Submitted:
31 March 2026
Posted:
01 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Monitoring
2.3. Statistical Analysis
3. Results
3.1. Monitoring of Environmental Parameters
3.1.1. Salinity
3.1.2. pH
3.1.3. Temperature (°C)
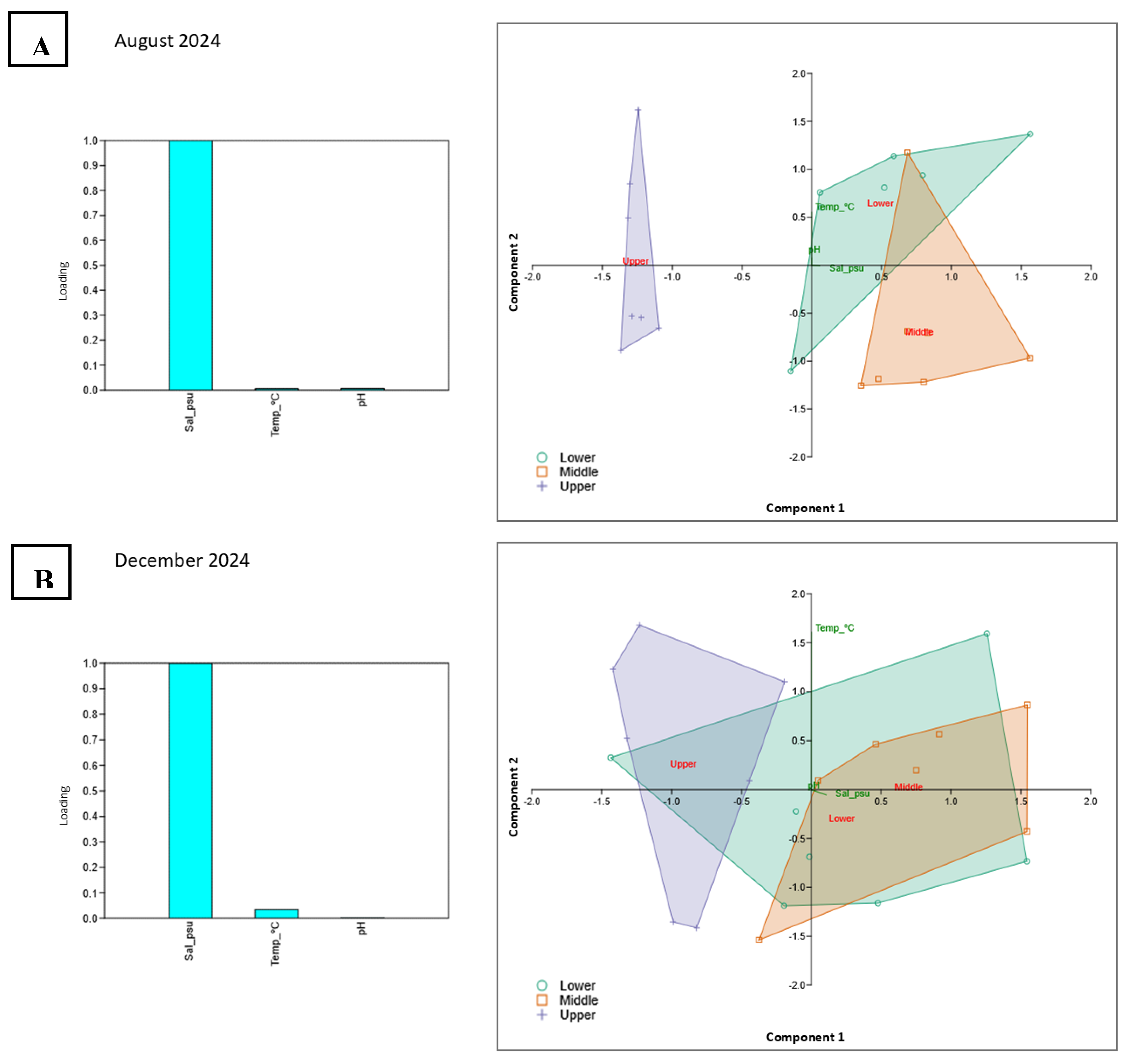
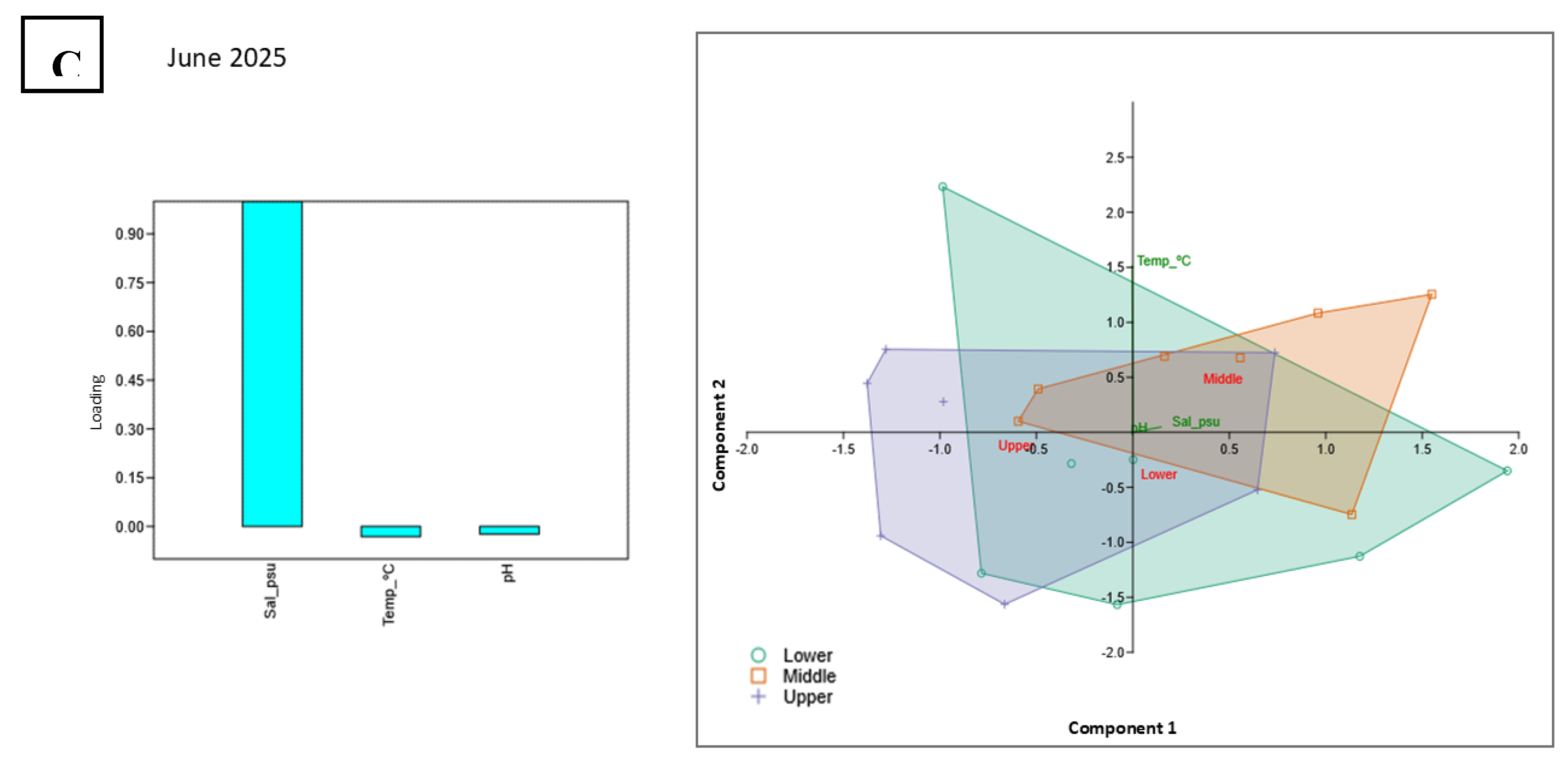
3.1.4. PCA
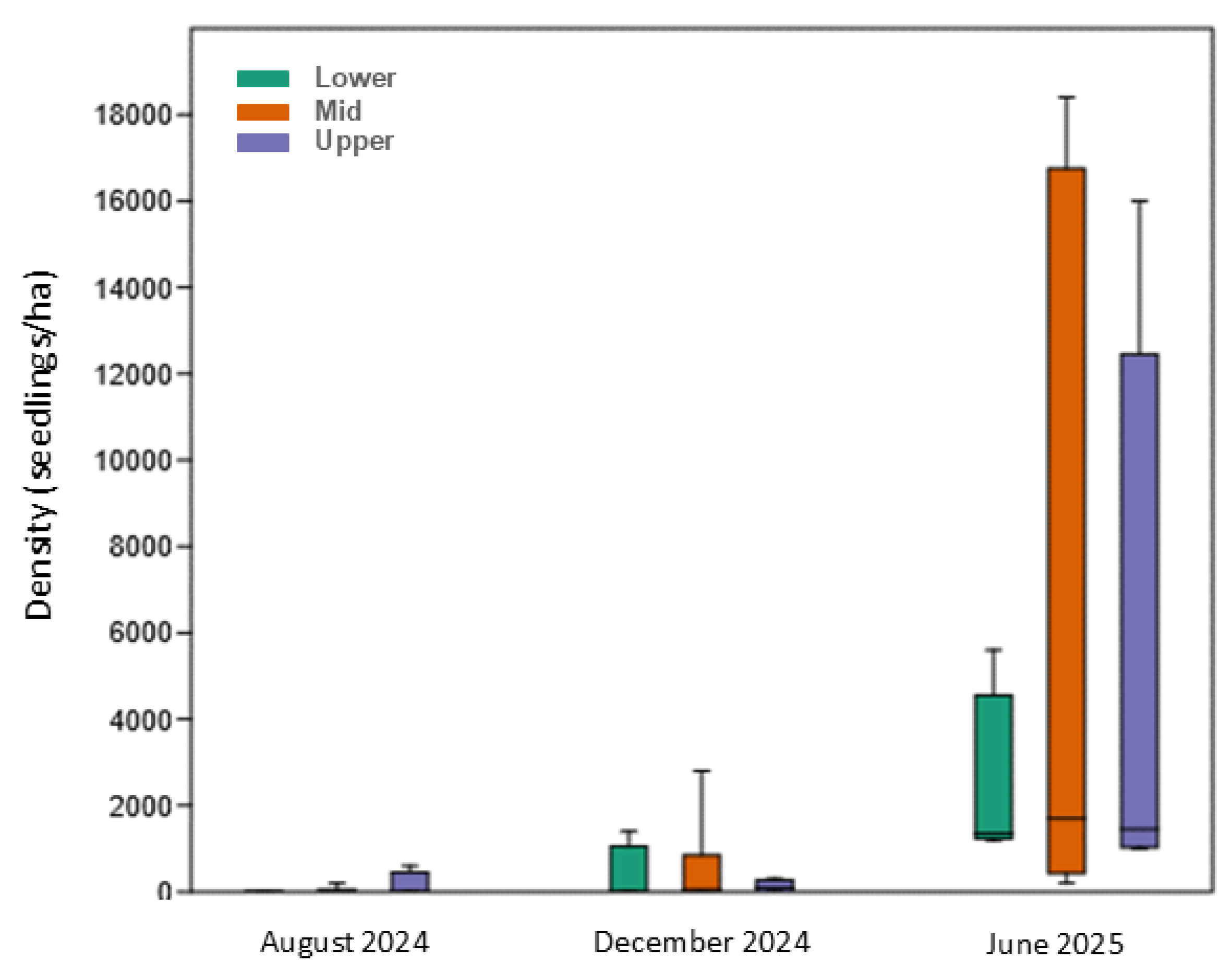
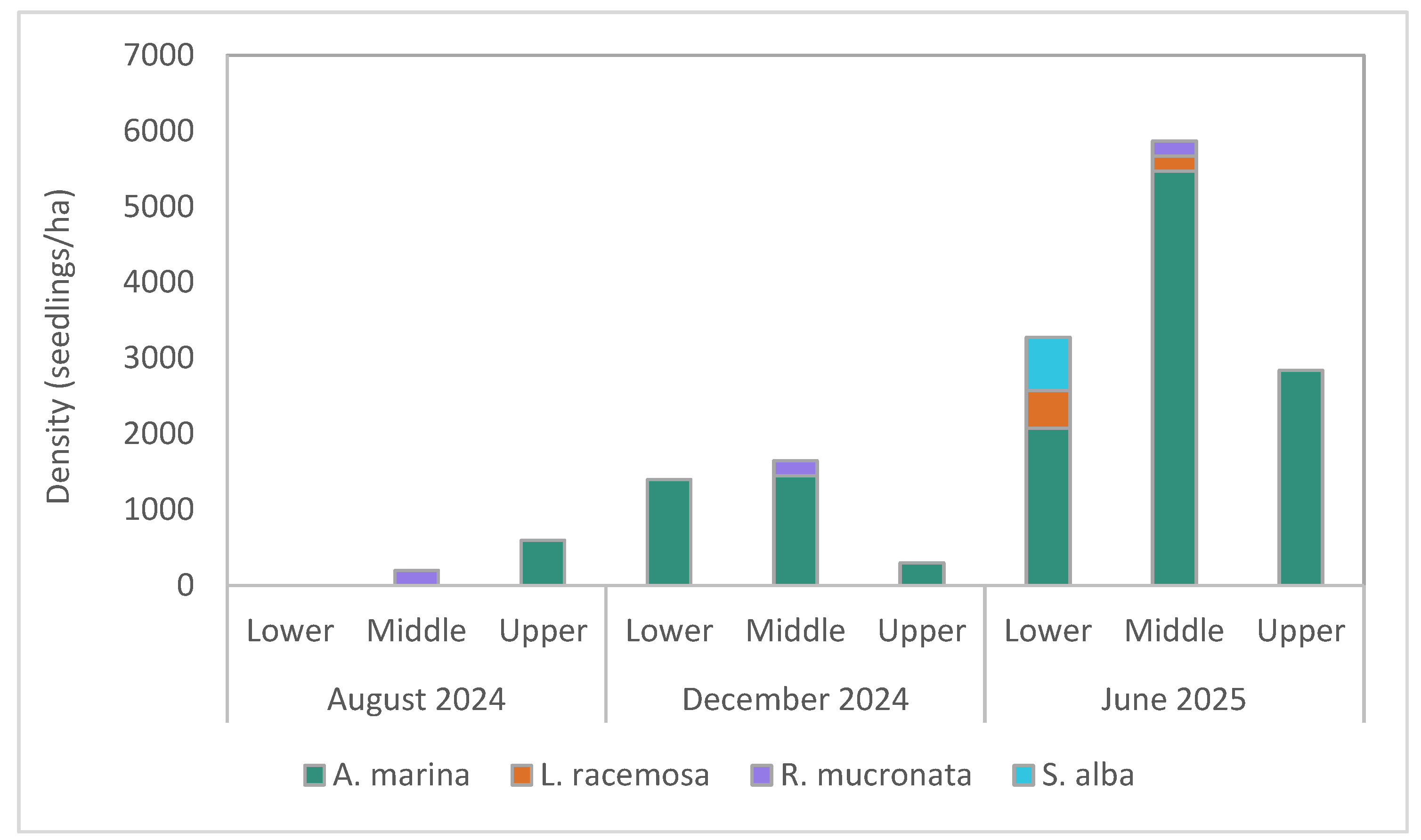
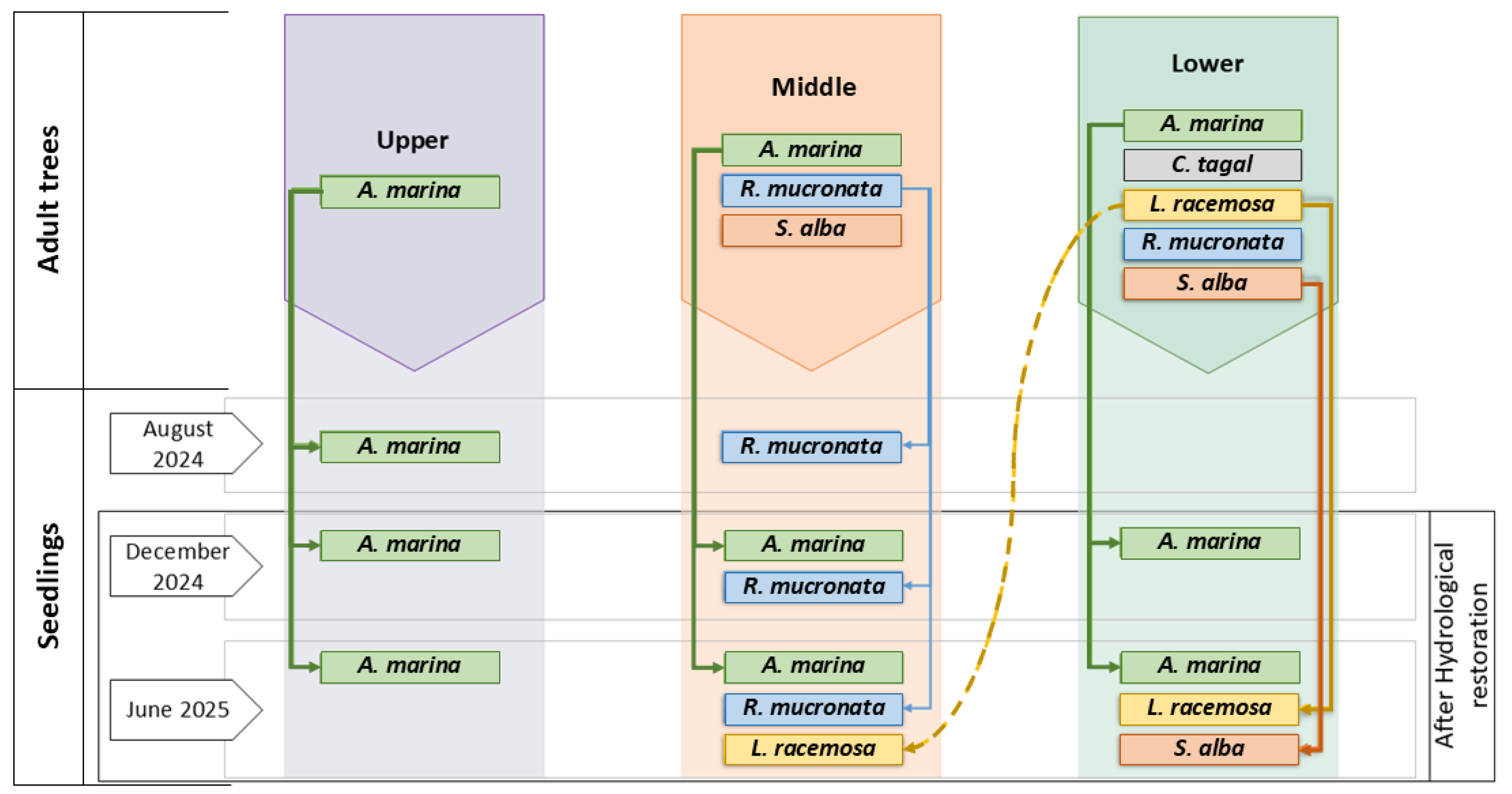
3.2. Monitoring of Natural Regeneration
3.2.1. Seedling Density
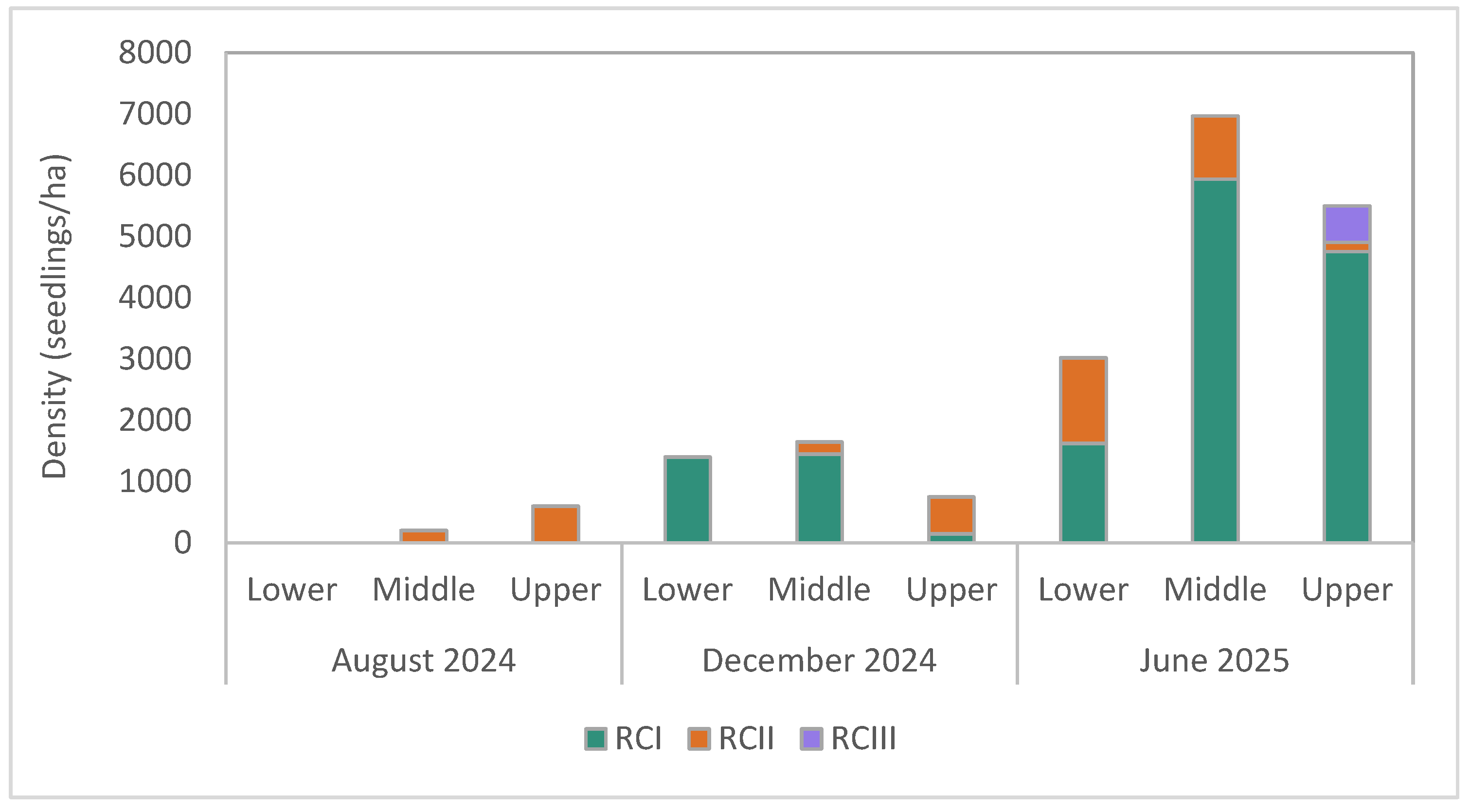
4. Discussion
4.1. Mangrove Degradation and the Need for Hydrological Restoration
4.2. Seed Recruitment and Natural Regeneration
5. Conclusion
Author Contributions
Acknowledgments
Conflicts of interest
References
- Ahidjo, S.; Hayatou, E.; Ayang, P. Evaluating the outcomes of small–scale cutting of mangrove forests in the ecosystem. African Journal of Ecology and Ecosystems 2015, 2(2), 095–102. Available online: https://www.Internationalscholarsjournals.org.
- Ahmed, S.; Sarker, S.; Friess, D.; Kamruzzaman, M.; Jacobs, M.; Islam, M.; Alam, A.; Suvo, M.; Sani, N.; Dey, T.; Naabeh, C.; Pretzsch, H. Salinity reduces site quality and mangrove forest functions. From monitoring to understanding. Science of The Total Environment 2022. [Google Scholar] [CrossRef] [PubMed]
- Alongi, D.; Carvalho, N. The effect of small-scale logging on stand characteristics and soil biogeochemistry in mangrove forests of Timor Leste. In Forest Ecology and Management; 2008. [Google Scholar] [CrossRef]
- Amade, U. Personal Communication . Mangrove nursery operator from the Geba Community, Memba District, Nampula, Mozambique, 2025. [Google Scholar]
- Asbridge, E.; Krause, C.; Lucas, R.; Owers, C.J.; Rogers, K.; Lymburner, L.; Mueller, N.; Ai, E.; Wong, S. Characterising the shortand long-term impacts of tropical cyclones on mangroves using the Landsat archive. Cambridge Prisms: Coastal Futures 2025, 3(e4), 1–14. [Google Scholar] [CrossRef]
- Balidy, H.; Bandeira, S.; Macamo, C.; Mechisso, M.; Laissone, J. Community-Based Mangrove Restoration: The Cases of the Limpopo Estuary and Quelimane Municipality. WIOMSA Symposium, 2019; Available online: https://symposium.wiomsa.org/wp-content/uploads/2019/06/551-906-1-SM.pdf.
- Bandeira, S.; Macamo, C.; Mahazule, R.; Mabilana, H. Estudo de Lições Aprendidas e Boas Práticas de Reabilitação do Mangal. Avaliação do programa de restauração de mangal no Estuário do Limpopo (Gaza), Tsolombane em Matutuine (Maputo), Nhangau (Sofala), Inhassunge e Macuze (Zambézia) e Mecúfi e Metuge (Cabo Delgado). 71PP. 2016. Available online: https://biblioteca.biofund.org.mz/wp-content/uploads/2021/02/1612900739-Li%C3%A7oes%20Aprendidas_Reabilita%C3%A7%C3%A3o%20mangais_em%20Mocambique-FINAL.pdf.
- Bandeira, S.; Balidy, H. Limpopo estuary mangrove transformation, rehabilitation and management. In Estuaries: a Lifeline of Ecosystem Services in the Western Indian Ocean; Salif, D, Scheren, P., Eds.; Springer, 2016; p. Chapter 14. 17p. [Google Scholar] [CrossRef]
- Best Practice Guidelines for Mangrove Restoration. In Global Mangrove Alliance; Beeston, M., Cameron, C., Hagger, V., Howard, J., Lovelock, C., Sippo, J., Tonneijk, F., van Bijsterveldt, C., van Eijk, P., Eds.; 2023; p. 278 pp. Available online: https://www.mangrovealliance.org/best-practice-guidelines-for-mangrove-restoration/.
- Blankespoor, R.; Dasgupta, S.; Lange, G. Mangroves as protection from storm surges in a changing climate. Ambio 2016, 46(4), 478–491. [Google Scholar] [CrossRef]
- Chen, Y.; Ye, Y. Effects of Salinity and Nutrient Addition on Mangrove Excoecaria agallocha. PLOS ONE 2014, 9(4), e93337. [Google Scholar] [CrossRef]
- Djamaluddin, R.; Brown, B.; Lewis, R., III. The practice of hydrological restoration to rehabilitate abandoned shrimp ponds in Bunaken National Park, North Sulawesi, Indonesia. Biodiversitas 2019, 20(1), 160–170. [Google Scholar] [CrossRef]
- Ellis, W.; Bell, S. Canopy gaps formed by mangrove trimming: an experimental test of impact on litter fall and standing litter stock in Southwest Florida (USA). Journal of Experimental Marine Biology and Ecology 2004, 311, 201–222. [Google Scholar] [CrossRef]
- Esguerra-Rodríguez, D.; De León-Lorenzana, A.; Teutli, C.; Prieto-Davó, A.; García-Maldonado, J.; Silveira, J.; Falcón, L. Do restoration strategies in mangroves recover microbial diversity? A case study in the Yucatan peninsula. PLOS ONE 2024, 19. [Google Scholar] [CrossRef]
- FAO. Mangrove Forest Management Guidelines. (FAO Forestry Paper 117). Rome, 1994. Available online: https://www.fao.org/4/ap428e/ap428e00.pdf.
- Ferreira, A.; Freire, F.; Rodrigues, J.; Bezerra, L. Mangrove Recovery in Semiarid Coast Shows Increase of Ecological Processes from Biotic and Abiotic Drivers in Response to Hydrological Restoration; Wetlands, 2022; p. 42. [Google Scholar] [CrossRef]
- Gatt, Y.; Walton, R.; Andradi-Brown, D.; Spalding, M.; Acosta-Velázquez, J.; Adame, F.; Barros, F.; Beeston, M.; Bernardino, A.; Buelow, C.; Cadier, C.; Vela, A.; Canty, S.; Dahdouh-Guebas, F.; Gomes, L.E.; Duncan, C.; Eger, A.; Enright, J.; Frausto-Illescas, T.; Worthington, T. The Mangrove Restoration Tracker Tool: Meeting local practitioner needs and tracking progress toward global targets. One Earth 2024, 7. [Google Scholar] [CrossRef]
- Gerona-Daga, M.E.; Salmo, S.G. A systematic review of mangrove restoration studies in Southeast Asia: Challenges and opportunities for the United Nation’s Decade on Ecosystem Restoration. Frontiers in Marine Science 2022, 9, 987737. [Google Scholar] [CrossRef]
- Glamore, W.; Rayner, D.; Ruprecht, J.; SadatNoori, M.; Khojasteh, D. Eco-hydrology as a driver for tidal restoration: Observations from a Ramsar wetland in eastern Australia. PLoS ONE 2021, 16(8), e0254701. [Google Scholar] [CrossRef] [PubMed]
- Herrera-Silveira, J. A.; Teutli-Hernandez, C.; Secaira-Fajardo, F.; Braun, R.; Bowman, J.; Geselbracht, L.; Musgrove, M.; Rogers, M.; Schmidt, J.; Robles-Toral, P. J.; Canul-Cabrera, J. A.; Guerra-Cano, L. Hurricane Damages to Mangrove Forests and Post-Storm Restoration Techniques and Costs; The Nature Conservancy: Arlington, VA, 2022. [Google Scholar] [CrossRef]
- INGD. Balanço da Época Chuvosa e Ciclónica 2023/2024. Instituto Nacional de Gestão e Redução do Risco de Desastres (INGD), 2024. Available online: https://ingd.gov.mz/wp-content/uploads/2025/07/Relatorio_do_Balanco_da_ECC_2023-2024-.pdf.
- INGD. PLANO ANUAL DE CONTINGÊNCIA 2025-202. Instituto Nacional de Gestão e Redução do Risco de Desastres (INGD), 2025. Available online: https://ingd.gov.mz/wp-content/uploads/2025/10/PC-2025-26-CM.pdf.
- Kairo, J.G.; Mangora, M. Guidelines on Mangrove Ecosystem Restoration for the Western Indian Ocean Region Western Indian Ocean Region. 2020. Available online: https://www.nairobiconvention.org/CHM%20Documents/WIOSAP/guidelines/GuidelinesonMangroveRestorationForTheWIO.pdf.
- Lewis, R., III. Ecological engineering for successful management and restoration of mangrove forests. Ecological Engineering 2005, 24, 403–418. [Google Scholar] [CrossRef]
- Lopez-Adame, H.; Zaragoza-Méndez, F.; Lara, A.; Alvarado-Barrientos, M.; Mariño-Tapia, I.; Sáinz-Hernández, E.; Rosela, P.; Zaldivar-Jiménez, A.; López-Portillo, J. Effectiveness of hydrological restoration in the mangrove of a coastal lagoon in the Gulf of Mexico. Ecological Engineering 2026, 223, 107848. [Google Scholar] [CrossRef]
- Macamo, C.; Massuanganhe, E.; Nicolau, D.; Bandeira, S.; Adams, J. Mangrove’s response to cyclone Eline (2000): What is happening 14 years later. Aquatic Botany 2016, 134. [Google Scholar] [CrossRef]
- Medina, E.; Cuevas, E.; Popp, M.; Lugo, A. Soil Salinity, Sun Exposure, and Growth of Acrostichum aureum, the Mangrove Fern. Botanical Gazette 1990, 151. [Google Scholar] [CrossRef]
- Medina-Calderón, J.; Mancera-Pineda, J.; Castañeda-Moya, E.; Rivera-Monroy, V. Hydroperiod and Salinity Interactions Control Mangrove Root Dynamics in a Karstic Oceanic Island in the Caribbean Sea (San Andres, Colombia). Frontiers in Marine Science 2021, 7(). [Google Scholar] [CrossRef]
- Monsalve, R.; Verduga, L. Water and Sediment Quality Changes in Mangrove Systems with Shrimp Farms in the Northern Ecuadorean Coast. In Applied Sciences; 2023. [Google Scholar] [CrossRef]
- Murdiyarso, D.; Sasmito, S.; Sillanpää, M.; Mackenzie, R.; Gaveau, D. Mangrove selective logging sustains biomass carbon recovery, soil carbon, and sediment. Scientific Reports 2021, 11, 12325. [Google Scholar] [CrossRef]
- Muttaqin, A.; Soemarno, S.; Purnomo, M.; Zakiyah, U. Analysis of the Mangrove Ecosystem Due to the Influence of Mount Bromo’s Cold Lava Material on Permata Pilang Beach, Probolinggo. Ecological Engineering & Environmental Technology 2024, 25, 272–281. [Google Scholar] [CrossRef]
- Ngole-Jeme, V.; Fonge, B.; Tabot, P.; Mumbang, C. Impact of logging activities in a tropical mangrove on ecosystem diversity and sediment heavy metal concentrations. Journal of Coastal Conservation 2016, 20(3). [Google Scholar] [CrossRef]
- Numbere, A. Natural Seedling Recruitment and Regeneration in Deforested and Sand-Filled Mangrove Forest at Eagle Island, Niger Delta Nigeria. In Ecology and Evolution; 2021. [Google Scholar] [CrossRef]
- Pérez-Ceballos, R.; Zaldívar-Jiménez, A.; Canales-Delgadillo, J.; López-Adame, H.; López-Portillo, J.; Merino-Ibarra, M. Determining hydrological flow paths to enhance restoration in impaired mangrove wetlands. PLOS ONE 2020, 15(1), e0227665. [Google Scholar] [CrossRef]
- Preferred by Nature. Ecosystem Restoration. Report for Eden Reforestation Projects. Sofala Province, Mozambique. 2022. Available online: https://www.preferredbynature.org/sites/default/files/publicsummaries/Eden%20Reforestation%20Projects%20Sofala%20ER%20ver%2022%20-%20PS_0.pdf.
- Radabaugh, K.; Dontis, E.; Chappel, A.; Russo, C.; Moyer, R. Early indicators of stress in mangrove forests with altered hydrology in Tampa Bay, Florida, USA. Estuarine, Coastal and Shelf Science 2021, 254. 107324. [Google Scholar] [CrossRef]
- Rivera-Monroy, V.; Lee, S.; Kristensen, E.; Twilley, R. Mangrove Ecosystems: A Global Biogeographic Perspective: Structure, Function, and Services; 2017. [Google Scholar] [CrossRef]
- Rodríguez- Rodríguez, A.; Mancera-Pineda, J.; Tavera, H. Mangrove restoration in Colombia: Trends and lessons learned. Forest Ecology and Management 2021, 496, 119414. [Google Scholar] [CrossRef]
- Rodrigues, M. Moçambique Preso a um Ciclo Vicioso de Reconstrução e Perdas pós-Ciclones. Instituto Nacional de Meteorologia (INAM) Relatório do Estado do Clima de Moçambique em 2023. 2024. Available online: https://www.inam.gov.mz.
- Ruiz-Guevara, S.; Quintero-Castañeda, C.; Hernandez, L.; Sierra Carrillo, M. Environmental Psychology and Mangrove Reforestation in the Ciénaga Grande de Santa Marta, Colombia: An Approach to Ecosystem Restoration. World 2025, 6(24). [Google Scholar] [CrossRef]
- Sánchez-Carrillo, S.; Garatuza-Payan, J.; Sánchez-Andrés, R.; Cervantes, F.J.; Bartolomé, M.C.; Merino-Ibarra, M.; Thalasso, F. Methane Production and Oxidation in Mangrove Soils Assessed by Stable Isotope Mass Balances. Water 2021, 13((13) 1867). [Google Scholar] [CrossRef]
- Silva, W.; Amarasinghe, M. Response of mangrove plant species to a saline gradient: Implications for ecological restoration. Acta Botanica Brasilica 2021, 35, 151–160. [Google Scholar] [CrossRef]
- Simon, L. Assessing ecosystem effects of small–scale cutting of Cameroon mangrove forests. Journal of Ecology and the Natural Environment 2012, 4(5). [Google Scholar] [CrossRef]
- Su, C.J.; Hsieh, S.Y.; Chiang, M.W.; Pang, K.L. Salinity, pH and temperature growth ranges of Halophytophthora isolates suggest their physiological adaptations to mangrove environments. Mycology 2020, 11(3), 256–262. [Google Scholar] [CrossRef]
- Sunkur, R.; Kantamaneni, K.; Bokhoree, C.; Ravan, S. Mangroves’ role in supporting ecosystem-based techniques to reduce disaster risk and adapt to climate change: A review. Journal of Sea Research 2023, 196. [Google Scholar] [CrossRef]
- Teutli-Hernández, C.; Herrera-Silveira, J.A.; Comín, F.A.; López, M.M. Nurse species could facilitate the recruitment of mangrove seedlings after hydrological rehabilitation. Ecological Engineering 2017. [Google Scholar] [CrossRef]
- Teutli-Hernández, C.; Herrera-Silveira, J.A.; Cisneros-de la Cruz, D.J.; Román-Cuesta, R. Mangrove ecological restoration guide: Lessons learned. Mainstreaming Wetlands into the Cli mate Agenda: A multilevel approach (SWAMP); CIFOR/CINVESTAV-IPN/UNAM-Sisal/PMC, 2020; p. 42p. [Google Scholar] [CrossRef]
- Turner, R.; Lewis, R. Hydrologic restoration of coastal wetlands. Wetlands Ecology and Management 1996, 4, 65–72. [Google Scholar] [CrossRef]
- Wodehouse, D.; Rayment, M. Mangrove area and propagule number planting targets produce sub-optimal rehabilitation and afforestation outcomes. Estuarine, Coastal and Shelf Science 2019, 222. 91–102. [Google Scholar] [CrossRef]
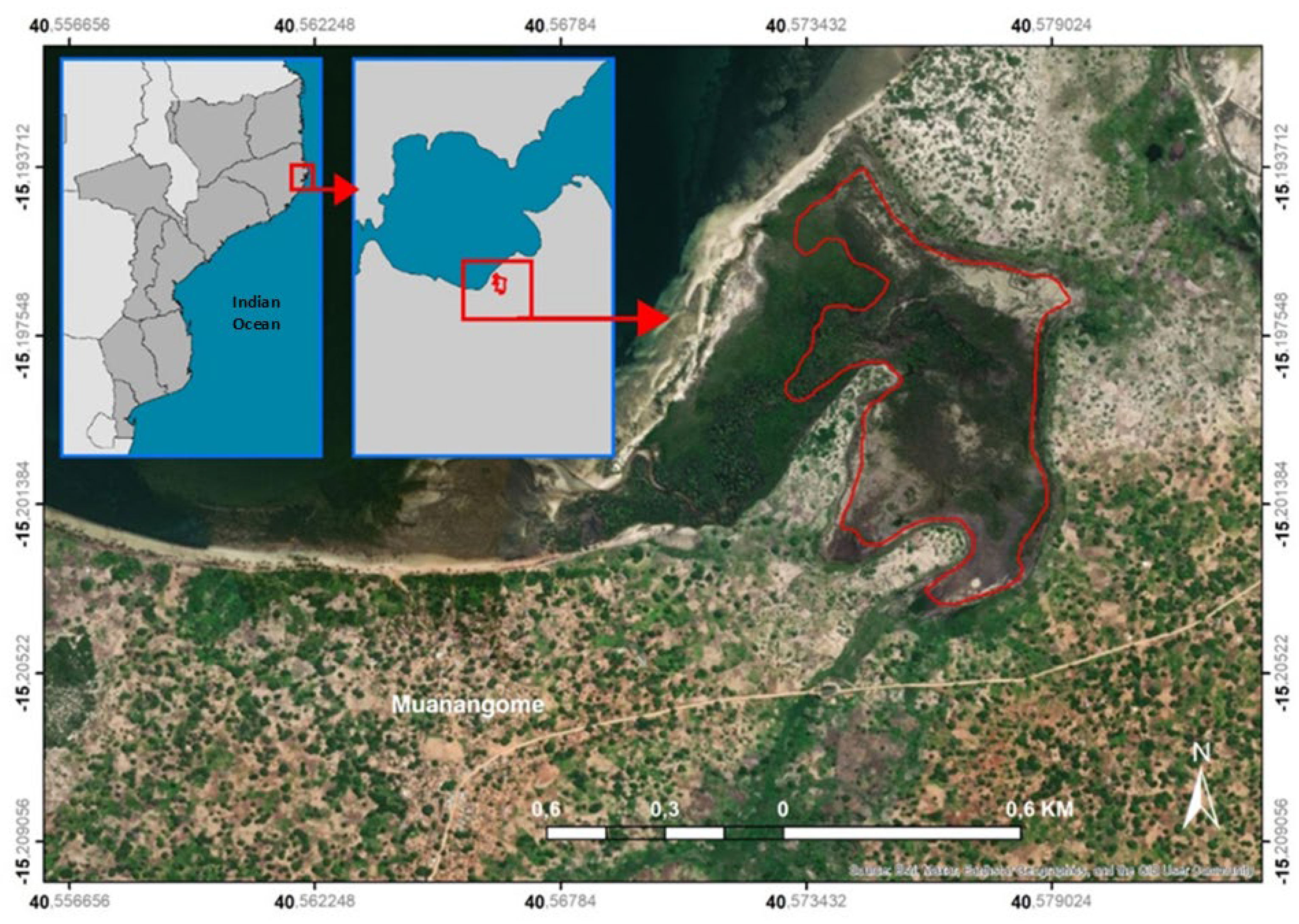


| Zones | Characteristics |
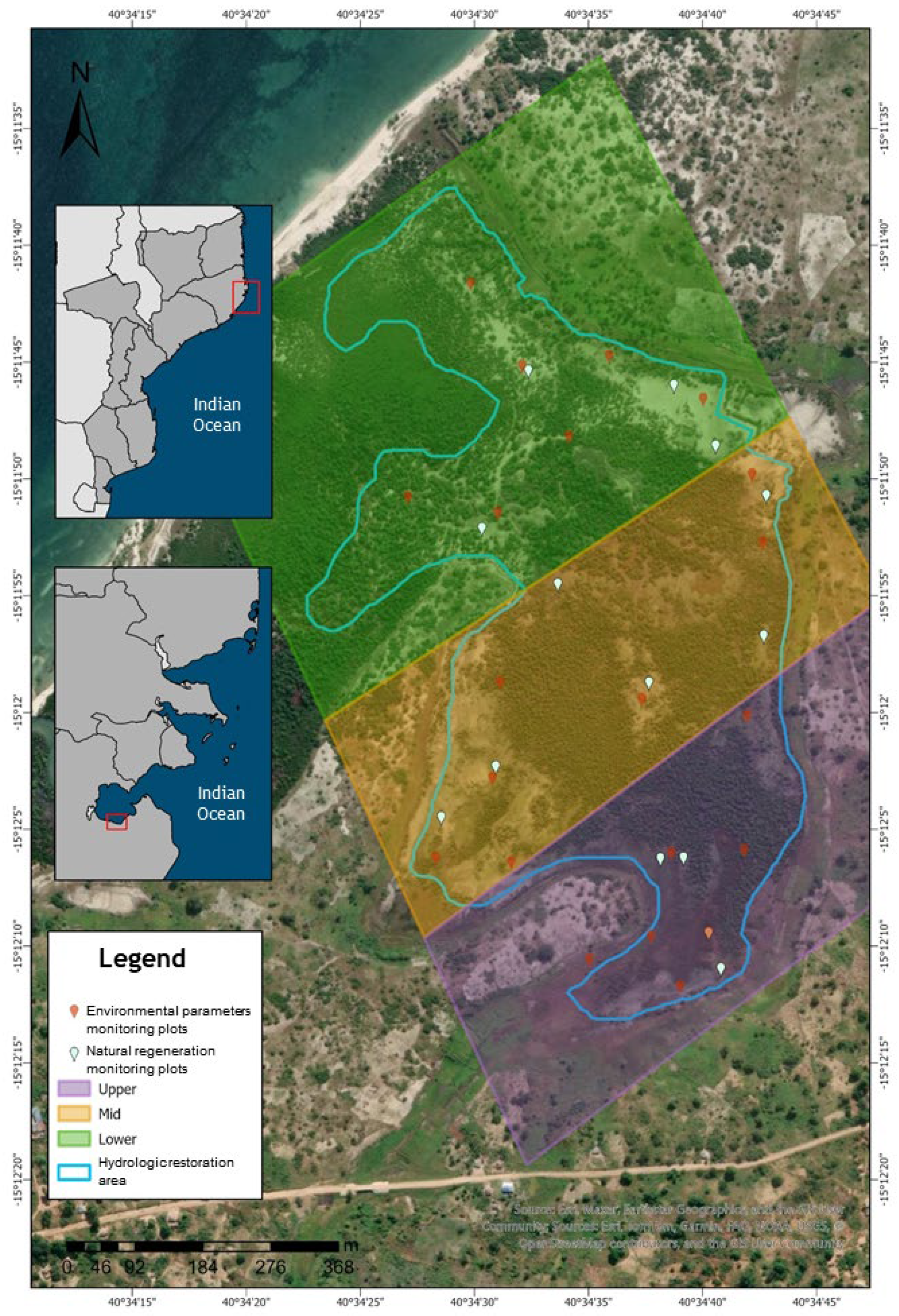 Figure 3. Main mangrove zones defined in the study area. Figure 3. Main mangrove zones defined in the study area. |
| Upper | Salinity: 0 to 30 Soil: sand and clay, firm to soft Mangrove species: A. marina. Other characteristics: dominated by associated species Typha latifolia and Cyperus sp.; river influence and stagnation of fresh water |
|
| Middle | Salinity: 50 to above 100; Soil: sand and clay, firm to soft; Mangrove species: A. marina, R. mucronata and S. alba; Other characteristics: intensive logging, canopy gaps; high density of associated species Juncus kraussii and Salicornia sp.; stagnant fresh and salt water |
|
| Lower | Salinity: 50 to 100 Soil: sandy, firm to soft Mangrove species: A. marina, C. tagal, R. mucronata, S. alba and L. racemosa; Other characteristics: intensive logging, canopy gaps |
| Zones | Baseline | Monitorings | |
|---|---|---|---|
| August 2024 | December 2024 | June 2025 | |
| Lower | 63.42 ±7.50 (a,1) | 42.64 ±7.77 | 27.73 ±7.28 (a) |
| Middle | 72.95 ± 5.02(b,2) | 52.55 ± 5.61 (3) | 33.84 ± 5.63 (b) |
| Upper | 3.59 ± 1.13 (c,1,2) | 19.23 ± 3.60 (c,3) | 14.26 ± 6.32 |
| Zones | Baseline | Monitorings | |
|---|---|---|---|
| August 2024 | December 2024 | June 2025 | |
| Lower | 7.77±0.081 2 | 7.33±0.093 4 | 6.84±0.09 |
| Middle | 6.63±0.211 | 6.54±0.063 | 6.50±0.13 |
| Upper | 6.67±0.322 | 6.56±0.164 | 7.30±0.32 |
| Zones | Baseline | Monitorings | |
|---|---|---|---|
| August 2024 | December 2024 | June 2025 | |
| Lower | 32.78±1.15 (a) | 32.73±0.61 | 27.06±0.94 (a) |
| Middle | 28.12±1.17 (b) | 33.52±0.56 (b,c) | 28.40±0.44 |
| Upper | 30.38±1.27 | 32.71±0.64 (d) | 27.93±0.62 (d) |
| Cyclone, year | Category (Saffir-Simpson scale) | Characteristics | Main impacts Nampula province (Mozambique) |
|---|---|---|---|
| JOKWE, March 2008 | 1 – Tropical cyclone | Landfall site: Nampula Province, between Nacala and Moz. Island |
|
| GOMBE, March 2022 | 3 – Tropical cyclone | Wind speeds up to 190 km/h; 200 mm in 24 hours; Most affected areas: Mozambique island, Lunga; |
Affected 642 383 people, 53 deaths and 77 injured; over 23 994 people displaced; damages to electricity infrastructure; 707 km of road impacted. Damage estimated at USD 81.9 million. |
| CHIDO, December 2024 | 4-equivalent – Tropical cyclone | Wind speeds up to 120km/h and reaching 260 km/h; 250 mm in 24hours; Most affected areas: Cabo Delgado, Nampula and Niassa; |
Affected 175 169 people, 493 injured and 45 people dead (37 in Cabo Delgado, 5 in Nampula and 3 in Niassa) |
| DIKELEDI, January 2025 | 2 – Tropical cyclone | Wind speed: 150 km/ha up to 180km/h; 210.4 to 247 mm; Most affected areas: Nampula Province, between Nacala Porto and Liupo |
Affected 283 333 people, 48 heath facilities, 221 schools |
| JUDE, March 2025 | 1 - Hurricane | Wind speed: 120 km/ha; more than 200mm in 24 hours; Most affected areas: Nampula Province between Memba and Mossuril |
Affected over 390 000 people, 13 deaths, 135 injured. 81 health facilities, 272 schools, 18 bridges, 48 water systems, and 73km of electricity lines |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




