Submitted:
31 March 2026
Posted:
01 April 2026
You are already at the latest version
Abstract
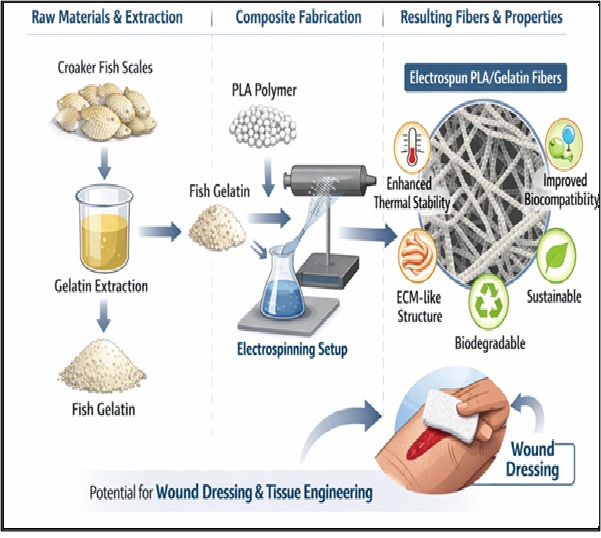
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Extraction of Gelatin
2.2.2. Electrospinning of PLA/Gelatin
2.3. Characterisation of Electrospun PLA/Gelatin Scaffolds
3. Results and Discussion
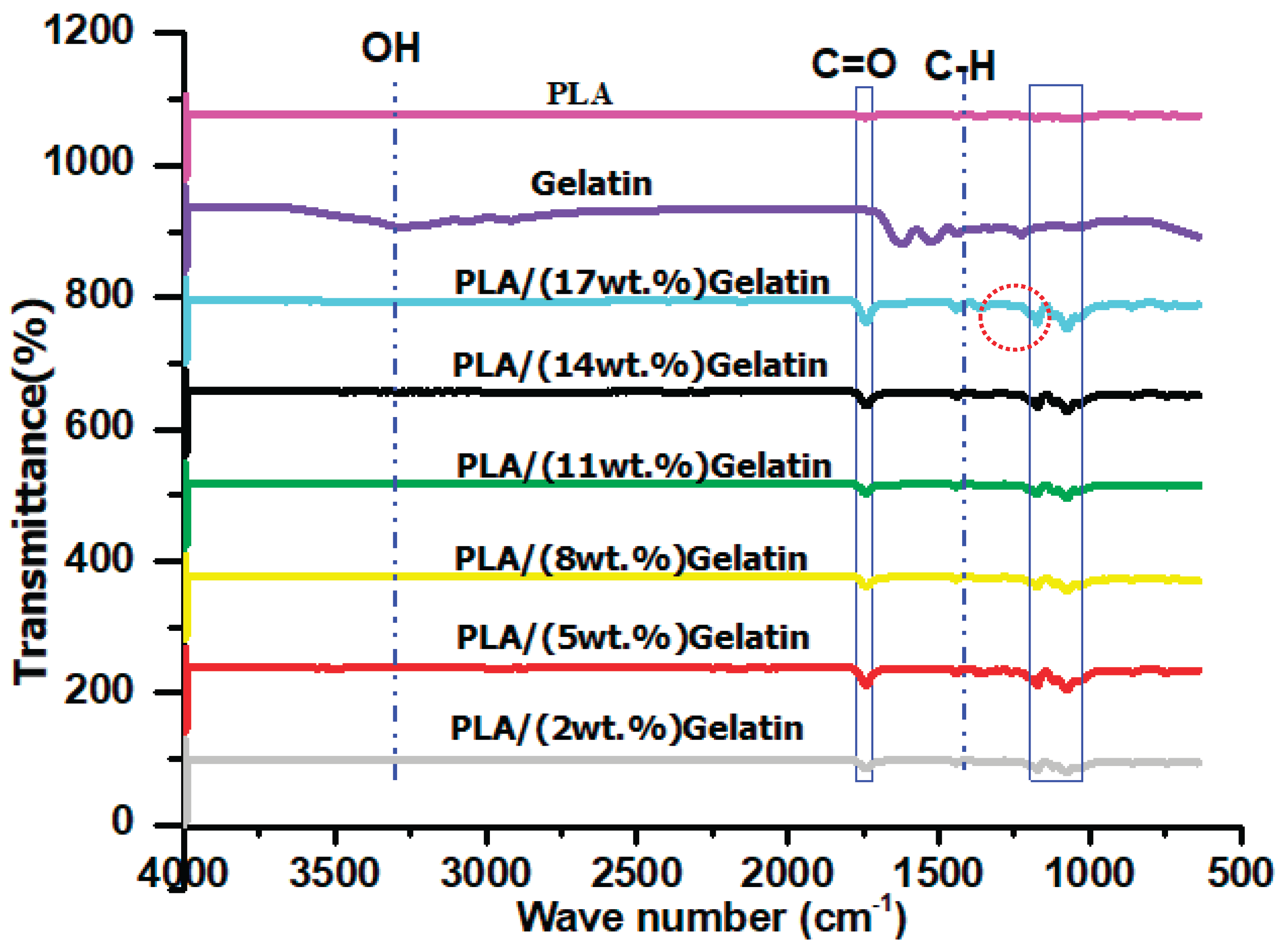
3.1. Chemical Analysis of Electrospun PLA and PLA/Gelatin Scaffold
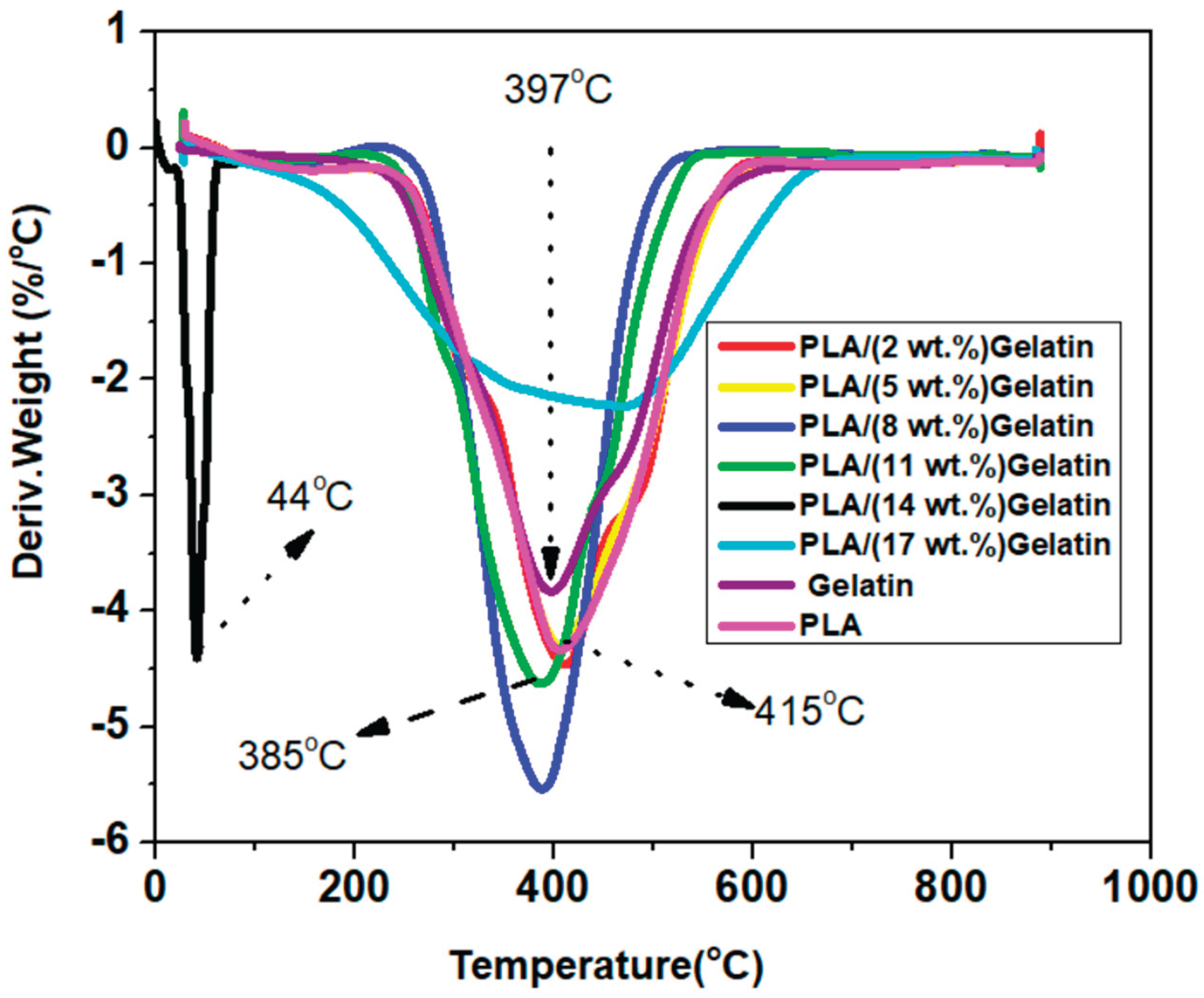
3.2. Thermal Properties of Electrospun PLA and PLA/Gelatin Scaffold
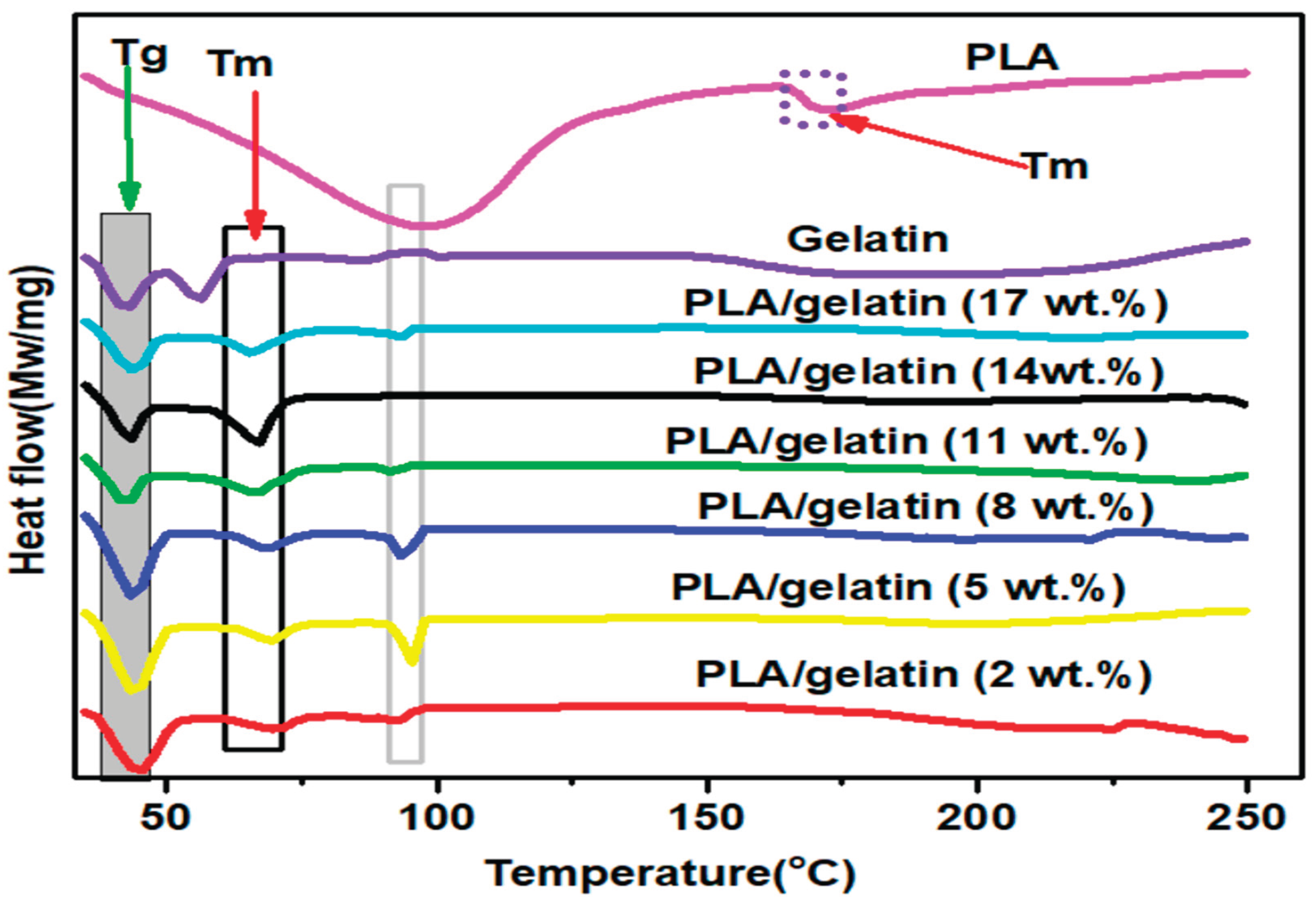
3.3. Glass Transition Properties of Electrospun PLA and PLA/Gelatin Scaffold
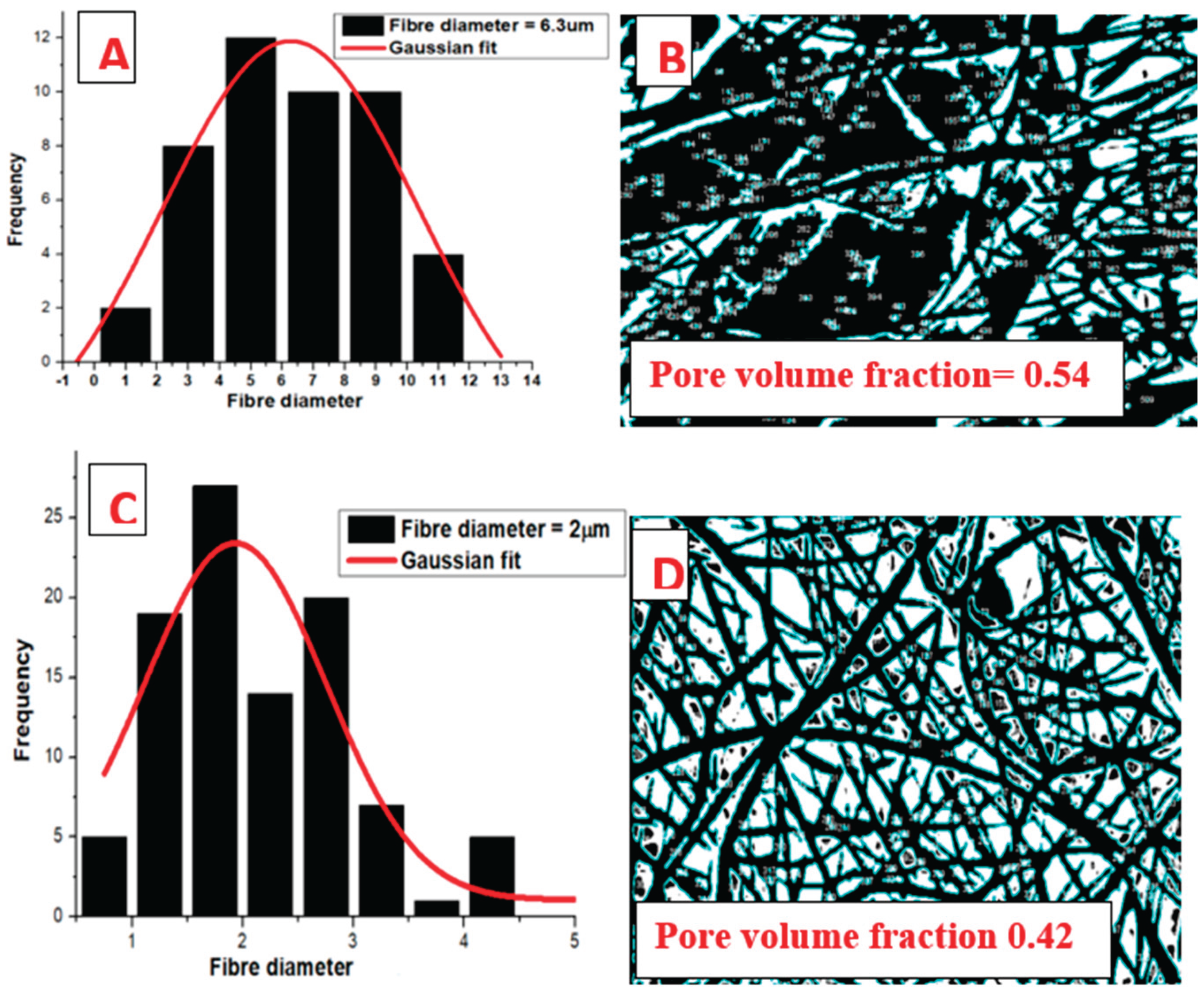
3.4. Morphological Evaluations of Electrospun PLA and PLA/Gelatin Scaffold
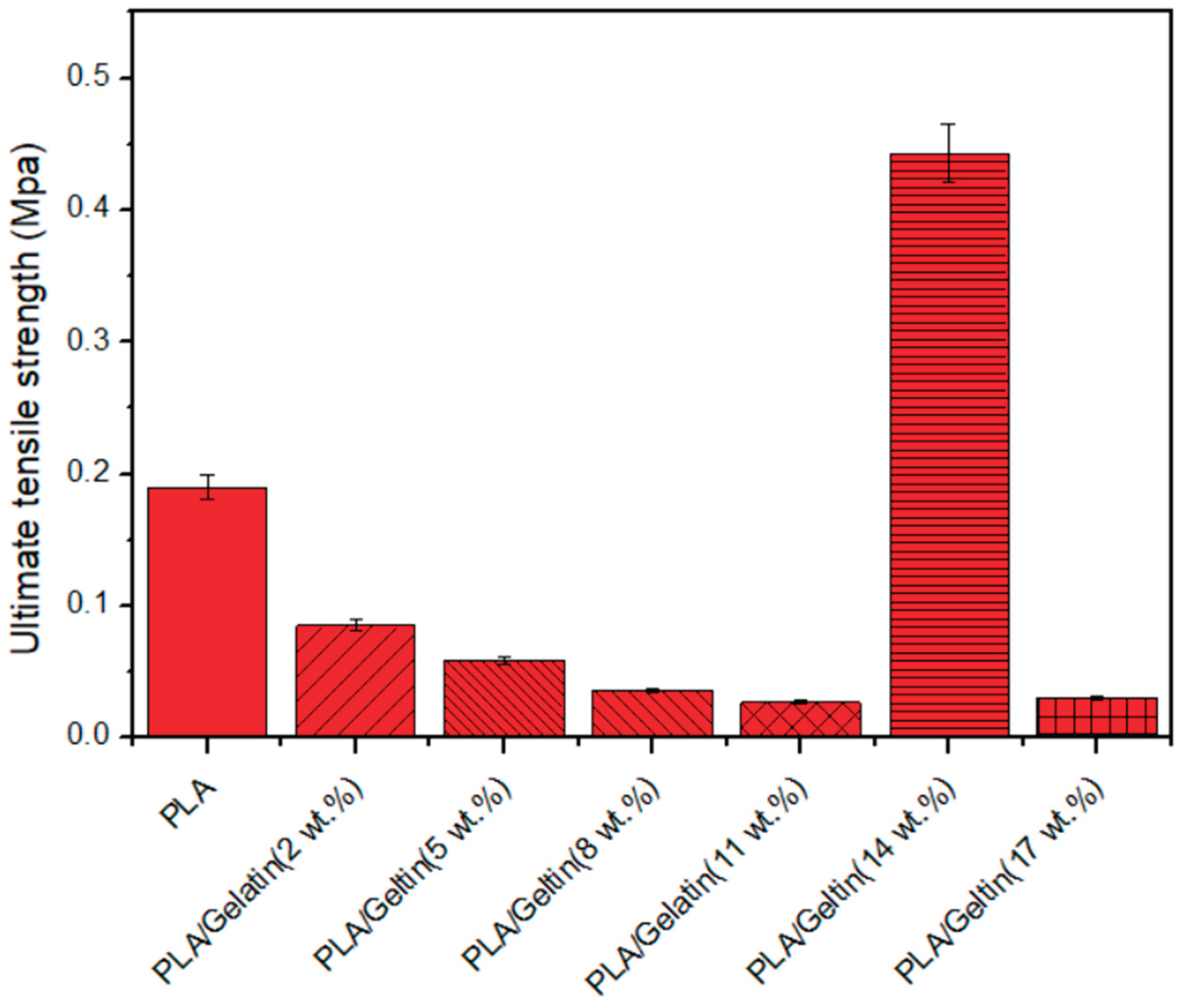
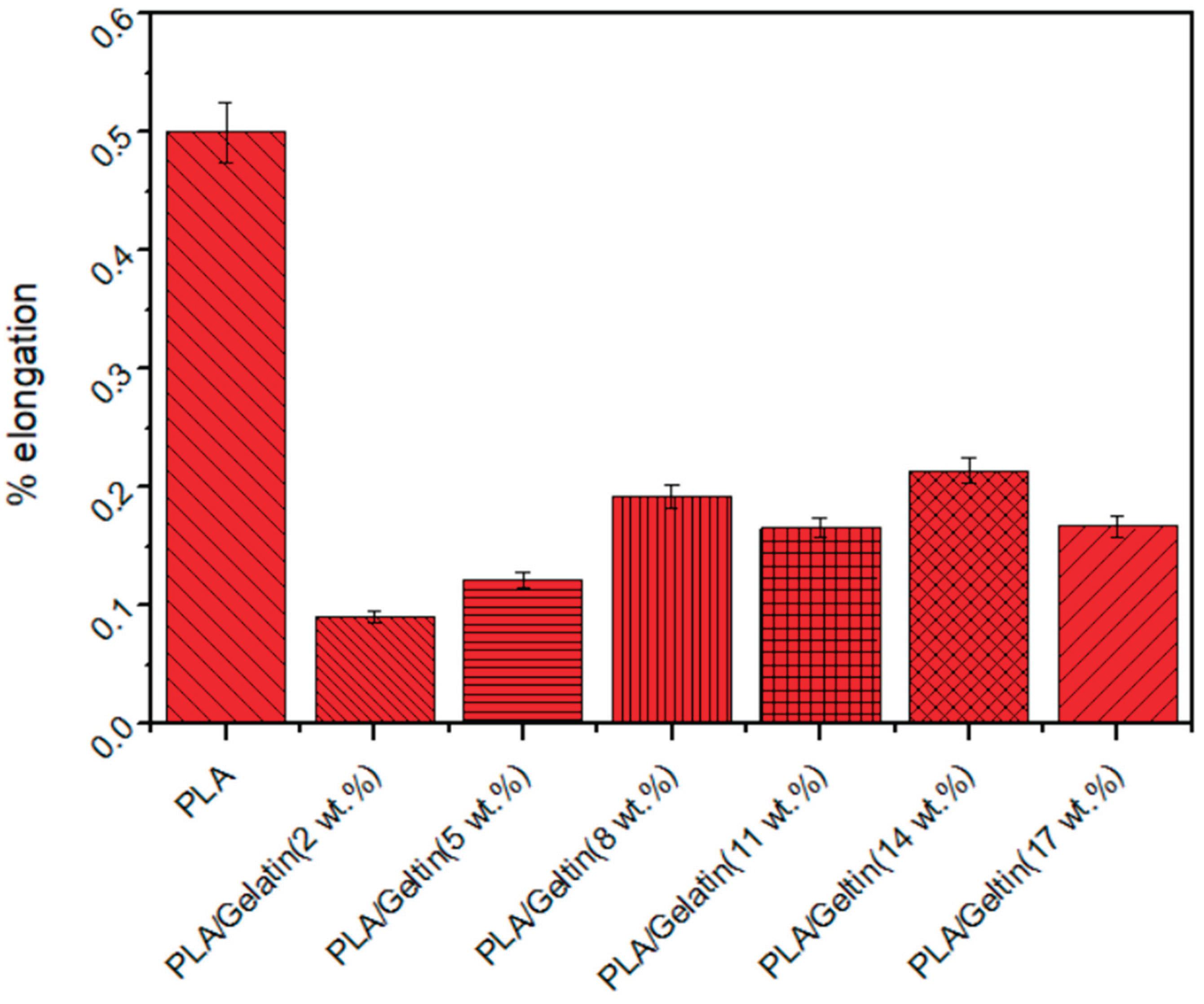
3.5. Strength Characteristics of Electrospun PLA and PLA/Gelatin Scaffold
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| TE | Tissue engineering |
| RM | Regenerative medicine |
References
- Imani, F.; Karimi-Soflou, R.; Shabani, I.; Karkhaneh, A. PLA electrospun nanofibers modified with polypyrrole-grafted gelatin as bioactive electroconductive scaffold. Polymer 2021, 218. [Google Scholar] [CrossRef]
- Huang, Z.-M.; Zhang, Y.; Ramakrishna, S.; Lim, C. Electrospinning and mechanical characterization of gelatin nanofibers. Polymer 2004, 45, 5361–5368. [Google Scholar] [CrossRef]
- Murphy, S.V.; Atala, A. 3D bioprinting of tissues and organs. Nat. Biotechnol. 2014, 32, 773–785. [Google Scholar] [CrossRef]
- El-Seedi, H.R.; Said, N.S.; Yosri, N.; Hawash, H.B.; El-Sherif, D.M.; Abouzid, M.; Abdel-Daim, M.M.; Yaseen, M.; Omar, H.; Shou, Q.; et al. Gelatin nanofibers: Recent insights in synthesis, bio-medical applications and limitations. Heliyon 2023, 9, e16228. [Google Scholar] [CrossRef]
- Bahraminasab, M.; Doostmohammadi, N.; Talebi, A.; Arab, S.; Alizadeh, A.; Ghanbari, A.; Salati, A. 3D printed polylactic acid/gelatin-nano-hydroxyapatite/platelet-rich plasma scaffold for critical-sized skull defect regeneration. Biomed. Eng. Online 2022, 21, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Wang, H.; Zhang, J.; Jiang, L.; Zhang, W.; Hu, Y. A facile design of EGF conjugated PLA/gelatin electrospun nanofibers for nursing care of in vivo wound healing applications. J. Ind. Text. 2020, 51, 420S–440S. [Google Scholar] [CrossRef]
- Muthukrishnan, L. An overview on electrospinning and its advancement toward hard and soft tissue engineering applications. Colloid Polym. Sci. 2022, 300, 875–901. [Google Scholar] [CrossRef]
- An, K.; Liu, H.; Guo, S.; Kumar, D.; Wang, Q. Preparation of fish gelatin and fish gelatin/poly(l-lactide) nanofibers by electrospinning. Int. J. Biol. Macromol. 2010, 47, 380–388. [Google Scholar] [CrossRef]
- Zhao, W.; Li, J.; Jin, K.; Liu, W.; Qiu, X.; Li, C. Fabrication of functional PLGA-based electrospun scaffolds and their applications in biomedical engineering. Mater. Sci. Eng. C 2016, 59, 1181–1194. [Google Scholar] [CrossRef]
- Khorshidi, S.; Solouk, A.; Mirzadeh, H.; Mazinani, S.; Lagaron, J.M.; Sharifi, S.; Ramakrishna, S. A review of key challenges of electrospun scaffolds for tissue-engineering applications. J. Tissue Eng. Regen. Med. 2015, 10, 715–738. [Google Scholar] [CrossRef]
- Zhang, Y.Z.; Venugopal, J.; Huang, Z.-M.; Lim, C.T.; Ramakrishna, S. Crosslinking of the electrospun gelatin nanofibers. Polymer 2006, 47, 2911–2917. [Google Scholar] [CrossRef]
- Chen, H.; Zhang, H.; Shen, Y.; Dai, X.; Wang, X.; Deng, K.; Long, X.; Liu, L.; Zhang, X.; Li, Y.; et al. Instant in-situ Tissue Repair by Biodegradable PLA/Gelatin Nanofibrous Membrane Using a 3D Printed Handheld Electrospinning Device. Front. Bioeng. Biotechnol. 2021, 9. [Google Scholar] [CrossRef] [PubMed]
- Magiera, A.; Markowski, J.; Menaszek, E.; Pilch, J.; Blazewicz, S. PLA-Based Hybrid and Composite Electrospun Fibrous Scaffolds as Potential Materials for Tissue Engineering. J. Nanomater. 2017, 2017, 1–11. [Google Scholar] [CrossRef]
- Torricelli, P.; Gioffrè, M.; Fiorani, A.; Panzavolta, S.; Gualandi, C.; Fini, M.; Focarete, M.L.; Bigi, A. Co-electrospun gelatin-poly(l-lactic acid) scaffolds: Modulation of mechanical properties and chondrocyte response as a function of composition. Mater. Sci. Eng. C 2014, 36, 130–138. [Google Scholar] [CrossRef]
- Bogdanova, A.; Pavlova, E.; Polyanskaya, A.; Volkova, M.; Biryukova, E.; Filkov, G.; Trofimenko, A.; Durymanov, M.; Klinov, D.; Bagrov, D. Acceleration of Electrospun PLA Degradation by Addition of Gelatin. Int. J. Mol. Sci. 2023, 24, 3535. [Google Scholar] [CrossRef]
- Karim, A.A.; Bhat, R. Fish gelatin: properties, challenges, and prospects as an alternative to mammalian gelatins. Food Hydrocoll. 2009, 23, 563–576. [Google Scholar] [CrossRef]
- Feng, X.; Zhu, H.; Wang, Y.; Yu, Y.; Dai, H.; Ma, L.; Zhang, Y. Clean and sustainable extraction of gelatin: Effects of microwave and freeze-thaw on the crosslinking degree and hydrogen bond of fish skin collagen. Food Chem. 2024, 470, 142682. [Google Scholar] [CrossRef]
- Yu, E.; Pan, C.; Chen, W.; Ruan, Q.; Luo, X.; Lv, M.; Fang, Y.; Jiang, L.; Ma, H. Gelatin from specific freshwater and saltwater fish extracted using six different methods: Component interactions, structural characteristics, and functional properties. LWT 2023, 191. [Google Scholar] [CrossRef]
- Rashedi, S.; Afshar, S.; Rostami, A.; Ghazalian, M.; Nazockdast, H. Co-electrospun poly(lactic acid)/gelatin nanofibrous scaffold prepared by a new solvent system: morphological, mechanical and in vitro degradability properties. Int. J. Polym. Mater. Polym. Biomater. 2020, 70, 545–553. [Google Scholar] [CrossRef]
- Bagrov, D.V.; Nikishin, I.I.; Pavlova, E.R.; Klinov, D.V. Distribution of polylactide and gelatin in single electrospun nanofibers studied by Raman spectroscopy. STATE-OF-THE-ART TRENDS OF SCIENTIFIC RESEARCH OF ARTIFICIAL AND NATURAL NANOOBJECTS, STRANN-2018; LOCATION OF CONFERENCE, RussiaDATE OF CONFERENCE; p. 040001.
- Nooeaid, P.; Chuysinuan, P.; Pengsuk, C.; Dechtrirat, D.; Lirdprapamongkol, K.; Techasakul, S.; Svasti, J. Polylactic acid microparticles embedded porous gelatin scaffolds with multifunctional properties for soft tissue engineering. J. Sci. Adv. Mater. Devices 2020, 5, 337–345. [Google Scholar] [CrossRef]
- Švachová, V.; Vojtová, L.; Pavliňák, D.; Vojtek, L.; Sedláková, V.; Hyršl, P.; Alberti, M.; Jaroš, J.; Hampl, A.; Jančář, J. Novel electrospun gelatin/oxycellulose nanofibers as a suitable platform for lung disease modeling. Mater. Sci. Eng. C 2016, 67, 493–501. [Google Scholar] [CrossRef] [PubMed]
- Ahlawat, J.; Kumar, V.; Gopinath, P. Carica papaya loaded poly (vinyl alcohol)-gelatin nanofibrous scaffold for potential application in wound dressing. Mater. Sci. Eng. C 2019, 103, 109834. [Google Scholar] [CrossRef]
- Chiou, B.-S.; Jafri, H.; Avena-Bustillos, R.; Gregorski, K.S.; Bechtel, P.J.; Imam, S.H.; Glenn, G.M.; Orts, W.J. Properties of electrospun pollock gelatin/poly(vinyl alcohol) and pollock gelatin/poly(lactic acid) fibers. Int. J. Biol. Macromol. 2013, 55, 214–220. [Google Scholar] [CrossRef]
- Deng, L.; Li, Y.; Zhang, A.; Zhang, H. Characterization and physical properties of electrospun gelatin nanofibrous films by incorporation of nano-hydroxyapatite. Food Hydrocoll. 2020, 103. [Google Scholar] [CrossRef]
- Kotatha, D.; Hirata, M.; Ogino, M.; Uchida, S.; Ishikawa, M.; Furuike, T.; Tamura, H. Preparation and Characterization of Electrospun Gelatin Nanofibers for Use as Nonaqueous Electrolyte in Electric Double-Layer Capacitor. J. Nanotechnol. 2019, 2019, 1–11. [Google Scholar] [CrossRef]
- Caseiro, A.R.; Silva, D.M.; Amorim, I.; Rêma, A.; Pedrosa, S.S.; Branquinho, M.V.; Gomes, P.S.; Fernandes, M.H.R.; Santos, J.D.; Mauricio, A.C.; et al. Processing, Characterization, and in Vivo Evaluation of Poly(l-lactic acid)-Fish Gelatin Electrospun Membranes for Biomedical Applications. ACS Appl. Bio Mater. 2018, 1, 226–236. [Google Scholar] [CrossRef]
- Zia, I.; Mirza, S.; Jolly, R.; Rehman, A.; Ullah, R.; Shakir, M. Trigonella foenum graecum seed polysaccharide coupled nano hydroxyapatite-chitosan: A ternary nanocomposite for bone tissue engineering. Int. J. Biol. Macromol. 2019, 124, 88–101. [Google Scholar] [CrossRef]
- Correia, D.; Padrão, J.; Rodrigues, L.; Dourado, F.; Lanceros-Méndez, S.; Sencadas, V. Thermal and hydrolytic degradation of electrospun fish gelatin membranes. Polym. Test. 2013, 32, 995–1000. [Google Scholar] [CrossRef]
- Leonés, A.; Salaris, V.; Mujica-Garcia, A.; Arrieta, M.P.; Lopez, D.; Lieblich, M.; Kenny, J.M.; Peponi, L. PLA Electrospun Fibers Reinforced with Organic and Inorganic Nanoparticles: A Comparative Study. Molecules 2021, 26, 4925. [Google Scholar] [CrossRef]
- Ortega-Toro, R.; López-Córdoba, A.; Avalos-Belmontes, F. Epoxidised sesame oil as a biobased coupling agent and plasticiser in polylactic acid/thermoplastic yam starch blends. Heliyon 2021, 7, e06176. [Google Scholar] [CrossRef] [PubMed]
- Salaris, V.; García-Obregón, I.S.F.; López, D.; Peponi, L. Fabrication of PLA-Based Electrospun Nanofibers Reinforced with ZnO Nanoparticles and In Vitro Degradation Study. Nanomaterials 2023, 13, 2236. [Google Scholar] [CrossRef] [PubMed]
- Odili, C.C.; Gbenebor, O.P.; Obisike, R.U.; Adeosun, S.O. Strengthening propensity of hydroxyapatite-reinforced polylactide fibre. Discov. Polym. 2025, 2, 1–12. [Google Scholar] [CrossRef]
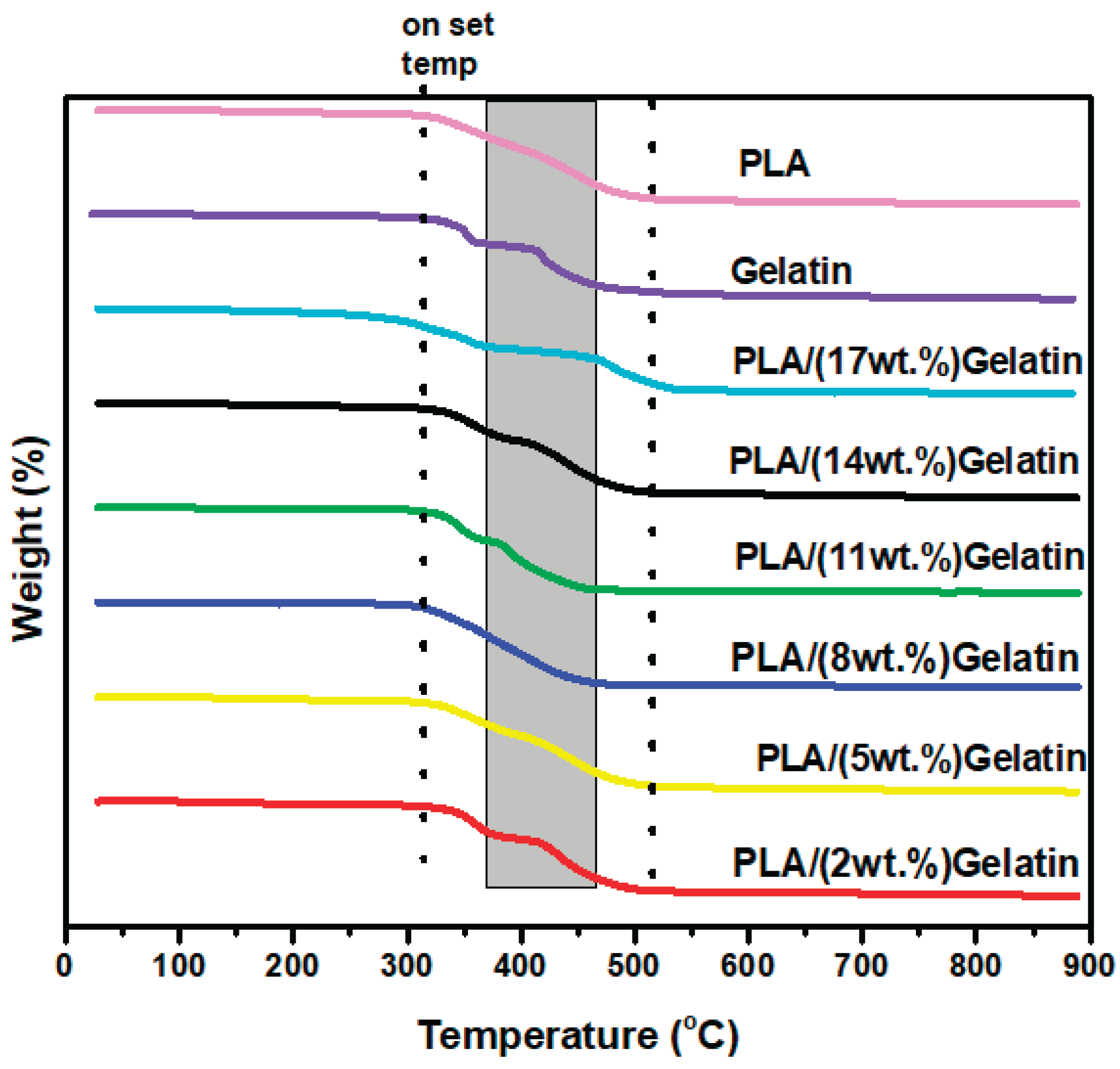

| Sample | On set | End set | Weight loss | First stage | 2nd stage | 3rd stage |
|---|---|---|---|---|---|---|
| PLA | 302 | 500 | 70 % | - | - | - |
| gelatin | 313 | 480 | 170 oc, & 2% | 360 oc, 25% | 43% | |
| PLA/2% gelatin | 321 | 502 | 203 oc ,3wt.% | 377oc, 39 wt.% | 502oc , 54 wt.% | |
| PLA/5% gelatin | 316 | 488 | 81wt.% | 187oc, 3wt.% | - | - |
| PLA/8% gelatin | 314 | 442 | 80WT.% | 161oc, 2wt.5 | - | - |
| PLA/11% gelatin | 322 | 430 | - | 158OC, 2WT.% | 361 oc, 26 wt.% | 44 wt.% |
| PLA/14% gelatin | 324 | 482 | -- | 173oc, 2wt.% | 357oc, 28 wt.% | 50 wt.% |
| PLA/17% gelatin | 271 | 533 | - | 466 | 38wt.% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




