Submitted:
31 March 2026
Posted:
01 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
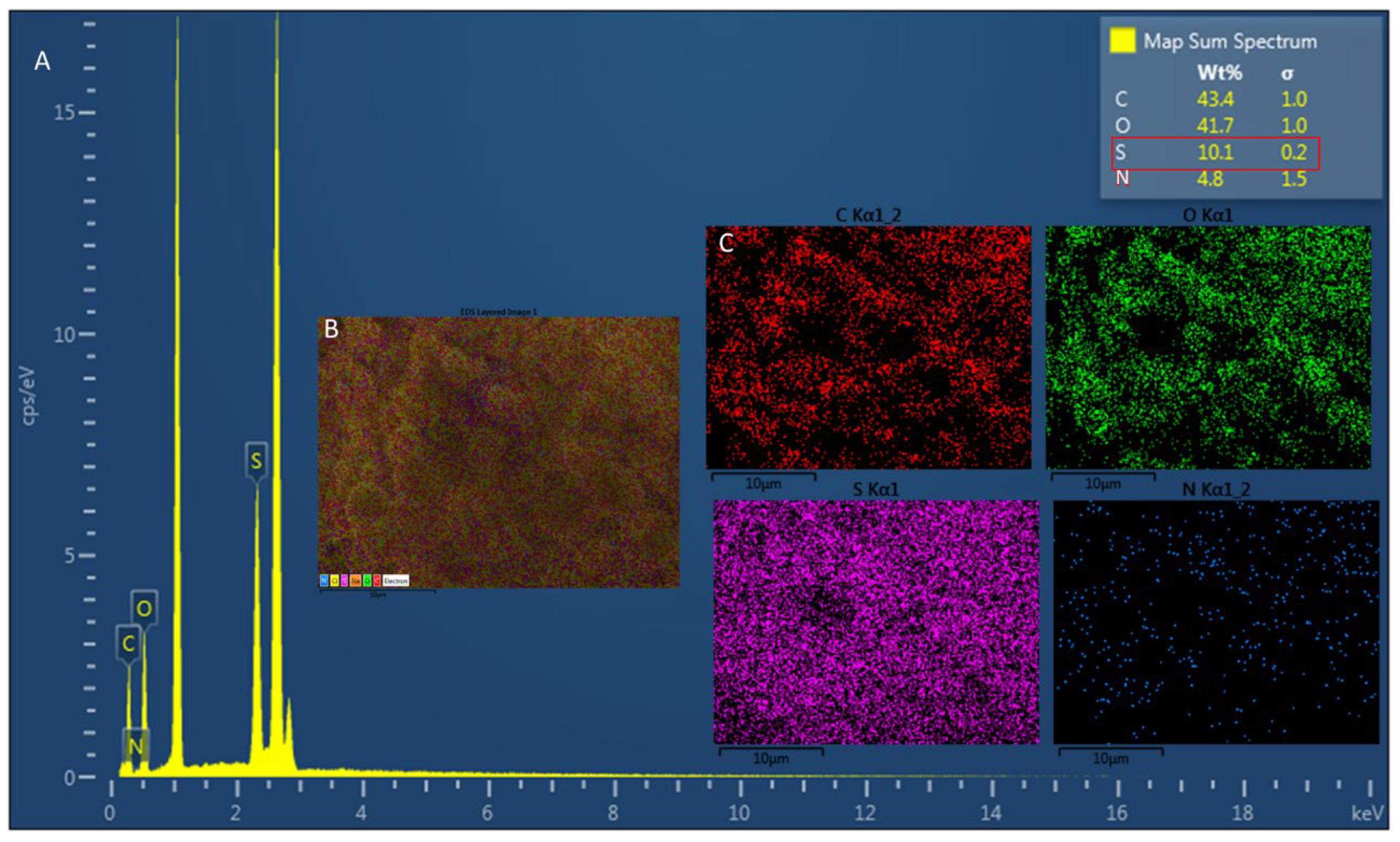
2.1. Elemental Characterization and Degree of Sulfation of Sulfated Chitosan
2.2. Antibacterial Activity
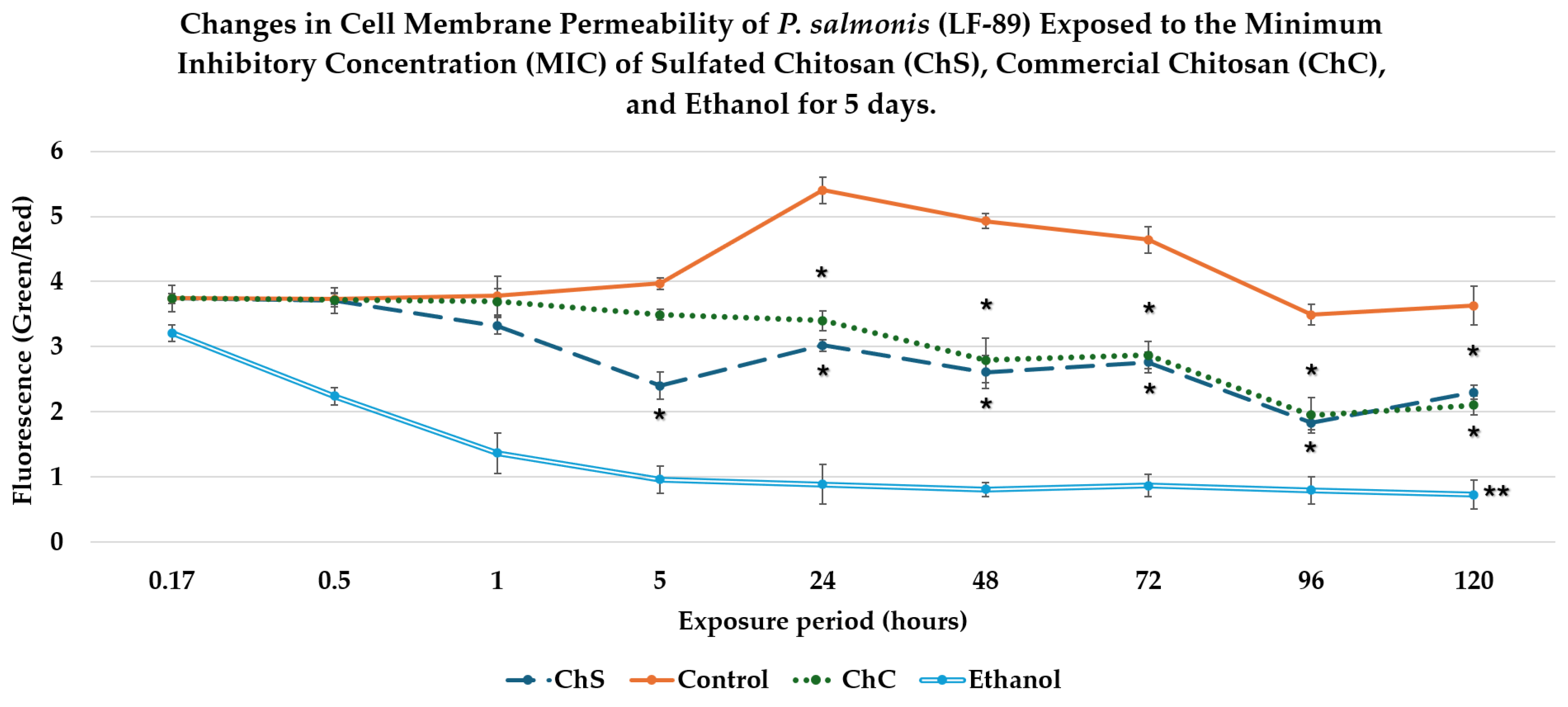
2.2.1. Bacterial Viability and Bacterial Membrane Permeability
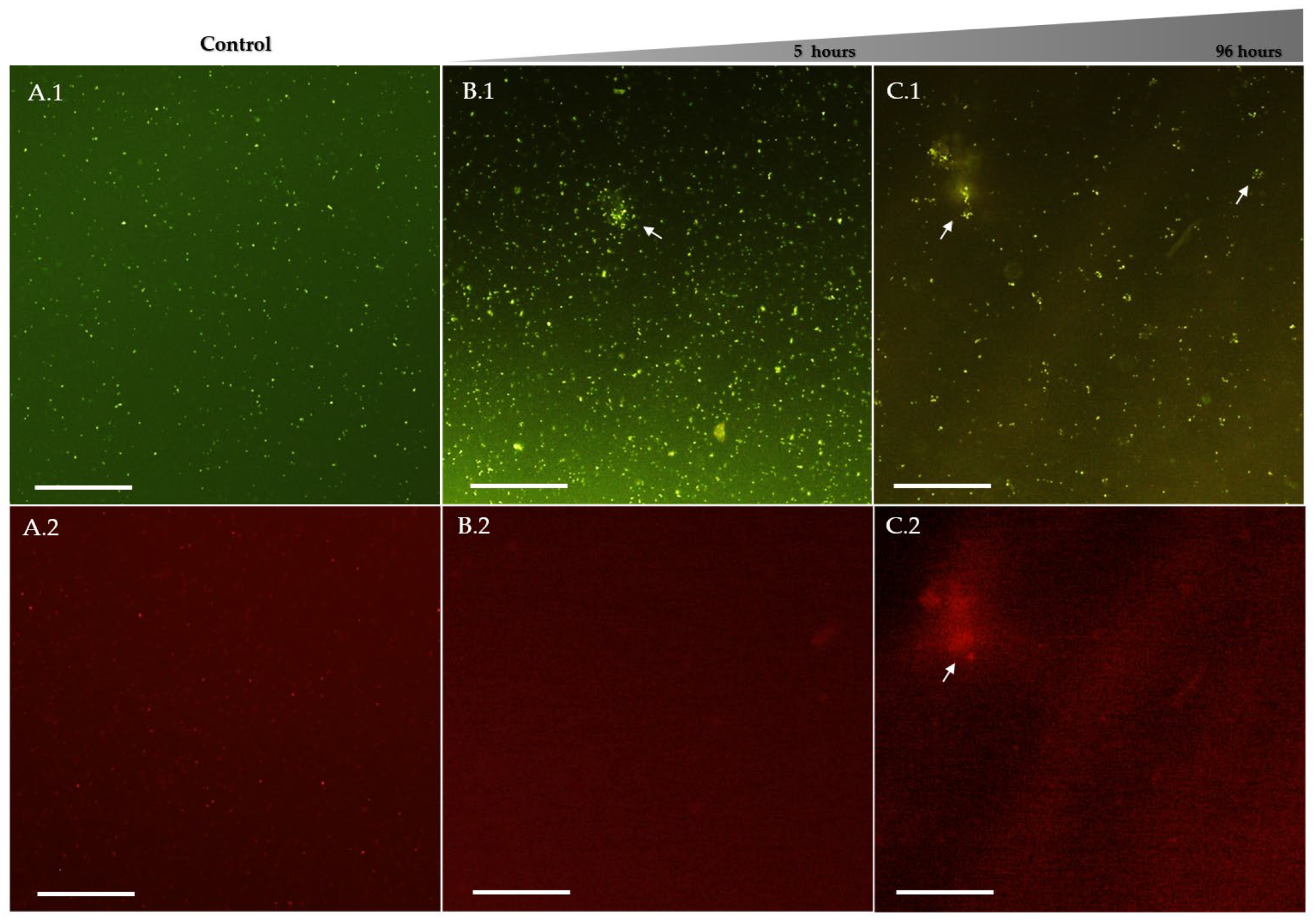
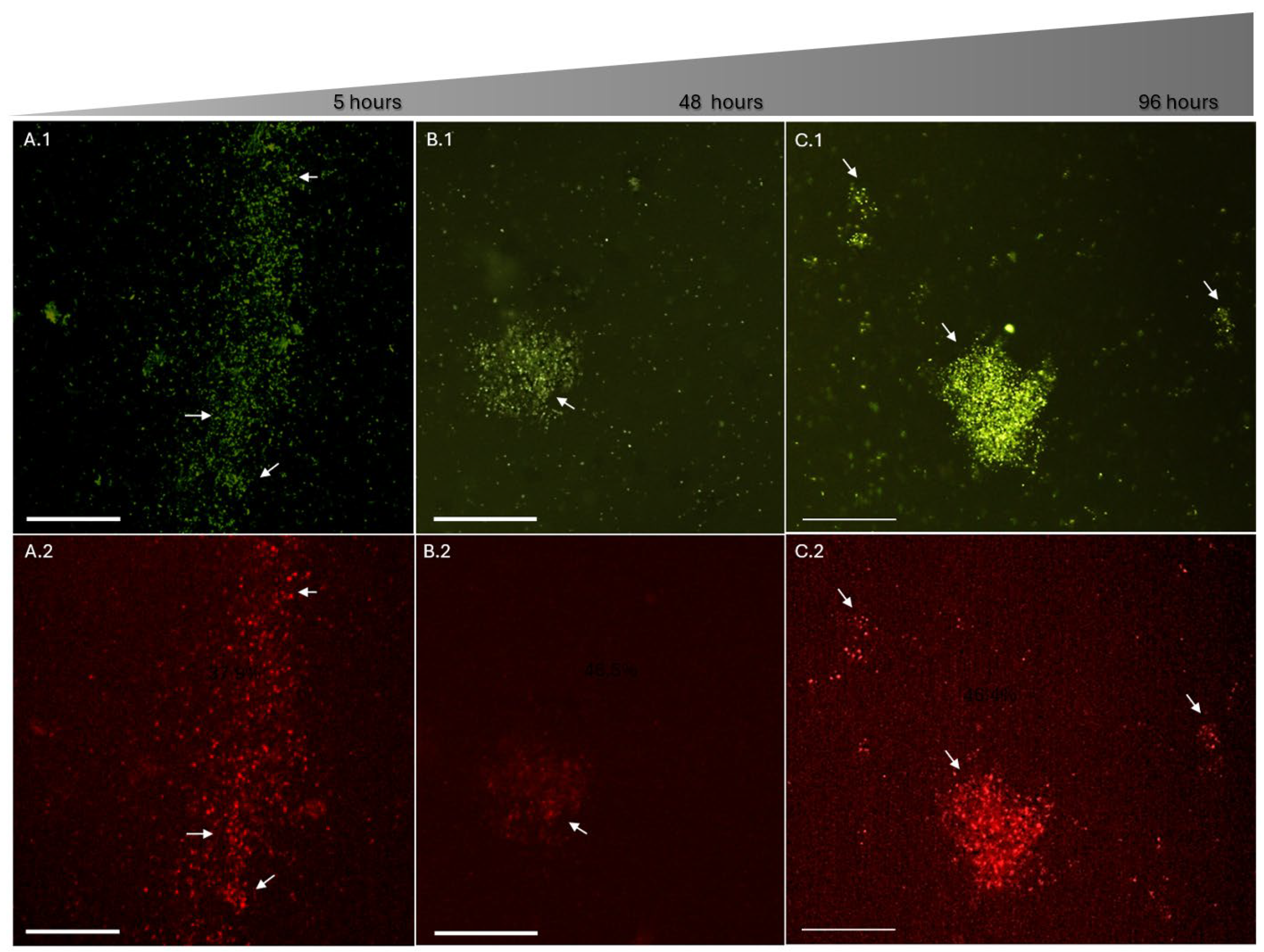
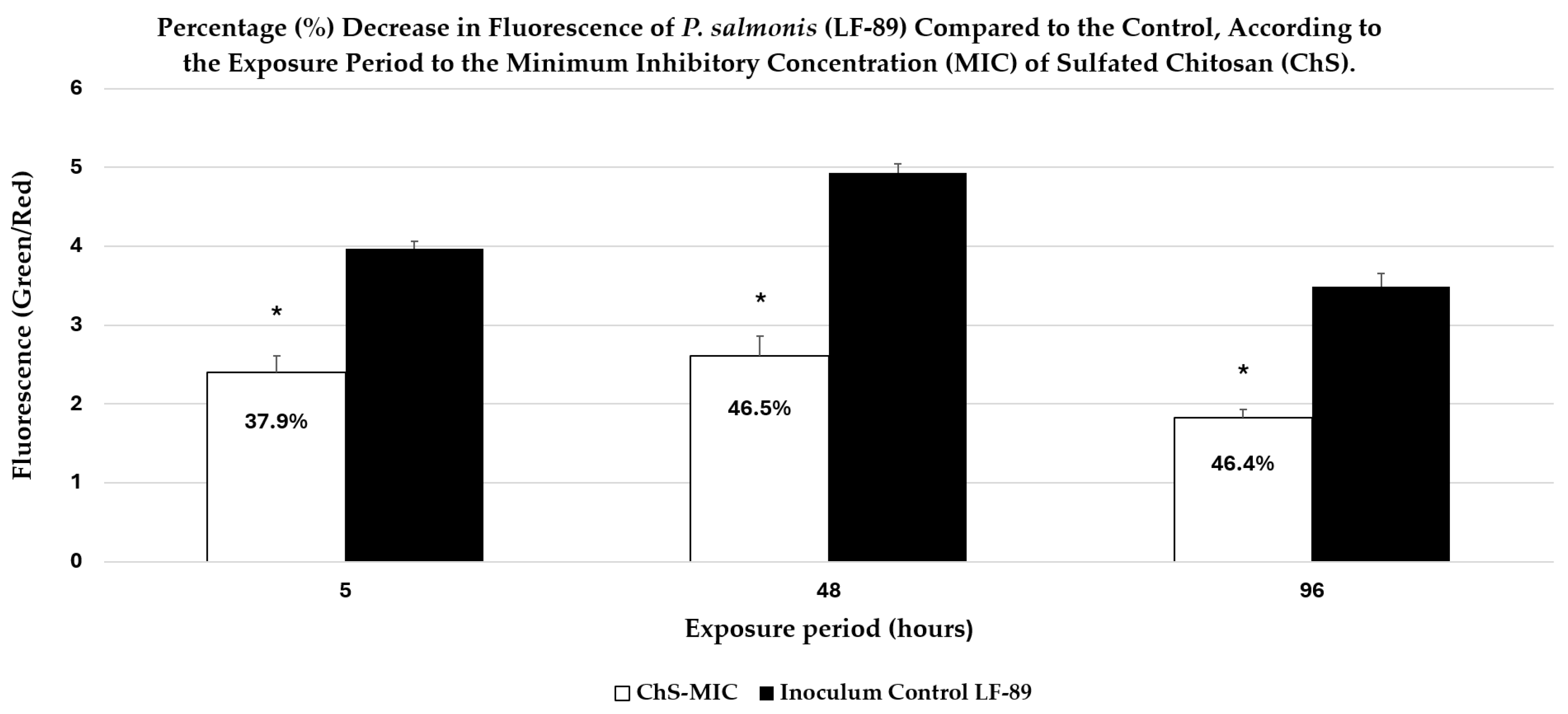
2.3. Fluorescence Microscopy Analysis
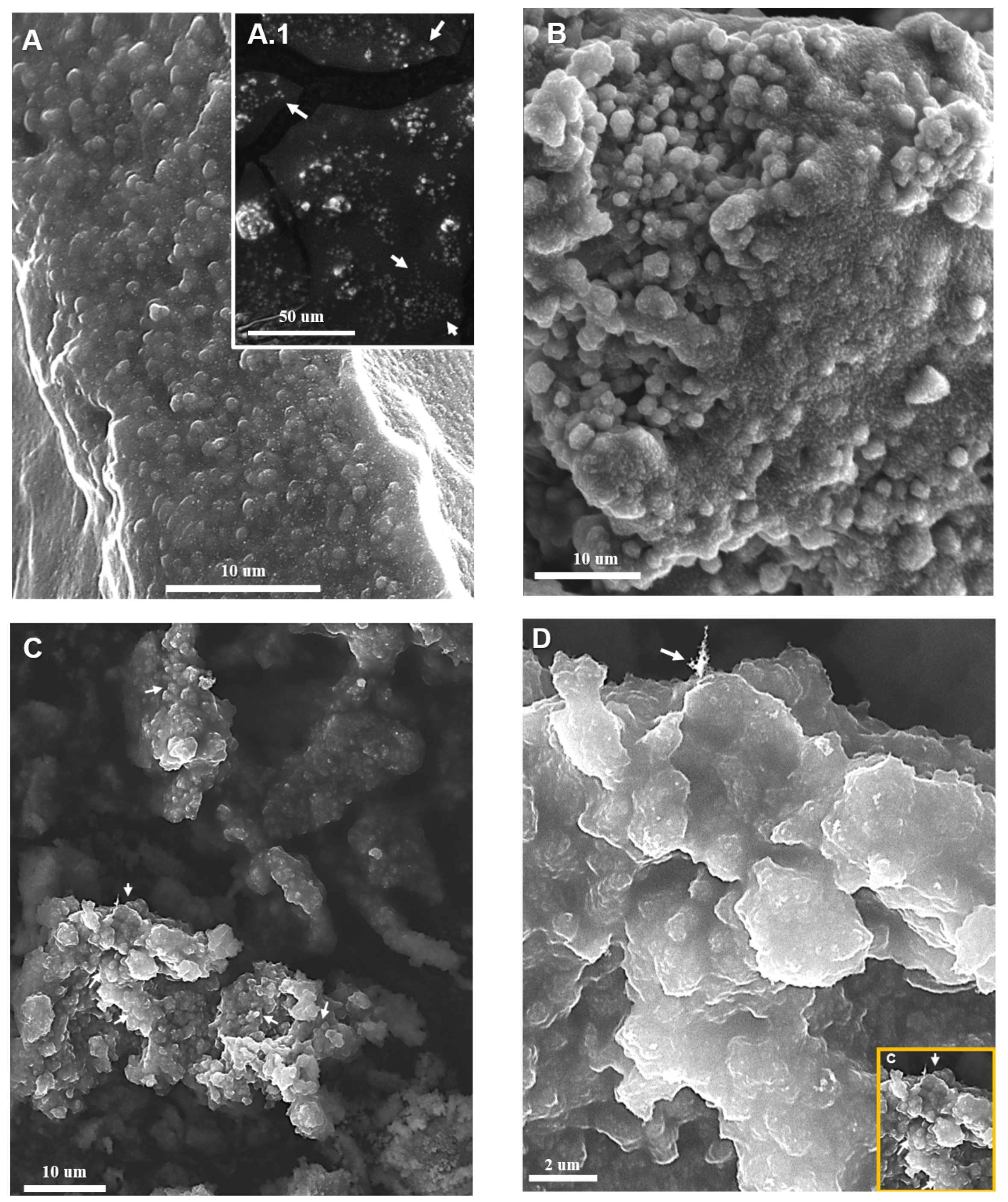
2.4. Scanning Electron Microscopy (SEM) Analysis
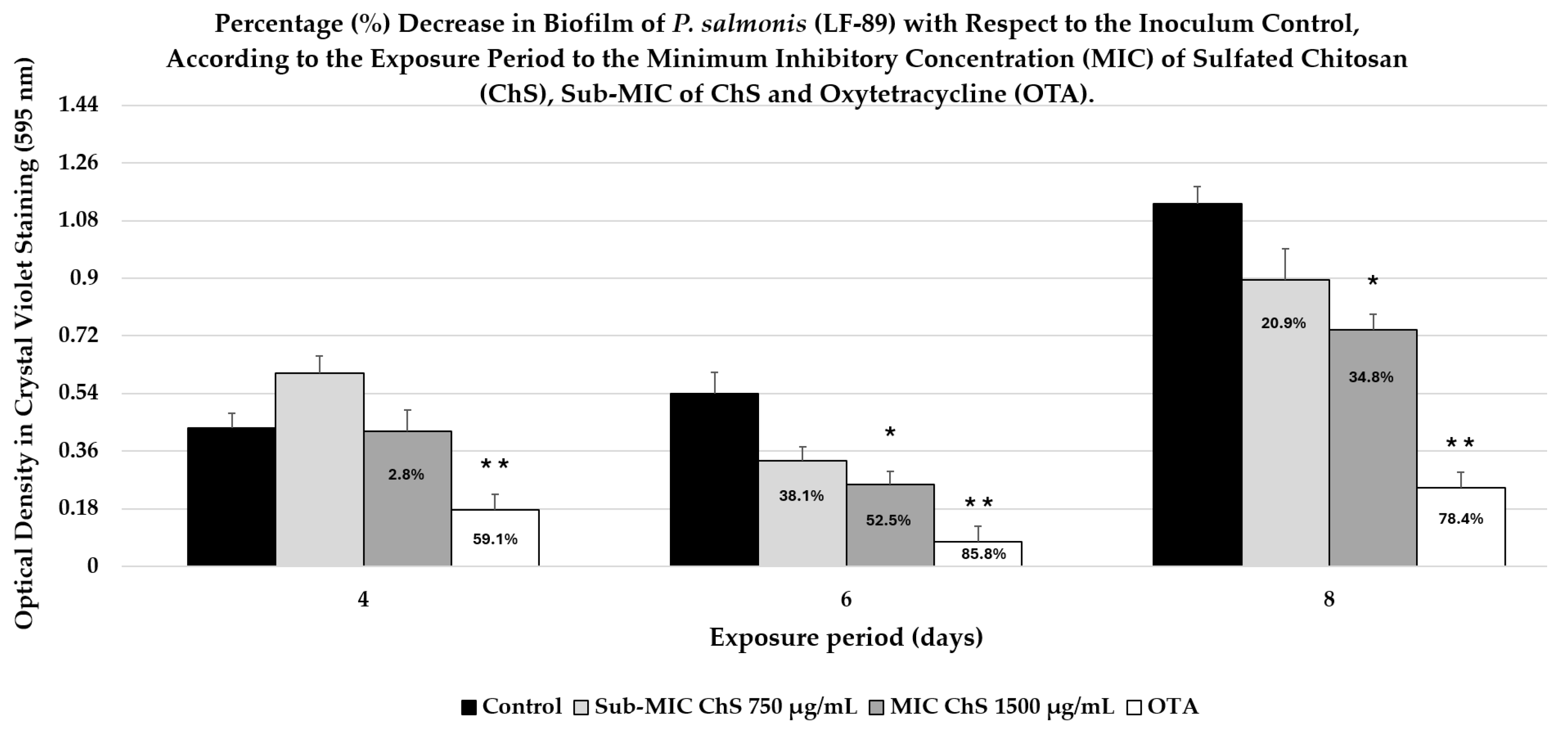
3. Evaluation of Biofilm Production
4. Discussion
5. Materials and Methods
5.1. Degree of Sulfation (DS) Estimation
5.2. Structural and Morphological Characterization of ChS
5.3. Scanning Electron Microscopy (SEM) of Polymer
- a.
- Biopolymer Preparation
- b.
- Antibacterial Activity Assays
- c.
- Minimum Inhibitory Concentration (MIC)
5.4. Minimum Bactericidal Concentration (MBC)
5.5. Biofilm Inhibition Assay
5.6. Bacterial Viability and Membrane Permeability by Fluorescence Microscopy (LIVE/DEAD Imaging)
5.7. Scanning Electron Microscopy (SEM) of Bacteria
5.8. Statistical Analysis
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rozas-Serri, M. Why Does Piscirickettsia Salmonis Break the Immunological Paradigm in Farmed Salmon? Biological Context to Understand the Relative Control of Piscirickettsiosis. Front. Immunol. 2022, 13, 856896. [Google Scholar] [CrossRef]
- Price, D.; Sánchez, J.; McClure, J.; McConkey, S.; Ibarra, R.; St-Hilaire, S. Assessing Concentration of Antibiotics in Tissue during Oral Treatments against Piscirickettsiosis. Preventive Veterinary Medicine 2018, 156, 16–21. [Google Scholar] [CrossRef]
- Diethelm-Varela, B.; Atero, N.; Córdova-Bührle, F.; Rezende, E.L.; Gelcich, S.; Sandoval, O.; Navarro, C.; Mardones, F.O. Epidemiology of Salmonid Rickettsial Septicemia ( SRS ) in Farmed Salmon: The Role of Sea Lice Infestations in Mortality Risk. Journal of Fish Diseases 2025, e70097. [Google Scholar] [CrossRef]
- Cabello, F.C.; Godfrey, H.P.; Tomova, A.; Ivanova, L.; Dölz, H.; Millanao, A.; Buschmann, A.H. Antimicrobial Use in Aquaculture Re-examined: Its Relevance to Antimicrobial Resistance and to Animal and Human Health. Environmental Microbiology 2013, 15, 1917–1942. [Google Scholar] [CrossRef] [PubMed]
- Millanao, A.R.; Barrientos-Schaffeld, C.; Siegel-Tike, C.D.; Tomova, A.; Ivanova, L.; Godfrey, H.P.; Dölz, H.J.; Buschmann, A.H.; Cabello, F.C. Resistencia a Los Antimicrobianos En Chile y El Paradigma de Una Salud: Manejando Los Riesgos Para La Salud Pública Humana y Animal Resultante Del Uso de Antimicrobianos En La Acuicultura Del Salmón y En Medicina. Rev. chil. infectol. 2018, 35, 299–308. [Google Scholar] [CrossRef]
- Souto Cavalli, L.; Tapia-Jopia, C.; Ochs, C.; López Gómez, M.A.; Neis, B. Salmon Mass Mortality Events and Occupational Health and Safety in Chilean Aquaculture. All Life 2023, 16, 2207772. [Google Scholar] [CrossRef]
- Farias, D.R.; Ibarra, R.; Tucca, F.; Jaramillo-Torres, A.; Cornejo, J.; Ibieta, P.; Mardones, F.O.; Avendaño-Herrera, R. Insights and Lessons from Chilean Salmon Aquaculture on Antimicrobial Use. Antibiotics 2025, 14, 1177. [Google Scholar] [CrossRef] [PubMed]
- Rozas, M.; Enríquez, R. Piscirickettsiosis and P Iscirickettsia Salmonis in Fish: A Review. Journal of Fish Diseases 2014, 37, 163–188. [Google Scholar] [CrossRef]
- Oliver, C.; Céspedes, C.; Santibañez, N.; Ruiz, P.; Romero, A. Subinhibitory Concentrations of Florfenicol Increase the Biofilm Formation of Piscirickettsia Salmonis. Journal of Fish Diseases 2023, 46, 591–596. [Google Scholar] [CrossRef]
- San Martín, B.; Fresno, M.; Cornejo, J.; Godoy, M.; Ibarra, R.; Vidal, R.; Araneda, M.; Anadón, A.; Lapierre, L. Optimization of Florfenicol Dose against Piscirickettsia Salmonis in Salmo Salar through PK/PD Studies. PLoS ONE 2019, 14, e0215174. [Google Scholar] [CrossRef]
- Bondad-Reantaso, M.G.; MacKinnon, B.; Karunasagar, I.; Fridman, S.; Alday-Sanz, V.; Brun, E.; Le Groumellec, M.; Li, A.; Surachetpong, W.; Karunasagar, I.; et al. Review of Alternatives to Antibiotic Use in Aquaculture. Reviews in Aquaculture 2023, 15, 1421–1451. [Google Scholar] [CrossRef]
- Valcarcel, J.; Novoa-Carballal, R.; Pérez-Martín, R.I.; Reis, R.L.; Vázquez, J.A. Glycosaminoglycans from Marine Sources as Therapeutic Agents. Biotechnology Advances 2017, 35, 711–725. [Google Scholar] [CrossRef]
- Arrieta-Mendoza, D.; Garces, B.; Hidalgo, A.A.; Neira, V.; Ramirez, G.; Neira-Carrillo, A.; Bucarey, S.A. Design of a New Vaccine Prototype against Porcine Circovirus Type 2 (PCV2), M. Hyopneumoniae and M. Hyorhinis Based on Multiple Antigens Microencapsulation with Sulfated Chitosan. Vaccines 2024, 12, 550. [Google Scholar] [CrossRef]
- Jiménez-Arriagada, D.; Hidalgo, A.A.; Neira, V.; Neira-Carrillo, A.; Bucarey, S.A. Low Molecular Weight Sulfated Chitosan Efficiently Reduces Infection Capacity of Porcine Circovirus Type 2 (PCV2) in PK15 Cells. Virol J 2022, 19, 52. [Google Scholar] [CrossRef] [PubMed]
- Bucarey, S.A.; Ramos, V.; Hidalgo, A.A.; Neira, V.; Neira-Carrillo, A.; Ferrer, P. Low-Molecular-Weight Sulfated Chitosan Microparticles Efficiently Bind HIV-1 In Vitro: Potential for Microbicide Applications. Molecules 2026, 31, 395. [Google Scholar] [CrossRef]
- Bucarey Vivanco, S.A.; Neira Carrillo, A.; Neira Ramirez, V.M. US20210059951 Vaccine Treatment and Control Infectious against Viral Pathogens Utilizing Heparan Sulfate (HS) as Cellular Receptor. 2022. [Google Scholar]
- Sahariah, P.; Másson, M. Antimicrobial Chitosan and Chitosan Derivatives: A Review of the Structure–Activity Relationship. Biomacromolecules 2017, 18, 3846–3868. [Google Scholar] [CrossRef] [PubMed]
- Dimassi, S.; Tabary, N.; Chai, F.; Blanchemain, N.; Martel, B. Sulfonated and Sulfated Chitosan Derivatives for Biomedical Applications: A Review. Carbohydrate Polymers 2018, 202, 382–396. [Google Scholar] [CrossRef] [PubMed]
- Doncel-Pérez, E.; Aranaz, I.; Bastida, A.; Revuelta, J.; Camacho, C.; Acosta, N.; Garrido, L.; Civera, C.; García-Junceda, E.; Heras, A.; et al. Synthesis, Physicochemical Characterization and Biological Evaluation of Chitosan Sulfate as Heparan Sulfate Mimics. Carbohydrate Polymers 2018, 191, 225–233. [Google Scholar] [CrossRef]
- Cesari, A.; Fabiano, A.; Piras, A.M.; Zambito, Y.; Uccello-Barretta, G.; Balzano, F. Binding and Mucoadhesion of Sulfurated Derivatives of Quaternary Ammonium-Chitosans and Their Nanoaggregates: An NMR Investigation. Journal of Pharmaceutical and Biomedical Analysis 2020, 177, 112852. [Google Scholar] [CrossRef]
- Revuelta, J.; Fraile, I.; Monterrey, D.T.; Peña, N.; Benito-Arenas, R.; Bastida, A.; Fernández-Mayoralas, A.; García-Junceda, E. Heparanized Chitosans: Towards the Third Generation of Chitinous Biomaterials. Mater. Horiz. 2021, 8, 2596–2614. [Google Scholar] [CrossRef]
- Žigrayová, D.; Mikušová, V.; Mikuš, P. Advances in Chitosan Derivatives: Preparation, Properties and Applications in Pharmacy and Medicine. Gels 2024, 10, 701. [Google Scholar] [CrossRef]
- Federer, C.; Kurpiers, M.; Bernkop-Schnürch, A. Thiolated Chitosans: A Multi-Talented Class of Polymers for Various Applications. Biomacromolecules 2021, 22, 24–56. [Google Scholar] [CrossRef]
- Muñoz-Bonilla, A.; Echeverria, C.; Sonseca, Á.; Arrieta, M.P.; Fernández-García, M. Bio-Based Polymers with Antimicrobial Properties towards Sustainable Development. Materials 2019, 12, 641. [Google Scholar] [CrossRef]
- Ke, C.-L.; Deng, F.-S.; Chuang, C.-Y.; Lin, C.-H. Antimicrobial Actions and Applications of Chitosan. Polymers 2021, 13, 904. [Google Scholar] [CrossRef]
- Si, Z.; Hou, Z.; Vikhe, Y.S.; Thappeta, K.R.V.; Marimuthu, K.; De, P.P.; Ng, O.T.; Li, P.; Zhu, Y.; Pethe, K.; et al. Antimicrobial Effect of a Novel Chitosan Derivative and Its Synergistic Effect with Antibiotics. ACS Appl. Mater. Interfaces 2021, 13, 3237–3245. [Google Scholar] [CrossRef]
- Kong, M.; Chen, X.G.; Xing, K.; Park, H.J. Antimicrobial Properties of Chitosan and Mode of Action: A State of the Art Review. International Journal of Food Microbiology 2010, 144, 51–63. [Google Scholar] [CrossRef]
- Muñoz-Bonilla, A.; Fernández-García, M. Polymeric Materials with Antimicrobial Activity. Progress in Polymer Science 2012, 37, 281–339. [Google Scholar] [CrossRef]
- Ke, C.-L.; Deng, F.-S.; Chuang, C.-Y.; Lin, C.-H. Antimicrobial Actions and Applications of Chitosan. Polymers 2021, 13, 904. [Google Scholar] [CrossRef] [PubMed]
- Egorov, A.R.; Kirichuk, A.A.; Rubanik, V.V.; Rubanik, V.V.; Tskhovrebov, A.G.; Kritchenkov, A.S. Chitosan and Its Derivatives: Preparation and Antibacterial Properties. Materials 2023, 16, 6076. [Google Scholar] [CrossRef] [PubMed]
- Laanoja, J.; Sihtmäe, M.; Vija, H.; Kurvet, I.; Otsus, M.; Šmits, K.; Kahru, A.; Kasemets, K. Particle-Driven Synergistic Antibacterial Effect of Silver–Chitosan Nanocomposites Against Escherichia Coli, Pseudomonas Aeruginosa, and Staphylococcus Aureus. ACS Omega 2025, 10, 27904–27919. [Google Scholar] [CrossRef]
- Raafat, D.; Sahl, H. Chitosan and Its Antimicrobial Potential – a Critical Literature Survey. Microbial Biotechnology 2009, 2, 186–201. [Google Scholar] [CrossRef]
- Yan, D.; Li, Y.; Liu, Y.; Li, N.; Zhang, X.; Yan, C. Antimicrobial Properties of Chitosan and Chitosan Derivatives in the Treatment of Enteric Infections. Molecules 2021, 26, 7136. [Google Scholar] [CrossRef]
- Helander, I.M.; Nurmiaho-Lassila, E.-L.; Ahvenainen, R.; Rhoades, J.; Roller, S. Chitosan Disrupts the Barrier Properties of the Outer Membrane of Gram-Negative Bacteria. International Journal of Food Microbiology 2001, 71, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Khalid, S.; Piggot, T.J.; Samsudin, F. Atomistic and Coarse Grain Simulations of the Cell Envelope of Gram-Negative Bacteria: What Have We Learned? Acc. Chem. Res. 2019, 52, 180–188. [Google Scholar] [CrossRef]
- Feng, P.; Luo, Y.; Ke, C.; Qiu, H.; Wang, W.; Zhu, Y.; Hou, R.; Xu, L.; Wu, S. Chitosan-Based Functional Materials for Skin Wound Repair: Mechanisms and Applications. Front. Bioeng. Biotechnol. 2021, 9, 650598. [Google Scholar] [CrossRef]
- Dasagrandhi, C.; Park, S.; Jung, W.-K.; Kim, Y.-M. Antibacterial and Biofilm Modulating Potential of Ferulic Acid-Grafted Chitosan against Human Pathogenic Bacteria. IJMS 2018, 19, 2157. [Google Scholar] [CrossRef]
- Wrońska, N.; Katir, N.; Miłowska, K.; Hammi, N.; Nowak, M.; Kędzierska, M.; Anouar, A.; Zawadzka, K.; Bryszewska, M.; El Kadib, A.; et al. Antimicrobial Effect of Chitosan Films on Food Spoilage Bacteria. IJMS 2021, 22, 5839. [Google Scholar] [CrossRef] [PubMed]
- Niu, B.; Zhang, G. Effects of Different Nanoparticles on Microbes. Microorganisms 2023, 11, 542. [Google Scholar] [CrossRef] [PubMed]
- Marshall, S.H.; Gómez, F.A.; Ramírez, R.; Nilo, L.; Henríquez, V. Biofilm Generation by Piscirickettsia Salmonis under Growth Stress Conditions: A Putative in Vivo Survival/Persistence Strategy in Marine Environments. Research in Microbiology 2012, 163, 557–566. [Google Scholar] [CrossRef]
- Manik, E.R.; Kaban, J. Synthesis of Sulfated Chitosan Through Sulfation Reaction of Chitosan with Chlorosulfonic Acid in N, N-Dimethylformamide, and Antibacterial Activity Test. JCNaR 2022, 4, 1–8. [Google Scholar] [CrossRef]
- Sun, Z.; Shi, C.; Wang, X.; Fang, Q.; Huang, J. Synthesis, Characterization, and Antimicrobial Activities of Sulfonated Chitosan. Carbohydrate Polymers 2017, 155, 321–328. [Google Scholar] [CrossRef]
- Fatoni, A.; Ahmad, I.N.; Utami, H.; Erjon; Rendowaty, A.; Hidayati, N. Synthesis, Characterization of Chitosan Sulfate Nanoparticles and Their Activity against Bacterial; Bogor, Indonesia, 2023; p. 020007. [Google Scholar]
- Ardean, C.; Davidescu, C.M.; Nemeş, N.S.; Negrea, A.; Ciopec, M.; Duteanu, N.; Negrea, P.; Duda-Seiman, D.; Musta, V. Factors Influencing the Antibacterial Activity of Chitosan and Chitosan Modified by Functionalization. IJMS 2021, 22, 7449. [Google Scholar] [CrossRef]
- Mania, S.; Banach-Kopeć, A.; Staszczyk, K.; Kulesza, J.; Augustin, E.; Tylingo, R. An Influence of Molecular Weight, Deacetylation Degree of Chitosan Xerogels on Their Antimicrobial Activity and Cytotoxicity. Comparison of Chitosan Materials Obtained Using Lactic Acid and CO2 Saturation. Carbohydrate Research 2023, 534, 108973. [Google Scholar] [CrossRef]
- Sila, A.; Bougatef, H.; Capitani, F.; Krichen, F.; Mantovani, V.; Amor, I.B.; Galeotti, F.; Maccari, F.; Nedjar, N.; Volpi, N.; et al. Studies on European Eel Skin Sulfated Glycosaminoglycans: Recovery, Structural Characterization and Anticoagulant Activity. International Journal of Biological Macromolecules 2018, 115, 891–899. [Google Scholar] [CrossRef]
- Revuelta, J.; Rusu, L.; Frances-Gomez, C.; Trapero, E.; Iglesias, S.; Pinilla, E.C.; Blázquez, A.-B.; Gutiérrez-Adán, A.; Konuparamban, A.; Moreno, O.; et al. Synthetic Heparan Sulfate Mimics Based on Chitosan Derivatives Show Broad-Spectrum Antiviral Activity. Commun Biol 2025, 8, 360. [Google Scholar] [CrossRef]
- Maïza, A.; Chantepie, S.; Vera, C.; Fifre, A.; Huynh, M.B.; Stettler, O.; Ouidja, M.O.; Papy-Garcia, D. The Role of Heparan Sulfates in Protein Aggregation and Their Potential Impact on Neurodegeneration. FEBS Letters 2018, 592, 3806–3818. [Google Scholar] [CrossRef]
- Ferreira, A.; Royaux, I.; Liu, J.; Wang, Z.; Su, G.; Moechars, D.; Callewaert, N.; De Muynck, L. The 3-O Sulfation of Heparan Sulfate Proteoglycans Contributes to the Cellular Internalization of Tau Aggregates. BMC Mol and Cell Biol 2022, 23, 61. [Google Scholar] [CrossRef]
- Liao, Y.-E.; Liu, J.; Arnold, K. Heparan Sulfates and Heparan Sulfate Binding Proteins in Sepsis. Front. Mol. Biosci. 2023, 10, 1146685. [Google Scholar] [CrossRef]
- Khan, S.; Faisal, S.; Shams, D.F.; Zia, M.; Nadhman, A. Photo-inactivation of Bacteria in Hospital Effluent via Thiolated Iron-doped Nanoceria. IET Nanobiotechnology 2019, 13, 875–879. [Google Scholar] [CrossRef]
- Schmitz, T.; Hombach, J.; Bernkop-Schnürch, A. Chitosan-N-Acetyl Cysteine Conjugates: In Vitro Evaluation of Permeation Enhancing and P-Glycoprotein Inhibiting Properties. Drug Delivery 2008, 15, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Rajan, A.; Robertson, M.J.; Carter, H.E.; Poole, N.M.; Clark, J.R.; Green, S.I.; Criss, Z.K.; Zhao, B.; Karandikar, U.; Xing, Y.; et al. Enteroaggregative E. Coli Adherence to Human Heparan Sulfate Proteoglycans Drives Segment and Host Specific Responses to Infection. PLoS Pathog 2020, 16, e1008851. [Google Scholar] [CrossRef]
- Sanchez, H.; Hopkins, D.; Demirdjian, S.; Gutierrez, C.; O’Toole, G.A.; Neelamegham, S.; Berwin, B. Identification of Cell-Surface Glycans That Mediate Motility-Dependent Binding and Internalization of Pseudomonas Aeruginosa by Phagocytes. Molecular Immunology 2021, 131, 68–77. [Google Scholar] [CrossRef]
- Zehr, E.P.; Erzen, C.L.; Oshima, K.; Langouet-Astrie, C.J.; LaRiviere, W.B.; Shi, D.; Zhang, F.; McCollister, B.D.; Windham, S.L.; Rizzo, A.N.; et al. Bacterial Pneumonia-Induced Shedding of Epithelial Heparan Sulfate Inhibits the Bactericidal Activity of Cathelicidin in a Murine Model. American Journal of Physiology-Lung Cellular and Molecular Physiology 2024, 326, L206–L212. [Google Scholar] [CrossRef]
- Day, C.J.; Tran, E.N.; Semchenko, E.A.; Tram, G.; Hartley-Tassell, L.E.; Ng, P.S.K.; King, R.M.; Ulanovsky, R.; McAtamney, S.; Apicella, M.A.; et al. Glycan:Glycan Interactions: High Affinity Biomolecular Interactions That Can Mediate Binding of Pathogenic Bacteria to Host Cells. Proc. Natl. Acad. Sci. U.S.A. 2015, 112. [Google Scholar] [CrossRef] [PubMed]
- Yeh, M.; Cheng, K.; Hu, C.; Huang, Y.; Young, J. Novel Protein-Loaded Chondroitin Sulfate–Chitosan Nanoparticles: Preparation and Characterization. Acta Biomaterialia 2011, 7, 3804–3812. [Google Scholar] [CrossRef] [PubMed]
- Serizawa, T.; Yamaguchi, M.; Akashi, M. Alternating Bioactivity of Polymeric Layer-by-Layer Assemblies: Anticoagulation vs Procoagulation of Human Blood. Biomacromolecules 2002, 3, 724–731. [Google Scholar] [CrossRef]
- Keast, D.; Janmohammad, A. The Hemostatic and Wound Healing Effect of Chitosan Following Debridement of Chronic Ulcers. Wounds 2021, 33, 263–270. [Google Scholar] [CrossRef]
- Riske, F.; Schroeder, J.; Belliveau, J.; Kang, X.; Kutzko, J.; Menon, M.K. The Use of Chitosan as a Flocculant in Mammalian Cell Culture Dramatically Improves Clarification Throughput without Adversely Impacting Monoclonal Antibody Recovery. Journal of Biotechnology 2007, 128, 813–823. [Google Scholar] [CrossRef]
- Wang, W.; Xue, C.; Mao, X. Chitosan: Structural Modification, Biological Activity and Application. International Journal of Biological Macromolecules 2020, 164, 4532–4546. [Google Scholar] [CrossRef]
- Ojeda-Hernández, D.D.; Canales-Aguirre, A.A.; Matias-Guiu, J.A.; Matias-Guiu, J.; Gómez-Pinedo, U.; Mateos-Díaz, J.C. Chitosan–Hydroxycinnamic Acids Conjugates: Emerging Biomaterials with Rising Applications in Biomedicine. IJMS 2022, 23, 12473. [Google Scholar] [CrossRef]
- Makshakova, O.N.; Zuev, Y.F. Interaction-Induced Structural Transformations in Polysaccharide and Protein-Polysaccharide Gels as Functional Basis for Novel Soft-Matter: A Case of Carrageenans. Gels 2022, 8, 287. [Google Scholar] [CrossRef]
- Holyavka, M.G.; Goncharova, S.S.; Sorokin, A.V.; Lavlinskaya, M.S.; Redko, Y.A.; Faizullin, D.A.; Baidamshina, D.R.; Zuev, Y.F.; Kondratyev, M.S.; Kayumov, A.R.; et al. Novel Biocatalysts Based on Bromelain Immobilized on Functionalized Chitosans and Research on Their Structural Features. Polymers 2022, 14, 5110. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Wang, D.; Liu, D.; Su, J.; Jin, Y.; Wang, D.; Han, B.; Jiang, Z.; Liu, B. Applications of Chitosan and Its Derivatives in Skin and Soft Tissue Diseases. Front. Bioeng. Biotechnol. 2022, 10, 894667. [Google Scholar] [CrossRef] [PubMed]
- Zeng, K.; Groth, T.; Zhang, K. Recent Advances in Artificially Sulfated Polysaccharides for Applications in Cell Growth and Differentiation, Drug Delivery, and Tissue Engineering. ChemBioChem 2019, 20, 737–746. [Google Scholar] [CrossRef] [PubMed]
- Herrera, V.; Olavarría, N.; Saavedra, J.; Yuivar, Y.; Bustos, P.; Almarza, O.; Mancilla, M. Complete Lipopolysaccharide of Piscirickettsia Salmonis Is Required for Full Virulence in the Intraperitoneally Challenged Atlantic Salmon, Salmo Salar, Model. Front. Cell. Infect. Microbiol. 2022, 12, 845661. [Google Scholar] [CrossRef]
- Oliver, C.; Sánchez, P.; Valenzuela, K.; Hernández, M.; Pontigo, J.P.; Rauch, M.C.; Garduño, R.A.; Avendaño-Herrera, R.; Yáñez, A.J. Subcellular Location of Piscirickettsia Salmonis Heat Shock Protein 60 (Hsp60) Chaperone by Using Immunogold Labeling and Proteomic Analysis. Microorganisms 2020, 8, 117. [Google Scholar] [CrossRef]
- Oliver, C.; Hernández, M.A.; Tandberg, J.I.; Valenzuela, K.N.; Lagos, L.X.; Haro, R.E.; Sánchez, P.; Ruiz, P.A.; Sanhueza-Oyarzún, C.; Cortés, M.A.; et al. The Proteome of Biologically Active Membrane Vesicles from Piscirickettsia Salmonis LF-89 Type Strain Identifies Plasmid-Encoded Putative Toxins. Front. Cell. Infect. Microbiol. 2017, 7, 420. [Google Scholar] [CrossRef]
- Valenzuela-Aviles, P.; Torrealba, D.; Figueroa, C.; Mercado, L.; Dixon, B.; Conejeros, P.; Gallardo-Matus, J. Why Vaccines Fail against Piscirickettsiosis in Farmed Salmon and Trout and How to Avoid It: A Review. Front. Immunol. 2022, 13, 1019404. [Google Scholar] [CrossRef]
- Gerlach, R.; Hensel, M. Protein Secretion Systems and Adhesins: The Molecular Armory of Gram-Negative Pathogens. International Journal of Medical Microbiology 2007, 297, 401–415. [Google Scholar] [CrossRef]
- Sánchez, P.; Oliver, C.; Hernández, M.; Cortés, M.; Cecilia Rauch, M.; Valenzuela, K.; Garduño, R.A.; Avendaño-Herrera, R.; Yáñez, A.J. In Vitro Genomic and Proteomic Evidence of a Type IV Pili-like Structure in the Fish Pathogen Piscirickettsia Salmonis. FEMS Microbiology Letters 2018, 365. [Google Scholar] [CrossRef]
- Levipan, H.A.; Irgang, R.; Opazo, L.F.; Araya-León, H.; Avendaño-Herrera, R. Collective Behavior and Virulence Arsenal of the Fish Pathogen Piscirickettsia Salmonis in the Biofilm Realm. Front. Cell. Infect. Microbiol. 2022, 12, 1067514. [Google Scholar] [CrossRef] [PubMed]
- Nag, M.; Lahiri, D.; Mukherjee, D.; Banerjee, R.; Garai, S.; Sarkar, T.; Ghosh, S.; Dey, A.; Ghosh, S.; Pattnaik, S.; et al. Functionalized Chitosan Nanomaterials: A Jammer for Quorum Sensing. Polymers 2021, 13, 2533. [Google Scholar] [CrossRef] [PubMed]
- Limayem, A.; Patil, S.B.; Mehta, M.; Cheng, F.; Nguyen, M. A Streamlined Study on Chitosan-Zinc Oxide Nanomicelle Properties to Mitigate a Drug-Resistant Biofilm Protection Mechanism. Front. Nanotechnol. 2020, 2, 592739. [Google Scholar] [CrossRef]
- Sahariah, P.; Papi, F.; Merz, K.L.; Sigurjonsson, O.E.; Meyer, R.L.; Nativi, C. Chitosan–Saccharide Conjugates for Eradication of Pseudomonas Aeruginosa Biofilms. RSC Appl. Polym. 2024, 2, 461–472. [Google Scholar] [CrossRef]
- Riahi, A.; Mabudi, H.; Tajbakhsh, E.; Roomiani, L.; Momtaz, H. Optimizing Chitosan Derived from Metapenaeus Affinis: A Novel Anti-Biofilm Agent against Pseudomonas Aeruginosa. AMB Expr 2024, 14, 77. [Google Scholar] [CrossRef]
- Patel, K.K.; Surekha, D.B.; Tripathi, M.; Anjum, Md.M.; Muthu, M.S.; Tilak, R.; Agrawal, A.K.; Singh, S. Antibiofilm Potential of Silver Sulfadiazine-Loaded Nanoparticle Formulations: A Study on the Effect of DNase-I on Microbial Biofilm and Wound Healing Activity. Mol. Pharmaceutics 2019, 16, 3916–3925. [Google Scholar] [CrossRef]
- Huang, J.; Liu, Y.; Yang, L.; Zhou, F. Synthesis of Sulfonated Chitosan and Its Antibiofilm Formation Activity against E. Coli and S. Aureus. International Journal of Biological Macromolecules 2019, 129, 980–988. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Jiang, Y.; Zhu, J.; Huang, J.; Zhang, H. Inhibition of Bacterial Adhesion and Biofilm Formation of Sulfonated Chitosan against Pseudomonas Aeruginosa. Carbohydrate Polymers 2019, 206, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Shen, H.-L.; Pan, J.-N.; Yu, T.; Zhou, W.-W. Ferrous Sulfate/Carboxymethyl Chitosan Agar-Based Film Triggers Ferroptosis in Pseudomonas Aeruginosa Planktonic and Biofilm Cells for Antibacterial Preservation of Fruits and Vegetables. International Journal of Biological Macromolecules 2025, 308, 142697. [Google Scholar] [CrossRef]
- Zúñiga, A.; Solis, C.; Cartes, C.; Nourdin, G.; Yañez, A.; Romero, A.; Haussmann, D.; Figueroa, J. Transcriptional Analysis of Metabolic and Virulence Genes Associated with Biofilm Formation in Piscirickettsia Salmonis Strains. FEMS Microbiology Letters 2020, 367, fnaa180. [Google Scholar] [CrossRef]
- Levipan, H.A.; Irgang, R.; Yáñez, A.; Avendaño-Herrera, R. Improved Understanding of Biofilm Development by Piscirickettsia Salmonis Reveals Potential Risks for the Persistence and Dissemination of Piscirickettsiosis. Sci Rep 2020, 10, 12224. [Google Scholar] [CrossRef]
- Oliver, C.; Ruiz, P.; Vidal, J.M.; Carrasco, C.; Escalona, C.E.; Barros, J.; Sepúlveda, D.; Urrutia, H.; Romero, A. Effect of Florfenicol on Piscirickettsia Salmonis Biofilm Formed in Materials Used in Salmonid Nets, Nylon and high-density Polyethylene. Journal of Fish Diseases 2024, 47, e13862. [Google Scholar] [CrossRef]
- Santibañez, N.; Vega, M.; Pérez, T.; Yáñez, A.; González-Stegmaier, R.; Figueroa, J.; Enríquez, R.; Oliver, C.; Romero, A. Biofilm Produced In Vitro by Piscirickettsia Salmonis Generates Differential Cytotoxicity Levels and Expression Patterns of Immune Genes in the Atlantic Salmon Cell Line SHK-1. Microorganisms 2020, 8, 1609. [Google Scholar] [CrossRef]
- Santibáñez, N.; Vega, M.; Pérez, T.; Enriquez, R.; Escalona, C.E.; Oliver, C.; Romero, A. In Vitro Effects of Phytogenic Feed Additive on Piscirickettsia Salmonis Growth and Biofilm Formation. Journal of Fish Diseases 2024, 47, e13913. [Google Scholar] [CrossRef] [PubMed]
- Escalona, C.E.; Santibañez, N.; Cortés, M.; Arriagada, V.; Ruiz, P.; Fuentes, D.; Romero, A.; Oliver, C. Sub-Inhibitory Concentrations of Florfenicol Modulate the Expression of Biofilm Formation and Antibiotic Resistance-Associated Genes in Biofilm-Embedded Piscirickettsia Salmonis. Journal of Fish Diseases 2026, e70166. [Google Scholar] [CrossRef] [PubMed]
- SERNAPESCA: SERVICIO NACIONAL DE PESCA Y ACUICULTURA; DEPARTAMENTO DE SALUD ANIMAL Programa Sanitario General para la Vigilancia de la Susceptibilidad a Antimicrobianos en la Salmonicultura: Procedimiento de toma de muestras para aislamiento de Piscirickettsia salmonis y análisis para determinación de concentración mínima inhibitoria (CMI), mediante microdilución en caldo; NORMA TÉCNICA No4; Servicio Nacional de Pesca y Acuicultura (SERNAPESCA): Santiago, Chile, 2020; p. 16.
- Mikalsen, J.; Skjærvik, O.; Wiik-Nielsen, J.; Wasmuth, M.A.; Colquhoun, D.J. Agar Culture of Piscirickettsia Salmonis, a Serious Pathogen of Farmed Salmonid and Marine Fish. FEMS Microbiology Letters 2008, 278, 43–47. [Google Scholar] [CrossRef]
- Vera, T.; Isla, A.; Cuevas, A.; Figueroa, J. Un Nuevo Medio de Cultivo Líquido Para El Patógeno Piscirickettsia Salmonis. Arch. med. vet. 2012, 44, 273–277. [Google Scholar] [CrossRef]
- Peeters, E.; Nelis, H.J.; Coenye, T. Comparison of Multiple Methods for Quantification of Microbial Biofilms Grown in Microtiter Plates. Journal of Microbiological Methods 2008, 72, 157–165. [Google Scholar] [CrossRef]
- Amador, C.I.; Stannius, R.O.; Røder, H.L.; Burmølle, M. High-Throughput Screening Alternative to Crystal Violet Biofilm Assay Combining Fluorescence Quantification and Imaging. Journal of Microbiological Methods 2021, 190, 106343. [Google Scholar] [CrossRef] [PubMed]
- Stepanović, S.; Vuković, D.; Hola, V.; Bonaventura, G.D.; Djukić, S.; Ćirković, I.; Ruzicka, F. Quantification of Biofilm in Microtiter Plates: Overview of Testing Conditions and Practical Recommendations for Assessment of Biofilm Production by Staphylococci. APMIS 2007, 115, 891–899. [Google Scholar] [CrossRef]
- Zhou, C.; Ding, Z.; Guo, Q.; Jiang, M. Visualization of Antimicrobial-Induced Bacterial Membrane Disruption with a Bicolor AIEgen. Chemosensors 2022, 10, 284. [Google Scholar] [CrossRef]
- Bucarey, S.A.; Pujol, M.; Poblete, J.; Nuñez, I.; Tapia, C.V.; Neira-Carrillo, A.; Martinez, J.; Bassa, O. Chitosan Microparticles Loaded with Yeast-Derived PCV2 Virus-like Particles Elicit Antigen-Specific Cellular Immune Response in Mice after Oral Administration. Virol J 2014, 11, 149. [Google Scholar] [CrossRef] [PubMed]
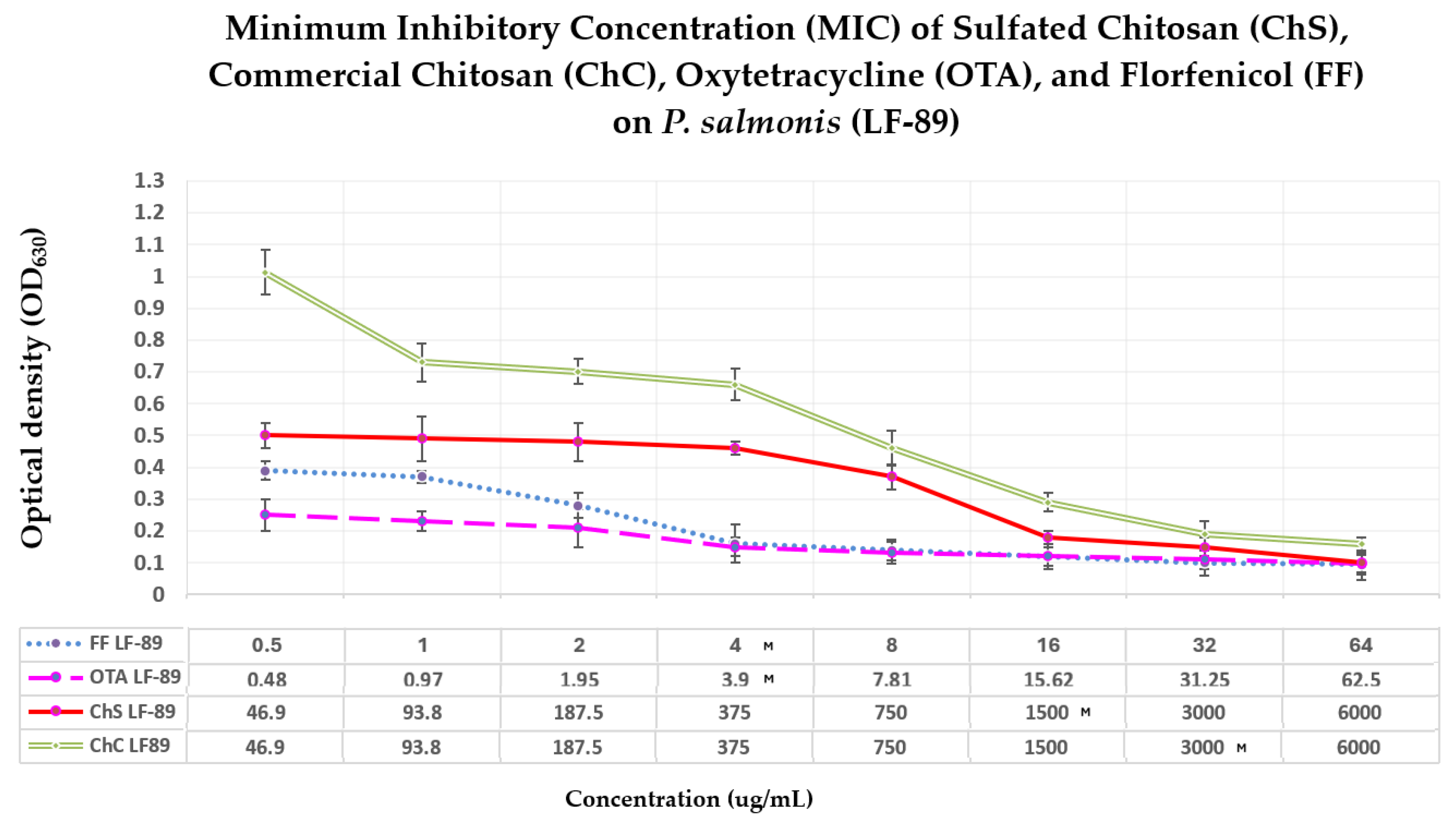
| Treatment | MIC (µg/mL) | MBC (µg/mL) | MBC (µg/mL) |
| ChS | 1500 | ≥ 1500 | Relative potency compared to the MIC of ChS |
| ChC | 3000 | ≥ 3000 | 2 times less potent |
| OTA | 3.9 | ≥ 3.9 | ~ 385 times more potent |
| FF | 4 | ≥ 4 | ~ 375 times more potent |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




