Submitted:
30 March 2026
Posted:
01 April 2026
You are already at the latest version
Abstract
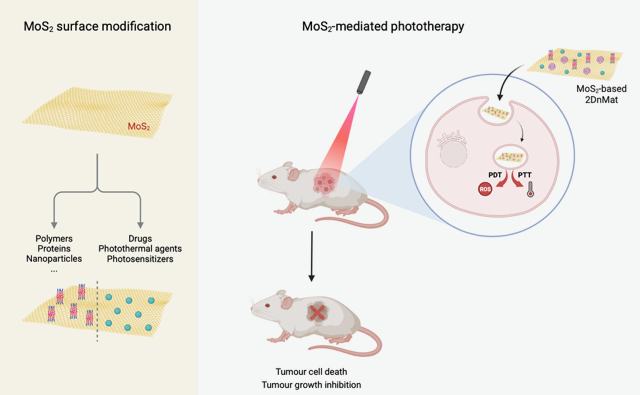
Keywords:
1. Introduction
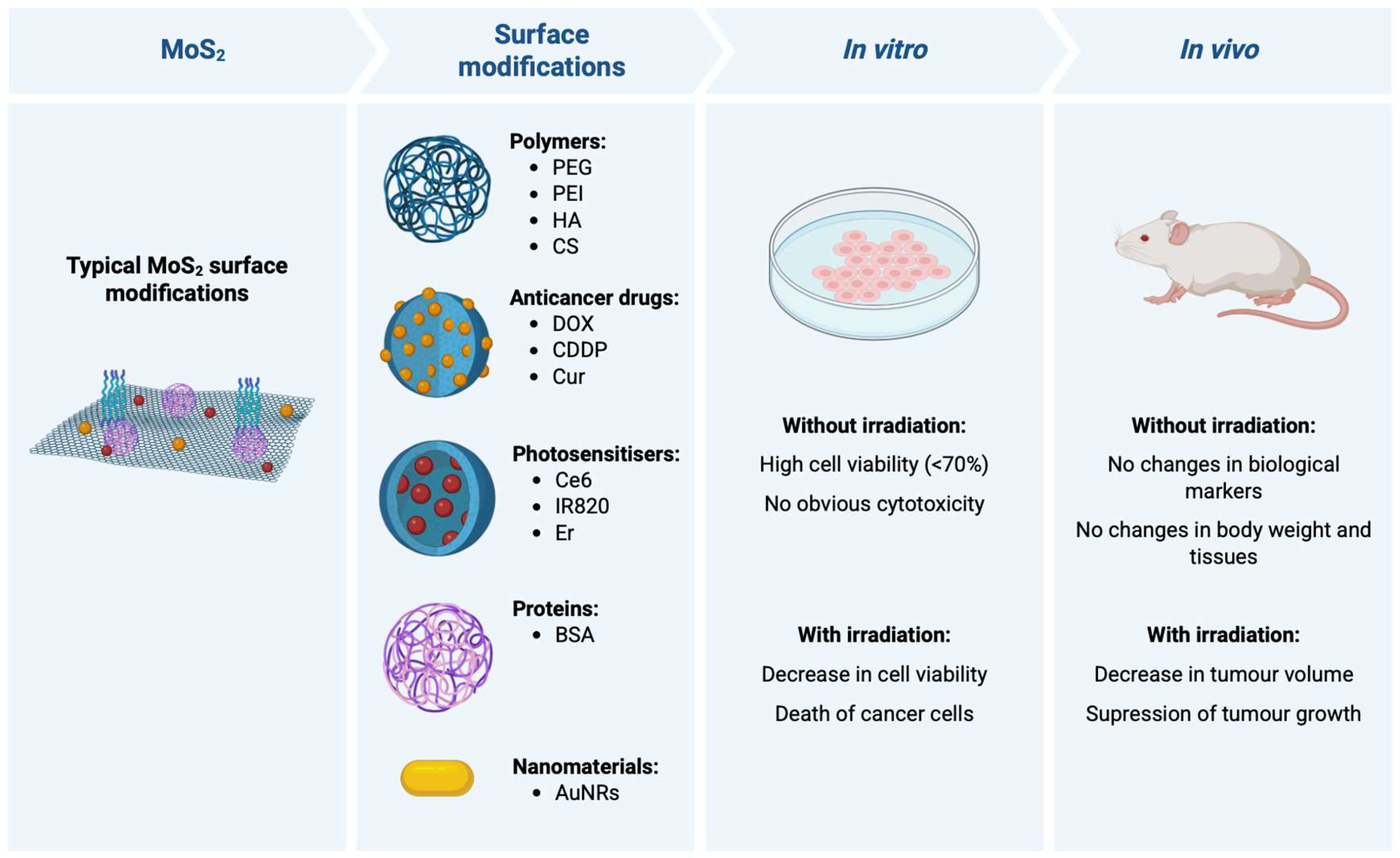
2. MoS2 Surface Functionalisation and Drug Conjugation Strategies
3. In Vitro Biocompatibility Studies
4. In Vivo Biocompatibility Studies
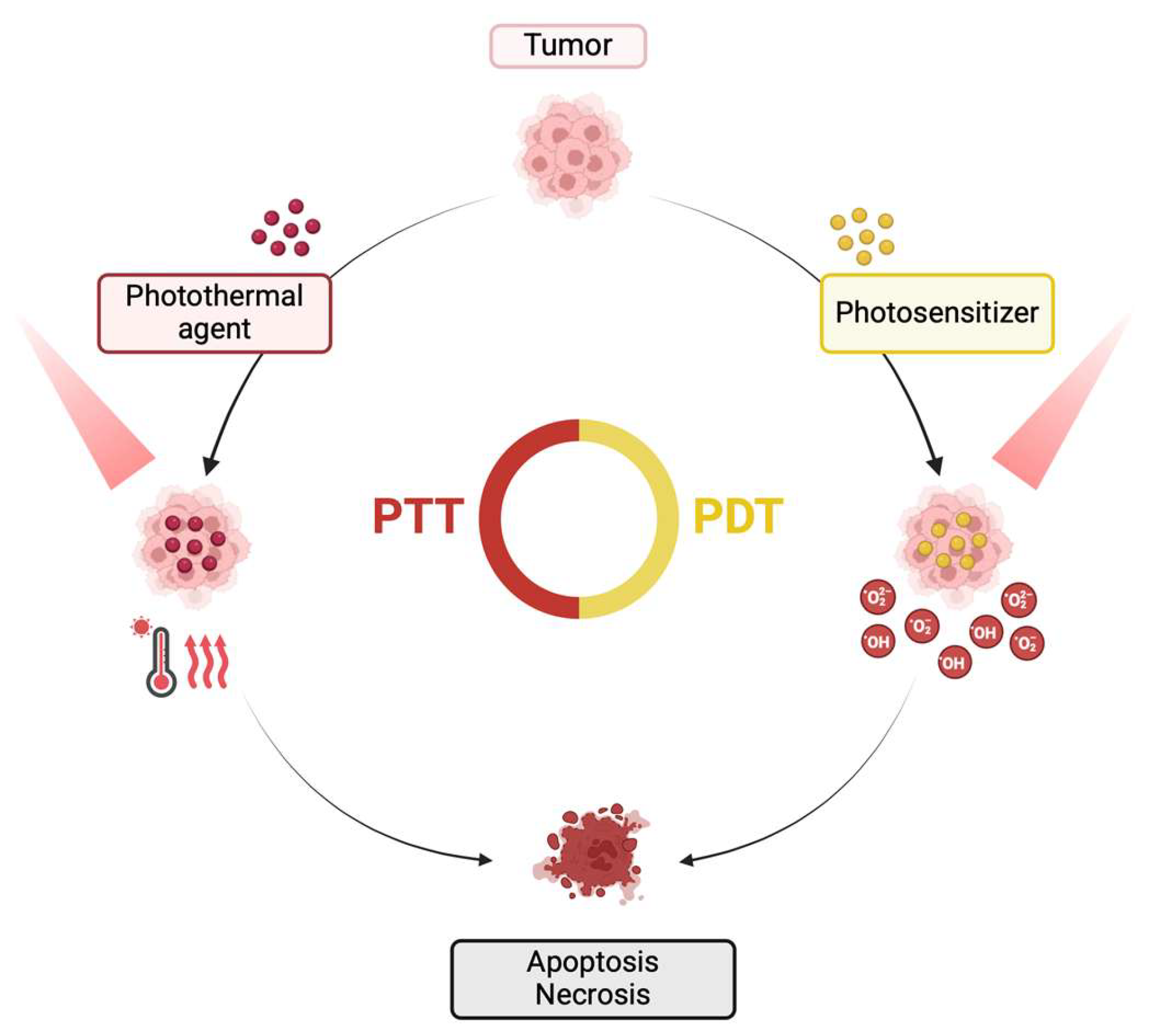
5. MoS2/drug Conjugates for Cancer Phototherapy – In Vitro Studies
6. MoS2/drug Conjugates for Cancer Phototherapy – In Vivo Studies
7. Challenges and Opportunities
8. Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brown, J.S.; et al. Updating the Definition of Cancer. Molecular Cancer Research 2023, 21, 1142–1147. [Google Scholar] [CrossRef]
- Saini, A.; et al. Cancer causes and treatments. Int. J. Pharm. Sci. Res 2020, 11, 3121–3134. [Google Scholar]
- ME, J.F.; et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. 2024. [Google Scholar]
- Zafar, A.; et al. Advancements and limitations in traditional anti-cancer therapies: a comprehensive review of surgery, chemotherapy, radiation therapy, and hormonal therapy. Discov Oncol 2025, 16, 607. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; et al. Phototherapy in cancer treatment: strategies and challenges. Signal Transduct Target Ther 2025, 10, 115. [Google Scholar] [CrossRef] [PubMed]
- Montaseri, H.; Kruger, C.A.; Abrahamse, H. Targeted photodynamic therapy using alloyed nanoparticle-conjugated 5-aminolevulinic acid for breast cancer. Pharmaceutics 2021, 13, 1375. [Google Scholar] [CrossRef]
- Liu, T.; et al. Combined photothermal and photodynamic therapy delivered by PEGylated MoS 2 nanosheets. Nanoscale 2014, 6, 11219–11225. [Google Scholar] [CrossRef]
- Ming, L.; et al. Enhancement of tumor lethality of ROS in photodynamic therapy. Cancer medicine 2021, 10, 257–268. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; et al. Applications of inorganic nanomaterials in photothermal therapy based on combinational cancer treatment. International Journal of Nanomedicine 2020, 1903–1914. [Google Scholar] [CrossRef]
- Algorri, J.F.; et al. Advanced Light Source Technologies for Photodynamic Therapy of Skin Cancer Lesions. Pharmaceutics 2023, 15, 2075. [Google Scholar] [CrossRef]
- Mallidi, S.; et al. In vivo evaluation of battery-operated light-emitting diode-based photodynamic therapy efficacy using tumor volume and biomarker expression as endpoints. J Biomed Opt 2015, 20, 048003. [Google Scholar] [CrossRef]
- Liu, H.; et al. Development and evaluation of a low-cost, portable, LED-based device for PDT treatment of early-stage oral cancer in resource-limited settings. Lasers Surg Med 2019, 51, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, R.K.; et al. Advancing Cancer Treatment and Diagnosis: A Review on Photodynamic Therapy Using OLED Technology. Molecules 2025, 30, 1305. [Google Scholar] [CrossRef]
- Banerjee, S.M.; et al. Photodynamic therapy in primary breast cancer. Journal of clinical medicine 2020, 9, 483. [Google Scholar] [CrossRef]
- Algorri, J.F.; et al. Light technology for efficient and effective photodynamic therapy: A critical review. Cancers 2021, 13, 3484. [Google Scholar] [CrossRef]
- Kwiatkowski, S.; et al. Photodynamic therapy–mechanisms, photosensitizers and combinations. Biomedicine & pharmacotherapy 2018, 106, 1098–1107. [Google Scholar]
- Dudzik, T.; Domański, I.; Makuch, S. The impact of photodynamic therapy on immune system in cancer - an update. Front Immunol 2024, 15, 1335920. [Google Scholar] [CrossRef]
- Han, Y.; et al. Clinical application of immunogenic cell death inducers in cancer immunotherapy: turning cold tumors hot. Front Cell Dev Biol 2024, 12, 1363121. [Google Scholar] [CrossRef]
- Lin, L.; et al. Nano-photosensitizers for enhanced photodynamic therapy. Photodiagnosis and Photodynamic Therapy 2021, 36, 102597. [Google Scholar] [CrossRef]
- Aebisher, D.; Szpara, J.; Bartusik-Aebisher, D. Advances in Medicine: Photodynamic Therapy. Int J Mol Sci 2024, 25. [Google Scholar] [CrossRef] [PubMed]
- Merlin, J.P.J.; Crous, A.; Abrahamse, H. Combining Photodynamic Therapy and Targeted Drug Delivery Systems: Enhancing Mitochondrial Toxicity for Improved Cancer Outcomes. Int J Mol Sci 2024, 25. [Google Scholar] [CrossRef] [PubMed]
- da Silva, D.B.; et al. Protoporphyrin IX (PpIX) loaded PLGA nanoparticles for topical Photodynamic Therapy of melanoma cells. Photodiagnosis and Photodynamic Therapy 2021, 35, 102317. [Google Scholar] [CrossRef]
- Deng, X.; Shao, Z.; Zhao, Y. Solutions to the Drawbacks of Photothermal and Photodynamic Cancer Therapy. Advanced Science 2021, 8, 2002504. [Google Scholar] [CrossRef] [PubMed]
- Huis In 't Veld, R.V.; et al. Current Challenges and Opportunities of Photodynamic Therapy against Cancer. Pharmaceutics 2023, 15. [Google Scholar] [CrossRef]
- Xiong, T.; et al. Recent Progress of Molecular Design in Organic Type I Photosensitizers. Small 2025, 21. [Google Scholar] [CrossRef]
- Zuo, T.; et al. Engineering tumor-oxygenated nanomaterials: advancing photodynamic therapy for cancer treatment. Frontiers in Bioengineering and Biotechnology 2024, 12, 2024. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; et al. Nanoplatform-enhanced photodynamic therapy for the induction of immunogenic cell death. J Control Release 2024, 365, 1058–1073. [Google Scholar] [CrossRef]
- Thiruppathi, J.; et al. Enhancing cancer immunotherapy with photodynamic therapy and nanoparticle: making tumor microenvironment hotter to make immunotherapeutic work better. Front Immunol 2024, 15, 1375767. [Google Scholar] [CrossRef]
- Liu, M.; et al. Functionalized MoS2-based nanomaterials for cancer phototherapy and other biomedical applications. ACS Materials Letters 2021, 3, 462–496. [Google Scholar] [CrossRef]
- Wang, J.; et al. MoS2-based nanocomposites for cancer diagnosis and therapy. Bioactive Materials 2021, 6, 4209–4242. [Google Scholar] [CrossRef]
- Jiang, W.; et al. Near-Infrared-II Nanomaterials for Activatable Photodiagnosis and Phototherapy. Chemistry – A European Journal 2024, 30, e202400816. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; et al. Applications of nanotheranostics in the second near-infrared window in bioimaging and cancer treatment. Nanoscale 2024, 16, 21697–21730. [Google Scholar] [CrossRef]
- Yang, G.-G.; et al. Multifunctional low-temperature photothermal nanodrug with in vivo clearance, ROS-Scavenging and anti-inflammatory abilities. Biomaterials 2019, 216, 119280. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; et al. Enhancing the efficiency of mild-temperature photothermal therapy for cancer assisting with various strategies. Pharmaceutics 2022, 14, 2279. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; et al. Recent advances in biomaterials based near-infrared mild photothermal therapy for biomedical application: A review. International Journal of Biological Macromolecules 2024, 278, 134746. [Google Scholar] [CrossRef]
- Lukácsi, S.; Munkácsy, G.; Győrffy, B. Harnessing Hyperthermia: Molecular, Cellular, and Immunological Insights for Enhanced Anticancer Therapies. Integr Cancer Ther 2024, 23, 15347354241242094. [Google Scholar] [CrossRef] [PubMed]
- Premji, T.P.; et al. Functionalized Nanomaterials for Inhibiting ATP-Dependent Heat Shock Proteins in Cancer Photothermal/Photodynamic Therapy and Combination Therapy. Nanomaterials (Basel) 2024, 14. [Google Scholar] [CrossRef]
- Debnath, M.; et al. Surface engineered nanohybrids in plasmonic photothermal therapy for cancer: Regulatory and translational challenges. Nanotheranostics 2024, 8, 202–218. [Google Scholar] [CrossRef]
- Costa-Almeida, R.; et al. Near-infrared radiation-based mild photohyperthermia therapy of non-melanoma skin cancer with PEGylated reduced nanographene oxide. Polymers 2020, 12, 1840. [Google Scholar] [CrossRef]
- Cheng, Z.; et al. Nanomaterials for cancer therapy: current progress and perspectives. Journal of Hematology & Oncology 2021, 14, 85. [Google Scholar] [CrossRef]
- Zhang, R.; et al. Recent advances in two-dimensional materials for drug delivery. Journal of Materials Chemistry B 2024, 12, 12437–12469. [Google Scholar] [CrossRef]
- Kuang, F.; et al. Post-Graphene 2D Materials: Structures, Properties, and Cancer Therapy Applications. Advanced Healthcare Materials 2024, 13, 2302604. [Google Scholar] [CrossRef]
- Li, C.; et al. Recent Advances of Emerging Metal-Containing Two-Dimensional Nanomaterials in Tumor Theranostics. Int J Nanomedicine 2024, 19, 805–824. [Google Scholar] [CrossRef]
- Mohammadpour, Z.; Majidzadeh-A, K. Applications of two-dimensional nanomaterials in breast cancer theranostics. ACS Biomaterials Science & Engineering 2020, 6, 1852–1873. [Google Scholar] [CrossRef]
- Silva, F.; et al. 2D Nanomaterials and Their Drug Conjugates for Phototherapy and Magnetic Hyperthermia Therapy of Cancer and Infections. Small 2024, 20, e2306137. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; et al. 2D nanomaterials-based delivery systems and their potentials in anticancer synergistic photo-immunotherapy. Colloids and Surfaces B: Biointerfaces 2024, 242, 114074. [Google Scholar] [CrossRef] [PubMed]
- Parihar, A.; Gaur, K.; Sarbadhikary, P. Advanced 2D Nanomaterials for Phototheranostics of Breast Cancer: A Paradigm Shift. Adv Biol (Weinh) 2025, 9, e2400441. [Google Scholar] [CrossRef] [PubMed]
- Rafieerad, A.; et al. Periodic Table of Immunomodulatory Elements and Derived Two-Dimensional Biomaterials. Advanced Science 2025, 12, 2406324. [Google Scholar] [CrossRef]
- Sukur, S.; Ranc, V. Magnetic 2D Transition-Metal-Based Nanomaterials in Biomedicine: Opportunities and Challenges in Cancer Therapy. Materials 2025, 18, 2570. [Google Scholar] [CrossRef]
- Cheng, L.; et al. 2D nanomaterials for cancer theranostic applications. Advanced Materials 2020, 32, 1902333. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, X.; Yue, H. Two-dimensional nanomaterials induced nano-bio interfacial effects and biomedical applications in cancer treatment. J Nanobiotechnology 2024, 22, 67. [Google Scholar] [CrossRef]
- Pourmadadi, M.; et al. A comprehensive review of synthesis, structure, properties, and functionalization of MoS2; emphasis on drug delivery, photothermal therapy, and tissue engineering applications. Journal of Drug Delivery Science and Technology 2022, 76, 103767. [Google Scholar] [CrossRef]
- Chakraborty, G.; Padmashree, R.; Prasad, A. Recent advancement of surface modification techniques of 2-D nanomaterials. Materials Science and Engineering: B 2023, 297, 116817. [Google Scholar] [CrossRef]
- Yu, X.; et al. Recent developments in two-dimensional molybdenum disulfide-based multimodal cancer theranostics. Journal of Nanobiotechnology 2024, 22, 515. [Google Scholar] [CrossRef]
- Ghosh, S.; Lai, J.Y. An insight into the dual role of MoS2-based nanocarriers in anticancer drug delivery and therapy. Acta Biomater 2024, 179, 36–60. [Google Scholar] [CrossRef]
- Sasanipoor, F.; Zhang, Z. Molybdenum Disulfide Nanocomposites for Cancer Diagnosis and Therapeutics: Biosensors, Bioimaging, and Phototherapy. Adv Healthc Mater 2025, 14, e2500655. [Google Scholar] [CrossRef]
- Samy, O.; et al. A Review on MoS2 Properties, Synthesis, Sensing Applications and Challenges. Crystals 2021, 11, 355. [Google Scholar] [CrossRef]
- Liu, J.; et al. Multifunctional MoS2 composite nanomaterials for drug delivery and synergistic photothermal therapy in cancer treatment. Ceramics International 2022, 48, 22378–22386. [Google Scholar] [CrossRef]
- Dong, X.; et al. Intelligent MoS2 nanotheranostic for targeted and enzyme-/pH-/NIR-responsive drug delivery to overcome cancer chemotherapy resistance guided by PET imaging. ACS applied materials & interfaces 2018, 10, 4271–4284. [Google Scholar]
- Li, J.-Q.; et al. An erythrocyte membrane-camouflaged biomimetic nanoplatform for enhanced chemo-photothermal therapy of breast cancer. Journal of materials chemistry B 2022, 10, 2047–2056. [Google Scholar] [CrossRef]
- Yin, W.; et al. High-throughput synthesis of single-layer MoS2 nanosheets as a near-infrared photothermal-triggered drug delivery for effective cancer therapy. ACS nano 2014, 8, 6922–6933. [Google Scholar] [CrossRef]
- Zeng, G.; et al. Surface modification and drug delivery applications of MoS2 nanosheets with polymers through the combination of mussel inspired chemistry and SET-LRP. Journal of the Taiwan Institute of Chemical Engineers 2018, 82, 205–213. [Google Scholar] [CrossRef]
- Wang, W.; et al. In situ growth of MoS2@ AuNRs nanoparticles with synergistically enhanced NIR response for controlled drug release. Materials Today Communications 2023, 37, 107448. [Google Scholar] [CrossRef]
- Liu, J.; et al. MoS2-based nanocomposites with high photothermal conversion efficiency for combinational photothermal/photodynamic tumor therapy. Journal of Alloys and Compounds 2024, 970, 172489. [Google Scholar] [CrossRef]
- Chen, Z.; et al. Targeted co-delivery of curcumin and erlotinib by MoS2 nanosheets for the combination of synergetic chemotherapy and photothermal therapy of lung cancer. Journal of Nanobiotechnology 2023, 21, 333. [Google Scholar] [CrossRef]
- Chibh, S.; et al. Photoresponsive and Shape-Switchable MoS2–Peptide-Hybrid Nanosystems for Enacting Photochemo and siRNA-Mediated Gene Therapy in Glioma. ACS Applied Materials & Interfaces 2025, 17, 29318–29340. [Google Scholar]
- Yin, Y.; et al. Thermo-responsive and biodegradable MoS2-based nanoplatform for tumor therapy and postoperative wound management. Journal of Colloid and Interface Science 2025(686), 634–649. [CrossRef]
- Gonçalves, M.; et al. A glance over doxorubicin based-nanotherapeutics: From proof-of-concept studies to solutions in the market. Journal of Controlled Release 2020, 317, 347–374. [Google Scholar] [CrossRef]
- Murali, A.; et al. Emerging 2D nanomaterials for biomedical applications. Materials Today 2021, 50, 276–302. [Google Scholar] [CrossRef]
- Kyriakides, T.R.; et al. Biocompatibility of nanomaterials and their immunological properties. Biomedical Materials 2021, 16, 042005. [Google Scholar] [CrossRef]
- Gruber, S.; Nickel, A. Toxic or not toxic? The specifications of the standard ISO 10993-5 are not explicit enough to yield comparable results in the cytotoxicity assessment of an identical medical device. Front Med Technol 2023, 5, 1195529. [Google Scholar] [CrossRef]
- Van Haute, D.; Liu, A.T.; Berlin, J.M. Coating Metal Nanoparticle Surfaces with Small Organic Molecules Can Reduce Nonspecific Cell Uptake. ACS Nano 2018, 12, 117–127. [Google Scholar] [CrossRef]
- Qie, Y.; et al. Surface modification of nanoparticles enables selective evasion of phagocytic clearance by distinct macrophage phenotypes. Sci Rep 2016, 6, 26269. [Google Scholar] [CrossRef]
- Zheng, H.; et al. Effect of Nanoparticle Surface Coating on Cell Toxicity and Mitochondria Uptake. J Biomed Nanotechnol 2017, 13, 155–166. [Google Scholar] [CrossRef]
- Almalik, A.; et al. Hyaluronic Acid Coated Chitosan Nanoparticles Reduced the Immunogenicity of the Formed Protein Corona. Sci Rep 2017, 7, 10542. [Google Scholar] [CrossRef]
- Almalik, A.; et al. Cellular responses of hyaluronic acid-coated chitosan nanoparticles. Toxicol Res (Camb) 2018, 7, 942–950. [Google Scholar] [CrossRef]
- Wu, J.; Yu, Y.; Su, G. Safety assessment of 2D MXenes: in vitro and in vivo. Nanomaterials 2022, 12, 828. [Google Scholar] [CrossRef]
- Zhang, Y.; et al. Recent Progress on NIR-II Photothermal Therapy. Front Chem 2021, 9, 728066. [Google Scholar] [CrossRef]
- Wu, X.; et al. Deep-Tissue Photothermal Therapy Using Laser Illumination at NIR-IIa Window. Nanomicro Lett 2020, 12, 38. [Google Scholar] [CrossRef]
- Toscano, F.; Torres-Arias, M. Nanoparticles cellular uptake, trafficking, activation, toxicity and in vitro evaluation. Curr Res Immunol 2023, 4, 100073. [Google Scholar] [CrossRef]
- Donahue, N.D.; Acar, H.; Wilhelm, S. Concepts of nanoparticle cellular uptake, intracellular trafficking, and kinetics in nanomedicine. Adv Drug Deliv Rev 2019, 143, 68–96. [Google Scholar] [CrossRef]
- Ngo, W.; et al. Why nanoparticles prefer liver macrophage cell uptake in vivo. Advanced Drug Delivery Reviews 2022, 185, 114238. [Google Scholar] [CrossRef]
- Hao, J.; et al. In Vivo Long-Term Biodistribution, Excretion, and Toxicology of PEGylated Transition-Metal Dichalcogenides MS(2) (M = Mo, W, Ti) Nanosheets. Adv Sci (Weinh) 2017, 4, 1600160. [Google Scholar] [CrossRef]
- Cisneros, E.P.; et al. The role of patient-specific variables in protein corona formation and therapeutic efficacy in nanomedicine. J Nanobiotechnology 2024, 22, 714. [Google Scholar] [CrossRef]
- Cao, M.; et al. Molybdenum derived from nanomaterials incorporates into molybdenum enzymes and affects their activities in vivo. Nature Nanotechnology 2021, 16, 708–716. [Google Scholar] [CrossRef]
- Dogra, P.; et al. Establishing the effects of mesoporous silica nanoparticle properties on in vivo disposition using imaging-based pharmacokinetics. Nature Communications 2018, 9, 4551. [Google Scholar] [CrossRef]
- López-Estévez, A.M.; et al. Personalized Cancer Nanomedicine: Overcoming Biological Barriers for Intracellular Delivery of Biopharmaceuticals. Advanced Materials 2024, 36, 2309355. [Google Scholar] [CrossRef]
- Saadh, M.J.; et al. Stealth Nanocarriers in Cancer Therapy: a Comprehensive Review of Design, Functionality, and Clinical Applications. AAPS PharmSciTech 2024, 25, 140. [Google Scholar] [CrossRef]
- Desai, N.; et al. Nanoparticle Therapeutics in Clinical Perspective: Classification, Marketed Products, and Regulatory Landscape. Small 2025, 21, 2502315. [Google Scholar] [CrossRef]
- Wong, K.-Y.; et al. Metal–Drug Coordination Nanoparticles and Hydrogels for Enhanced Delivery. Advanced Materials 2024, 36, 2404053. [Google Scholar] [CrossRef]
- Yan, Z.; et al. Long-term exposure of molybdenum disulfide nanosheets leads to hepatic lipid accumulation and atherogenesis in apolipoprotein E deficient mice. NanoImpact 2023, 30, 100462. [Google Scholar] [CrossRef]
- Qi, Z.; et al. Molybdenum disulfide induces growth inhibition and autophagy-dependent hepatocyte cell death through directly binding and regulating the activity of MST2. Materials Today Bio 2025, 35, 102394. [Google Scholar] [CrossRef]
- Li, J.; et al. Dissolution of 2D Molybdenum Disulfide Generates Differential Toxicity among Liver Cell Types Compared to Non-Toxic 2D Boron Nitride Effects. Small 2021, 17, e2101084. [Google Scholar] [CrossRef]
- Lima, A.F.; Justo, G.Z.; Sousa, A.A. Realizing active targeting in cancer nanomedicine with ultrasmall nanoparticles. Beilstein J Nanotechnol 2024, 15, 1208–1226. [Google Scholar] [CrossRef]
- Long, X.; et al. Injectable 2D-MoS2-integrated Bioadhesive Hydrogel as Photothermal-Derived and Drug-Delivery Implant for Colorectal Cancer Therapy. Advanced Healthcare Materials 2025, 14, 2404842. [Google Scholar] [CrossRef] [PubMed]
- Sun, R.; et al. Nanomaterials and their composite scaffolds for photothermal therapy and tissue engineering applications. Sci Technol Adv Mater 2021, 22, 404–428. [Google Scholar] [CrossRef]
- Wang, X.; et al. A 3D-printed scaffold with MoS2 nanosheets for tumor therapy and tissue regeneration. NPG Asia Materials 2017, 9, e376–e376. [Google Scholar] [CrossRef]
- Sasanipoor, F.; Zhang, Z. Molybdenum Disulfide Nanocomposites for Cancer Diagnosis and Therapeutics: Biosensors, Bioimaging, and Phototherapy. Advanced Healthcare Materials 2025, 14, 2500655. [Google Scholar] [CrossRef] [PubMed]
- Mülhopt, S.; et al. Characterization of Nanoparticle Batch-To-Batch Variability. Nanomaterials 2018, 8, 311. [Google Scholar] [CrossRef]
- Faria, M.; et al. Minimum information reporting in bio–nano experimental literature. Nature Nanotechnology 2018, 13, 777–785. [Google Scholar] [CrossRef]
- Sharifi, S.; et al. Importance of Standardizing Analytical Characterization Methodology for Improved Reliability of the Nanomedicine Literature. Nano-Micro Letters 2022, 14, 172. [Google Scholar] [CrossRef]
- Camacho Vieira, C.; et al. Is it advantageous to use quality by design (QbD) to develop nanoparticle-based dosage forms for parenteral drug administration? Int J Pharm 2024, 657, 124163. [Google Scholar] [CrossRef] [PubMed]
- Nagpal, S.; et al. Revisiting nanomedicine design strategies for follow-on products: A model-informed approach to optimize performance. Journal of Controlled Release 2024, 376, 1251–1270. [Google Scholar] [CrossRef]
- Csóka, I.; et al. Regulatory Considerations, Challenges and Risk-based Approach in Nanomedicine Development. Curr Med Chem 2021, 28, 7461–7476. [Google Scholar] [CrossRef]
- Nag, S.; et al. Nanomaterials-assisted photothermal therapy for breast cancer: State-of-the-art advances and future perspectives. Photodiagnosis and Photodynamic Therapy 2024, 45, 103959. [Google Scholar] [CrossRef]
- Zhu, L.; et al. A Biodegradable 2D Metallic MoS(2) Genesheet for Synergistic NIR-II Photothermal Immunotherapy. Small 2025, 21, e2502577. [Google Scholar] [CrossRef]
- Younas, A.; et al. Recent advances in cancer nanomedicine: From smart targeting to personalized therapeutics - pioneering a new era in precision oncology. Materials Today Bio 2026, 36, 102660. [Google Scholar] [CrossRef]
- Administration, U.S.F.D. Drug Products, Including Biological Products, that Contain Nanomaterials - Guidance for Industry. 2022. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/drug-products-including-biological-products-contain-nanomaterials-guidance-industry (accessed on 8 March 2026).
- Agency, E.M. Reflection paper on surface coatings: general issues for consideration regarding parenteral administration of coated nanomedicine products. 2013. Available online: https://www.ema.europa.eu/en/news/european-medicines-agency-publishes-reflection-paper-general-issues-consideration-regarding-coated-nanomedicines (accessed on 8 March 2026).
- Liu, T.; et al. Ultra-small MoS2 nanodots with rapid body clearance for photothermal cancer therapy. Nano Research 2016, 9, 3003–3017. [Google Scholar] [CrossRef]
- Nieves, L.M.; et al. Renally Excretable Molybdenum Disulfide Nanoparticles as Contrast Agents for Dual-Energy Mammography and Computed Tomography. Bioconjugate Chemistry 2024, 35, 2006–2014. [Google Scholar] [CrossRef] [PubMed]
| Material | Therapeutic cargo | Functionalisation strategy | Key effects | Refs. |
|---|---|---|---|---|
| Biotin-BSA-PEI-LA-MoS2-LA-PEG | DOX Drug loading: EE = 23% DL = 34% (drug:material ratio = 2.5:1, pH 5.5) Drug release: 15% (pH 7.0, 72 h) 30% (pH 5.5, 72 h) Drug release with irradiation (laser 808 nm, 1.5 W·cm-2, 30 min) 23% (pH 7.0, 72 h) 42% (pH 5.5, 72 h) |
1) LA-PEI adsorption on MoS2 2) PEI-LA-MoS2-LA-PEG synthesized by adsorption 3) Biotin-BSA obtained by EDC/NHS and adsorption on PEI-LA-MoS2-LA-PEG 4) DOX adsorption on Biotin-BSA-PEI-LA-MoS2-LA-PEG |
Photothermal output unchanged over four laser on/off cycles at 1.5 W·cm⁻² (10 min heating/10 min cooling per cycle) Increased cellular uptake (biotin-mediated targeting) |
[58] |
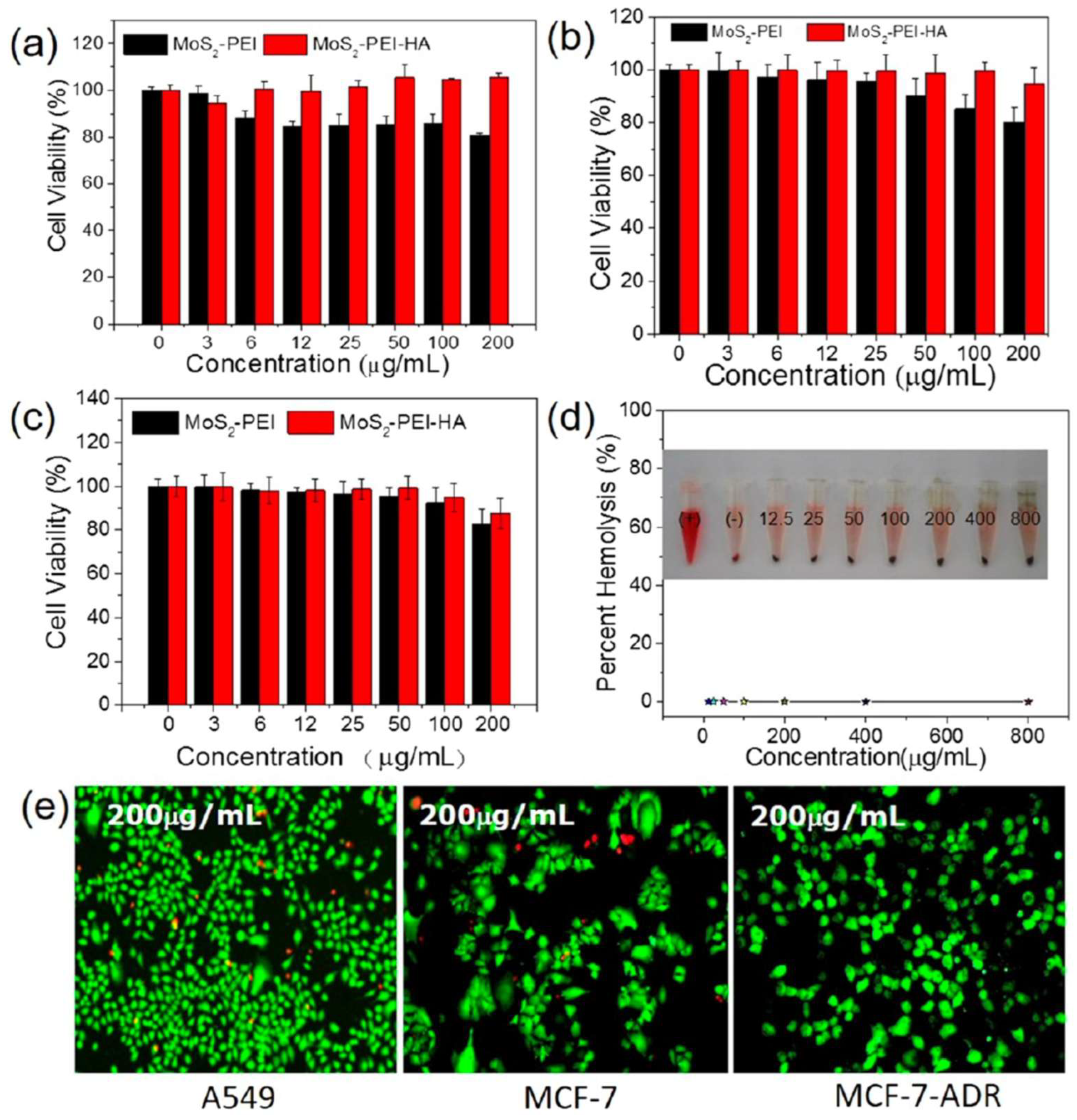
| MoS2-PEI-HA | DOX Drug loading: DL = 34% (drug:material weight ratio n/s, pH 8.0) Drug release: 11% (pH 7.4, 6 h) 25% (pH 5.0, 6 h) Drug release with HAase: 23% (pH 7.4, 6 h) 42% (pH 5.0, 6 h) Drug release with irradiation (laser 808 nm, 0.6 W·cm-2, 10 min) 35.8% (pH 5.0, 6 h) Drug release with irradiation + HAase: 77% (pH 5.0, 6 h) |
1) MoS2-PEI formation through electrostatic interaction 2) MoS2-PEI-HA synthesized by coupling -NH2 with -COOH of HA 3) DOX adsorption on MoS2-PEI-HA |
Maintained photothermal performance over four 600 s irradiation/cooling cycles at 1.0 W·cm⁻² Improved biocompatibility (HA) CD44 targeting (via HA) Enzyme-responsive DOX release (HA degradation) Zeta Potential: MoS2 = −20 mV MoS2-PEI = 20 mV MoS2-PEI-HA = −18 mV |
[59] |
| MoS2-RBC | DOX Drug loading: DL = 99% (drug:material ratio = 2:1, pH 7.4) Drug release: ~14.5% (pH 5.5, 4 h) ~7% (pH 7.4, 4 h) Drug release with irradiation (laser 808 nm, 2 W·cm-2) ~15% (pH 5.5, 4 h) ~8% (pH 7.4, 4 h) |
1) MoS2-RBC formation by ultrasonic uniform dispersion 2) DOX adsorption on MoS2-RBC |
Improved stability in water (evenly dispersed in water, PBS, and RPMI-1640 + 10% serum for 48 h, whereas unmodified MoS2 showed visible sedimentation by 24 h) Immune evasion (reduced macrophage phagocytosis and protein adsorption) Zeta Potential: MoS2 = −47.97 mV MoS2-RBC = −36.43 mV MoS2-RBC-DOX = −17.37 mV |
[60] |
| MoS2-CS | DOX Drug loading: DL = 32% (drug:material ratio n/s, pH 8.0) Drug release: 6% (pH 8.0, 1 h) Drug release with irradiation (laser 808 nm, 0.8 W·cm-2, 10 min) 12.4% (pH 8.0, 1 h) |
1) MoS2-CS formation during the exfoliation process 2) DOX is noncovalently loaded onto MoS2-CS |
High physiological stability (well dispersed in deionized water up to 1 mg·mL⁻¹ and stable in various physiological solutions for at least 1 week) Improved biocompatibility Zeta Potential: MoS2-CS = 15 mV DOX = −15 mV |
[61] |
| MoS2-PDA-poly(MPC-IA) | CDDP Drug loading: DL = 55% (drug:material ratio = 0.27:1, pH 7.4) Drug release: 49% (pH 5.5, 48 h) 15% (pH 7.4, 48 h) |
1) MoS2-PDA formation through the self-polymerization of dopamine 2) Surface-initiated SET-LRP of MPC and IA monomers 3) CDDP loaded on via coordination with carboxyl groups |
Improved dispersibility in PBS (stable in PBS for at least 24 h; unmodified MoS2 precipitated within 5 min, while MoS2-PDA was almost completely deposited by 12 h) Zeta Potential: MoS2-PDA-poly(MPC-IA) = −32.3 eV |
[62] |
| MoS2-PEG | Ce6 Drug loading: DL = 30% (drug:material ratio = 2.5:1, pH 6.0) |
1) PEG adsorption on MoS2 2) MoS2-PEG load Ce6 by physical adsorption |
Reported as stable in physiological solutions and highly water-soluble after PEGylation Enhanced uptake Generation of ROS |
[7] |
| MoS2-AuNRs-HAP-PDA | DOX Drug loading: EE = 80% (drug:material ratio = 0.075:1, pH 7.4) Drug release: 31% (pH 4.5, 10 h) ~12% (pH 7.4, 10 h) Drug release with irradiation (laser 808 nm, 4 W·cm-2) 49% (pH 4.5, 10 h) ~15% (pH 7.4, 10 h) |
1) In situ growth of AuNRs on MoS2 2) MoS2-AuNRs-HAP-PDA formation by self-assembly of HAP and PDA 3) DOX loaded on MoS2-AuNRs-HAP-PDA via PDA adhesion and HAP electrostatic interaction |
Increased stability in aqueous solutions (MA remained uniformly dispersed in deionized water for 6 days, whereas MoS2 showed distinct aggregation after 3 days) Enhanced PTT efficacy (AuNRs) Zeta Potential: MoS2 = −20.63 mV MoS2-AuNRs = 44.57 mV MoS2-AuNRs-HAP = −2.05 mV MoS2-AuNRs-HAP-PDA = −7.45 mV |
[63] |
| MoS2-PEI | IR820 Drug loading: DL = 9% (drug:material ratio = 1:3, pH 7.4) |
1) PEI adsorption on MoS2 2) IR820 loading on MoS2-PEI via electrostatic interaction |
Generation of ROS Zeta Potential: MoS2 = −47.6 eV MoS2-PEI = 28.3 mV MoS2-PEI-IR820 = −19.5 mV |
[64] |
| MoS2-PEG-Biotin | Cur Drug loading: DL = 22% (drug:material ratio n/s) Drug release: 7% (2 h) Drug release with irradiation (laser 808 nm, 0.6 W·cm-2, 10 min) 17% (2 h) Er Drug loading: DL = 10% (drug:material ratio n/s) Drug release: 5% (2 h) Drug release with irradiation (laser 808 nm, 0.6 W·cm-2, 10 min) 12% (2 h) |
1) PEG-Biotin grafted onto MoS2 via thiol binding 2) Cur and Er adsorption on MoS2-PEG-Biotin |
Remarkable physiological stability (well dispersed in DI water, PBS, and cell medium for 7 days) Increased cellular uptake (biotin-mediated targeting) Zeta Potential: MoS2 = −44 mV MoS2-PEG-Biotin = −36 mV |
[65] |
| FA-MoS2-peptide | DOX Drug loading: EE = 30% (drug:material ratio n/s) Drug release: ~63% (8 h) Drug release with irradiation (laser 808 nm, 1.35 W·cm-2, 3 min) ~90% (8 h) Anti-Gal-1 siRNA Drug loading: EE = 67% (drug:material ratio n/s) |
1) Bulk MoS2 exfoliated in aqueous self-assembled peptide NSs 2) FA conjugated via EDC/NHS 3) DOX post loaded and anti-Gal-1 siRNA loaded electrostatically |
Enhanced targeting and cellular uptake (FA) siRNA protection against ribonuclease degradation |
[66] |
| MoS2-LAU | AIPH Drug loading: DL = ~70% (drug:material ratio 25:1) Drug release: ~2% (50 min) Drug release with irradiation (laser 808 nm, 1.0 W·cm-2, 5 min on and 20 min off cycles) ~17% (50 min) |
1) Hydrothermal MoS2 synthesis 2) AIPH physically adsorbed onto MoS2 3) LA coating |
Thermo-responsive release (LA) Improved colloidal stability (hydrodynamic diameter showed no significant fluctuation over 5 days in DMEM, PBS, saline, or water) ROS generation in hypoxic environment |
[67] |
| Material | Size | Culture conditions and cell viability | Uptake and location |
Additional outcomes | Refs. |
|---|---|---|---|---|---|
| Biotin-BSA-PEI-LA-MoS2-LA-PEG | 200 nm (thickness: < 10 nm) |
HeLa, 24 h incubation ~105% (500 µg·mL-1) Concentrations tested: 0–500 µg·mL-1 |
Biotin-mediated endocytosis | - | [58] |
| MoS2-PEI-HA | 30-50 nm (thickness: 5-7 nm) |
A549, 24 h incubation ~105% (200 µg·mL-1) MCF-7, 24 h incubation ~95% (200 µg·mL-1) Concentrations tested: 0–200 µg·mL-1 |
CD44-mediated uptake | Negligible haemolysis (800 µg·mL-1, 3 h incubation with RBC) | [59] |
| MoS2-RBC | 232 nm | MCF-7, 24 h incubation ~101% (50 µg·mL-1) Concentrations tested: 2.5–50 µg·mL-1 |
Evenly distributed in cytoplasm | Reduced phagocytosis by macrophages Reduced BSA adsorption |
[60] |
| MoS2-CS | 80 nm (thickness: 4-6 nm) |
KB, 24 h incubation 85% (400 µg·mL-1) Panc-1, 24 h incubation ~99% (400 µg·mL-1) Concentrations tested: 0–400 µg·mL-1 |
Distributed in cytoplasm | Negligible haemolysis (800 µg·mL-1, 3 h incubation with RBC) | [61] |
| MoS2-PDA-poly(MPC-IA) | n/s | L929, 24 h incubation 90% (100 µg·mL-1) Concentrations tested: 0–100 µg·mL-1 |
n/s | - | [62] |
| MoS2-PEG | n/s (thickness: 1 nm) |
4T1, 12 h incubation 85% (4 mg·mL-1) Concentrations tested: 0.25–8 mg·mL-1 |
Distributed in cytoplasm | - | [7] |
| MoS2-AuNRs-HAP-PDA | 100-200 nm | EA.hy926, 24 h incubation 85.32% (50 µg·mL-1) MCF-7, 24 h incubation 86.92% (50 µg·mL-1) Concentrations tested: 0–50 µg·mL-1 |
n/s | - | [63] |
| MoS2-PEI | 340 nm | L929, 24 h incubation 91% (125 µg·mL-1) 4T1, 24 h incubation 98% (125 µg·mL-1) Concentrations tested: 0–125 µg·mL-1 |
Distributed in cytoplasm | - | [64] |
| MoS2-PEG-Biotin | 120 nm (thickness: 2.3 nm) |
HELF, 72 h incubation 90% (500 µg·mL-1) A549, 72 h incubation 90% (500 µg·mL-1) Concentrations tested: 25–500 µg·mL-1 |
Biotin-mediated uptake Distributed in cytoplasm |
Negligible haemolysis (500 µg·mL-1, 2 h incubation with RBC) | [65] |
| FA-MoS2-peptide | n/s | L929, 24 h incubation ~90% (2.5 µg·mL-1) Concentrations tested: 0.625–2.5 µg·mL-1 |
FA increased uptake to ~4.2-fold Lysosomal localization initially and substantial escape by 12 h |
- | [66] |
| MoS2-LAU | 120 nm | L929, 24 h incubation ~95% (200 µg·mL-1) Concentrations tested: 0–200 µg·mL-1 |
n/s | Negligible haemolysis (200 µg·mL-1, 2 h incubation with RBC) | [67] |
| Material | Animal model | Toxicokinetics | Histology | Additional observations | Refs. |
|---|---|---|---|---|---|
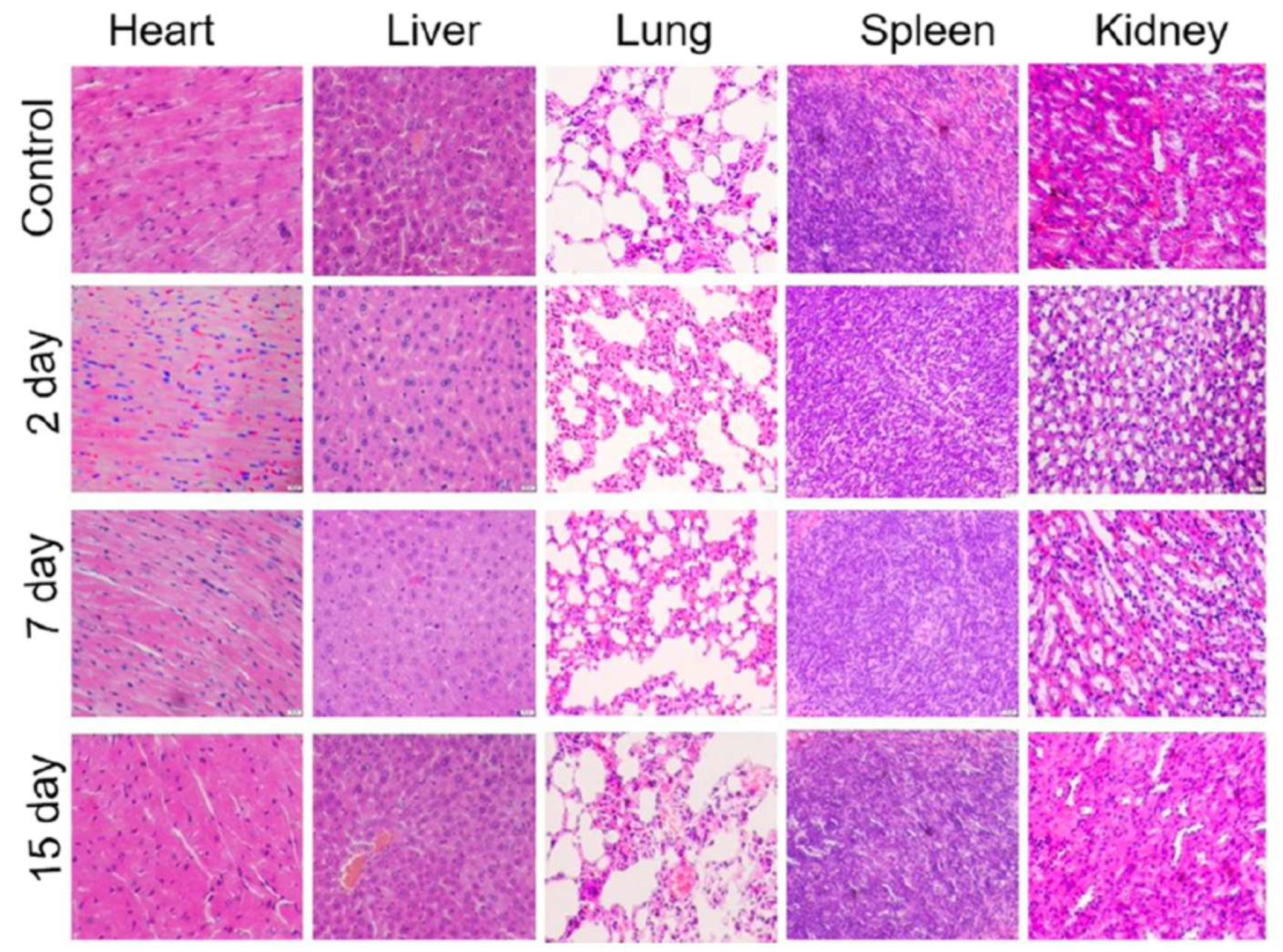
| MoS2-PEI-HA | Female BALB/c mice (15 days) |
IV administration (10 mg·kg-1) Accumulation in liver, spleen, lung, and tumour |
No damage or inflammation in major organs | No significant weight loss | [59] |
| MoS2-RBC | BALB/c mice (24 h) |
IV administration (0.8 mg·mL-1, 200 μL) Long circulation Accumulation in liver, spleen, and tumour |
- | No significant weight loss | [60] |
| MoS2-CS | Male BALB/c nude mice (24 days) |
IT administration (2 mg·kg-1) |
- | No significant weight loss | [61] |
| MoS2-PEG | 4T1 tumour-bearing BALB/c mice (21 days) |
IV administration (6.85 mg·kg-1) Accumulation in tumour |
No damage or inflammation in major organs | No significant weight loss | [7] |
| MoS2-PEI | 4T1 tumour-bearing BALB/c mice (14 days) |
IV administration (100 μg·mL-1, 100 µL) Accumulation in tumour Normal serum biochemistry markers |
No damage or inflammation in major organs | No significant weight loss | [64] |
| MoS2-PEG-Biotin | A549 tumour-bearing BALB/c nude female mice (21 days) |
IV administration (8 mg·kg-1) Accumulation in liver, spleen, and tumour |
No damage or inflammation in major organs | No significant weight loss | [65] |
| FA-MoS2-peptide | Male Wistar rats (7 days) |
IV administration Accumulation in liver, kidney, lung, spleen, brain, and tumour |
No damage or inflammation in major organs | No significant weight loss | [66] |
| MoS2-LAU | Healthy KM mice (14 days) |
IV administration (1 mg·mL-1, 200 µL) Accumulation in liver, spleen, and tumour |
No damage or inflammation in major organs | No significant weight loss | [67] |
| Material | Irradiation method | Energy | Irradiation time | Culture conditions and cell viability | Refs. |
|---|---|---|---|---|---|
| DOX@Biotin-BSA-PEI-LA-MoS2-LA-PEG | Laser (808 nm) |
1.5 W·cm−2 | 20 min | MCF-7-ADR, 24 h incubation 2DnMat only: 70% 2DnMat + drug: 15% (DOX 6 µg·mL-1) Concentrations tested (DOX): 0.1–6 µg·mL-1 |
[58] |
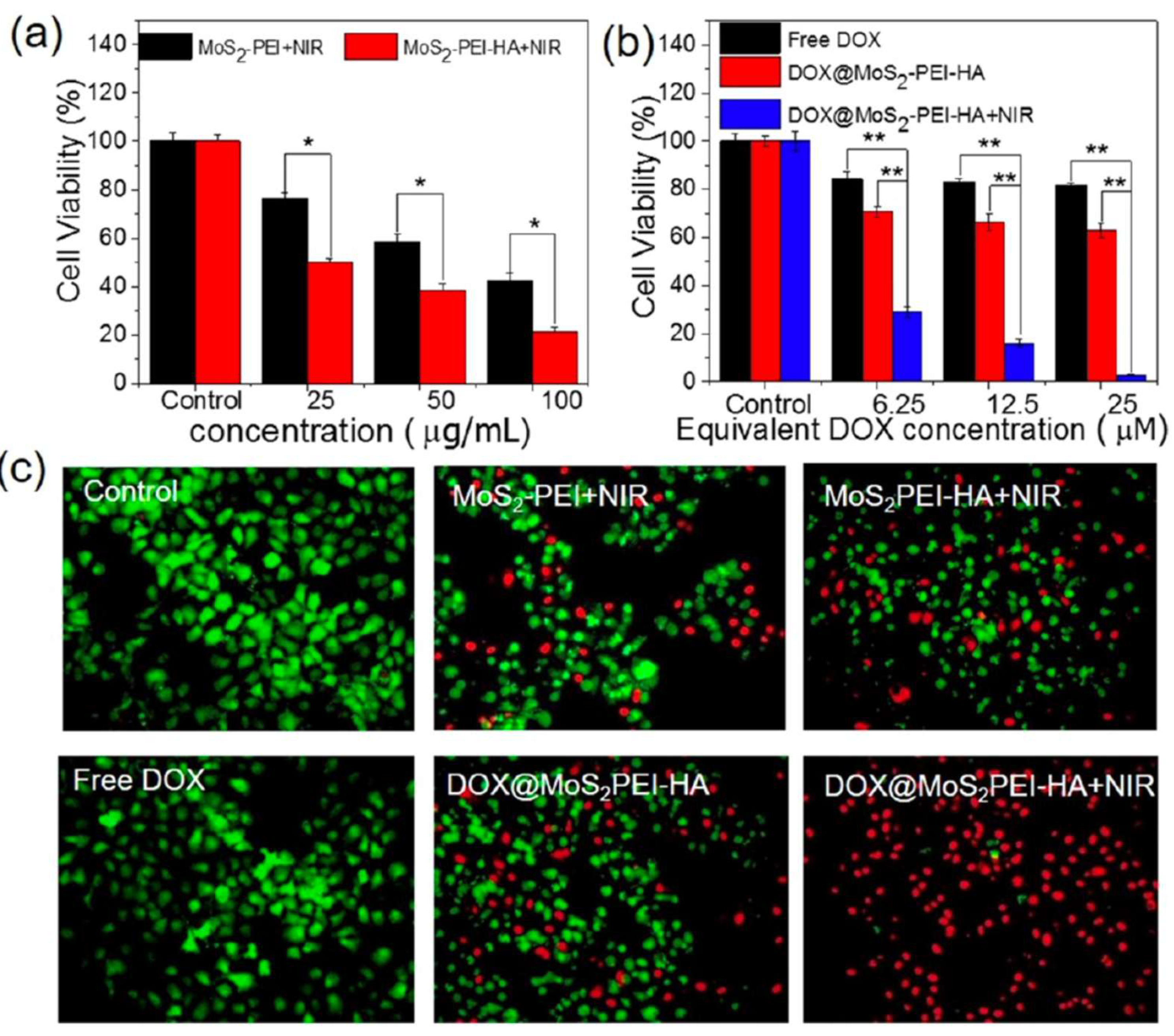
| DOX@MoS2-PEI-HA | Laser (808 nm) |
0.6 W·cm−2 | 10 min | MCF-7-ADR, 24 h incubation 2DnMat only: 20% (100 µg·mL-1) 2DnMat + drug: 2.9% (DOX 25 µM) Concentrations tested: 25–100 µg·mL-1 |
[59] |
| DOX@MoS2-RBC | Laser (808 nm) |
2 W·cm−2 | 10 min | MCF-7, 24 h incubation 2DnMat only: 35% (50 µg·mL-1) 2DnMat + drug: 20% (DOX 25 µg·mL-1) Concentrations tested: 12.5–50 µg·mL-1 |
[60] |
| DOX@MoS2-CS | Laser (808 nm) |
1 W·cm−2 | 7 min | KB, 24 h incubation 2DnMat only: 10% (50 µg·mL-1) 2DnMat + drug: 5% (DOX 50 µg·mL-1) Panc-1, 24 h incubation 2DnMat only: 25% (50 µg·mL-1) 2DnMat + drug: 5% (DOX 50 µg·mL-1) Concentrations tested: 0–50 µg·mL-1 |
[61] |
| Ce6@MoS2-PEG | PTT - Laser (808 nm) PDT - Light (660 nm) |
0.45 W·cm−2 (PTT) 0.005 W·cm−2 (PDT) |
20 min | 4T1, 24 h incubation 2DnMat only: 100% (6.4 µg·mL-1) 2DnMat + drug: 15% (Ce6 2 µM) Concentrations tested: 0–50 µg·mL-1 |
[7] |
| DOX@MoS2-AuNRs-HAP-PDA | Laser (808 nm) |
0.8 W·cm−2 | n/s | MCF-7, 24 h incubation 44.85% (50 µg·mL-1) Concentrations tested: 0–50 µg·mL-1 |
[63] |
| IR820@MoS2-PEI | Laser (808 nm) |
1 W·cm−2 | 5 min | 4T1, 24 h incubation 10.94% (125 µg·mL-1) Concentrations tested: 0–125 µg·mL-1 |
[64] |
| Cur/Er@MoS2-PEG-Biotin | Laser (808 nm) |
1 W·cm−2 | 10 min | A549, 48 h incubation 2DnMat only: 41.4% (100 µg·mL-1) 2DnMat + drug: 10% (Cur 20 µg·mL-1, Er 10 µg·mL-1) |
[65] |
| siRNA/DOX@FA-MoS2-peptide | Laser (808 nm) |
1.35 W·cm−2 | 3 min | C6, 48 h incubation 2DnMat only: ~68% (2.5 µg·mL-1) 2DnMat + drug: ~22% (2.5 µg·mL-1) |
[66] |
| MoS2-AIPH@LAU | Laser (808 nm) |
1 W·cm−2 | 5 min | HT29, 24 h incubation 2DnMat only: 32.9% (200 µg·mL-1) 2DnMat + drug: 6.1% (200 µg·mL-1) |
[67] |
| Material | Irradiation conditions | Animal model | Tumour growth | Additional observations | Refs. |
|---|---|---|---|---|---|
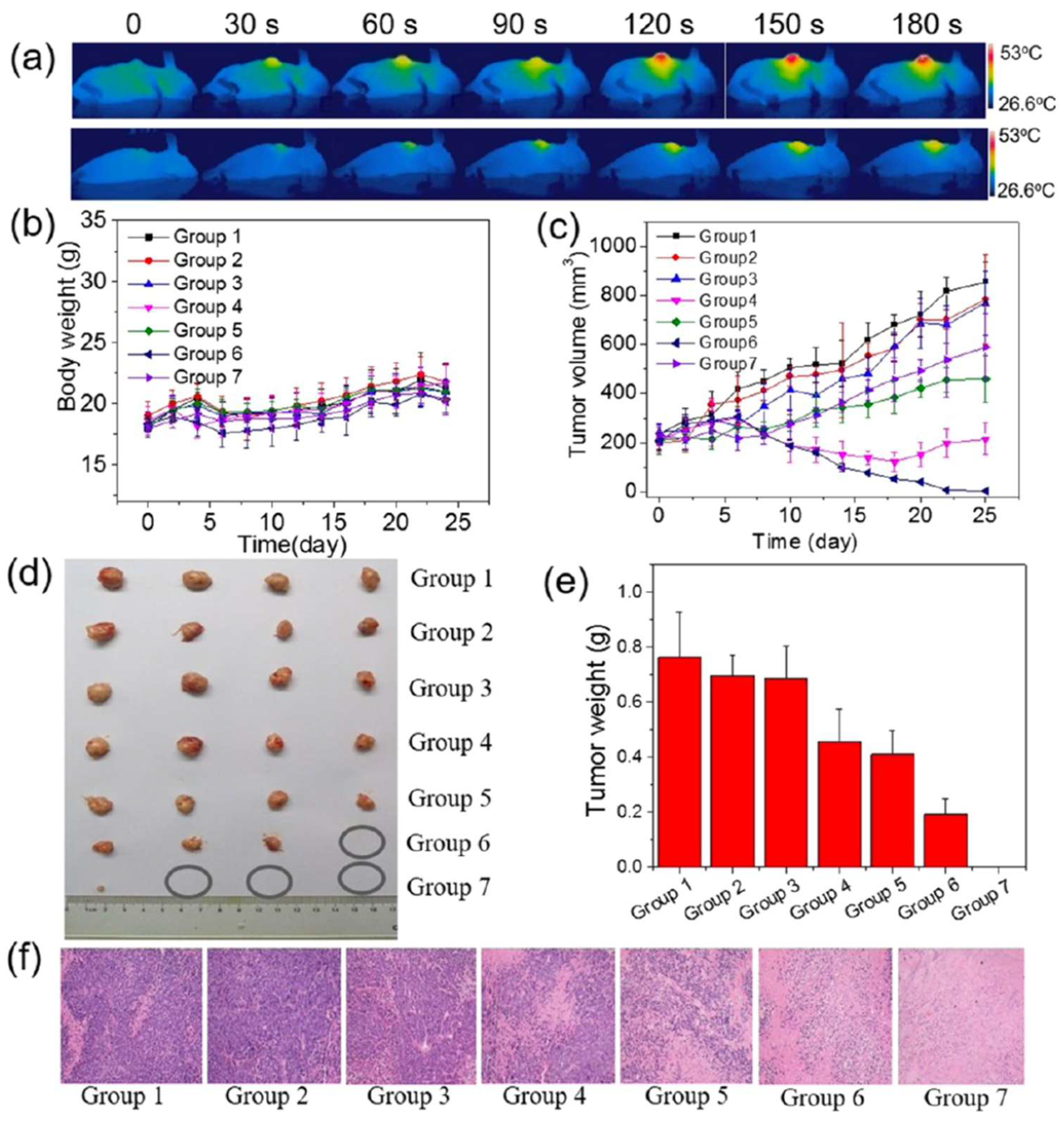
| DOX@MoS2-PEI-HA | Laser (808 nm) 0.6 W·cm−2 10 min |
MCF-7-ADR tumour-bearing BALB/c nude mice (25 days) |
IV administration (10 mg·kg-1) Tumour growth inhibition: 96% No tumour regrowth |
No significant weight loss Tumour-site temperature reached ~45 °C Cell shrinkage and nuclear damage |
[59] |
| DOX@MoS2-RBC | Laser (808 nm) 1.5 W·cm−2 5 min |
4T1 tumour-bearing BALB/c mice (15 days) |
IV administration (MoS2 0.8 mg·mL-1, DOX 0.75 mg·mL-1) Tumour growth inhibition: ~96% |
No significant weight loss Tumour-site temperature reached 45.3 °C |
[60] |
| DOX@MoS2-CS | Laser (808 nm) 0.9 W·cm−2 7 min |
Panc-1 tumour-bearing male BALB/c nude mice (24 days) |
IT administration (MoS2-CS 2 mg·kg-1, DOX 0.95 mg·kg-1) Tumour growth inhibition: ~85% |
No significant weight loss Tumour-site temperature reached ~55 °C Organized vacuolar degeneration, eosinophilic cytoplasm, nuclear damage, and necrosis |
[61] |
| Ce6@MoS2-PEG | PTT - Laser (808 nm) 0.45 W·cm−2 20 min PDT - Light (660 nm) 0.005 W·cm−2 20 min |
4T1 tumour-bearing BALB/c mice (21 days) |
IV administration (6.85 mg·kg-1, Ce6 2.0 mg·kg-1) Tumour growth inhibition: ~70% |
No significant weight loss Tumour-site temperature reached 44.8 °C |
[7] |
| IR820@MoS2-PEI | Laser (808 nm) 1 W·cm−2 5 min |
4T1 tumour-bearing BALB/c mice (14 days) |
IV administration (100 μg·mL-1, 100 µL) Tumour growth inhibition: 98.3% No tumour regrowth |
No significant weight loss Tumour-site temperature reached ~56 °C Severe nuclear damage and reduced cell proliferation |
[64] |
| Cur/Er@MoS2-PEG-Biotin | Laser (808 nm) 1 W·cm−2 10 min |
A549 tumour-bearing BALB/c nude mice (21 days) |
IV administration (MoS2-PEG-Biotin 8 mg·kg-1, Cur 1.6 mg·kg-1, Er 0.8 mg·kg-1) Tumour growth inhibition: 95.6% |
No significant weight loss Tumour-site temperature reached ~48 °C Tumour cell necrosis and lysis |
[65] |
| siRNA/DOX@FA-MoS2-peptide | Laser (808 nm) 1 W·cm−2 10 min |
C6 syngeneic glioma rats (21 days) |
IV administration 15-fold reduction in tumour volume |
No significant weight loss Decreased expression of PCNA |
[66] |
| MoS2-AIPH@LAU | Laser (808 nm) 1 W·cm−2 5 min |
HT29 tumour-bearing BALB/c nude mice (14 days) |
IV/IT administration Complete tumour eradication No recurrence |
No significant weight loss Tumour-site temperature reached ~50 °C |
[67] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




