Submitted:
31 March 2026
Posted:
01 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
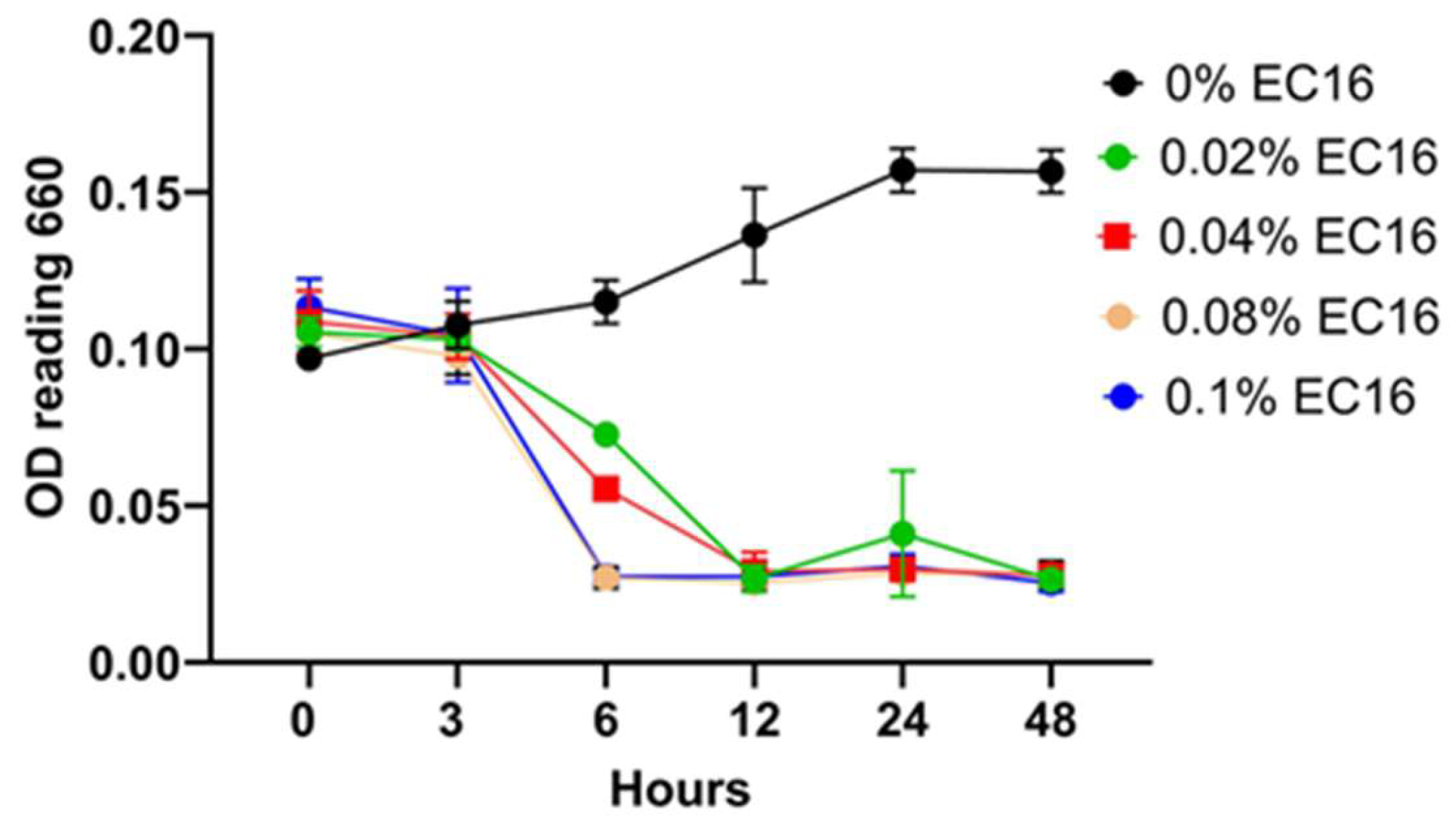
2.2. Porphyromonas Gingivalis (Pg) and Response to EC16 NPs In Vitro
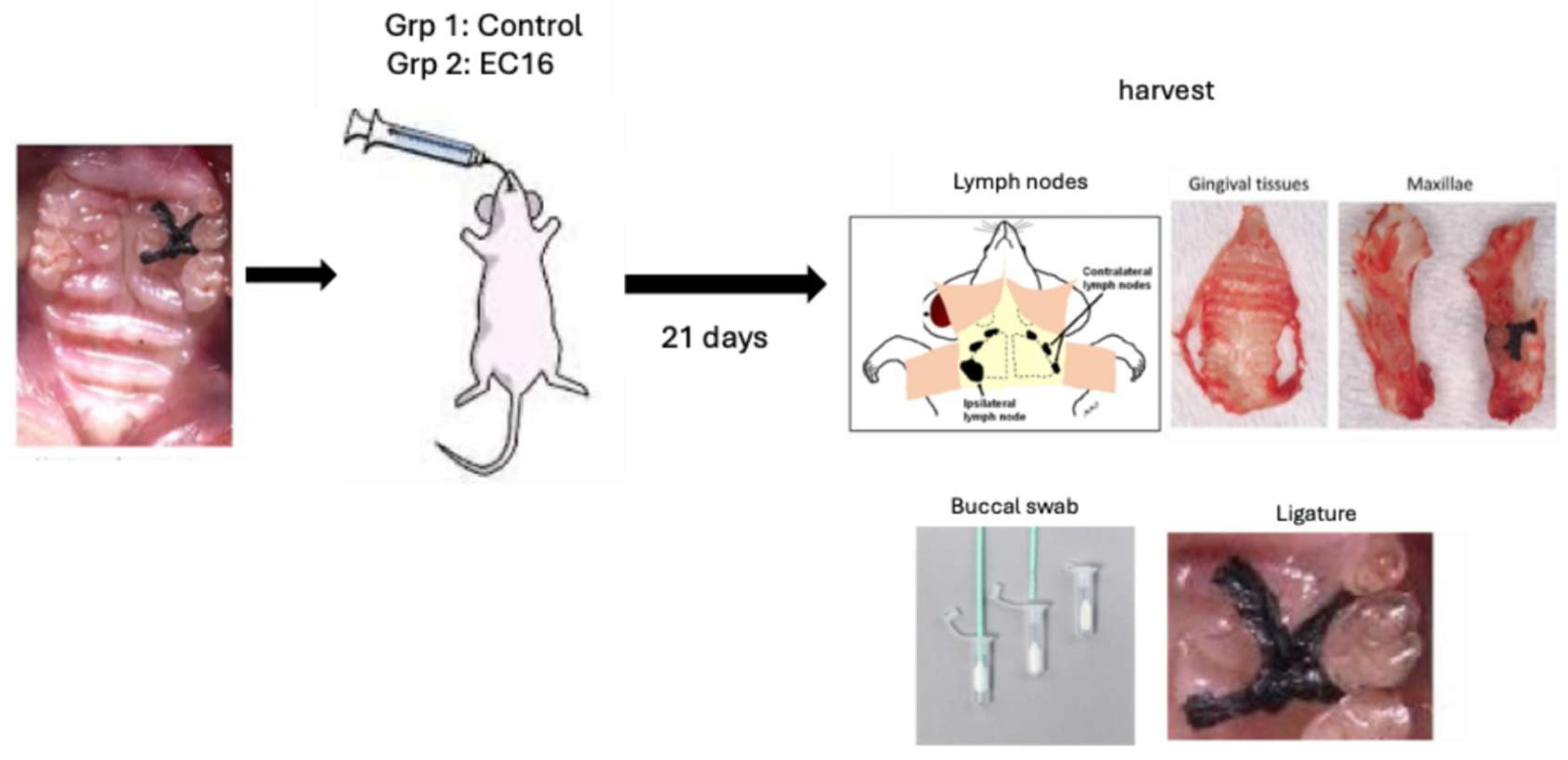
2.3. Animals and Procedures
2.4. Nanoparticle Tracking Analysis of EC16
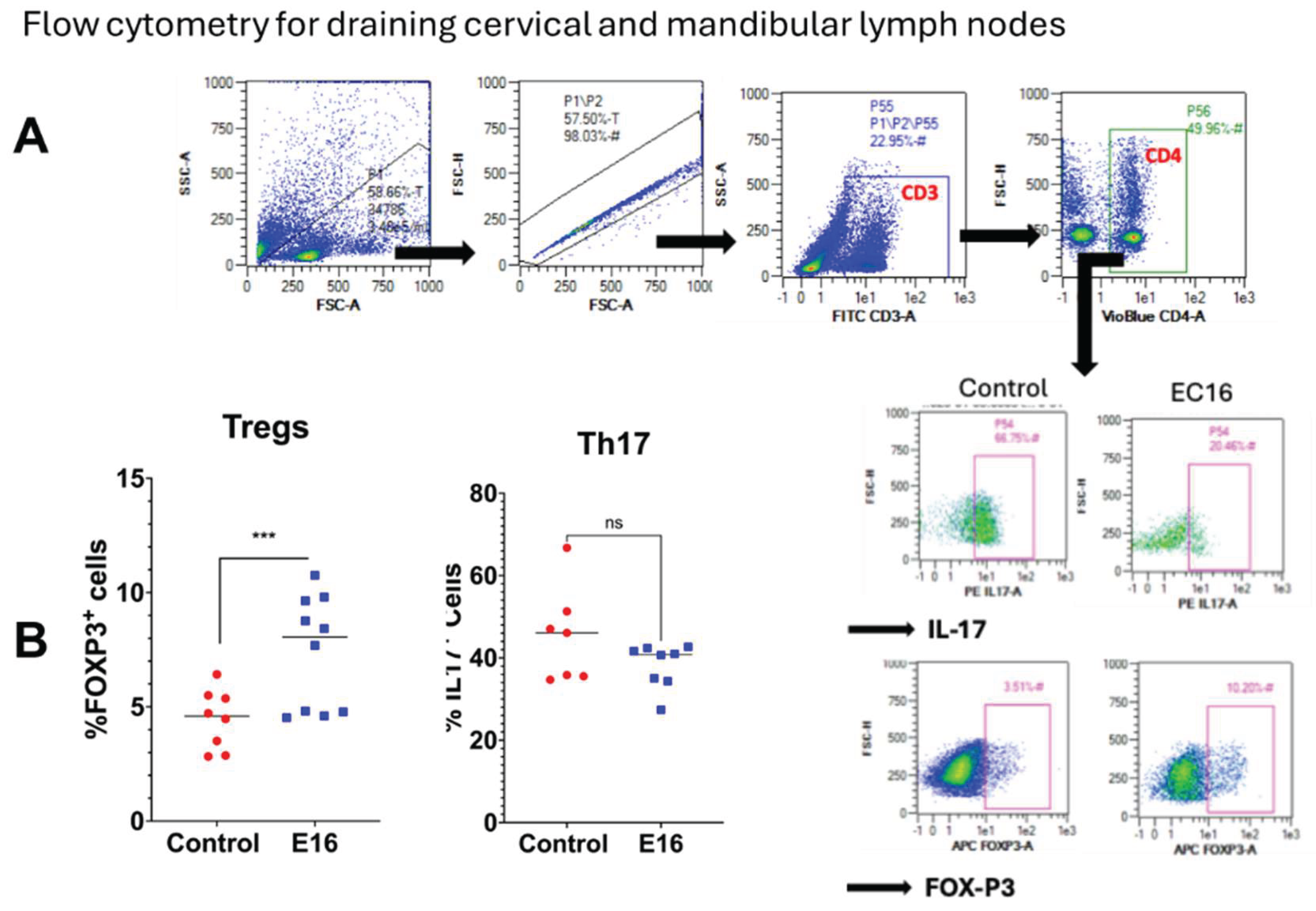
2.5. Flow Cytometry and Antibodies
2.6. Western Blotting and Antibodies
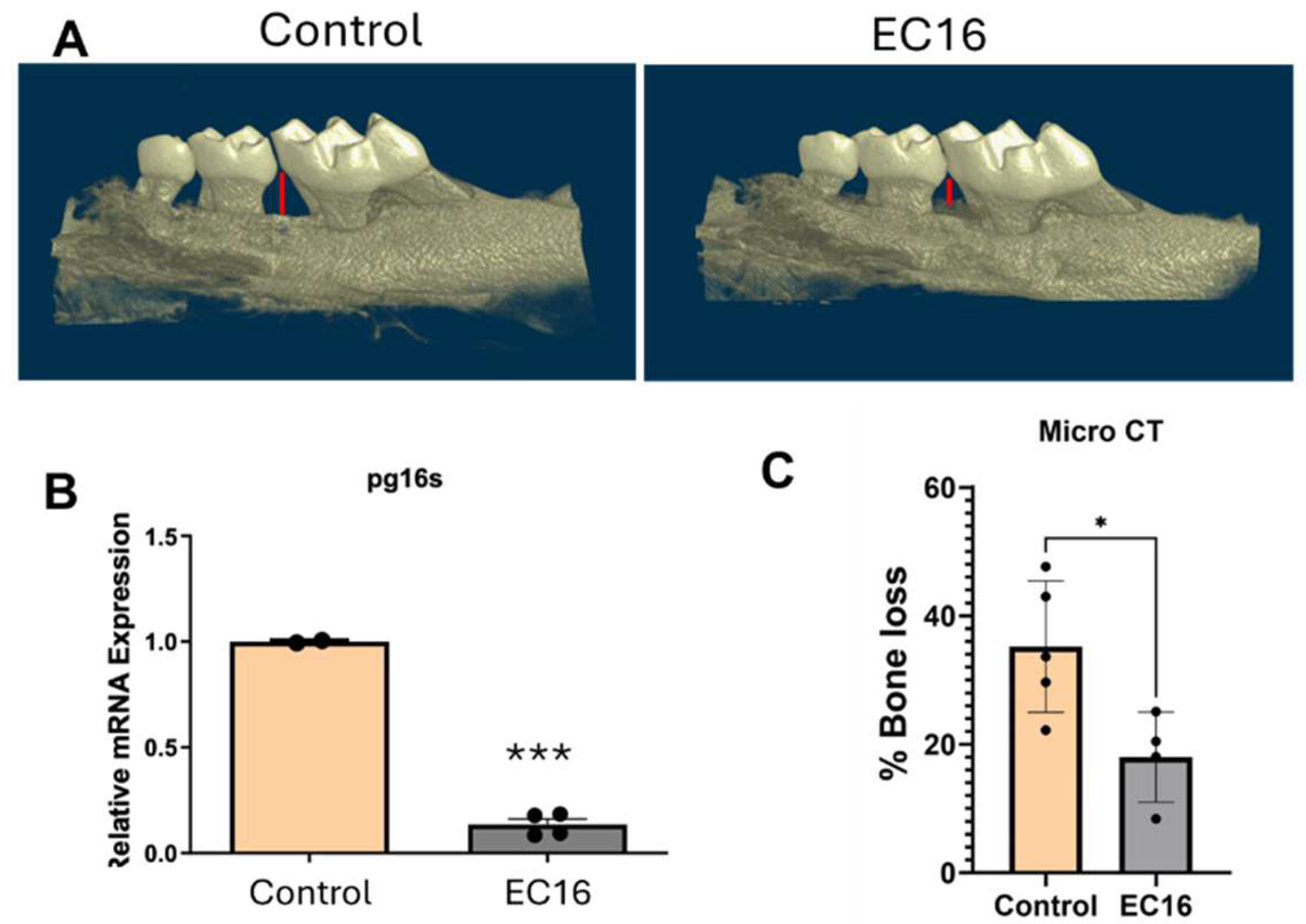
2.7. Micro-CT Imaging and Bone Parameter Analysis
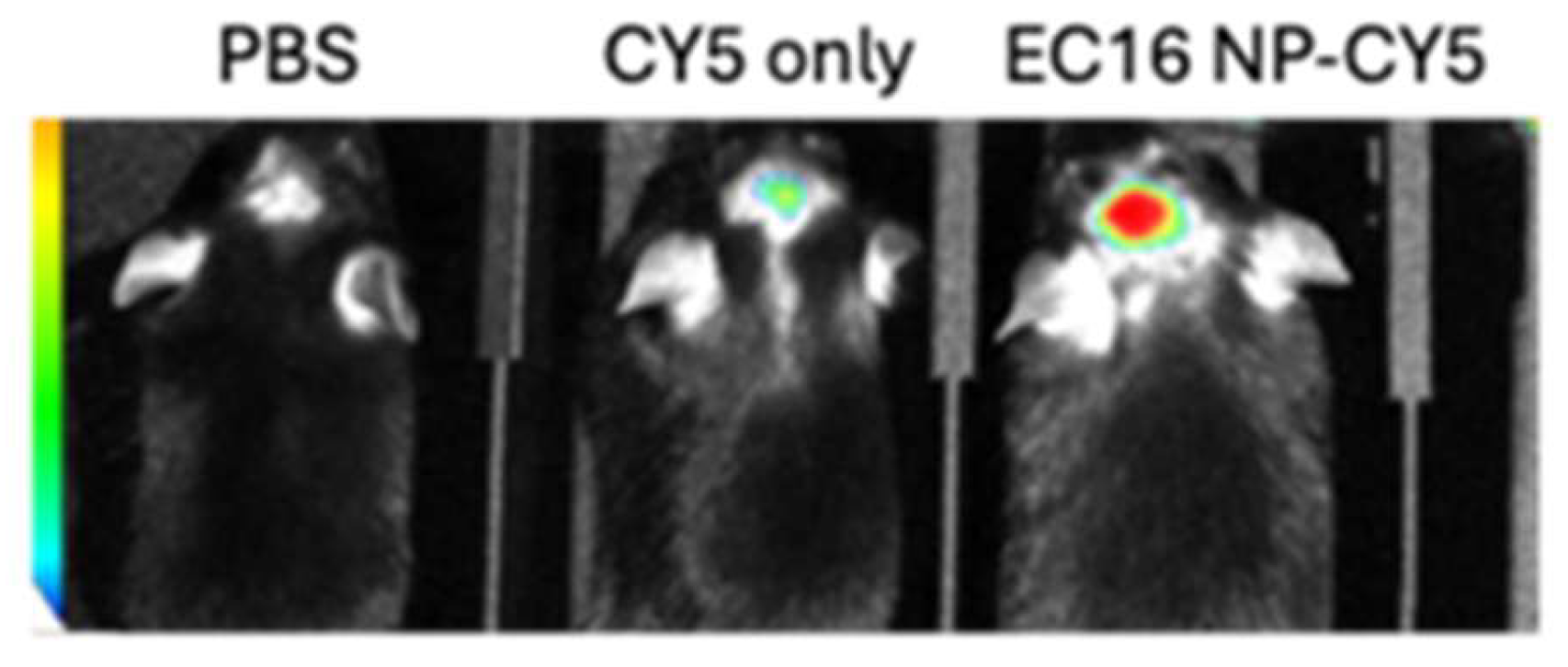
2.8. In Vivo Live Animal Imaging System (IVIS) Imaging
2.9. Statistical Analysis
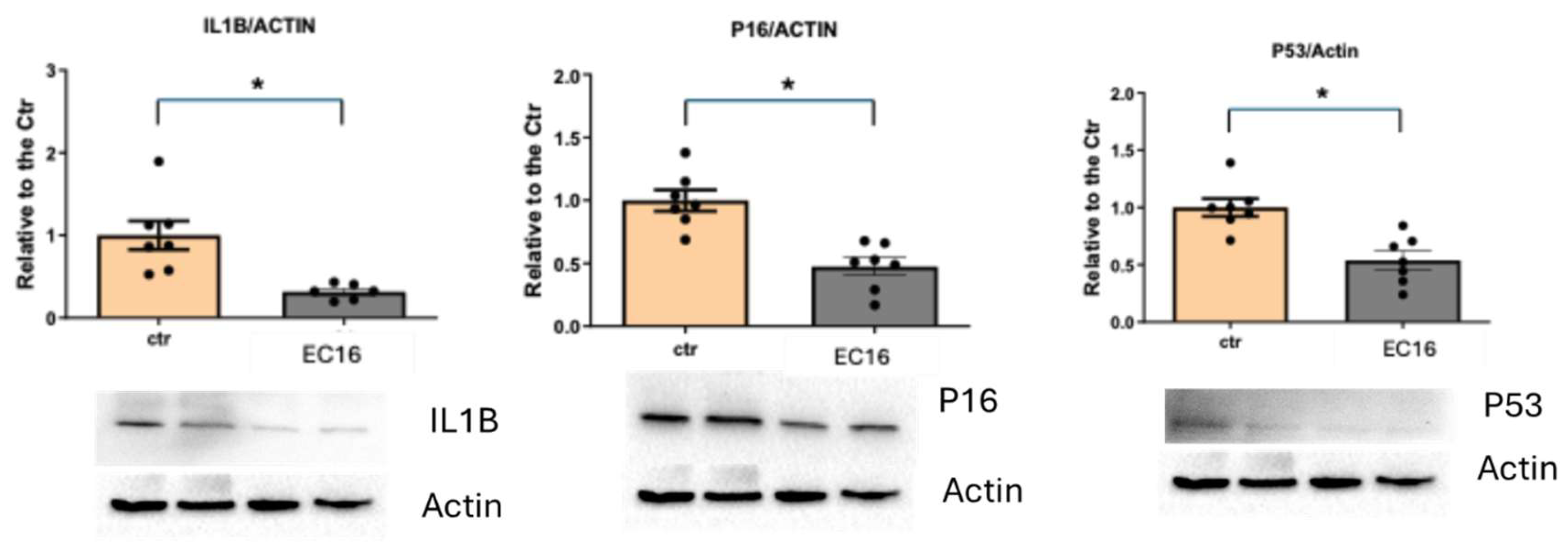
3. Results
Discussion
Conclusion
Institutional Review Board Statement
Conflicts of Interest
References
- Gasner, N. S., & Schure, R. S. (2025). Periodontal Disease. In StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing.
- Kamer, A. R., Craig, R. G., Dasanayake, A. P., et al. (2008). Inflammation and Alzheimer’s disease: Possible role of periodontal diseases. Alzheimer’s & Dementia, 4(4), 242–250. [CrossRef]
- Costa, F. O., Cota, L. O. M., Lages, E. J. P., et al. (2023). Chronic Periodontitis Exposure and the Risk of Alzheimer’s Disease: A Retrospective Cohort Study. Dentistry Journal, 12(10), 331.
- Alba-Castro, V., Fabbricatore, M., Gaviraghi, A., Piras, G., Rossi, V., Sartoris, C., Scarlata, D., & Tonoli, G. (2024). Reciprocal Interactions Between Periodontal Disease and Alzheimer’s Disease: Implications for Mutual Triggering, Exacerbation, and Treatment Interventions—A Comprehensive Review of the Literature. Life, 14(6), 81. [CrossRef]
- Chen, C.-K., Wu, Y.-T., & Chang, Y.-C. (2017). Association between chronic periodontitis and the risk of Alzheimer’s disease: A retrospective, population-based, matched-cohort study. Alzheimer’s Research & Therapy, 9, 56. [CrossRef]
- Leira, Y., Domínguez, C., Seoane, J., Seoane-Romero, J., Pías-Peleteiro, J. M., Takkouche, B., ... & Aldrey, J. M. (2017). Is periodontal disease associated with Alzheimer’s disease? A systematic review with meta-analysis. Neuroepidemiology, 48(1-2), 21–31. [CrossRef]
- Sparks Stein, P., Steffen, M. J., Smith, C., Jicha, G., Ebersole, J. L., Abner, E., & Dawson, D. (2012). Serum antibodies to periodontal pathogens are a risk factor for Alzheimer’s disease. Alzheimer’s & Dementia, 8(3), 196–203. [CrossRef]
- Ide, M., Harris, M., Stevens, A., Sussams, R., Hopkins, V., Culliford, D., ... & Holmes, C. (2016). Periodontitis and cognitive decline in Alzheimer’s disease. PLoS One, 11(3), e0151081. [CrossRef]
- Dominy, S. S., Lynch, C., Ermini, F., et al. (2019). Porphyromonas gingivalis in Alzheimer’s disease brains: Evidence for disease causation and treatment with small-molecule inhibitors. Science Advances, 5(1), eaau3333. [CrossRef]
- Hosseini, H., Arab, S., Kiani, Z., Zafarian, N., Taghizadeh-Boroujeni, S., & Shokouhi, P. (2024). Periodontal Disease and Alzheimer’s: Insights from a Systematic Literature Network Analysis. The Journal of Prevention of Alzheimer’s Disease. 2024;4(11):1148-1165. [CrossRef]
- Bouziane, A., Yahyaoui, H., El-Hassani, A., et al. (2023). Effect of Periodontal Disease on Alzheimer’s Disease: A Systematic Review. Dentistry Journal, 11(1), 14.
- Teixeira, F. B., Saito, M. T., Matheus, F. C., Prediger, R. D., Yamada, E. S., Maia, C. S. F., & Lima, R. R. (2017). Periodontitis and Alzheimer’s disease: A possible comorbidity between oral chronic inflammatory condition and neuroinflammation. Frontiers in Aging Neuroscience, 9, 327. [CrossRef]
- Elashiry, M., Carroll, A., Yuan, J., Liu, Y., Hamrick, M., Cutler, C. W., Wang, Q., & Elsayed, R. (2024). Oral microbially-induced small extracellular vesicles cross the blood–brain barrier. International Journal of Molecular Sciences, 25(8), 4509. [CrossRef]
- Elsayed, R., Carroll, A., Elashiry, M., Cutler, C., Hamrick, M., Liu, Y., & Wang, Q. (2024). Oral microbially-induced exosomes cross blood-brain barrier contributing to neuroinflammation and Alzheimer’s disease pathogenesis. The Journal of Immunology, 212(1_Supplement), 1340–6038.
- Wang, R. P. H., Huang, J., Chan, K. W. Y., Leung, W. K., Goto, T., Ho, Y. S., & Chang, R. C. C. (2023). IL-1β and TNF-α play an important role in modulating the risk of periodontitis and Alzheimer’s disease. Journal of Neuroinflammation, 20, 71. [CrossRef]
- Seyedmoalemi, M. A., & Saied-Moallemi, Z. (2025). Association between periodontitis and Alzheimer’s disease: A narrative review. IBRO Neuroscience Reports, 18, 360–365. [CrossRef]
- Jimenez-Harrison DM, Butler MJ, Ijaz H. et al. (2025). Ligature-induced periodontitis in a transgenic mouse model of Alzheimer’s disease dysregulates neuroinflammation, exacerbates cognitive impairment, and accelerates amyloid pathology. Brain, Behavior, & Immunity - Health. 44, 100969. [CrossRef]
- Kwon T, Lamster IB, Levin L. Current Concepts in the Management of Periodontitis. Int Dent J. 2021 Dec;71(6):462-476. Epub 2021 Feb 19. [CrossRef]
- Graziani, F., Cei, S., La Ferla, F., Vano, M., Gennai, S., Filice, N., Gabriele, M., & D’Aiuto, F. (2010). Systemic inflammation following non-surgical periodontal therapy: A systematic review and meta-analysis. Journal of Clinical Periodontology, 46(3), 241–254.
- Yue H, Xu X, Liu Q, et al. Effects of non-surgical periodontal therapy on systemic inflammation and metabolic markers in patients undergoing haemodialysis and/or peritoneal dialysis: a systematic review and meta-analysis. BMC Oral Health. 2020 Jan 22;20:18. [CrossRef]
- Herrera, D., Montero Sánchez, T., Loyola-Rodríguez, J. P., Roldán, S., Martín, C., & Sanz, M. (2022). Treatment of stage IV periodontitis: The EFP S3-level clinical practice guideline. Journal of Clinical Periodontology, 49(S24), 165–181. [CrossRef]
- Heinz-Dieter Müller, Sigrun Eick, Andreas Moritz, Adrian Lussi, Reinhard Gruber. Cytotoxicity and Antimicrobial Activity of Oral Rinses In Vitro. Biomed Res Int. 2017 Mar 19;2017:4019723. [CrossRef]
- Cai J, Chen Z, Liu H et al. Green tea epigallocatechin-3-gallate alleviates Porphyromonas gingivalis-induced periodontitis in mice. Int Immunopharmacol. 2015 Dec;29(2):839-845. [CrossRef]
- Joshi, V., Singh, K., Singh, G., Sharma, D., & Sharma, M. (2018). EGCG nanoparticles attenuate aluminum chloride induced neurobehavioral deficits, beta amyloid and tau pathology in a rat model of Alzheimer’s disease. Frontiers in Aging Neuroscience, 10, 244. [CrossRef]
- Cano A, Ettcheto M, Chang JH, Barroso E et al. Dual-drug loaded nanoparticles of Epigallocatechin-3-gallate (EGCG)/Ascorbic acid enhance therapeutic efficacy of EGCG in a APPswe/PS1dE9 Alzheimer’s disease mice model. J Control Release. 2019 May 10;301:62–75. Polymer, surfactant (Tween 80). [CrossRef]
- Cano A, Ettcheto M, Espina M et al. Epigallocatechin-3-gallate loaded PEGylated-PLGA nanoparticles: A new anti-seizure strategy for temporal lobe epilepsy. Nanomedicine: Nanotechnology, Biology and Medicine. 14 (4) 2018, 1073-1085. [CrossRef]
- Tian M, Chen G, Xu J, et al. Epigallocatechin gallate-based nanoparticles with reactive oxygen species scavenging property for effective chronic periodontitis treatment. Chemical Engineering Journal Volume 433, Part 2, 1 April 2022, 132197. Formaldehyde subgingival injection. [CrossRef]
- Jiarong Zhong, Douglas Dickinson, Lester Sampath, Stephen Hsu. Effects of Epigallocatechin-3-Gallate-Palmitate (EC16) on In Vitro Norovirus Infection. Microbiol Infect Dis. 2021; 5(5): 1-7. [CrossRef]
- Nicolette Frank, Douglas Dickinson, Yutao Liu, Hongfang Yu, Jingwen Cai and Stephen Hsu. Facilitated Self-Assembling Technology (FAST) for the Preparation of Nanoparticles to Increase the Solubility and Bioavailability of Hydrophobic Molecules. Fortune Journal of Health Sciences, 8 (2025): 283-295. [CrossRef]
- Nicolette Frank, Douglas Dickinson, Caroline Dudish, Wesley James, Garrison Lovett, Yutao Liu, Hongfang Yu, Jingwen Cai, Bo Yao, Xiacui Jiang and Stephen Hsu. Potential Therapeutic Use of EGCG-Palmitate Nanoparticles for Norovirus Infection. Biomed J Sci & Tech Res 59(1)-2024. BJSTR. [CrossRef]
- Frank, N.; Dickinson, D.; Lovett, G.; Liu, Y.; Yu, H.; Cai, J.; Yao, B.; Jiang, X.; Hsu, S. Evaluation of Novel Nasal Mucoadhesive Nanoformulations Containing Lipid-Soluble EGCG for Long COVID Treatment. Pharmaceutics 2024, 16, 791. [CrossRef]
- Nicolette Frank, Douglas Dickinson, William Garcia, Yutao Liu, Hongfang Yu, Jingwen Cai, Sahaj Patel, Bo Yao, Xiaocui Jiang, and Stephen Hsu. Feasibility Study of Developing a Saline-Based Antiviral Nanoformulation Containing Lipid-Soluble EGCG: A Potential Nasal Drug to Treat Long COVID. 2024. Viruses. 16:196. [CrossRef]
- Dickinson D, Marsh B, Shao X, Sampath L, Yao B, Jiang X, and Stephen Hsu S. Virucidal activities of novel hand hygiene and surface disinfectant formulations containing EGCG-palmitate (EC16). American Journal of Infection Control. 2022; 50 (11), 1212-1219. [CrossRef]
- Dickinson D, Tufo MD, Liu E, Shao X and Hsu S. Bactericidal and Fungicidal Activities of Novel ProtecTeaV Formulations - Alcohol-Based Hand Hygiene and Surface Disinfectant Prototypes Containing Epigallocatechin-3-Gallate-Palmitate (EC16). Microbiol Infect Dis. 2021; 5(5): 1-8. [CrossRef]
- Tinchun Chu, Lee H. Lee, Ayuni Yussof, Sabrina Lopez, Gabriela Herrera, Priscilla Luna, Mahfuza Uddin, Laying Wu, John A. Murzaku, Douglas Dickinson, Stephen Hsu. Enhanced Sporicidal Activity of Alcohol and Epigallocatechin-Palmitate-Based Hand Hygiene Formulations Comprised of Plant-Derived Compounds. Journal of Biosciences and Medicines, 2020, 8, 89-99.
- Tinchun Chu, Lee H. Lee, Theresa Aponte, Sabrina Lopez, Giselle Lalata, Gabriela Herrera, Ayuni Yussof, Douglas Dickinson, and Stephen Hsu. Sporicidal Activity of Novel Formulations Containing Lipophilic Epigallocatechin-3-Gallate and Natural Ingredients. Microbiology & Infectious Diseases. 2019; 3:1-6. [CrossRef]
- Elsayed, R.; Kurago, Z.; Cutler, C.W.; Arce, R.M.; Gerber, J.; Celis, E.; Sultan, H.; Elashiry, M.; Meghil, M.; Sun, C.; et al. Role of dendritic cell-mediated immune response in oral homeostasis: A new mechanism of osteonecrosis of the jaw. Faseb j 2020, 34, 2595-2608. [CrossRef]
- Elashiry, M.; Elashiry, M.M.; Elsayed, R.; Rajendran, M.; Auersvald, C.; Zeitoun, R.; Rashid, M.H.; Ara, R.; Meghil, M.M.; Liu,Y.; et al. Dendritic cell derived exosomes loaded with immunoregulatory cargo reprogram local immune responses and inhibit degenerative bone disease in vivo. J. Extracell. Vesicles 2020, 9, 1795362. [CrossRef]
- Xiaogang Chu, Mahmoud Elashiry, Angelica Carroll, Celine Joyce Cornelius Timothius, Christopher W Cutler, Ranya Elsayed. The Role of Senescence in Experimental Periodontitis at the Causal Level: An in Vivo Study. Cells. 2025 Feb 5;14(3):226. [CrossRef]
- Jingwen Cai, Caroline Dudish, Amani Mouna, Angelena Jacob, Wesley James, Douglas Dickinson, Hongfang Yu, Yutao Liu, Ashish K. Sarker, Mustafa Culha, Diya Garrepally, Mizuho Kittaka, Stephen. Food-Grade Preparation of Nutraceutical Nanoparticles Using Facilitated Self-Assembling Technology (FAST) for A New Generation of Nutritional Supplements. Journal of Nanotechnology Research 8 (2026): 01-11. [CrossRef]
- Elsayed, R.; Elashiry, M.; Liu, Y.; Morandini, A.C.; El-Awady, A.; Elashiry, M.M.; Hamrick, M.;Cutler, C.W. Microbially-Induced Exosomes from Dendritic Cells Promote Paracrine Immune Senescence: Novel Mechanism of Bone Degenerative Disease in Mice. Aging Dis. 2023, 14, 136-151. [CrossRef]
- Nicolette Frank, Douglas Dickinson, Yutao Liu, Hongfang Yu, Jingwen Cai and Stephen Hsu. Facilitated Self-Assembling Technology (FAST) for the Preparation of Nanoparticles to Increase the Solubility and Bioavailability of Hydrophobic Molecules. Fortune Journal of Health Sciences, 8 (2025): 283-295. [CrossRef]
- Magdalena Paczkowska-Walendowska, Jan Grzegorzewski, Jakub Kwiatek, Marta Leśna, Judyta Cielecka-Piontek. Green Tea: A Novel Perspective on the Traditional Plant’s Potential in Managing Periodontal Diseases. Pharmaceuticals (Basel). 2025 Mar 14;18(3):409. [CrossRef]
- Yadong Wang a, Yadong Wu b, Jukun Song b. Global and Regional Burden of Periodontal Disease in Adults (1990-2021). Int Dent J. 2025 Sep 3;75(6):103883. [CrossRef]
- M Jevdjevic 1, S Listl. Global, Regional, and Country-Level Economic Impacts of Oral Conditions in 2019. J Dent Res. 2024 Nov 13;104(1):17–21. [CrossRef]
- Kamboj N, Sharma S, Kumar R. Neuroprotective insights into epigallocatechin gallate (EGCG) for neurodegenerative disorders. Explor Neurosci. 2025;4:100673. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



