Submitted:
01 April 2026
Posted:
02 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
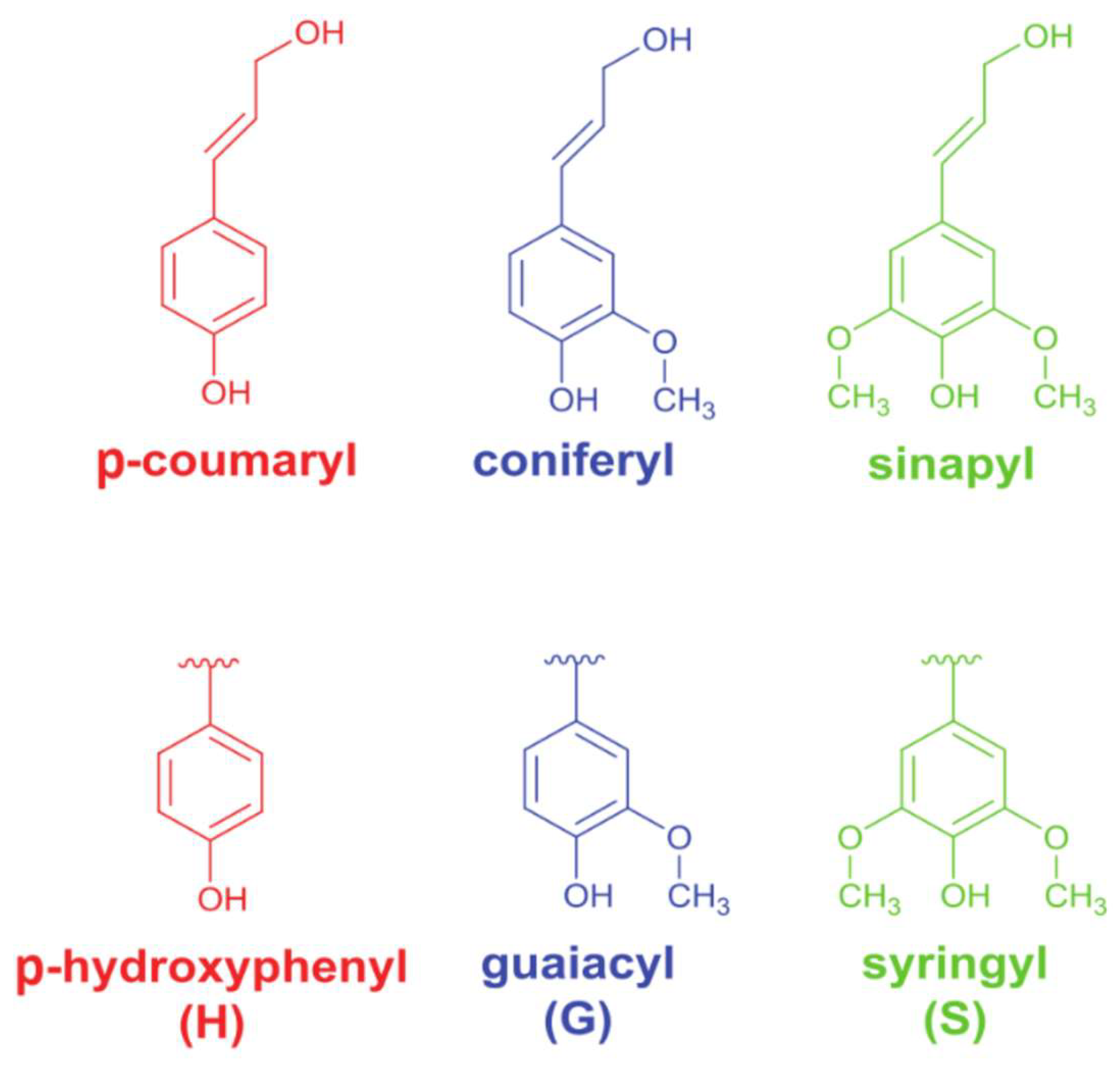
2.1. Characterization of Technical Lignins
2.1.1. Elemental Composition
2.1.2. Compositional Analysis
2.1.3. Molecular Weight Distribution
2.2. Substrate Selection
2.3. Solvent Selection
2.4. Catalyst Selection
2.5. Influence of Reaction Time
2.6. Influence of Oxygen Partial Pressure
2.7. Upscaling and Further Improvements
2.8. Influence of Catalyst Loading
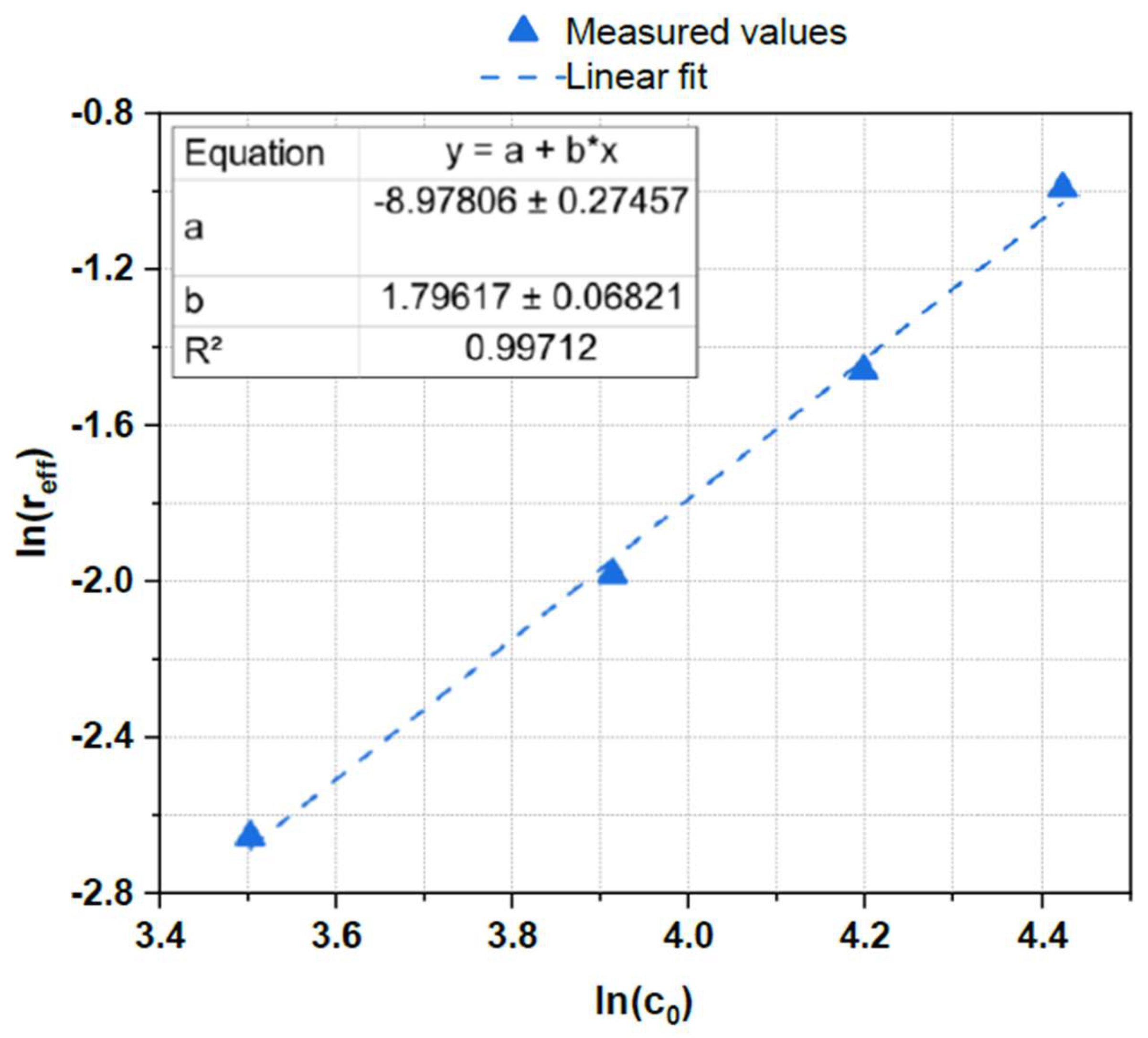
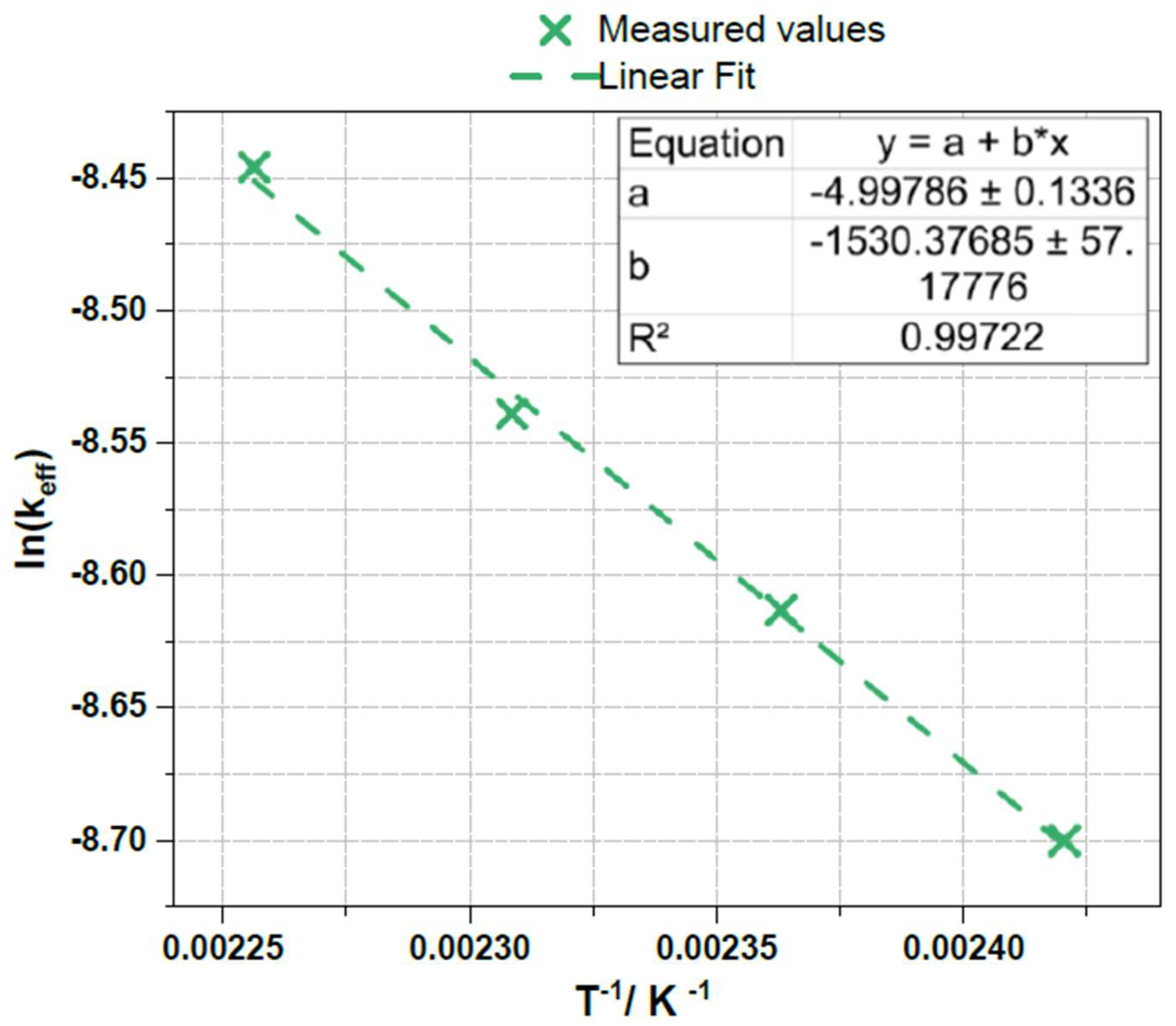
2.9. Determination of Reaction Order and Activation Energy
- Reaction temperature: 160 °C
- Residence time: 16 h
- Stirring speed: 50 rpm (to enable homogeneity)
- O2 partial pressure at T0: 14 bar
- Solvent system: Methanol/water at 8:2 v/v ratio
- Solvent volume: 30 mL
- Substrate: Softwood organosolv lignin S1
- Substrate mass: 1,500 mg
- Catalyst: HPMo-Ni3
- Catalyst mass: 150 mg
3. Materials and Methods
3.1. Catalyst Synthesis
| Category | Chemical Formula | Abbreviation |
| Commercial | H3PMo12O40 | HPMo-0 |
| H3PW12O40 | HPW-0 | |
| H3SiW12O40 | HSiW-0 | |
| Vanadium | H4PV1Mo11O40 | HPMo-V1 |
| H5PV2Mo10O40 | HPMo-V2 | |
| H6PV3Mo9O40 | HPMo-V3 | |
| H7PV4Mo8O40 | HPMo-V4 | |
| H8PV5Mo7O40 | HPMo-V5 | |
| Cobalt | H7PCo1Mo11O40 | HPMo-Co1 |
| H11PCo2Mo10O40 | HPMo-Co2 | |
| H15PCo3Mo9O40 | HPMo-Co3 | |
| Na7PCo1Mo11O40 | NaPMo-Co1 | |
| Na15PCo3W9O40 | NaPW-Co3 | |
| K10P2Co1W17O62 | WD-Co1 | |
| Manganese | H7PMn1Mo11O40 | HPMo-Mn1 |
| H11PMn2Mo10O40 | HPMo-Mn2 | |
| K10P2Mn1W17O62 | WD-Mn1 | |
| Nickel | H7PNi1Mo11O40 | HPMo-Ni1 |
| H11PNi2Mo10O40 | HPMo-Ni2 | |
| H15PNi3Mo9O40 | HPMo-Ni3 | |
| Niobium | Na6PNb3Mo9O40 | NaPMo-Nb3 |
| Indium | H15PIn4Mo8O40 | HPMo-In4 |
| Disubstituted | H8PV1Mn1Mo10O40 | HPMo-V1Mn1 |
| H12PV1Mn2Mo9O40 | HPMo-V1Mn2 | |
| H14PV3Mn2Mo7O40 | HPMo-V3Mn2 | |
| H12PV5Mn1Mo6O40 | HPMo-V5Mn1 | |
| H11PNi1Mn1Mo10O40 | HPMo-Ni1Mn1 | |
| H11PNi1Co1Mo10O40 | HPMo-Ni1Co1 |
3.2. Catalytic Experiments
4. Conclusions
5. Patents
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BTX | Benzene, Toluene, Xylene |
| D | Polydispersity |
| EtOH | Ethanol |
| G | Guaiacyl unit |
| GC-MS | Gas chromatography coupled with mass spectroscopy |
| GPC | Gel-permeation chromatography |
| H | p-hydroxyphenyl unit |
| ICP-OES | Inductively coupled plasma optical emission spectroscopy |
| MeOH | Methanol |
| MeSy | Methyl syringate |
| MeVa | Methyl vanillate |
| Mn | number average molecular weight |
| Mw | weight average molecular weights |
| NREL | National renewable energy laboratory |
| POM | Polyoxometalate |
| PTFE | Polytetrafluoroethylene |
| RedOx | Reduction-oxidation-reaction |
| S | Syringyl unit |
| Sy | Syringaldehyde |
| Va | Vanillin |
References
- Behr, T. Seidensticker, Chemistry of Renewables; Springer Berlin Heidelberg: Berlin, Heidelberg, 2020. [Google Scholar]
- Cai, J.; He, Y.; Yu, X.; Banks, S. W.; Yang, Y.; Zhang, X.; Yu, Y.; Liu, R.; Bridgwater, A. V. Renewable and Sustainable Energy Reviews 2017, 76, 309–322. [CrossRef]
- Steinbrecher, T.; Albert, J.; Kaltschmitt, M. MDPI Sustain. Chem. 2025, 6, 38. [CrossRef]
- Kumaravel, S.; Thiruvengetam, P.; Karthick, K.; Sankar, S. S.; Karmakar, A.; Kundu, S. Biotechnology progress 2021, 37(2), e3111. [CrossRef]
- Steinbrecher, T.; Sherbi, M.; Bonk, F.; Lüdtke, O.; Albert, J.; Kaltschmitt, M. Biomass & Bioenergy 2024, 183, 107136. [CrossRef]
- Functional materials from lignin: Methods and advances. In Sustainable chemistry series; Loh, X.J., Kai, D., Li, Z., Eds.; World Scientific: Singapore, 2018; volume 3. [Google Scholar]
- Kaltschmitt, M.; Hartmann, H.; Hofbauer, H. Energie aus Biomasse; Springer-Verlag: Heidelberg, 2016. [Google Scholar]
- Menon, V.; Rao, M. Progress in energy and combustion science 2012, 38, 522–550. [CrossRef]
- J. D. Gargulak, S. E. Lebo, T. J. McNally, in Kirk-Othmer Encyclopedia of Chemical Technology (Ed: J. W. &. S. Inc), John Wiley & Sons, Inc, Hoboken, NJ, USA 2000.
- Figueiredo, P.; Lintinen, K.; Hirvonen, J. T.; Kostiainen, M. A.; Santos, H. A. Progress in Materials Science 2018, 93, 233–269. [CrossRef]
- Sen, S.; Patil, S.; Argyropoulos, D. S. Green Chem. 2015, 17(11), 4862–4887. [CrossRef]
- Saito, T.; Brown, R. H.; Hunt, M. A.; Pickel, D. L.; Pickel, J. M.; Messman, J. M.; Baker, F. S.; Keller, M.; Naskar, A. K. Green Chem. 2012, 14(12), 3295. [CrossRef]
- Conversion of Lignin into Bio-Based Chemicals and Materials. In Green Chemistry and Sustainable Technology; Xu, C., Ferdosian, F., Eds.; Springer Berlin Heidelberg, Berlin: Heidelberg, 2017. [Google Scholar]
- Industrial Lignin Production and Applications: Status Quo and Future Directions, 1st ed.; Pye, E.K., Ed.; Wiley-VCH: Weinheim, Bergstr, 2008. [Google Scholar]
- Kirk-Othmer encyclopedia of chemical technology: Vanillin. In A John Wiley & Sons, Inc., publication, 5th ed.; Havkin-Frenkel, D., Seidel, A., Kirk, R.E., Othmer, D.F., Eds.; Wiley-Interscience: Hoboken, NJ, 2005; Vol.12. [Google Scholar]
- Schutyser, W.; Renders, T.; van den Bosch, S.; Koelewijn, S.-F.; Beckham, G. T.; Sels, B. F. Chemical Society reviews 2018, 47(3), 852–908. [CrossRef]
- Walton, N. J.; Mayer, M. J.; Narbad, A. Phytochemistry 2003, 63(5), 505–515. [CrossRef]
- Cabeza Sánchez, Á.; Trygve Berglihn, O.; Ottaviano, E.; Rücker, T.; Pettersen, T.; Wittgens, B.; Aliko, A.; Gálvez, L.; López, M. Open Res Europe 2024, 4, 5. [CrossRef]
- Hocking, M. B. J. Chem. Educ. 1997, 74(9), 1055. [CrossRef]
- Ranadive, A. S.; Agric, J. Food Chem. 1992, 40(10), 1922–1924. [CrossRef]
- UN Food and Agriculture Organization. Vanilla production in 2017. 2017. [Google Scholar]
- Fache, M.; Boutevin, B.; Caillol, S. ACS Sustainable Chem. Eng. 2016, 4(1), 35–46. [CrossRef]
- Marić, I.; Guo, Y.; Fürst, M. J. L. J.; van Aelst, K.; van den Bosch, S.; de Simone, M.; Martins, L. O.; Sels, B. F.; Fraaije, M. W. Adv Synth Catal 2023, 365(22), 3987–3995. [CrossRef]
- Fache, M.; Boutevin, B.; Caillol, S. European Polymer Journal 2015, 68, 488–502. [CrossRef]
- Fache, M.; Darroman, E.; Besse, V.; Auvergne, R.; Caillol, S.; Boutevin, B. Green Chem 2014, 16(4), 1987–1998. [CrossRef]
- Schlemmer, W.; Nothdurft, P.; Petzold, A.; Riess, G.; Frühwirt, P.; Schmallegger, M.; Gescheidt-Demner, G.; Fischer, R.; Freunberger, S. A.; Kern, W.; Spirk, S. Angewandte Chemie (International ed. in English) 2020, 59(51), 22943–22946. [CrossRef] [PubMed]
- Elbs, K.; Lerch, H. J. Prakt. Chem. 1916, 93(1), 1–9. [CrossRef]
- Sriprasanthi, R. B.; Mohamad Ibrahim, M. N.; Shamsudeen, S.; Adam, F.; Bhawani, S. A. BioRes 2012, 7(3), 4377–4399. [CrossRef]
- Lee, J.; Li, Y.; Cheng, J.-T.; Liu, I.-M.; Cheng, K.-C. Pharmaceuticals 2024, 17, 538. [CrossRef]
- Llevot, A.; Grau, E.; Carlotti, S.; Grelier, S.; Cramail, H. Macromolecular rapid communications 2016, 37(1), 9–28. [CrossRef] [PubMed]
- Weng, T.-H.; Chen, T.-H.; Zheng, Q.; Weng, W.-H.; Huang, L.; Lai, D.; Fu, Y.-S.; Weng, C.-F. Biomedicine & pharmacotherapy 2021, 141, 111865. [CrossRef]
- Wu, J.; Fu, Y.-S.; Lin, K.; Huang, X.; Chen, Y.-J.; Lai, D.; Kang, N.; Huang, L.; Weng, C.-F. Biomedecine & pharmacotherapie 2022, 153, 113339. [CrossRef]
- Zhou, J.; Zhang, H.; Deng, J.; Wu, Y. Macromol. Chem. Phys. 2016, 217(21), 2402–2408. [CrossRef]
- Dai, G.; Li, Q.; Zang, D.; Wei, Y. Green Chem. 2023, 25, 6263–6269. [CrossRef]
- Zhong, J.; Pérez-Ramírez, J.; Yan, N. Green Chem. 2021, 23, 18–36. [CrossRef]
- Fan, C.; Guan, H.; Zhang, H.; Wang, J.; Wang, S.; Wang, X. Biomass Bioenergy 2011, 35, 2659–2665. [CrossRef]
- Song, Y.; Wang, X.; Qu, Y.; Huang, C.; Li, Y.; Chen, B. Catalysts 2016, 6, 49. [CrossRef]
- Albert, J.; Wasserscheid, P. Green Chem. 2015, 17(12), 5164–5171. [CrossRef]
- Albert, J. Faraday Discussions 2017, 202, 99–109. [CrossRef]
- Bertleff, *!!! REPLACE !!!*; Claußnitzer, J.; Korth, W.; Wasserscheid, P.; Jess, A.; Albert, J. ACS Sustain. Chem. Eng. 2017, 5, 4110–4118. [CrossRef]
- Raabe, J.; Poller, M. J.; Voß, D.; Albert, J. ChemSusChem 2023, 16, 16. [CrossRef]
- Preuster, P.; Albert, J. Energy Tech 2018, 6(3), 501–509. [CrossRef]
- Bukowski, A.; Esau, D.; Rafat Said, A. A.; Brandt-Talbot, A.; Albert, J. ChemPlusChem 2020, 85(2), 373–386. [CrossRef]
- Feng, N.; She, S.; Tang, F.; Zhao, X.; Chen, J.; Wang, P.; Wu, Q.; Rojas, O. J. Biomacromolecules 2023, 24(6), 2541–2548. [CrossRef]
- Zhang, H.; Fu, S.; Chen, Y. International journal of biological macromolecules 2020, 147, 607–615. [CrossRef] [PubMed]
- Ruwoldt, J.; Syverud, K.; Tanase-Opedal, M. Sustainable Chemistry for the Environment 2024, 6, 100102. [CrossRef]
- Deng, W.; Zhang, H.; Wu, X.; Li, R.; Zhang, Q.; Wang, Y. Green Chem 2015, 17(11), 5009–5018. [CrossRef]
- De Gregorio, G. F.; Prado, R.; Vriamont, C.; Erdocia, X.; Labidi, J.; Hallett, J. P.; Welton, T. ACS Sustainable Chem. Eng. 2016, 4(11), 6031–6036. [CrossRef]
- Prado, R.; Erdocia, X.; de Gregorio, G. F.; Labidi, J.; Welton, T. ACS Sustainable Chem. Eng. 2016, 4(10), 5277–5288. [CrossRef]
- Stärk, K.; Taccardi, N.; Bösmann, A.; Wasserscheid, P. ChemSusChem 2010, 3, 719–723. [CrossRef]
- Albert, J.; Lüders, D.; Bösmann, A.; Guldi, D. M.; Wasserscheid, P. Green Chem 2014, 16(1), 226–237. [CrossRef]
- Voitl, T.; Nagel, M. V.; von Rohr, P. R. Holzforschung 2010, 64, 13–19. [CrossRef]
- Voitl, T.; von Rohr, P. R. Ind. Eng. Chem. Res. 2010, 49(2), 520–525. [CrossRef]
- Wassenberg, A.; Esser, T.; Poller, M. J.; Albert, J. Materials 2023, 16(7), 2864. [CrossRef]
- Wassenberg, A.; Esser, T.; Poller, M. J.; Voß, D.; Albert, J. ChemSusChem 2025, e202401973. [CrossRef]
- Mürtz, S. D.; Raabe, J.-C.; Poller, M. J.; Palkovits, R.; Albert, J.; Kurig, N. ChemCatChem 2024, 16, 5. [CrossRef]
- Fargues, C.; Mathias, Á.; Rodrigues, A. Ind. Eng. Chem. Res. 1996, 35(1), 28–36. [CrossRef]
- Werhan, H.; Mir, J. M.; Voitl, T.; Rudolf von Rohr, P. Holzforschung 2011, 65(5), 703–709. [CrossRef]
- Odyakov, V. F.; Zhizhina, E. G.; Maksimovskaya, R. I. Applied Catalysis A: General 2008, 342(1-2), 126–130. [CrossRef]
- Raabe, J.-C.; Aceituno Cruz, J.; Albert, J.; Poller, M. J. Inorganics 2023, 11(4), 138. [CrossRef]
- Raabe, J.-C.; Jameel, F.; Stein, M.; Albert, J.; Poller, M. J. Dalton Transactions 2024, 53, 454–466. Available online: https://pubs.rsc.org/en/content/articlelanding/2023/dt/d3dt03883a. [CrossRef] [PubMed]
- Raabe, J.; Esser, T.; Jameel, F.; Stein, M.; Albert, J.; Poller, M. J. Inorganic Chemistry Frontiers 2023, 10, 4854–4868. [CrossRef]
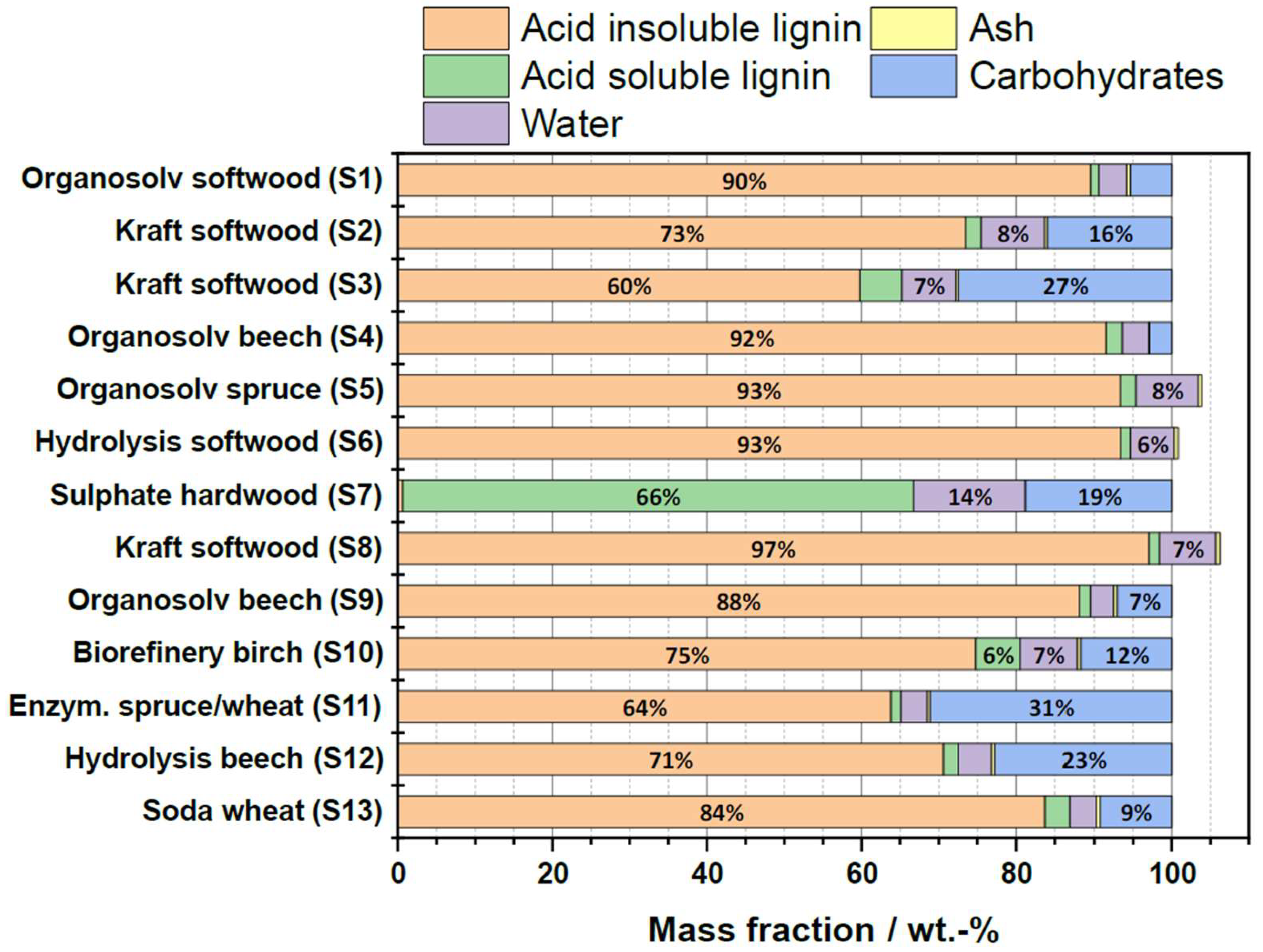
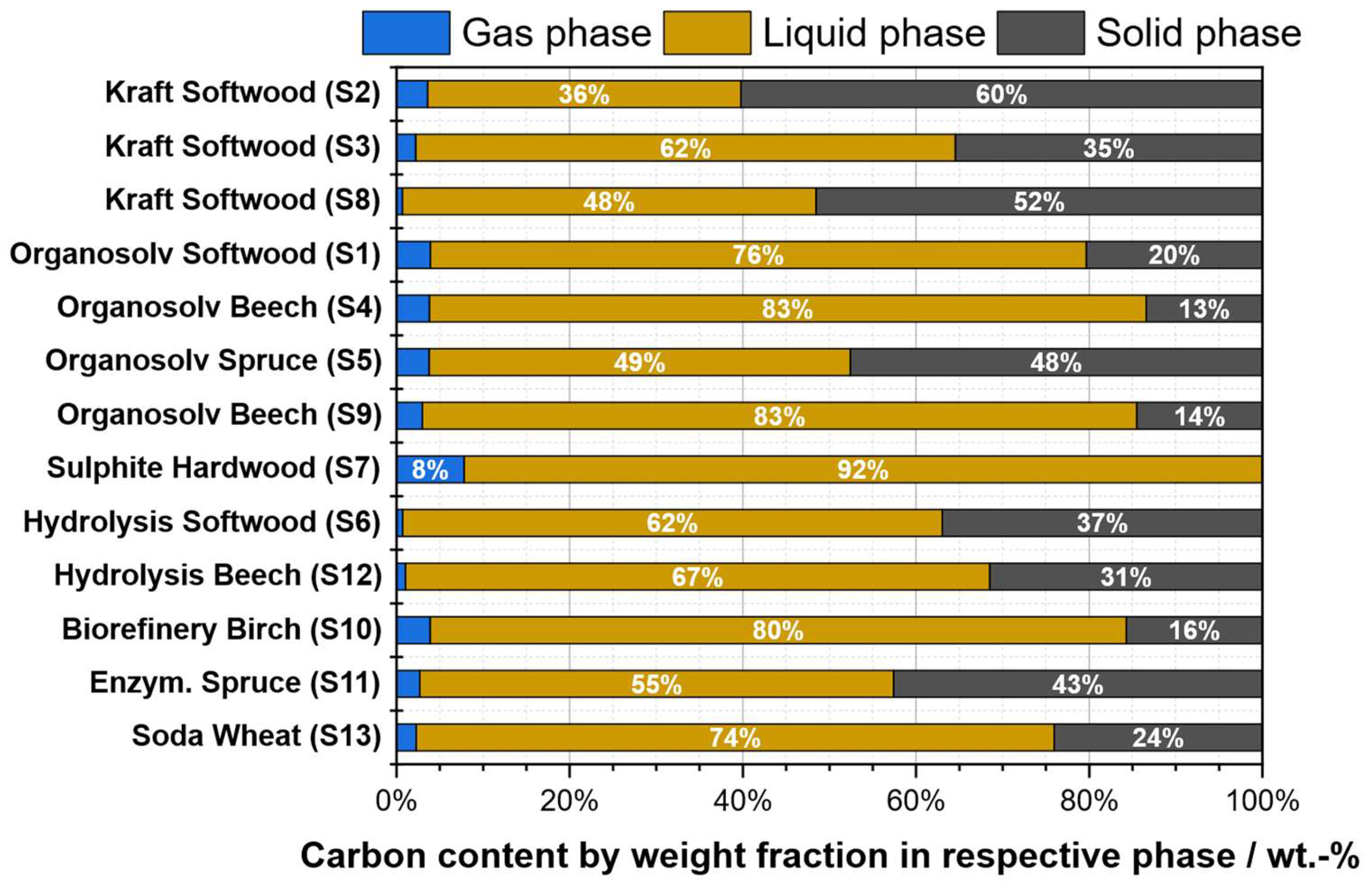
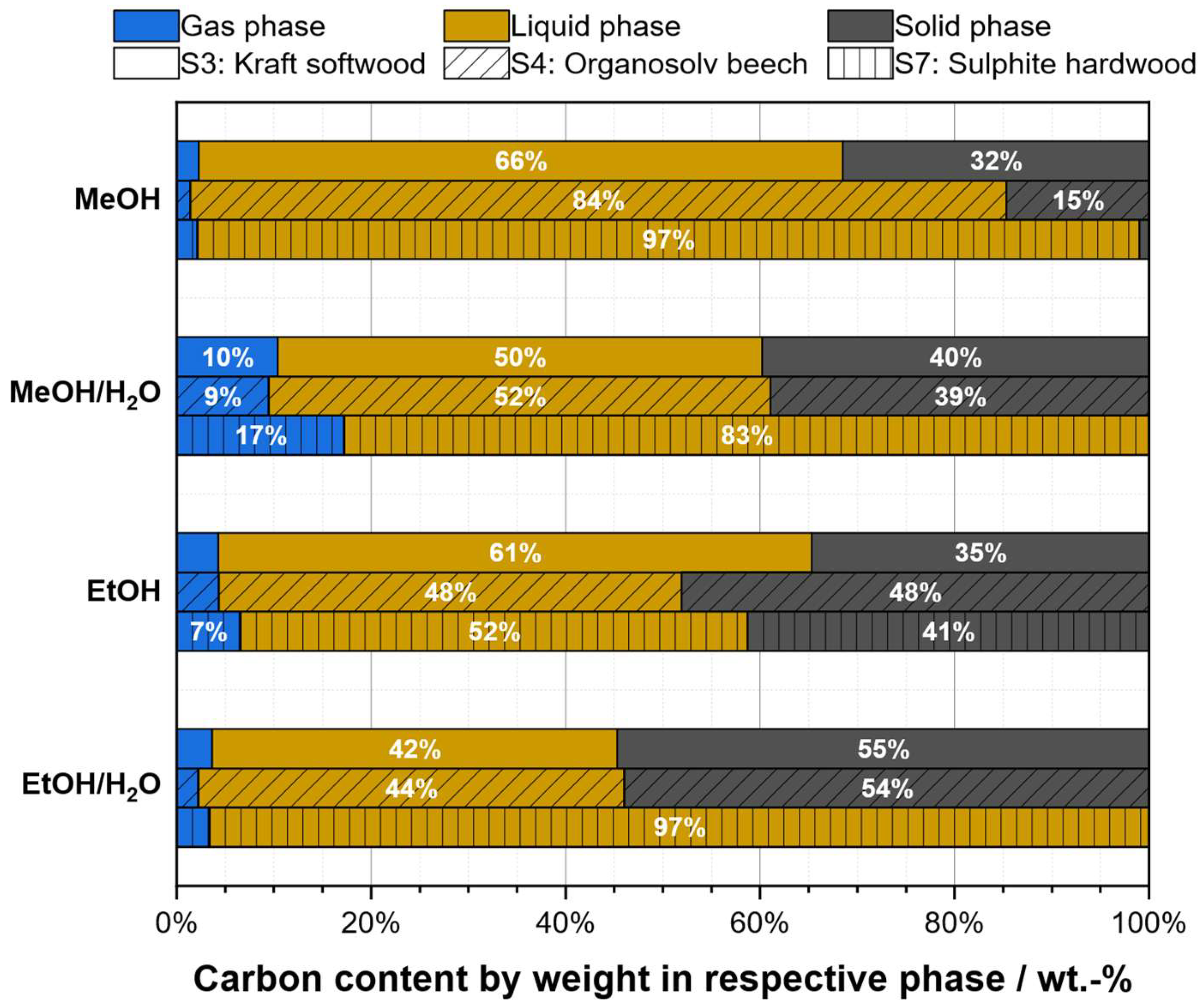
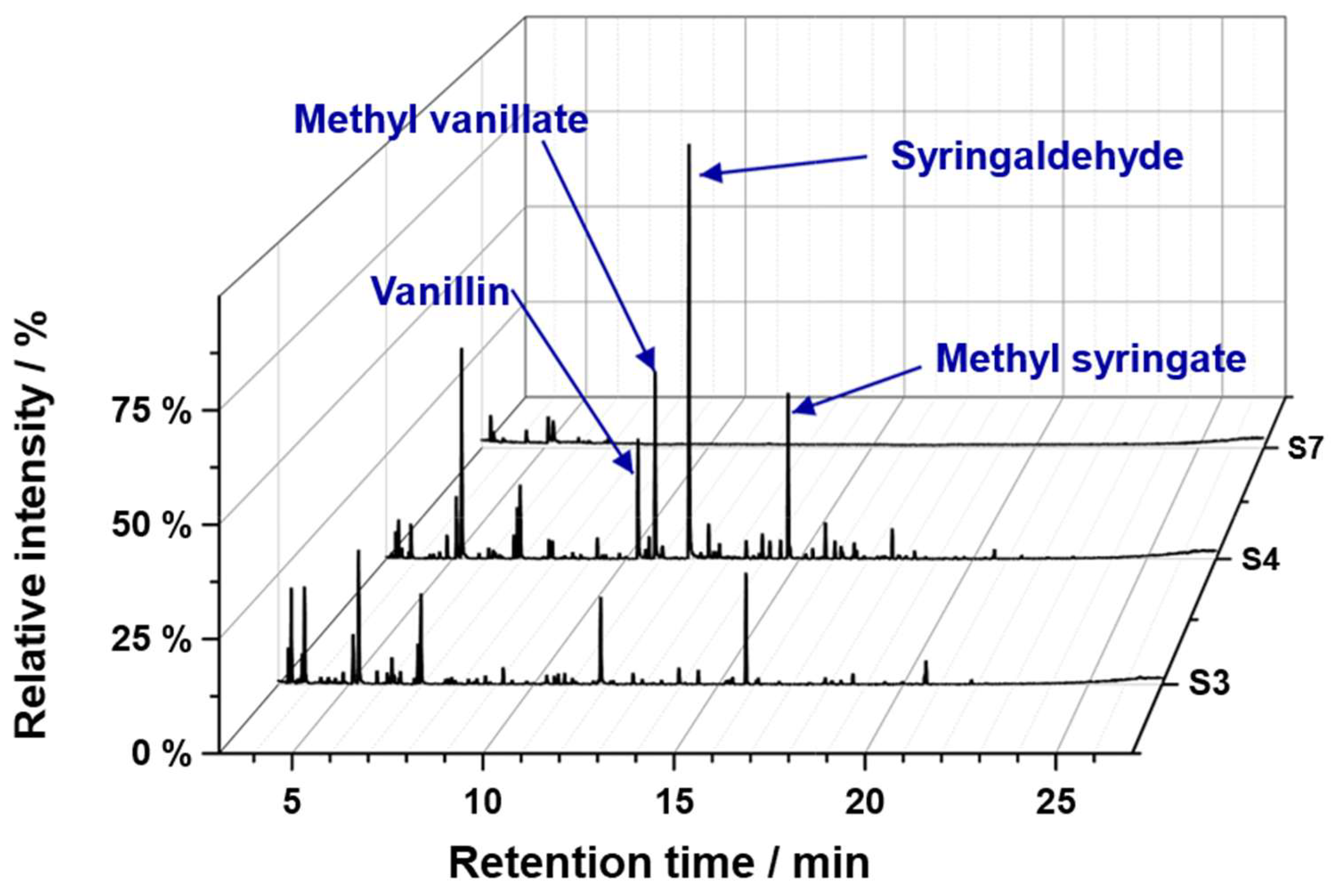
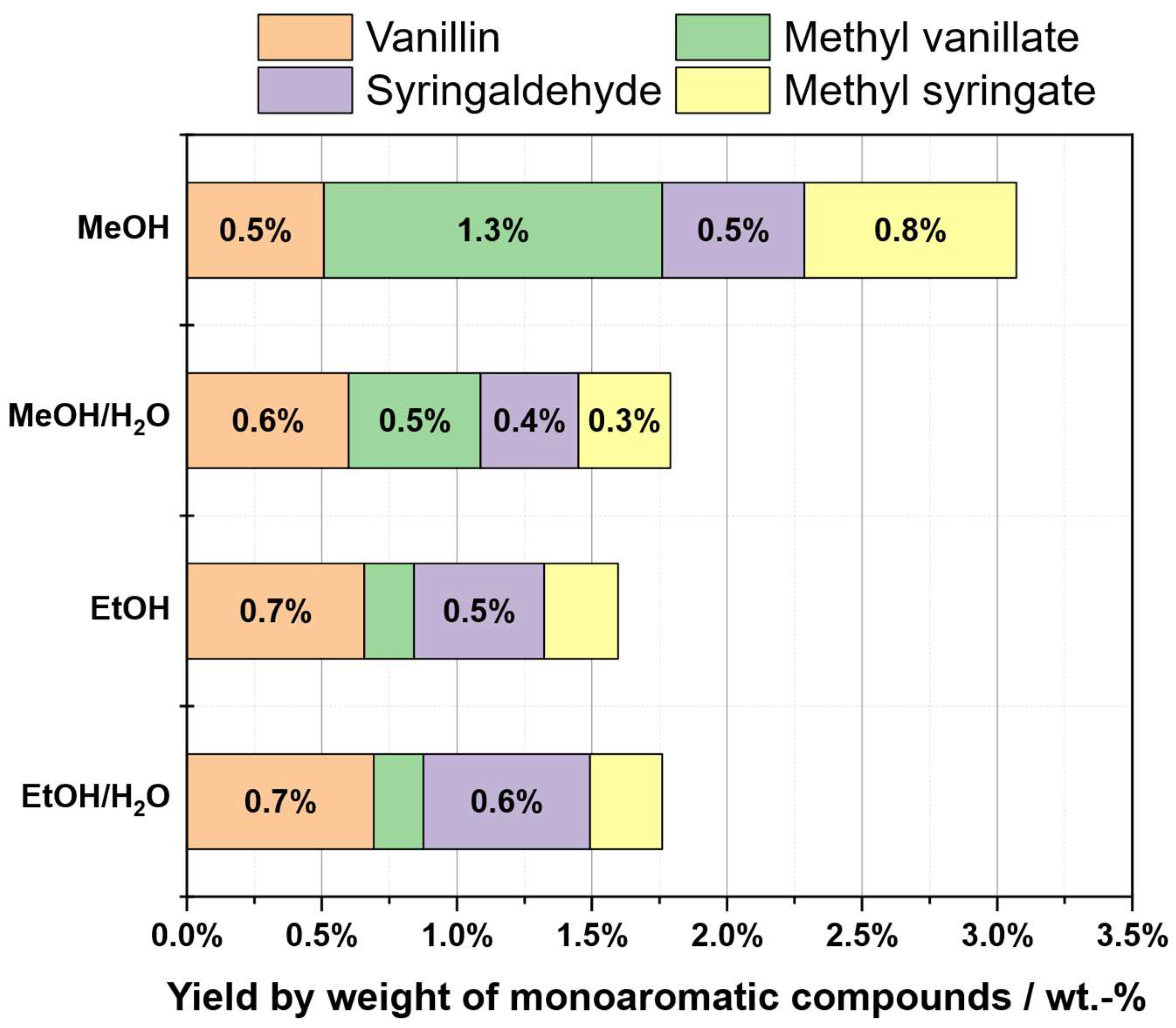
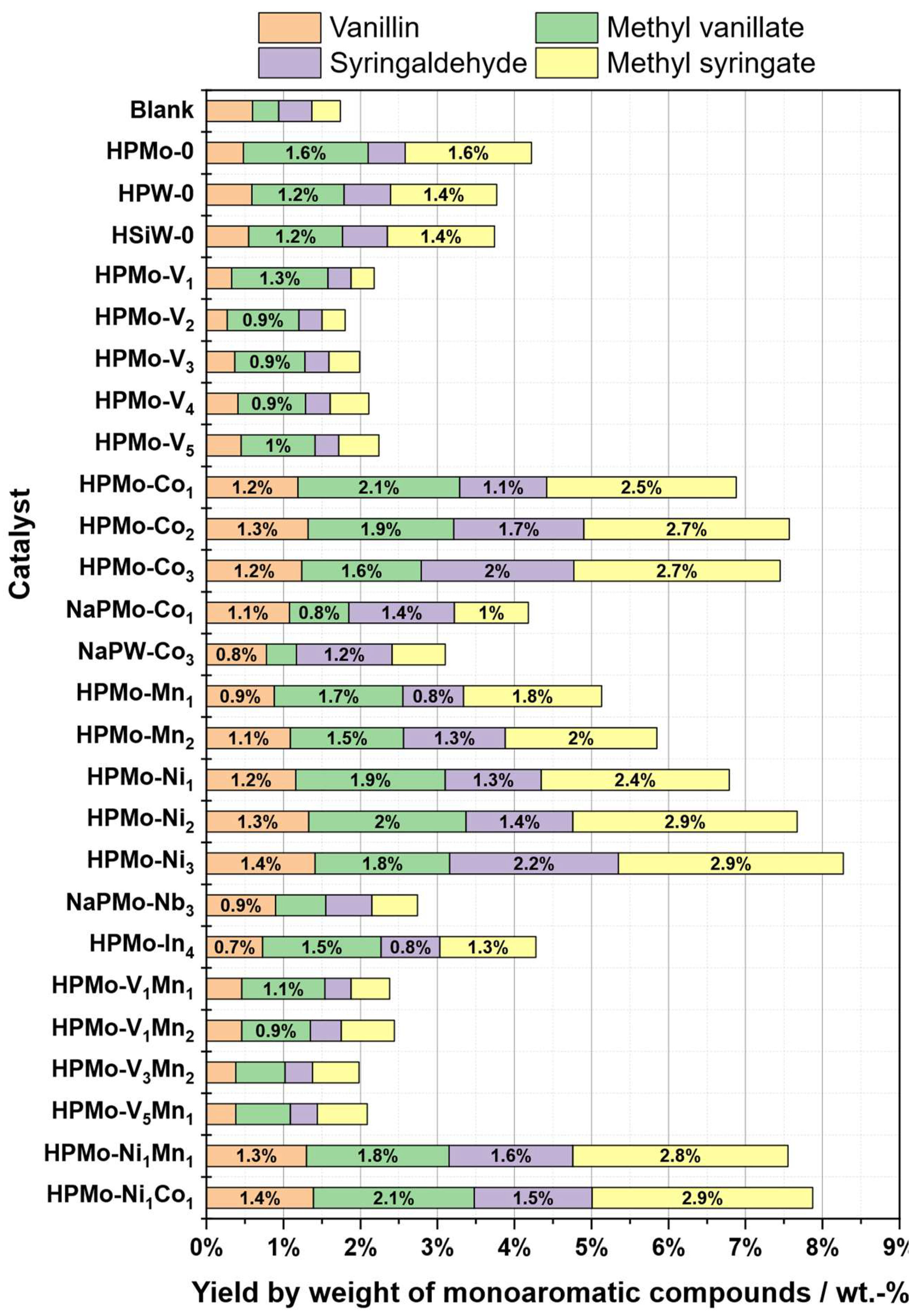
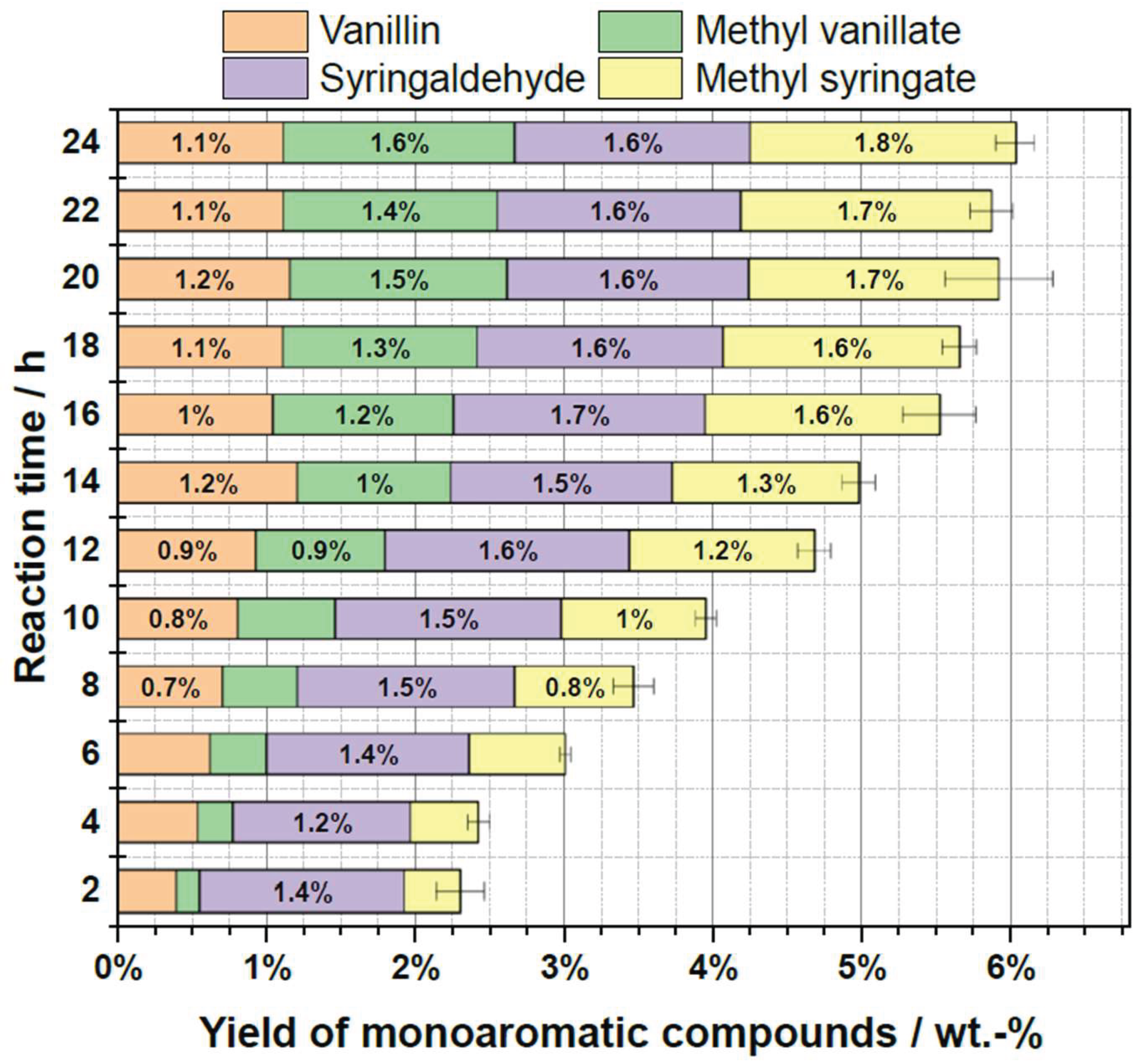
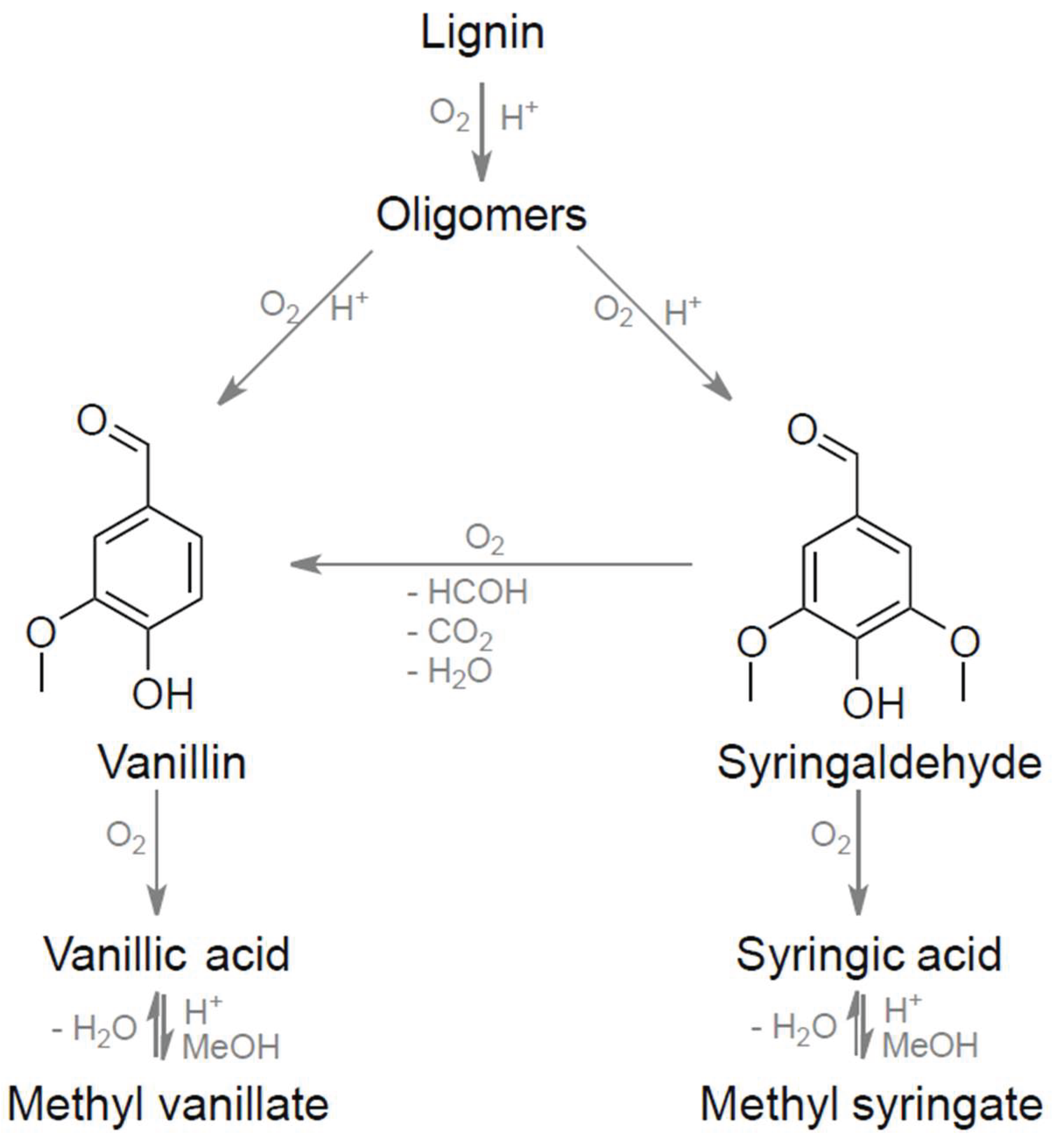
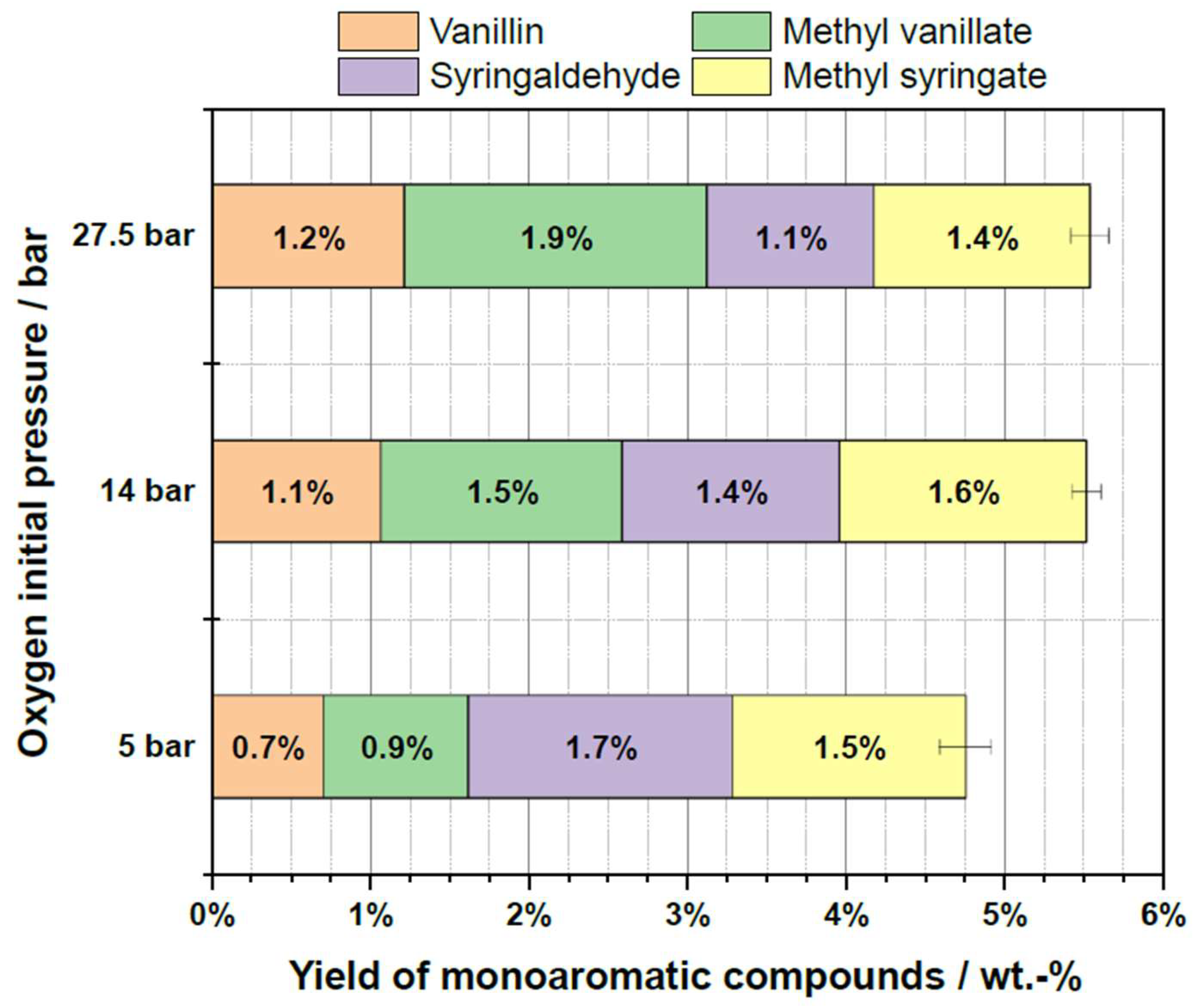
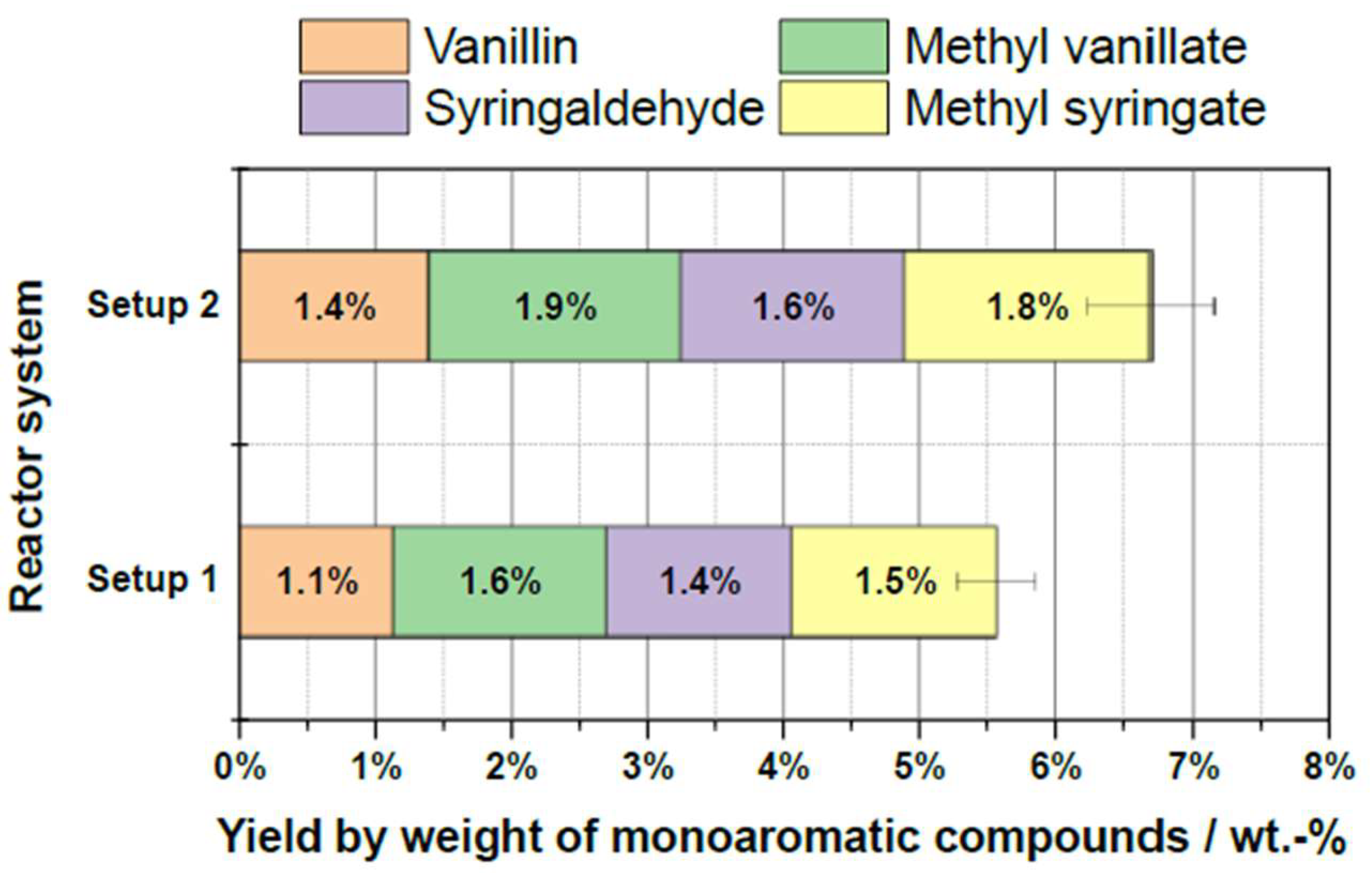
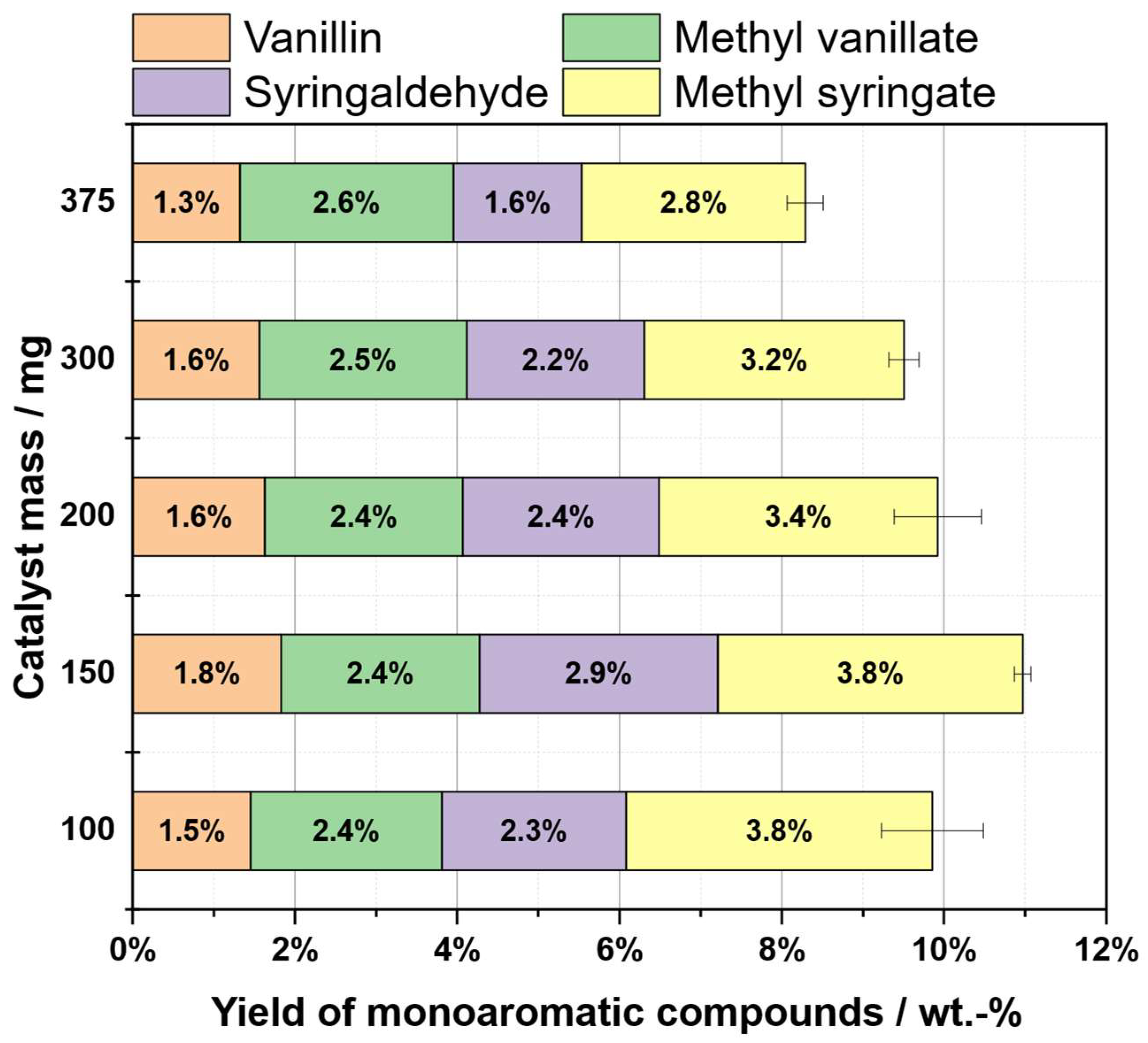
| Substrate # | Provider | Biomass type | Pulping type |
| S1 | FAU Erlangen | Softwood | Organosolv |
| S2 | Merck | Softwood | Kraft |
| S3 | Merck | Softwood | Kraft |
| S4 | Fraunhofer CBP, Leuna | Beech wood | Organosolv |
| S5 | Fraunhofer CBP, Leuna | Spruce wood | Organosolv |
| S6 | LignoPure, Hamburg | Softwood | Hydrolysis |
| S7 | LignoPure, Hamburg | Hardwood | Sulphate |
| S8 | LignoPure, Hamburg | Softwood | Kraft |
| S9 | Fraunhofer CBP, Leuna | Beech wood | Organosolv |
| S10 | LignoPure, Hamburg | Birch wood | 2G Biorefinery |
| S11 | LignoPure, Hamburg | Spruce wood & wheat straw | Enzymatic |
| S12 | LignoPure, Hamburg | Beech wood | Hydrolysis |
| S13 | LignoPure, Hamburg | Wheat straw | Purified Soda |
| Substrate # | Lignin description | Mw / Da | Mn / Da | D / - |
| S1 | Organosolv softwood | 2513 | 864 | 2.9 |
| S2 | Kraft softwood | 5391 | 1260 | 4.3 |
| S3 | Kraft softwood | 6177 | 1378 | 4.5 |
| S4 | Organosolv beech | 2234 | 800 | 2.8 |
| S5 | Organosolv spruce | 3607 | 1133 | 3.2 |
| S6 | Hydrolysis softwood | 1111 | 475 | 2.3 |
| S7 | Sulphate hardwood | 3717 | 783 | 4.7 |
| S8 | Kraft softwood | 6680 | 1456 | 4.6 |
| S9 | Organosolv beech | 3049 | 852 | 3.6 |
| S10 | 2G Biorefinery birch | 7215 | 844 | 8.6 |
| S11 | Enzymatic spruce&wheat | 1613 | 705 | 2.3 |
| S12 | Hydrolysis beech | 9348 | 977 | 9.6 |
| S13 | Soda wheat | 5910 | 1212 | 4.9 |
| Entry # | Initial lignin concentration / mg mL-1 | Monoaromatic concentration / mg mL-1 |
| 1 | 33.2 | 1.1 |
| 2 | 50.2 | 2.2 |
| 3 | 66.7 | 3.7 |
| 4 | 83.4 | 5.9 |
| Entry # | Reaction temperature / °C | Monoaromatic concentration / mg mL-1 |
| 1 | 140 | 3.0 |
| 2 | 150 | 3.3 |
| 3 | 160 | 3.5 |
| 4 | 170 | 3.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




