Submitted:
31 March 2026
Posted:
01 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
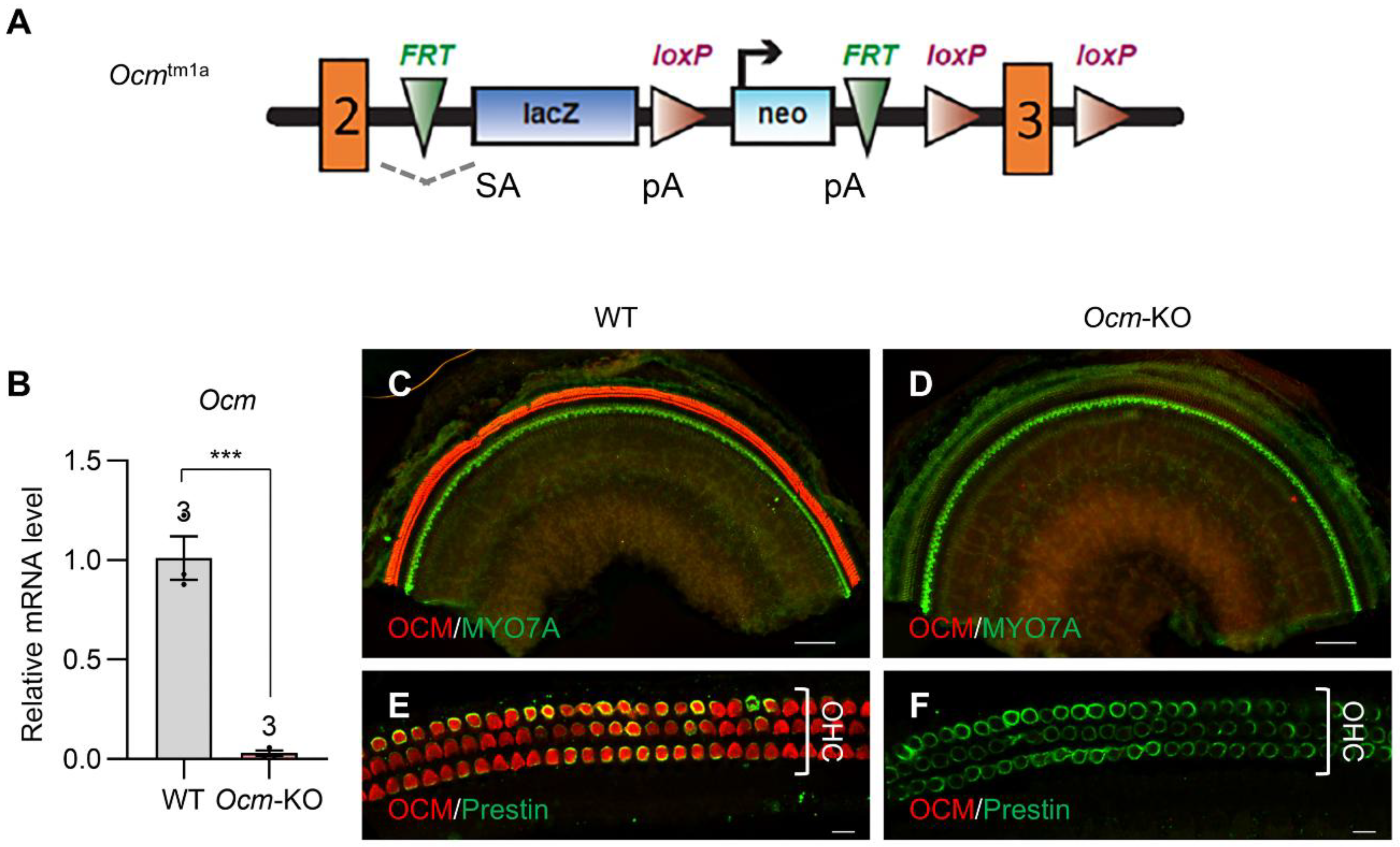
2.1. Establishment of a New Ocm-KO Mouse Model: Ocmtm1a/tm1a
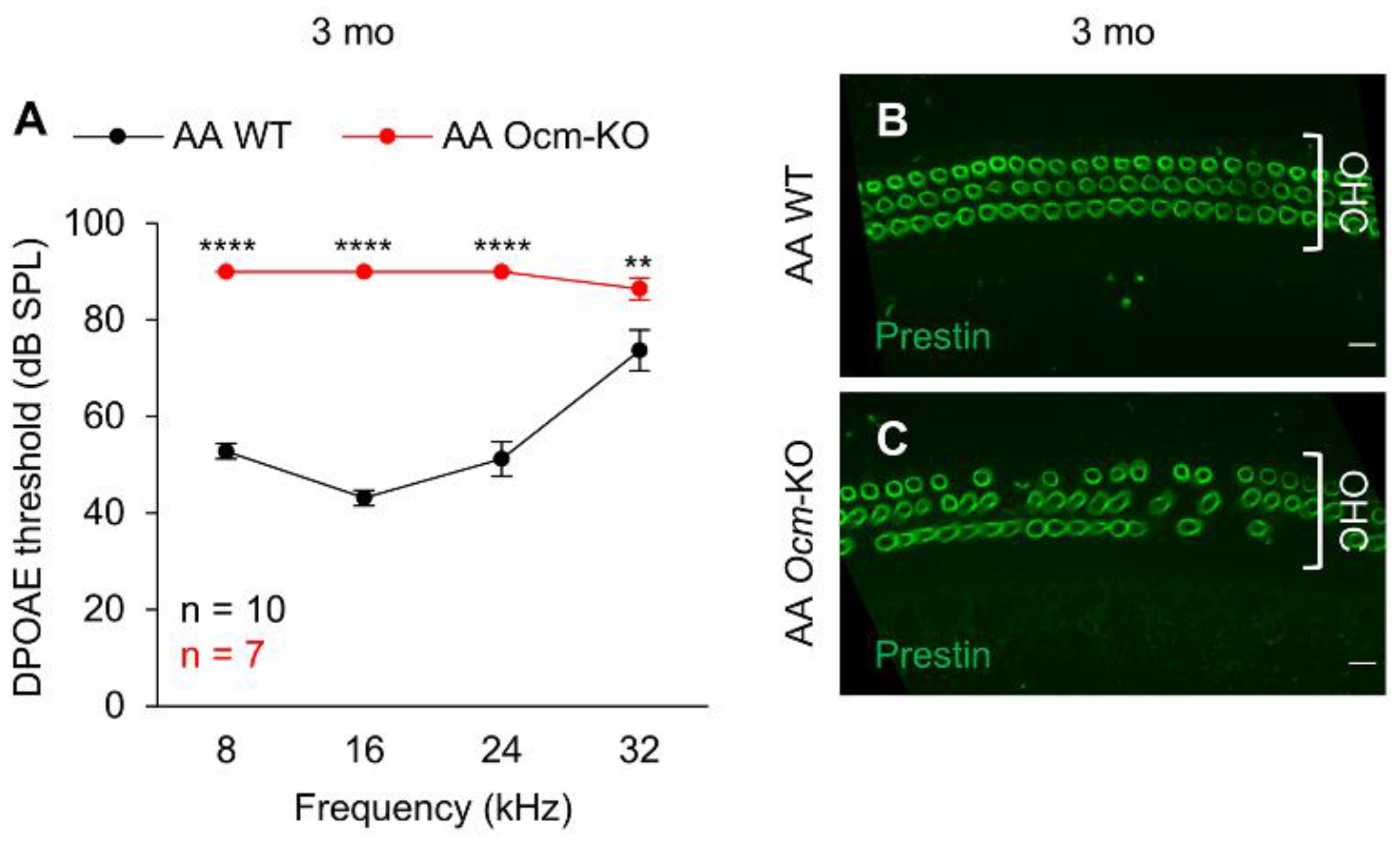
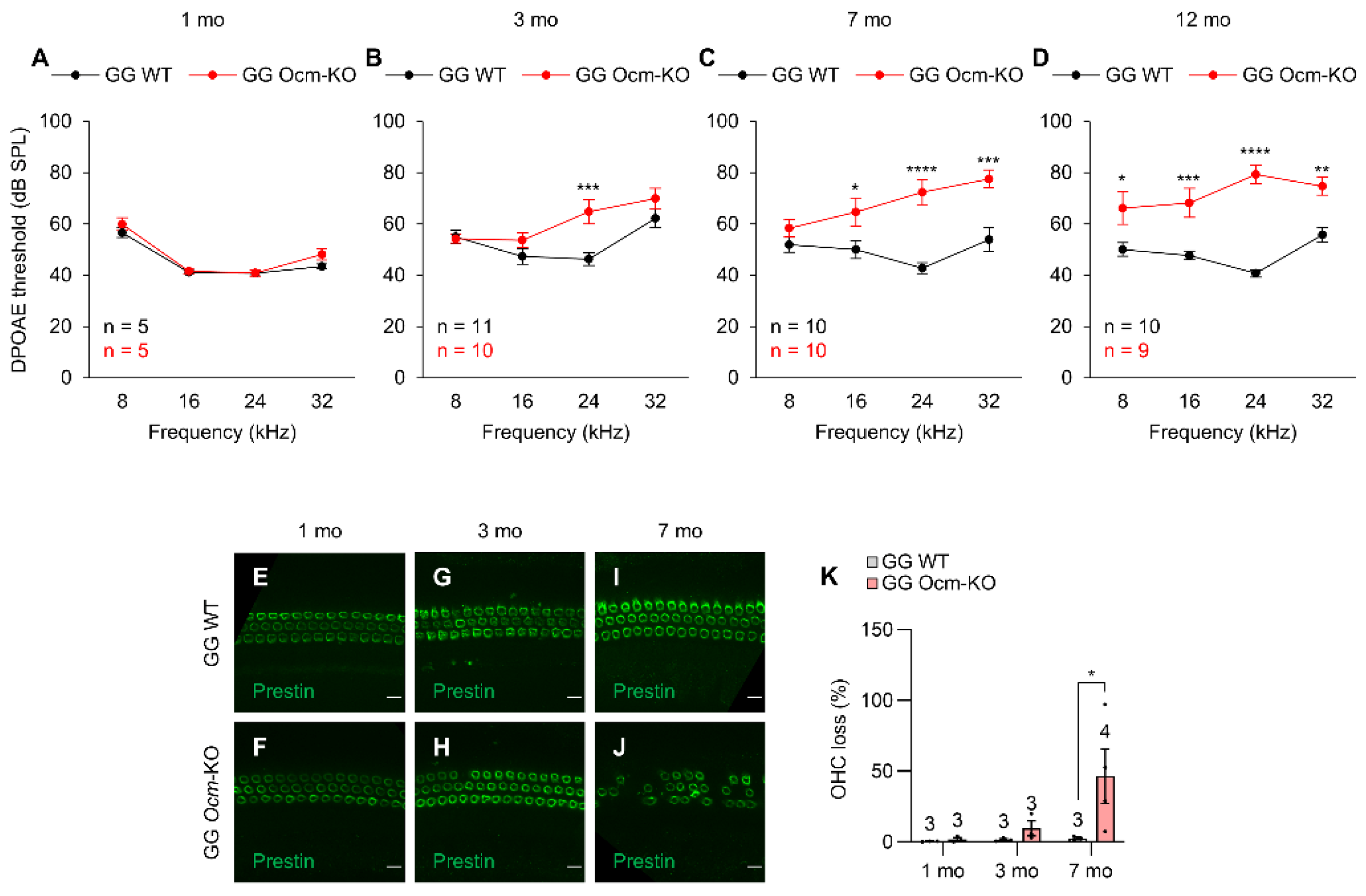
2.2. Ocmtm1a/tm1a Mice Display Early Progressive Hearing Loss and OHC Degeneration
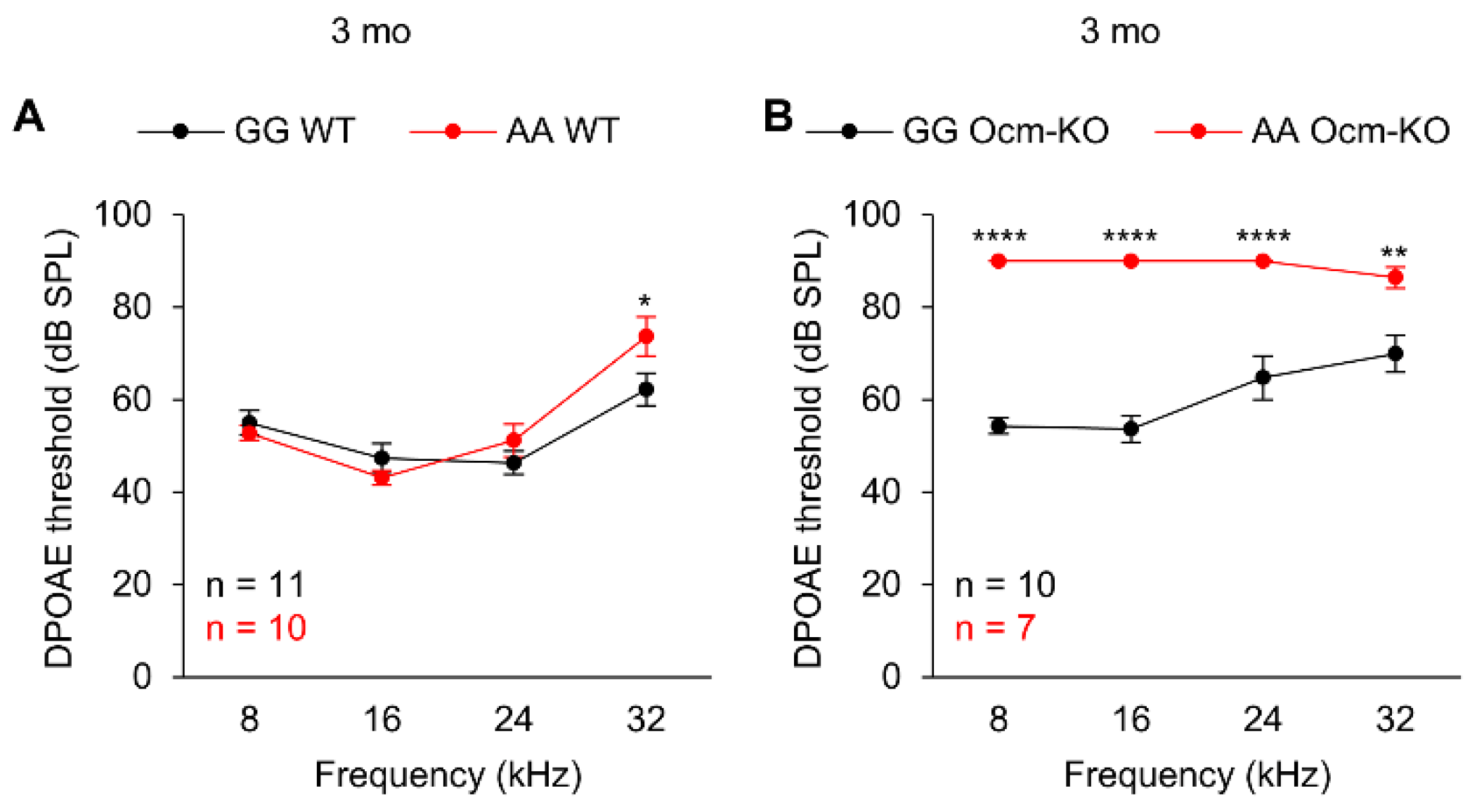
2.3. Cdh23753A Alone Further Accelerates Age-Related Hearing Loss in Ocmtm1a/tm1a Mice
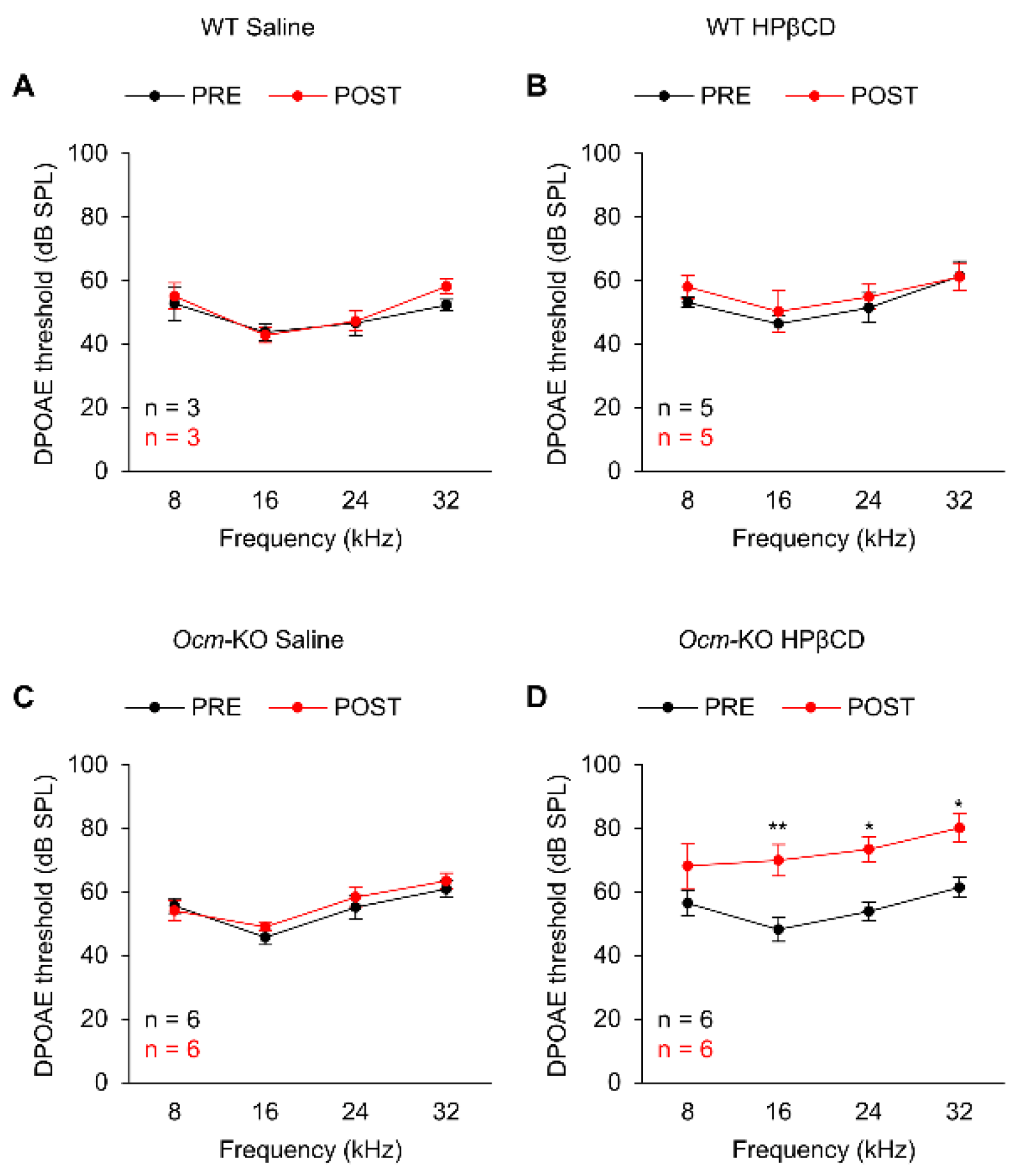
2.4. Ocmtm1a/tm1a Mice Are More Susceptible to HPβCD-Induced Hearing Loss
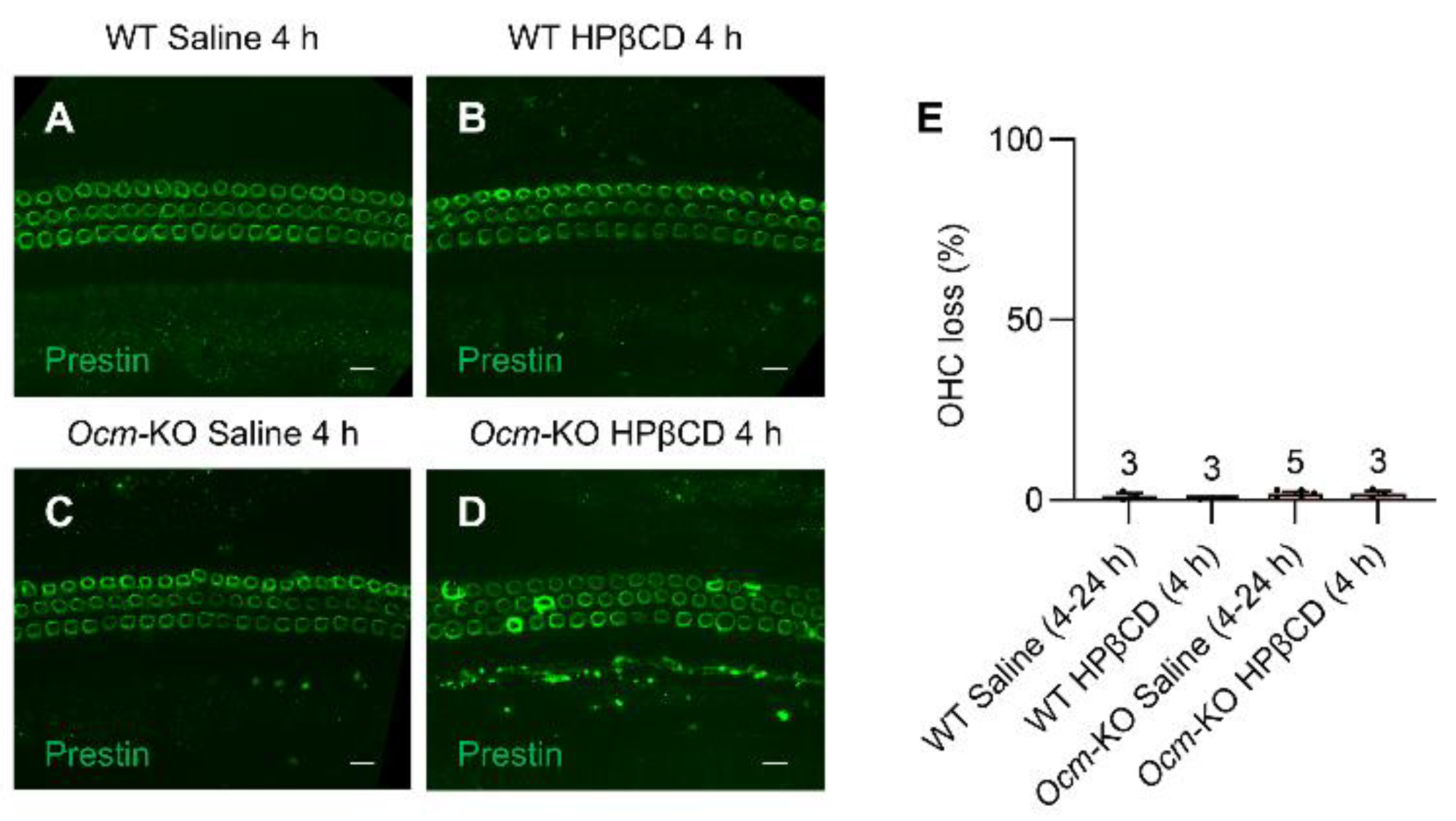
2.5. Ocmtm1a/tm1a OHCs Are More Susceptible to HPβCD-Induced Structural Deterioration In Vivo
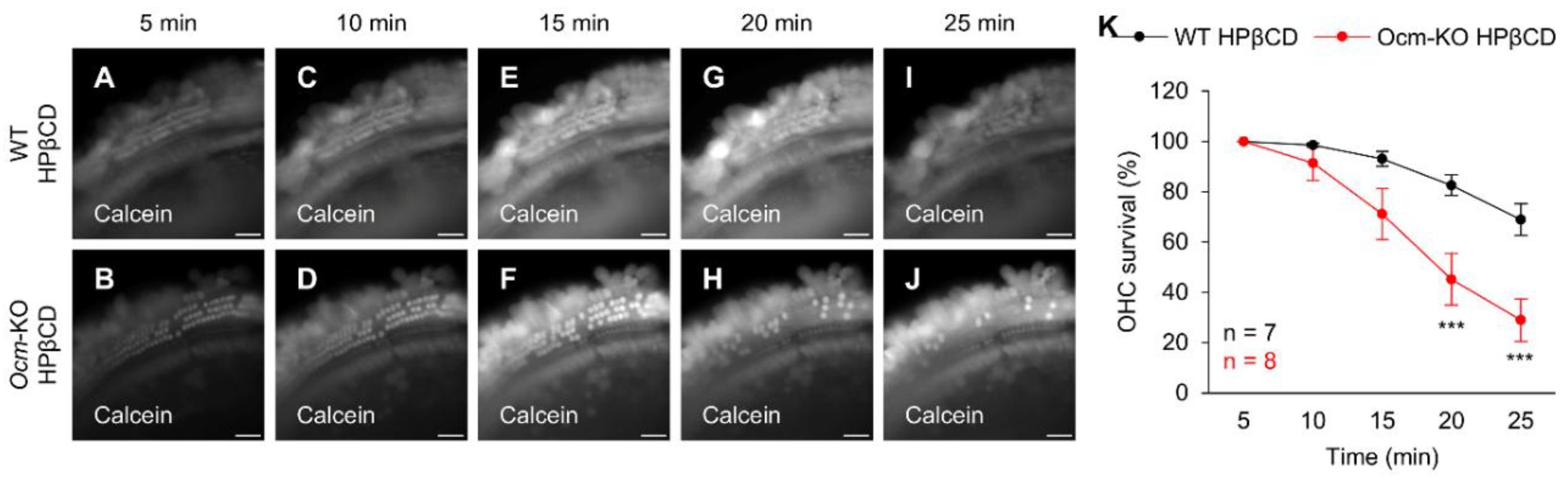
2.6. Ocmtm1a/tm1a OHCs Are More Susceptible to HPβCD-Induced Rupture In Vitro
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. RT-qPCR
4.3. Immunofluorescence
4.4. DPOAE
4.5. HPβCD Treatment In Vivo
4.6. HPβCD Treatment In Vitro
4.7. Statistics
Supplementary Materials
Author Contributions
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Skarnes, W.C.; Rosen, B.; West, A.P.; Koutsourakis, M.; Bushell, W.; Iyer, V.; Mujica, A.O.; Thomas, M.; Harrow, J.; Cox, T.; et al. A conditional knockout resource for the genome-wide study of mouse gene function. Nature 2011, 474, 337–342. [Google Scholar] [CrossRef]
- Reed, N.S.; Jiang, K.; Deal, J.A. Hearing Loss Among Older Adults: Epidemiology, Disparities, and Gaps in Research. Annu. Rev. Public Health 2025. [Google Scholar] [CrossRef]
- Siemens, J.; Lillo, C.; Dumont, R.A.; Reynolds, A.; Williams, D.S.; Gillespie, P.G.; Muller, U. Cadherin 23 is a component of the tip link in hair-cell stereocilia. Nature 2004, 428, 950–955. [Google Scholar] [CrossRef] [PubMed]
- Kazmierczak, P.; Sakaguchi, H.; Tokita, J.; Wilson-Kubalek, E.M.; Milligan, R.A.; Muller, U.; Kachar, B. Cadherin 23 and protocadherin 15 interact to form tip-link filaments in sensory hair cells. Nature 2007, 449, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Brownell, W.E.; Bader, C.R.; Bertrand, D.; de Ribaupierre, Y. Evoked mechanical responses of isolated cochlear outer hair cells. Science 1985, 227, 194–196. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Shen, W.; He, D.Z.; Long, K.B.; Madison, L.D.; Dallos, P. Prestin is the motor protein of cochlear outer hair cells. Nature 2000, 405, 149–155. [Google Scholar] [CrossRef]
- Dallos, P.; Wu, X.; Cheatham, M.A.; Gao, J.; Zheng, J.; Anderson, C.T.; Jia, S.; Wang, X.; Cheng, W.H.; Sengupta, S.; et al. Prestin-based outer hair cell motility is necessary for mammalian cochlear amplification. Neuron 2008, 58, 333–339. [Google Scholar] [CrossRef]
- Fettiplace, R. Hair Cell Transduction, Tuning, and Synaptic Transmission in the Mammalian Cochlea. Compr. Physiol. 2017, 7, 1197–1227. [Google Scholar] [CrossRef]
- Keithley, E.M. Pathology and mechanisms of cochlear aging. J. Neurosci. Res. 2020, 98, 1674–1684. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.Z.; O'Malley, J.T.; de Gruttola, V.; Liberman, M.C. Age-Related Hearing Loss Is Dominated by Damage to Inner Ear Sensory Cells, Not the Cellular Battery That Powers Them. J. Neurosci. 2020, 40, 6357–6366. [Google Scholar] [CrossRef]
- Fettiplace, R.; Nam, J.H. Tonotopy in calcium homeostasis and vulnerability of cochlear hair cells. Hear. Res. 2019, 376, 11–21. [Google Scholar] [CrossRef]
- Senarita, M.; Thalmann, I.; Shibasaki, O.; Thalmann, R. Calcium-binding proteins in organ of Corti and basilar papilla: CBP-15, an unidentified calcium-binding protein of the inner ear. Hearing research 1995, 90, 169–175. [Google Scholar] [CrossRef]
- Thalmann, I.; Shibasaki, O.; Comegys, T.H.; Henzl, M.T.; Senarita, M.; Thalmann, R. Detection of a beta-parvalbumin isoform in the mammalian inner ear. Biochem. Biophys. Res. Commun. 1995, 215, 142–147. [Google Scholar] [CrossRef]
- Henzl, M.T.; Shibasaki, O.; Comegys, T.H.; Thalmann, I.; Thalmann, R. Oncomodulin is abundant in the organ of Corti. Hear. Res. 1997, 106, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Sakaguchi, N.; Henzl, M.T.; Thalmann, I.; Thalmann, R.; Schulte, B.A. Oncomodulin is expressed exclusively by outer hair cells in the organ of Corti. J. Histochem. Cytochem. 1998, 46, 29–40. [Google Scholar] [CrossRef]
- Hackney, C.M.; Mahendrasingam, S.; Penn, A.; Fettiplace, R. The concentrations of calcium buffering proteins in mammalian cochlear hair cells. J. Neurosci. Off. J. Soc. Neurosci. 2005, 25, 7867–7875. [Google Scholar] [CrossRef]
- Pangrsic, T.; Gabrielaitis, M.; Michanski, S.; Schwaller, B.; Wolf, F.; Strenzke, N.; Moser, T. EF-hand protein Ca2+ buffers regulate Ca2+ influx and exocytosis in sensory hair cells. Proc. Natl. Acad. Sci. USA 2015, 112, E1028-1037. [Google Scholar] [CrossRef] [PubMed]
- Tong, B.; Hornak, A.J.; Maison, S.F.; Ohlemiller, K.K.; Liberman, M.C.; Simmons, D.D. Oncomodulin, an EF-Hand Ca2+ Buffer, Is Critical for Maintaining Cochlear Function in Mice. J. Neurosci. 2016, 36, 1631–1635. [Google Scholar] [CrossRef]
- Climer, L.K.; Hornak, A.J.; Murtha, K.; Yang, Y.; Cox, A.M.; Simpson, P.L.; Le, A.; Simmons, D.D. Deletion of Oncomodulin Gives Rise to Early Progressive Cochlear Dysfunction in C57 and CBA Mice. Front. Aging Neurosci. 2021, 13, 749729. [Google Scholar] [CrossRef] [PubMed]
- Lachgar-Ruiz, M.; Ingham, N.J.; Martelletti, E.; Chen, J.; James, E.; Panganiban, C.; Lewis, M.A.; Steel, K.P. Two new mouse alleles of Ocm and Slc26a5. Hear. Res. 2024, 452, 109109. [Google Scholar] [CrossRef]
- Li, H.S.; Borg, E. Age-related loss of auditory sensitivity in two mouse genotypes. Acta Otolaryngol. 1991, 111, 827–834. [Google Scholar] [CrossRef]
- Zheng, Q.Y.; Johnson, K.R.; Erway, L.C. Assessment of hearing in 80 inbred strains of mice by ABR threshold analyses. Hear. Res. 1999, 130, 94–107. [Google Scholar] [CrossRef]
- Johnson, K.R.; Zheng, Q.Y.; Noben-Trauth, K. Strain background effects and genetic modifiers of hearing in mice. Brain Res. 2006, 1091, 79–88. [Google Scholar] [CrossRef]
- Johnson, K.R.; Erway, L.C.; Cook, S.A.; Willott, J.F.; Zheng, Q.Y. A major gene affecting age-related hearing loss in C57BL/6J mice. Hear. Res. 1997, 114, 83–92. [Google Scholar] [CrossRef]
- Johnson, K.R.; Zheng, Q.Y.; Erway, L.C. A major gene affecting age-related hearing loss is common to at least ten inbred strains of mice. Genomics 2000, 70, 171–180. [Google Scholar] [CrossRef]
- Noben-Trauth, K.; Zheng, Q.Y.; Johnson, K.R. Association of cadherin 23 with polygenic inheritance and genetic modification of sensorineural hearing loss. Nat. Genet. 2003, 35, 21–23. [Google Scholar] [CrossRef] [PubMed]
- Newton, S.; Aguilar, C.; Bowl, M.R. C57BL/6-derived mice and the Cdh23(ahl) allele - Background matters. Hear. Res. 2025, 462, 109278. [Google Scholar] [CrossRef] [PubMed]
- Kane, K.L.; Longo-Guess, C.M.; Gagnon, L.H.; Ding, D.; Salvi, R.J.; Johnson, K.R. Genetic background effects on age-related hearing loss associated with Cdh23 variants in mice. Hear. Res. 2012, 283, 80–88. [Google Scholar] [CrossRef]
- Coisne, C.; Tilloy, S.; Monflier, E.; Wils, D.; Fenart, L.; Gosselet, F. Cyclodextrins as Emerging Therapeutic Tools in the Treatment of Cholesterol-Associated Vascular and Neurodegenerative Diseases. Molecules 2016, 21. [Google Scholar] [CrossRef] [PubMed]
- Ishitsuka, Y.; Irie, T.; Matsuo, M. Cyclodextrins applied to the treatment of lysosomal storage disorders. Adv. Drug Deliv. Rev. 2022, 191, 114617. [Google Scholar] [CrossRef]
- Braga, S.S. Molecular Mind Games: The Medicinal Action of Cyclodextrins in Neurodegenerative Diseases. Biomolecules 2023, 13. [Google Scholar] [CrossRef]
- Crumling, M.A.; King, K.A.; Duncan, R.K. Cyclodextrins and Iatrogenic Hearing Loss: New Drugs with Significant Risk. Front. Cell Neurosci. 2017, 11, 355. [Google Scholar] [CrossRef] [PubMed]
- Testa, G.; Schaft, J.; van der Hoeven, F.; Glaser, S.; Anastassiadis, K.; Zhang, Y.; Hermann, T.; Stremmel, W.; Stewart, A.F. A reliable lacZ expression reporter cassette for multipurpose, knockout-first alleles. Genesis 2004, 38, 151–158. [Google Scholar] [CrossRef]
- Coleman, J.L.; Brennan, K.; Ngo, T.; Balaji, P.; Graham, R.M.; Smith, N.J. Rapid Knockout and Reporter Mouse Line Generation and Breeding Colony Establishment Using EUCOMM Conditional-Ready Embryonic Stem Cells: A Case Study. Front. Endocrinol. 2015, 6, 105. [Google Scholar] [CrossRef]
- Crumling, M.A.; Liu, L.; Thomas, P.V.; Benson, J.; Kanicki, A.; Kabara, L.; Halsey, K.; Dolan, D.; Duncan, R.K. Hearing loss and hair cell death in mice given the cholesterol-chelating agent hydroxypropyl-beta-cyclodextrin. PloS one 2012, 7, e53280. [Google Scholar] [CrossRef]
- Cronin, S.; Lin, A.; Thompson, K.; Hoenerhoff, M.; Duncan, R.K. Hearing Loss and Otopathology Following Systemic and Intracerebroventricular Delivery of 2-Hydroxypropyl-Beta-Cyclodextrin. J. Assoc. Res. Otolaryngol. JARO 2015, 16, 599–611. [Google Scholar] [CrossRef]
- Takahashi, S.; Homma, K.; Zhou, Y.; Nishimura, S.; Duan, C.; Chen, J.; Ahmad, A.; Cheatham, M.A.; Zheng, J. Susceptibility of outer hair cells to cholesterol chelator 2-hydroxypropyl-beta-cyclodextrine is prestin-dependent. Sci. Rep. 2016, 6, 21973. [Google Scholar] [CrossRef]
- Zhou, Y.; Takahashi, S.; Homma, K.; Duan, C.; Zheng, J.; Cheatham, M.A.; Zheng, J. The susceptibility of cochlear outer hair cells to cyclodextrin is not related to their electromotile activity. Acta Neuropathol. Commun. 2018, 6, 98. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.Y.; Kabara, L.L.; Swiderski, D.L.; Raphael, Y.; Duncan, R.K.; Kim, Y.H. ROS Scavenger, Ebselen, Has No Preventive Effect in New Hearing Loss Model Using a Cholesterol-Chelating Agent. J. Audiol. Otol. 2019, 23, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Zhou, Y.; Fuentes, R.J.; Tan, X. Verification of Outer Hair Cell Motor Protein, Prestin, as a Serological Biomarker for Mouse Cochlear Damage. Int. J. Mol. Sci. 2024, 25. [Google Scholar] [CrossRef]
- Liu, X.; Ding, D.; Chen, G.D.; Li, L.; Jiang, H.; Salvi, R. 2-Hydroxypropyl-beta-cyclodextrin Ototoxicity in Adult Rats: Rapid Onset and Massive Destruction of Both Inner and Outer Hair Cells Above a Critical Dose. Neurotox. Res. 2020, 38, 808–823. [Google Scholar] [CrossRef]
- Ding, D.; Manohar, S.; Jiang, H.; Salvi, R. Hydroxypropyl-beta-cyclodextrin causes massive damage to the developing auditory and vestibular system. Hear. Res. 2020, 396, 108073. [Google Scholar] [CrossRef] [PubMed]
- Ding, D.; Jiang, H.; Salvi, R. Cochlear spiral ganglion neuron degeneration following cyclodextrin-induced hearing loss. Hear. Res. 2021, 400, 108125. [Google Scholar] [CrossRef] [PubMed]
- Ding, D.; Jiang, H.; Manohar, S.; Liu, X.; Li, L.; Chen, G.D.; Salvi, R. Spatiotemporal Developmental Upregulation of Prestin Correlates With the Severity and Location of Cyclodextrin-Induced Outer Hair Cell Loss and Hearing Loss. Front. Cell Dev. Biol. 2021, 9, 643709. [Google Scholar] [CrossRef]
- Manohar, S.; Ding, D.; Jiang, H.; Li, L.; Chen, G.D.; Kador, P.; Salvi, R. Combined antioxidants and anti-inflammatory therapies fail to attenuate the early and late phases of cyclodextrin-induced cochlear damage and hearing loss. Hear. Res. 2022, 414, 108409. [Google Scholar] [CrossRef]
- Bratosin, D.; Mitrofan, L.; Palii, C.; Estaquier, J.; Montreuil, J. Novel fluorescence assay using calcein-AM for the determination of human erythrocyte viability and aging. Cytometry A 2005, 66, 78–84. [Google Scholar] [CrossRef]
- Lowenheim, H.; Kil, J.; Gultig, K.; Zenner, H.P. Determination of hair cell degeneration and hair cell death in neomycin treated cultures of the neonatal rat cochlea. Hear. Res. 1999, 128, 16–26. [Google Scholar] [CrossRef]
- Cartagena-Rivera, A.X.; Le Gal, S.; Richards, K.; Verpy, E.; Chadwick, R.S. Cochlear outer hair cell horizontal top connectors mediate mature stereocilia bundle mechanics. Sci. Adv. 2019, 5, eaat9934. [Google Scholar] [CrossRef] [PubMed]
- Kolarova, H.; Mosinger, J.; Lenobel, R.; Kejlova, K.; Jirova, D.; Strnad, M. In vitro toxicity testing of supramolecular sensitizers for photodynamic therapy. Toxicol. In Vitro 2003, 17, 775–778. [Google Scholar] [CrossRef]
- Tanaka, Y.; Ishitsuka, Y.; Yamada, Y.; Kondo, Y.; Takeo, T.; Nakagata, N.; Higashi, T.; Motoyama, K.; Arima, H.; Matsuo, M.; et al. Influence of Npc1 genotype on the toxicity of hydroxypropyl-beta-cyclodextrin, a potentially therapeutic agent, in Niemann-Pick Type C disease models. Mol. Genet. Metab. Rep. 2014, 1, 19–30. [Google Scholar] [CrossRef]
- Ignat, S.R.; Dinescu, S.; Varadi, J.; Fenyvesi, F.; Nguyen, T.L.P.; Ciceu, A.; Hermenean, A.; Costache, M. Complexation with Random Methyl-beta-Cyclodextrin and (2-Hydroxypropyl)-beta-Cyclodextrin Promotes Chrysin Effect and Potential for Liver Fibrosis Therapy. Materials (Basel) 2020, 13. [Google Scholar] [CrossRef]
- Yin, Y.; Henzl, M.T.; Lorber, B.; Nakazawa, T.; Thomas, T.T.; Jiang, F.; Langer, R.; Benowitz, L.I. Oncomodulin is a macrophage-derived signal for axon regeneration in retinal ganglion cells. Nat. Neurosci. 2006, 9, 843–852. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Cui, Q.; Gilbert, H.Y.; Yang, Y.; Yang, Z.; Berlinicke, C.; Li, Z.; Zaverucha-do-Valle, C.; He, H.; Petkova, V.; et al. Oncomodulin links inflammation to optic nerve regeneration. Proc. Natl. Acad. Sci. U S A 2009, 106, 19587–19592. [Google Scholar] [CrossRef] [PubMed]
- Kurimoto, T.; Yin, Y.; Habboub, G.; Gilbert, H.Y.; Li, Y.; Nakao, S.; Hafezi-Moghadam, A.; Benowitz, L.I. Neutrophils express oncomodulin and promote optic nerve regeneration. J. Neurosci. Off. J. Soc. Neurosci. 2013, 33, 14816–14824. [Google Scholar] [CrossRef]
- Kwon, M.J.; Kim, J.; Shin, H.; Jeong, S.R.; Kang, Y.M.; Choi, J.Y.; Hwang, D.H.; Kim, B.G. Contribution of macrophages to enhanced regenerative capacity of dorsal root ganglia sensory neurons by conditioning injury. J. Neurosci. 2013, 33, 15095–15108. [Google Scholar] [CrossRef] [PubMed]
- Kwon, M.J.; Seo, Y.; Cho, H.; Kim, H.S.; Oh, Y.J.; Geniscan, S.; Kim, M.; Park, H.H.; Joe, E.H.; Kwon, M.H.; et al. Nanogel-mediated delivery of oncomodulin secreted from regeneration-associated macrophages promotes sensory axon regeneration in the spinal cord. Theranostics 2022, 12, 5856–5876. [Google Scholar] [CrossRef]
- Bork, J.M.; Peters, L.M.; Riazuddin, S.; Bernstein, S.L.; Ahmed, Z.M.; Ness, S.L.; Polomeno, R.; Ramesh, A.; Schloss, M.; Srisailpathy, C.R.; et al. Usher syndrome 1D and nonsyndromic autosomal recessive deafness DFNB12 are caused by allelic mutations of the novel cadherin-like gene CDH23. Am. J. Hum. Genet. 2001, 68, 26–37. [Google Scholar] [CrossRef]
- Astuto, L.M.; Bork, J.M.; Weston, M.D.; Askew, J.W.; Fields, R.R.; Orten, D.J.; Ohliger, S.J.; Riazuddin, S.; Morell, R.J.; Khan, S.; et al. CDH23 mutation and phenotype heterogeneity: a profile of 107 diverse families with Usher syndrome and nonsyndromic deafness. Am. J. Hum. Genet. 2002, 71, 262–275. [Google Scholar] [CrossRef]
- Mianne, J.; Chessum, L.; Kumar, S.; Aguilar, C.; Codner, G.; Hutchison, M.; Parker, A.; Mallon, A.M.; Wells, S.; Simon, M.M.; et al. Correction of the auditory phenotype in C57BL/6N mice via CRISPR/Cas9-mediated homology directed repair. Genome Med. 2016, 8, 16. [Google Scholar] [CrossRef]
- Miyasaka, Y.; Shitara, H.; Suzuki, S.; Yoshimoto, S.; Seki, Y.; Ohshiba, Y.; Okumura, K.; Taya, C.; Tokano, H.; Kitamura, K.; et al. Heterozygous mutation of Ush1g/Sans in mice causes early-onset progressive hearing loss, which is recovered by reconstituting the strain-specific mutation in Cdh23. Hum. Mol. Genet. 2016, 25, 2045–2059. [Google Scholar] [CrossRef]
- Johnson, K.R.; Tian, C.; Gagnon, L.H.; Jiang, H.; Ding, D.; Salvi, R. Effects of Cdh23 single nucleotide substitutions on age-related hearing loss in C57BL/6 and 129S1/Sv mice and comparisons with congenic strains. Sci. Rep. 2017, 7, 44450. [Google Scholar] [CrossRef]
- Yasuda, S.P.; Seki, Y.; Suzuki, S.; Ohshiba, Y.; Hou, X.; Matsuoka, K.; Wada, K.; Shitara, H.; Miyasaka, Y.; Kikkawa, Y. c. Hear. Res. 2020, 389, 107926. [CrossRef]
- Esterberg, R.; Hailey, D.W.; Coffin, A.B.; Raible, D.W.; Rubel, E.W. Disruption of intracellular calcium regulation is integral to aminoglycoside-induced hair cell death. J. Neurosci. 2013, 33, 7513–7525. [Google Scholar] [CrossRef]
- Esterberg, R.; Hailey, D.W.; Rubel, E.W.; Raible, D.W. ER-mitochondrial calcium flow underlies vulnerability of mechanosensory hair cells to damage. J. Neurosci. 2014, 34, 9703–9719. [Google Scholar] [CrossRef] [PubMed]
- Esterberg, R.; Linbo, T.; Pickett, S.B.; Wu, P.; Ou, H.C.; Rubel, E.W.; Raible, D.W. Mitochondrial calcium uptake underlies ROS generation during aminoglycoside-induced hair cell death. J. Clin. Invest. 2016, 126, 3556–3566. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Wang, W.; Liu, H.; Liu, H.; Wu, H. Cisplatin induces calcium ion accumulation and hearing loss by causing functional alterations in calcium channels and exocytosis. Am. J. Transl. Res. 2019, 11, 6877–6889. [Google Scholar]
- Zhao, H.; Xu, Y.; Song, X.; Zhang, Q.; Wang, Y.; Yin, H.; Bai, X.; Li, J. Cisplatin induces damage of auditory cells: Possible relation with dynamic variation in calcium homeostasis and responding channels. Eur. J. Pharmacol. 2022, 914, 174662. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.S.; et al. Cisplatin drives mitochondrial dysregulation in sensory hair cells. bioRxiv 2024. [Google Scholar] [CrossRef]
- Murtha, K.E.; Sese, W.D.; Sleiman, K.; Halpage, J.; Padyala, P.; Yang, Y.; Hornak, A.J.; Simmons, D.D. Absence of oncomodulin increases susceptibility to noise-induced outer hair cell death and alters mitochondrial morphology. Front. Neurol. 2024, 15, 1435749. [Google Scholar] [CrossRef]
- Murtha, K.E.; Yang, Y.; Ceriani, F.; Jeng, J.Y.; Climer, L.K.; Jones, F.; Charles, J.; Devana, S.K.; Hornak, A.J.; Marcotti, W.; et al. Oncomodulin (OCM) uniquely regulates calcium signaling in neonatal cochlear outer hair cells. Cell Calcium 2022, 105, 102613. [Google Scholar] [CrossRef]
- Yang, Y.; Murtha, K.; Climer, L.K.; Ceriani, F.; Thompson, P.; Hornak, A.J.; Marcotti, W.; Simmons, D.D. Oncomodulin regulates spontaneous calcium signalling and maturation of afferent innervation in cochlear outer hair cells. J. Physiol. 2023, 601, 4291–4308. [Google Scholar] [CrossRef] [PubMed]
- Cheatham, M.A.; Edge, R.M.; Homma, K.; Leserman, E.L.; Dallos, P.; Zheng, J. Prestin-Dependence of Outer Hair Cell Survival and Partial Rescue of Outer Hair Cell Loss in PrestinV499G/Y501H Knockin Mice. PLoS One 2015, 10, e0145428. [Google Scholar] [CrossRef]
- Takahashi, S.; Sun, W.; Zhou, Y.; Homma, K.; Kachar, B.; Cheatham, M.A.; Zheng, J. Prestin Contributes to Membrane Compartmentalization and Is Required for Normal Innervation of Outer Hair Cells. Front. Cell. Neurosci. 2018, 12, 211. [Google Scholar] [CrossRef] [PubMed]
- Lewis, M.A.; Quint, E.; Glazier, A.M.; Fuchs, H.; De Angelis, M.H.; Langford, C.; van Dongen, S.; Abreu-Goodger, C.; Piipari, M.; Redshaw, N.; et al. An ENU-induced mutation of miR-96 associated with progressive hearing loss in mice. Nat. Genet. 2009, 41, 614–618. [Google Scholar] [CrossRef] [PubMed]
- Chessum, L.; Matern, M.S.; Kelly, M.C.; Johnson, S.L.; Ogawa, Y.; Milon, B.; McMurray, M.; Driver, E.C.; Parker, A.; Song, Y.; et al. Helios is a key transcriptional regulator of outer hair cell maturation. Nature 2018, 563, 696–700. [Google Scholar] [CrossRef]
- Melgar-Rojas, P.; Alvarado, J.C.; Fuentes-Santamaria, V.; Gabaldon-Ull, M.C.; Juiz, J.M. Validation of Reference Genes for RT-qPCR Analysis in Noise-Induced Hearing Loss: A Study in Wistar Rat. PLoS One 2015, 10, e0138027. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Tan, X.; Zhou, Y.; Agarwal, A.; Lim, M.; Xu, Y.; Zhu, Y.; O'Brien, J.; Tran, E.; Zheng, J.; Gius, D.; et al. Systemic application of honokiol prevents cisplatin ototoxicity without compromising its antitumor effect. Am. J. Cancer Res. 2020, 10, 4416–4434. [Google Scholar]
- Zheng, J.; Du, G.G.; Matsuda, K.; Orem, A.; Aguinaga, S.; Deak, L.; Navarrete, E.; Madison, L.D.; Dallos, P. The C-terminus of prestin influences nonlinear capacitance and plasma membrane targeting. Journal of cell science 2005, 118, 2987–2996. [Google Scholar] [CrossRef]
- Tan, X.; Jahan, I.; Xu, Y.; Stock, S.; Kwan, C.C.; Soriano, C.; Xiao, X.; Garcia-Anoveros, J.; Fritzsch, B.; Richter, C.P. Auditory Neural Activity in Congenitally Deaf Mice Induced by Infrared Neural Stimulation. Sci. Rep. 2018, 8, 388. [Google Scholar] [CrossRef]
- Chung, Y.; Koehler, S.D.; Cancelarich, S.; Gibson, T.M.; Pregernig, G.; Becker, L.; Artinian, Q.A.; Goodliffe, J.W.; Pan, N.; Quigley, T.M.; et al. Functional, sustained recovery of hearing in Otoferlin-deficient mice using DB-OTO, a hair-cell-specific AAV-based gene therapy. Mol. Ther. Methods Clin. Dev. 2025, 33, 101577. [Google Scholar] [CrossRef] [PubMed]
- Edelstein, A.D.; Tsuchida, M.A.; Amodaj, N.; Pinkard, H.; Vale, R.D.; Stuurman, N. Advanced methods of microscope control using muManager software. J. Biol. Methods 2014, 1. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



