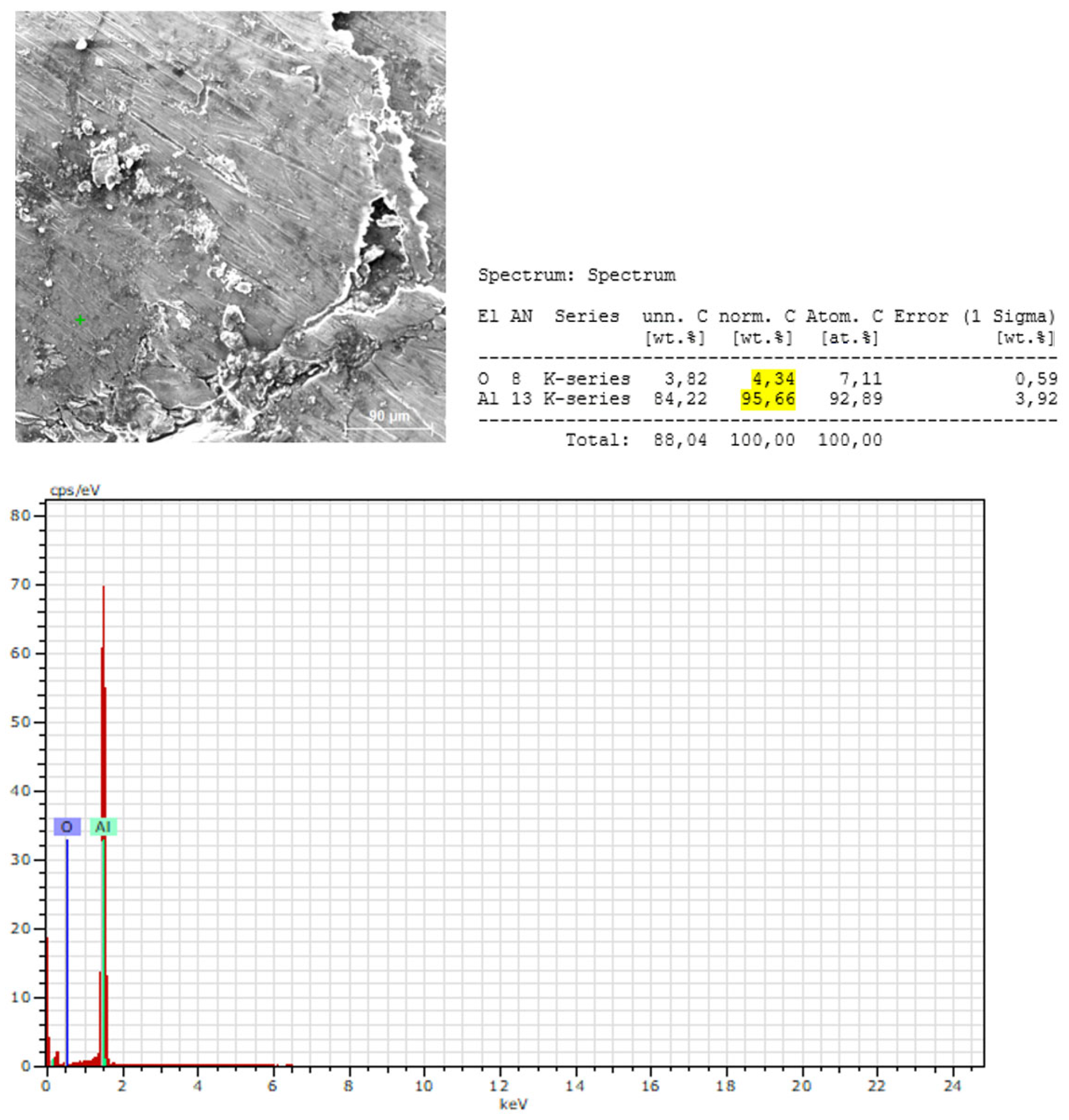
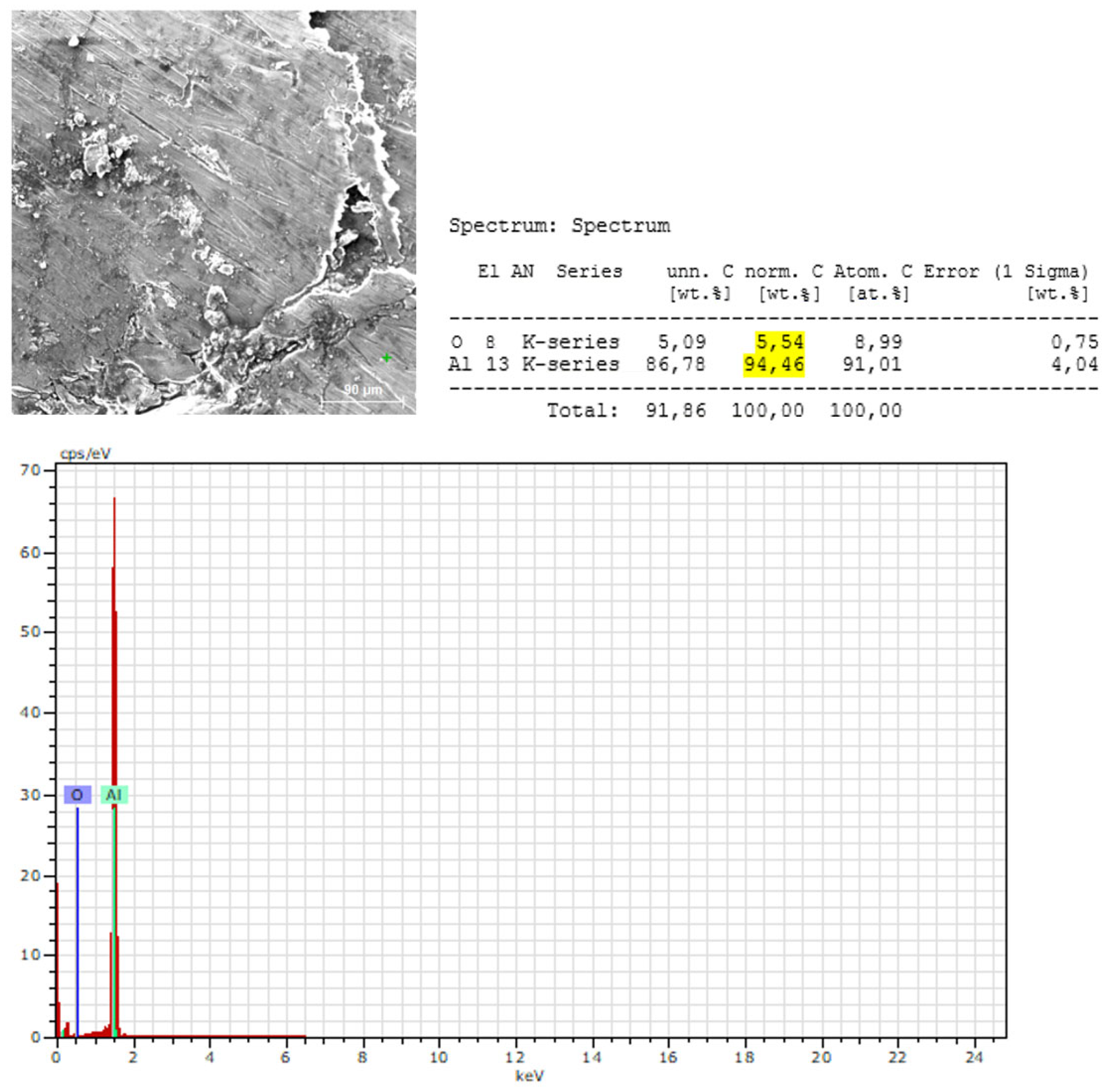
3.2.2. Microstructural and Local Chemical Analysis (SEM–EDS) of Sample B
- a fine grained metallic structure typical of AlSiMg type aluminum alloys,
- a regular texture with oriented grinding and polishing traces resulting from sample preparation,
- local discontinuities, fine micropores, and small material pull outs that represent potential nucleation sites for oxidation,
- occasional darker inclusions corresponding to Fe rich particles or oxide agglomerates.
- The surface of Sample B is covered by a thin Al₂O₃ oxide layer:
- 2.
- Aluminum is the dominant element in all measurement points:
- 3.
- EDS does not confirm the presence of Mg, Si, Mn, or Fe at the analyzed points:
- 4.
- The surface is microstructurally homogeneous, without inclusions or segregated phases:
- a uniform texture,
- fine pores typical of secondary aluminum,
- no Fe-rich or Si-rich intermetallic particles,
- no oxide agglomerates.
- 5.
- The oxygen content at the measured points indicates the presence of a surface oxidation layer typical of aluminum alloys:
- 6.
- EDS confirms the XRF-based conclusions regarding chemical homogeneity:
- local enrichment by Fe, Mn, Si, or Mg,
- intermetallic phases,
- segregation phenomena after remelting.
3.2.3. Density, Porosity, and Degree of Compaction of Sample B
- weight loss:
- metallurgical metal yield:
- dross fraction (oxide residues):
- the melting process led to a significant reduction in porosity from 12.1% (Sample A) to 8.15% (Sample B),
- the density increased to 2.388 g·cm⁻³, confirming the complete transformation of the chip-based material into a compact metallic volume,
- the yield of 94.2% confirms the high quality of the briquette as an input raw material,
- the residual dross fraction of 2.25% indicates a standard level of oxidation during the melting of chip-based recycled material.
4. Discussion
4.1. Changes in Density and Porosity During the Transition from Pressed Briquette to Casting
4.2. Mechanisms of Oxidation and Dross Formation
- a dross fraction of 2.25%,
- a relative melting loss of 3.55%,
- a yield of 94.2%.
4.3. Chemical Homogeneity After Remelting and Its Significance for Metallurgical Applications
4.4. Influence of Process-Related Contaminants and Gas Porosity
- reduce the density of the briquette,
- decompose and release gas during heating,
- promote the formation of gas porosity in the casting,
- increase metal burn off,
- reduce the overall yield.
4.5. Industrial Implications for the Use of Recycled Briquettes
- the compaction level of the briquette (85–88% for Sample A),
- the density after melting (2.388 g.cm-³ for Sample B).
- fragment rapidly,
- submerge beneath the molten metal surface when added appropriately,
- provide a large reactive surface area,
- achieve yields of 90–96%.
4.6. Key Findings of the Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Laurent Brocq, M.; Lilensten, L.; Pinot, C.; Schulze, A.; Duchaussoy, A.; Bourgon, J.; Leroy, E.; Tekkaya, A.E. Solid State Recycling of Aluminium Chips: Multi Technique Characterization and Analysis of Oxidation. Materialia 2023, 31, 101864. [Google Scholar] [CrossRef]
- Chen, X.; Ben Saada, M.; Lavisse, B.; Ammar, A. Recent Advances in the Remelting Process for Recycling Aluminium Alloy Chips: A Critical Review. Int. J. Mater. Form. 2025, 18, 42. [Google Scholar] [CrossRef]
- Gudić, S.; Vrsalović, L.; Krolo, J.; Nagode, A.; Labetić, I.D.; Lela, B. Corrosion Behaviour of Recycled Aluminium AlSi9Cu3(Fe) Machining Chips by Hot Extrusion and Thixoforming. Sustainability 2024, 16, 1358. [Google Scholar] [CrossRef]
- Pavlasek, L.; Bernatik, M.; Cervenakova, T.; Trojan, J. Behaviour of High Density Aluminium Briquette During Melting in Laboratory Conditions. Arch. Foundry Eng. 2025, 25(2). [Google Scholar] [CrossRef]
- Vlach, T.; Cais, J.; Horký, R. Influence of Aluminium Waste Chips Contenton the Final Properties of AlSi7Mg0.3 Alloy Casts. Manufacturing Technology 2024. [Google Scholar] [CrossRef]
- Penchev, T.; Altaparmakov, I.; Karastojanov, D. Briquetting of Metal Chips by Controlled Impact: Experimental Study. Available online: https://www.inase.org/library/2014/athens/bypaper/ENVEC/ENVEC-10.pdf.
- Vallejo Olivares, A.; Philipson, H.; Gökelma, M.; Roven, H.; Furu, T.; Kvithyld, A.; Tranell, G. Compaction of Aluminium Foil and Its Effect on Oxidation and Recycling Yield. In Light Metals; Springer, 2021. [Google Scholar] [CrossRef]
- Cinkilic, E.; Moodispaw, M.; Zhang, J.; Miao, J.; Luo, A.A. A New Recycled Al–Si–Mg Alloy for Sustainable Structural Die Casting Applications. Metall. Mater. Trans. A 2022, 53, 2861–2873. [Google Scholar] [CrossRef]
- Nunes, H.; Madureira, R.; Vieira, M.F.; Reis, A.; Emadinia, O. Excessive Fe Contamination in Secondary Al Alloys: Microstructure, Porosity, and Corrosion Behaviour. Metals 2025, 15, 451. [Google Scholar] [CrossRef]
- Javidani, M.; Larouche, D..; Chen, X. G. Assessment of Post-eutectic Reactions in Multicomponent Al-Si Foundry Alloys Containing Cu, Mg, and Fe. Metallurgical and Materials Transactions A 2015, 46(7), 1–14. [Google Scholar] [CrossRef]
- ISBN 10.1007/s11661-015-2856-x.
- Sasai, K. Kinetics on Formation, Growth, and Removal of Alumina Inclusions in Molten Steel. ISIJ Int. 2022, 62, 426–436. [Google Scholar] [CrossRef]
- Zhang, L.; Thomas, B. Alumina Inclusion Behavior During Steel Deoxidation. 7th European Electric Steelmaking Conference, May 26-29, 2002; pp. 2.77–2.86. [Google Scholar]
- Wu, Z.; Zheng, W.; Li, G.; Matsuura, H.; Tsukihashi, F. Effect of Inclusions’ Behavior on the Microstructure in Al–Ti Deoxidized and Mg Treated Steel. Metall. Mater. Trans. B 2015, 46, 1226–1241. [Google Scholar] [CrossRef]
- Jin, P.; Yang, Y.; Cao, L.; Yuan, X.; Wang, G. Effects of S Content on Inclusion Formation in Al and Ti–Mg Complex Deoxidized Steel. Steel Research International 2023, 94(7). [Google Scholar] [CrossRef]
- Wang, D.; Li, B.; Huang, L.; Zhan, D.; Gao, H. Effects of Deoxidation Processes on Inclusions in Environmentally Friendly Free Cutting Steel. Metals 2025, 15, 1018. [Google Scholar] [CrossRef]
- Kem, A.Y. Applications of Powder Metallurgy to the Production of Recycled Iron and Aluminum Deoxidation Briquettes for In Ladle Use. Metallurgist 2019. [Google Scholar] [CrossRef]
- Wang, J.; Wang, L.; Li, X.; Li, J.; Chen, C.; Li, C.; Zhuang, C. Study on the Thermodynamic Stability and Evolution of Inclusions in Al–Ti Deoxidized Steel. High Temp. Mater. Processes 2022, 41, 206–2015. [Google Scholar] [CrossRef]
- Cha, W.-Y.; Kim, W.-Y. In situ Observation of Inclusion Formation Behavior in Ultra Low Carbon Steel with Al–Ti Complex Deoxidation. ISIJ Int. 2025, 65, 1631–1641. [Google Scholar] [CrossRef]
- Vallejo-Olivares, A.; Philipson, H.; Gökelma, M.; Roven, H.J.; Furu, T.; Kvithyld, A.; Tranell, G. Compaction of Aluminium Foil and Its Effect on Oxidation and Recycling Yield. In Light Metals 2021; Springer Nature: Cham, Switzerland, 2021; pp. 735–741. [Google Scholar] [CrossRef]
- Ulus, A.; Ekici, H.; Güler, E. Optimization of Recovery Efficiency for Briquetted Aluminum Chips up to Briquetting Parameters. In Light Metals 2017; Ratvik, A.P., Ed.; Springer: Cham, Switzerland, 2017; pp. 925–932. [Google Scholar] [CrossRef]




| Measurement Location | Description of Measurement Position | Chemical Composition [%] | |||||
| Al | Mg | Si | Cr | Mn | Fe | ||
| AM 1 | Center | 96.70 | 1.00 | 1.15 | 0.15 | 0.31 | - |
| AM 2 | Center | 96.62 | 1.07 | 1.12 | 0.14 | 0.33 | 0.02 |
| AM 3 | Center | 96.78 | 0.96 | 1.18 | 0.16 | 0.30 | 0.01 |
| Measurement Location | Description of Measurement Position | Chemical Composition [%] | |||||
| Al | Mg | Si | Cr | Mn | Fe | ||
| BM 1 | Bottom-left corner | 97.21 | 0.49 | 0.77 | 0.091 | 0.36 | 0.41 |
| BM 2 | Top-left corner | 97.29 | 0.38 | 0.85 | 0.051 | 0.41 | 0.44 |
| BM 3 | Center – left face | 97.51 | 0.55 | 0.78 | 0.088 | 0.38 | 0.53 |
| BM 4 | Center – right face | 97.50 | 0.41 | 0.67 | 0.043 | 0.36 | 0.63 |
| BM 5 | Top-right corner | 97.50 | 0.33 | 0.78 | 0.038 | 0.36 | 0.38 |
| BM 6 | Bottom-right corner | 97.46 | 0.38 | 0.84 | 0.054 | 0.38 | 0.59 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




