1. Introduction
Colorectal cancer (CRC) continues to represent one of the leading causes of cancer-related morbidity and mortality worldwide. The increasing prevalence of the disease, together with the growing number of therapies used throughout the course of the neoplastic process, has led to a greater demand for healthcare resources [
1,
2]. In this context, cytotoxic chemotherapy based on fluoropyrimidines, oxaliplatin, and irinotecan constitutes the main therapeutic cornerstone, whether in adjuvant, neoadjuvant, or palliative settings, and is frequently combined with targeted therapies according to the tumor’s molecular profile [
3].
Among the available regimens, FOLFOX and XELOX have been widely used as first-line treatments, while irinotecan—an inhibitor of topoisomerase I—plays a key role in later treatment lines or as a therapeutic alternative in regimens such as FOLFIRI, particularly in patients who progress after exposure to oxaliplatin [
4] or when oxaliplatin is contraindicated. However, the toxicity profile of irinotecan differs substantially from that of oxaliplatin-based regimens, with a predominance of gastrointestinal and systemic adverse effects, particularly severe diarrhea, anorexia, asthenia, and persistent fatigue [
5,
6].
Beyond their immediate impact on treatment tolerance, these adverse effects have clinically relevant consequences on the patient’s nutritional status and functional capacity. Irinotecan-induced toxicity is associated with alterations in food intake, malabsorption, accelerated loss of muscle mass, and progressive functional decline—factors that directly influence treatment adherence, quality of life, and overall prognosis [
7,
8]. This impact is especially significant in patients previously treated with FOLFOX or XELOX, in whom cumulative toxicity may further increase the risk of malnutrition and clinical frailty [
9].
Cancer-related malnutrition is an independent prognostic factor associated with increased toxicity, a higher rate of complications, reduced overall survival, and significant deterioration in quality of life [
10,
11]. In patients with CRC undergoing chemotherapy, its prevalence may exceed 60–70%, particularly when persistent gastrointestinal symptoms and progressive functional decline coexist [
12]. Despite this high incidence, systematic nutritional assessment is still not fully integrated into many oncological care protocols.
In this context, the Patient-Generated Subjective Global Assessment (PG-SGA) has become the reference instrument for nutritional evaluation in oncology, as it allows a comprehensive assessment including weight loss, changes in dietary intake, nutrition-impact symptoms, and—particularly relevant—functional capacity [
13]. This functional dimension plays a central role in patients treated with irinotecan, as it reflects not only nutritional status but also the interaction between pharmacological toxicity, systemic inflammation, and loss of muscle mass.
In a previous study, Sánchez-Diestro et al. systematically evaluated nutritional status and symptoms associated with nutritional risk in patients with colorectal cancer treated mainly with oxaliplatin-based regimens such as FOLFOX and XELOX, using the PG-SGA as the central screening and assessment tool [
14]. That study identified a high prevalence of malnutrition and characterized differential toxicity profiles associated with these regimens. However, although irinotecan was present in a limited proportion of the cohort, its specific analysis was not the primary objective of the study, which limits the extrapolation of the results to this drug.
Irinotecan presents a distinctive toxicity profile characterized by a high burden of gastrointestinal symptoms and a potentially greater impact on functional capacity, particularly when administered in later lines of treatment. Evidence specifically focused on its nutritional and functional repercussions remains scarce, and few studies have addressed this impact from a comprehensive perspective using validated tools such as the PG-SGA.
Therefore, it is necessary to further investigate the adverse effects of irinotecan beyond its classical toxicity, incorporating its influence on the nutritional status and functional capacity of patients with colorectal cancer. Early identification of these effects through structured diagnostic instruments could facilitate earlier and more personalized nutritional and clinical interventions, optimizing treatment tolerance and clinical outcomes. Within this framework, the present study is conceived as an extension of previously published work, with the aim of specifically analyzing the impact of chemotherapy regimens including irinotecan on functional capacity and nutritional status in patients with colorectal cancer.
2. Materials and Methods
2.1. Study Population and Data Sources
An analytical observational cross-sectional study was conducted in a cohort of adult patients with a histopathologically confirmed diagnosis of colorectal adenocarcinoma in locally advanced or metastatic stages (stage III–IV) who were receiving active systemic chemotherapy regimens that included irinotecan as one of their components.
The study was carried out in the Medical Oncology Department of the University Hospital of Badajoz (Spain) within the framework of routine clinical practice between November 2023 and June 2024. Patients aged ≥18 years with sufficient cognitive capacity to participate in the structured clinical interview and to complete the nutritional assessment instruments were included, provided that they gave written informed consent.
Patients with severe concomitant medical conditions unrelated to the oncological process that could significantly alter baseline nutritional status or functional capacity were excluded. These included advanced organ failure, active systemic inflammatory diseases, or severe infectious processes.
Data were obtained through a directed clinical interview, systematic review of the electronic medical record, and nutritional and body composition assessment using validated tools.
2.2. Baseline Demographic and Clinical Variables
Baseline demographic and clinical variables were recorded, including age, sex, body mass index (BMI), location of the primary tumor (colon or rectum), and tumor stage, categorized as stage III or IV according to the current TNM classification.
Age was analyzed both as a continuous and categorical variable due to its clinical relevance as a factor influencing irinotecan-related toxicity, functional reserve, and nutritional vulnerability.
2.3. Variables Related to Therapeutic Exposure
A detailed characterization of the systemic oncological treatments administered was performed, with particular emphasis on the inclusion of irinotecan within specific therapeutic regimens and its potential contribution to the overall toxicity profile of treatment.
The complete therapeutic sequence was documented, including prior or subsequent exposure to oxaliplatin-based regimens (FOLFOX or XELOX), in order to explore whether the administration of irinotecan within a given treatment sequence could be associated with nutritional and functional deterioration that might lead to poorer clinical tolerance to FOLFOX, as indirectly suggested in the previous study.
Therapeutic intent (adjuvant, neoadjuvant, or palliative) and the concomitant use of targeted therapies (bevacizumab, cetuximab, or panitumumab), when clinically indicated, were also recorded.
From a clinical standpoint, the occurrence of adverse events typically associated with irinotecan was systematically evaluated, including early and late diarrhea, anorexia, nausea, vomiting, mucositis, abdominal pain, marked asthenia, persistent fatigue, and deterioration of functional capacity. These effects were considered potential mediators of the negative impact of irinotecan administration on nutritional status and overall treatment tolerability.
2.4. Nutritional Status and Body Composition Assessment
Nutritional status was assessed using the Patient-Generated Subjective Global Assessment (PG-SGA), a validated tool in oncological populations that integrates information on weight loss, changes in dietary intake, nutrition-impact symptoms, and evaluation of functional capacity, a component of particular interest in patients exposed to irinotecan.
The total score was interpreted according to internationally accepted cut-off points, determining the level of nutritional intervention required.
Additionally, body composition analysis was performed using multifrequency segmental bioelectrical impedance analysis with the TANITA® RD-545 device (TANITA Europe). This equipment estimates body composition based on the measurement of impedance generated by the passage of a low-intensity alternating current, allowing the assessment of parameters such as fat-free mass, total fat mass, skeletal muscle mass, bone mass, and basal metabolic rate, with segmental analysis of the trunk and extremities.
Results were interpreted according to reference values adjusted for age and sex, as well as consensus clinical cut-off points for the identification of muscle mass loss and risk of malnutrition.
2.5. Statistical Analysis
Statistical analysis was performed using JASP software (version 0.17.0.0), based on the R environment. Continuous variables were described using mean and standard deviation or median and interquartile range depending on data distribution, which was assessed using the Kolmogorov–Smirnov test.
Categorical variables were expressed as absolute frequencies and percentages. To analyze the association between the inclusion of irinotecan in the therapeutic regimen and nutritional, functional, and body composition parameters, χ2 tests or Fisher’s exact test were used.
Additionally, the relationship between exposure to irinotecan and clinical and functional tolerance to oxaliplatin-based regimens (FOLFOX) was explored through comparative analyses of nutritional and functional indicators. A p-value < 0.05 was considered statistically significant.
2.6. Ethical Considerations
The study was conducted in accordance with the ethical principles of the Declaration of Helsinki and current biomedical research legislation. All participants received detailed information about the objectives and procedures of the study and signed informed consent prior to inclusion.
The protocol was approved by the Ethics Committee of the Badajoz Health Area Management. Data were anonymized and treated confidentially, and were used exclusively for scientific purposes.
3. Results
3.1. Demographic and Clinical Characteristics of the Cohort
A total of 91 patients with a diagnosis of colorectal adenocarcinoma receiving active chemotherapy treatment were included. No losses or refusals were recorded in the completion of the Patient-Generated Subjective Global Assessment (PG-SGA); therefore, all subjects were included in the final analysis.
Of the total sample, 35 patients (38.46%) were women and 56 (61.54%) were men.
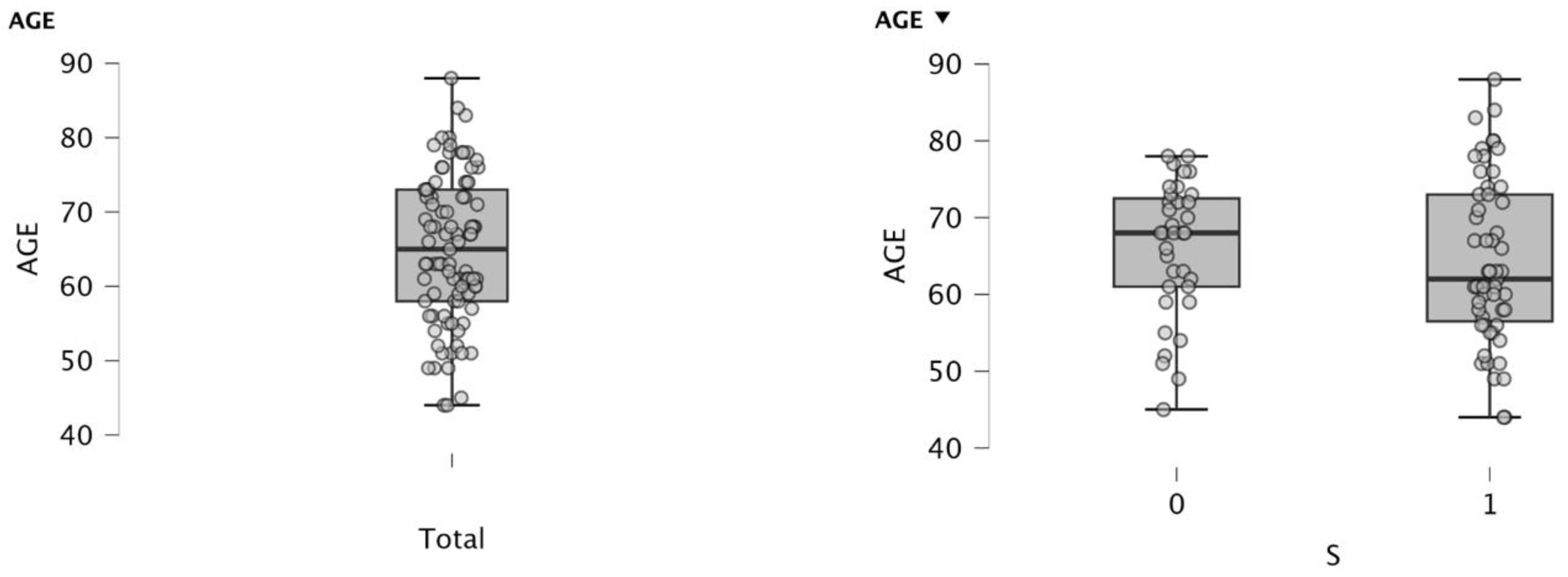
The overall mean age was 64.97 ± 9.92 years, with an interquartile range between 58 years (P25) and 73 years (P75) (
Figure 1).
When stratified by sex, the mean age in men was 64.27 ± 10.69 years, while in women it was 66.00 ± 8.77 years. No clinically relevant differences were observed in the age distribution between the two groups (
Figure 1).
3.2. Distribution of Therapeutic Regimens
Regarding the treatment received, 42 patients (46.2%) were undergoing treatment with XELOX and 36 patients (39.6%) with FOLFOX. Thirteen patients (14.3%) were receiving irinotecan, administered in combination with FOLFOX in 10 cases (76.9% of those treated with irinotecan) and in combination with XELOX in 3 cases (23.1%).
Therefore, the subgroup treated exclusively with FOLFOX consisted of 26 patients, while the FOLFOX–irinotecan subgroup included 10 patients. All patients were evaluated using multifrequency bioelectrical impedance analysis and the PG-SGA.
3.3. PG-SGA Results According to Irinotecan Exposure
Analysis of the total PG-SGA score showed a greater unfavorable nutritional burden in patients treated with irinotecan, particularly in those receiving it in combination with FOLFOX.
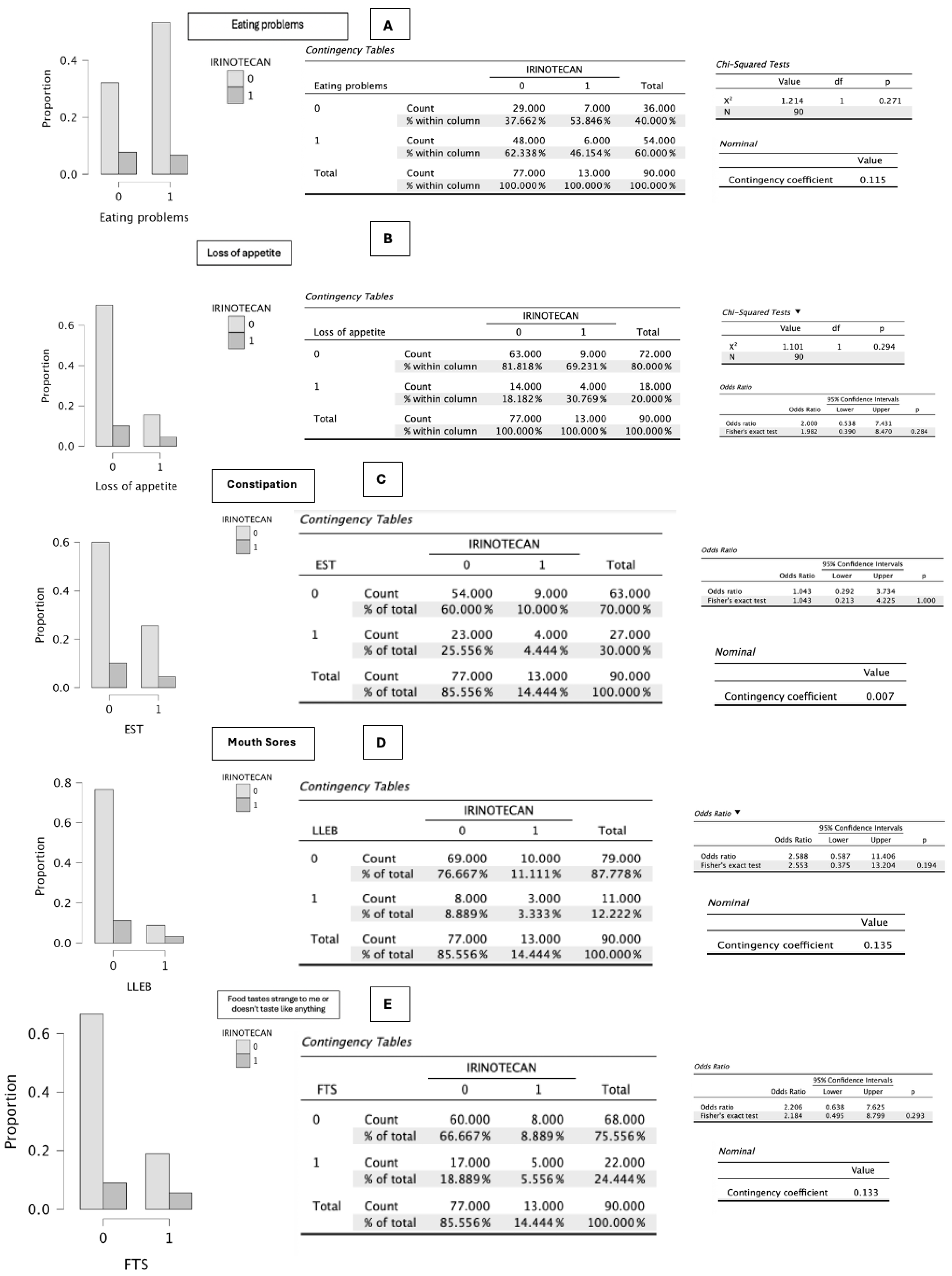
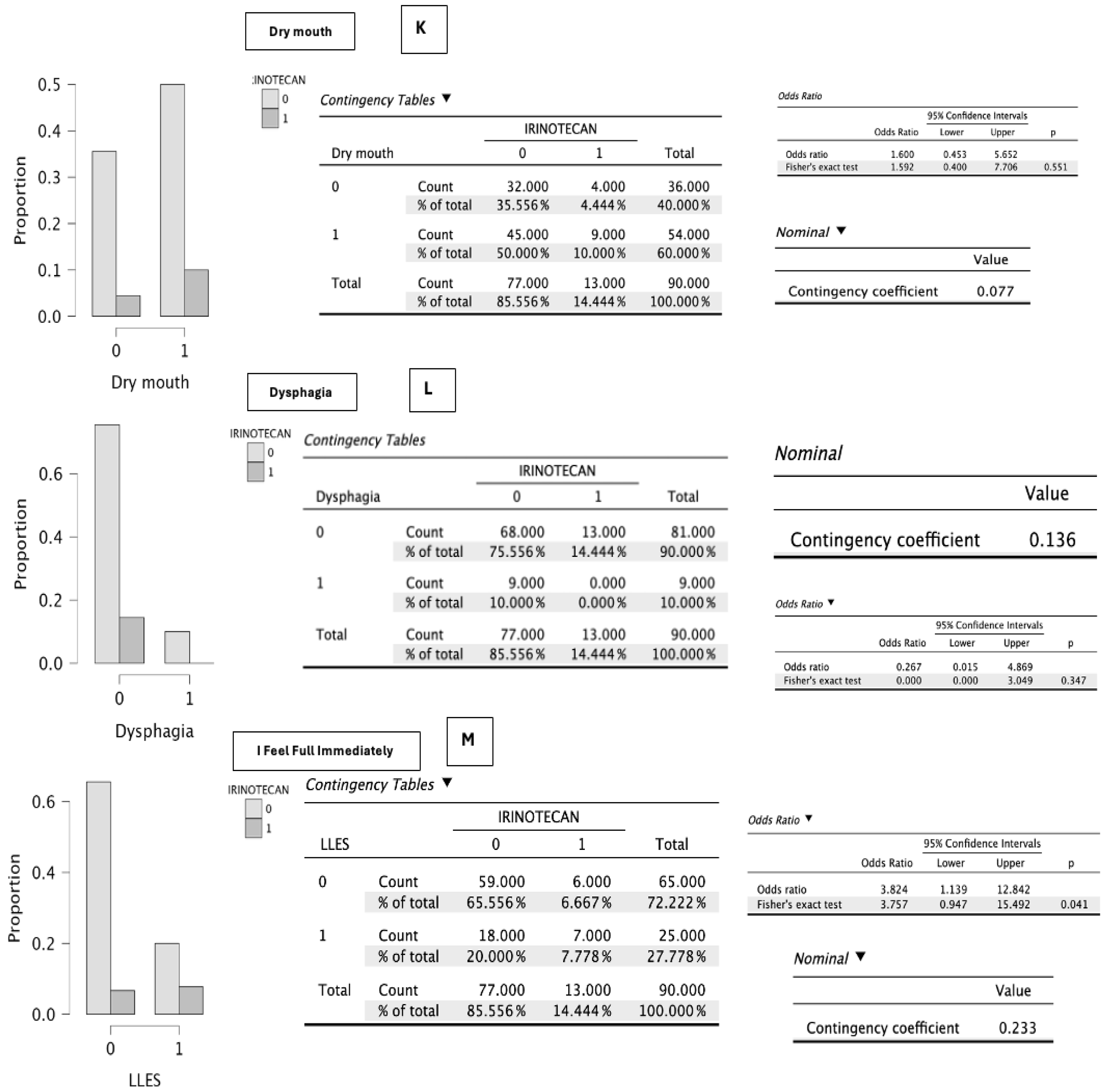
In the specific analysis of symptomatic items related to irinotecan administration (n = 13), no statistically significant differences were observed in: eating problems (p = 0.271), loss of appetite (p = 0.294), constipation (p = 1.000), oral sores (p = 0.194), taste alterations (p = 0.293), unpleasant smells (p = 1.000), pain (p = 0.537), diarrhea (p = 0.209), dry mouth (p = 0.551), and dysphagia (p = 0.136) (
Figure 2,
Figure 3,
Figure 4 and
Figure 5).
Values close to statistical significance were observed for nausea (p = 0.089), associated with an increase of one point in the PG-SGA score, and vomiting (p = 0.087), which increased the score by up to three points (
Figure 2,
Figure 3,
Figure 4 and
Figure 5).
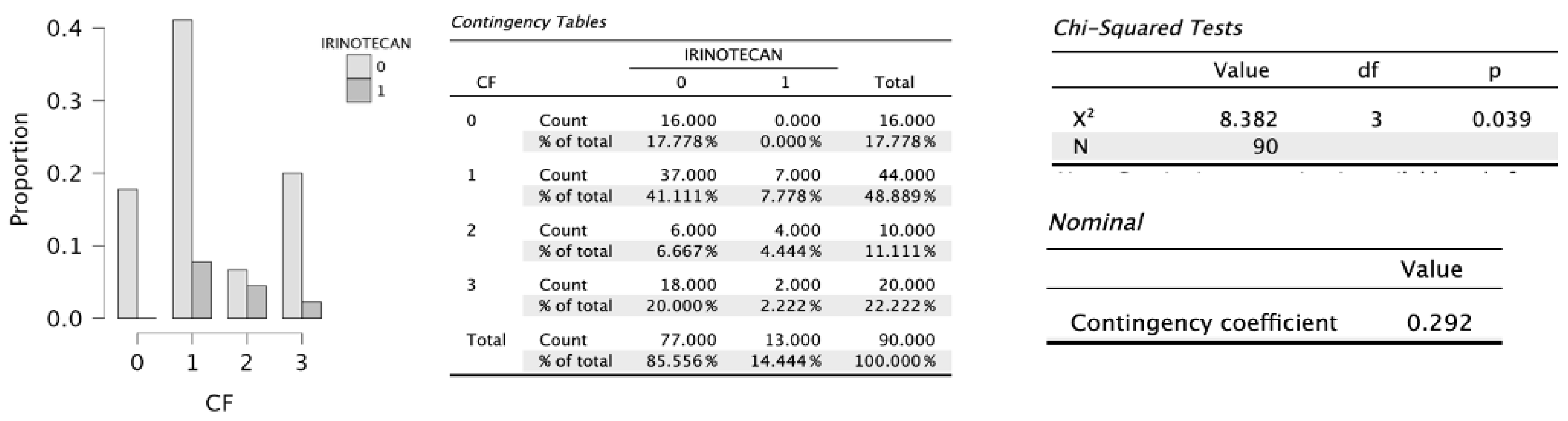
On the other hand, statistical significance was reached for early satiety (p = 0.041), which added one point to the scale score, and for progressive deterioration of functional capacity (p = 0.039), an item that may negatively increase the score by up to three points (
Figure 2,
Figure 3,
Figure 4 and
Figure 5).
When specifically comparing FOLFOX–irinotecan (n = 10) with FOLFOX alone (n = 26), a higher frequency of functional deterioration was observed in the combination group, contributing to higher overall PG-SGA scores.
3.4. Global Integration of Nutritional Scores
The combination of symptoms approaching significance (nausea and vomiting) together with the significant associations observed for early satiety and functional deterioration resulted in higher total PG-SGA scores in patients treated with irinotecan, particularly in combination with FOLFOX, compared with FOLFOX alone and XELOX. These findings were evident in the cohort assessed through both bioelectrical impedance analysis and PG-SGA, allowing characterization of a differential nutritional profile according to the administered therapeutic regimen.
4. Discussion
4.1. Prevalence and Prognostic Relevance of Malnutrition in Colorectal Cancer
In the analyzed cohort, a higher overall nutritional deterioration burden was observed in patients exposed to irinotecan, particularly those treated with the FOLFOX–irinotecan combination, where the increase in total PG-SGA scores was mainly driven by functional decline and the emergence of gastrointestinal symptoms impacting nutrition, such as early satiety, nausea, and vomiting. This finding positions functional capacity as the main determinant of nutritional deterioration in this therapeutic context, beyond isolated weight loss, reinforcing the concept of cancer-related malnutrition as a multidimensional process.
The high nutritional vulnerability observed in patients treated with irinotecan aligns with available literature in advanced colorectal cancer, where malnutrition prevalence ranges from 40% to 70%, depending on the diagnostic method used and the line of systemic treatment. Multicenter studies using PG-SGA have shown that high scores are independently associated with poorer treatment tolerance, dose reductions, and lower overall survival, even after adjusting for tumor stage and performance status [
15,
16,
17]. Our results extend these findings by identifying a nutritional profile specifically linked to irinotecan use.
From a prognostic standpoint, functional deterioration observed in patients treated with irinotecan has notable clinical relevance, as loss of functionality has been established as one of the most robust predictors of mortality and toxicity in gastrointestinal oncology, with a greater impact than BMI or isolated weight loss [
18,
19]. The relationship between functional status, muscle mass, and survival has been widely described in L3-level CT studies in metastatic colorectal cancer treated with fluoropyrimidine- and irinotecan-based chemotherapy [
20,
21].
4.2. Pathophysiology and Underlying Mechanisms
The nutritional deterioration profile observed in patients treated with irinotecan can be explained by a complex interaction between gastrointestinal toxicity, systemic inflammation, and accelerated muscle mass loss. Unlike oxaliplatin-only regimens, irinotecan induces more prolonged and clinically debilitating digestive toxicity, mediated by intestinal mucosal damage, activation of inflammatory pathways, and microbiota alterations, promoting anorexia, malabsorption, and early satiety [
22,
23].
The active metabolite SN-38 causes intestinal epithelial injury and release of proinflammatory cytokines, generating a hypercatabolic state that accelerates muscle proteolysis and reduces functional capacity. This process is amplified in advanced treatment lines, where cumulative toxicity from previous therapies diminishes patient physiological reserve, explaining the greater impact observed in the FOLFOX–irinotecan subgroup of our cohort. Pharmacokinetic studies have also shown that lean body mass is a key determinant of systemic exposure to irinotecan and its active metabolite, so patients with lower muscle mass experience higher toxicity for the same body surface area dose [
24,
25,
26].
4.3. Influence of Age and Comorbidities
The mean age of the cohort, close to 65 years, situates the studied population in a clinical scenario where functional reserve and body composition directly affect treatment tolerance. Although no significant age differences between sexes were observed in our analysis, literature shows that older patients have a higher risk of functional decline during irinotecan treatment, even in the absence of significant hematologic toxicity differences [
27,
28].
The coexistence of sarcopenia and frailty in metastatic colorectal cancer patients is associated with higher incidence of dose-limiting toxicity and early treatment interruptions, especially in irinotecan-containing regimens [
20,
29]. In this context, functional capacity assessed by PG-SGA could act as an integrative clinical marker of biological age, metabolic reserve, and therapeutic tolerance.
4.4. Nutritional Screening and Diagnostic Tools
Use of the PG-SGA enabled the identification of a nutritional deterioration pattern that would not have been detected by isolated anthropometric parameters. The relevance of the functional component was particularly evident in patients treated with irinotecan, where it was the main driver of the global score increase.
This finding aligns with international studies showing the superior predictive capacity of PG-SGA compared with simplified screening tools in actively treated oncology patients [
15,
30]. The inclusion of multifrequency bioelectrical impedance analysis in our cohort adds value by providing an objective measure of body composition, consistent with evidence indicating muscle mass as a primary determinant of chemotherapy toxicity and survival [
20,
31].
4.5. Clinical and Strategic Implications
The results have direct clinical implications for planning treatment in advanced colorectal cancer. Early identification of functional and nutritional deterioration in patients eligible for irinotecan could allow implementation of nutritional interventions and therapeutic exercise programs before dose reductions or treatment interruptions occur. Recent clinical trials have shown that multimodal interventions combining nutrition and exercise can improve chemotherapy tolerance and reduce toxicity in gastrointestinal cancer patients [
31,
32].
Our findings also support the need to systematically incorporate nutritional and body composition assessment into therapeutic decision-making, especially in the oxaliplatin–irinotecan sequence, where cumulative toxicity significantly affects functional capacity.
4.6. Limitations
Although the sample size allows for consistent estimates aligned with previously published evidence, expanding the cohort in future studies would increase statistical power to detect smaller associations that may still be clinically relevant. Similarly, the single-center design, while favoring standardization of procedures and consistency in diagnostic criteria and data collection, limits extrapolation to other care settings with different case mixes and organizational structures; multicenter studies would improve external validity and incorporate greater population heterogeneity.
The cross-sectional design provides an accurate snapshot of nutritional and functional status at a specific treatment time but prevents analysis of temporal trajectories and causal relationships between nutritional deterioration and clinical outcomes. Longitudinal studies with serial assessments would allow characterization of changes in body composition, functional capacity, and nutrition-impact symptoms across systemic treatment lines, particularly regarding cumulative irinotecan exposure.
Additionally, while this work focuses on clinical, functional, and validated nutritional assessment variables, the lack of detailed information on socioeconomic factors, lifestyle, habitual physical activity, and psychosocial factors may partially influence interpretation, given their recognized role in malnutrition risk, intervention adherence, and treatment tolerance. Future incorporation of these dimensions, together with systematic patient-reported outcomes and quality-of-life assessments using validated instruments, would allow a more comprehensive, patient-centered understanding of the clinical impact of nutritional deterioration.
5. Conclusions
This study demonstrates that exposure to irinotecan-containing regimens in patients with advanced colorectal cancer is associated with a differential pattern of nutritional deterioration, primarily characterized by worsening functional capacity and gastrointestinal symptoms directly affecting intake, especially early satiety, nausea, and vomiting. This pattern cannot be explained solely by weight loss but reflects a complex process involving pharmacologic toxicity, reduced metabolic reserve, and progressive physical activity limitation.
The combination of irinotecan with FOLFOX identifies a particularly vulnerable subgroup, where symptom burden and functional deterioration result in higher PG-SGA scores, suggesting a greater risk of clinically relevant malnutrition and potentially lower systemic treatment tolerance. These findings reinforce the role of functional capacity as a central component in oncological nutritional assessment and as a potential integrative marker of biological frailty and physiological reserve.
Systematic use of validated tools such as PG-SGA, combined with multifrequency bioelectrical impedance analysis, allows detection of nutritional alterations not identifiable through conventional anthropometric parameters and provides a more accurate assessment of the true impact of chemotherapy. Nutritional assessment should therefore be considered a structural element in the management of colorectal cancer patients, especially those candidates for irinotecan or previously exposed to oxaliplatin-based regimens.
From a clinical perspective, early identification of functional and nutritional deterioration enables implementation of individualized multimodal strategies integrating nutritional intervention, symptom optimization, and therapeutic exercise programs to improve treatment tolerance, maintain dose intensity, and preserve quality of life.
Overall, our results provide evidence that the impact of irinotecan extends beyond its classic toxicity profile to affect the patient’s nutritional and functional status, highlighting the need for systematic nutritional evaluation in therapeutic decision-making and follow-up algorithms for advanced colorectal cancer.
Author Contributions
L.E.S.-D.: conception, design, writing, investigation, formal analysis and methodology; R.M.-M.: conception, design and revision; Y.M.-G.: revision; M.A.-B.: revision; M.S.P.-V.: revision; J.G.-M.: supervision and revision. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of the Health Area of Badajoz of the Extremadura Health Service (SES) on 1 February 2024.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.:.
Data Availability Statement
The original contributions presented in this study are included in the article. The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024 May;74(3):229–63. [CrossRef]
- Arnold M, Sierra MS, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global patterns and trends in colorectal cancer incidence and mortality. Gut. 2017 Apr 1;66(4):683–91. [CrossRef]
- Van Cutsem E, Cervantes A, Adam R, Sobrero A, Van Krieken JH, Aderka D, et al. ESMO consensus guidelines for the management of patients with metastatic colorectal cancer. Ann Oncol. 2016 Aug 1;27(8):1386–422. [CrossRef]
- Tournigand C, André T, Achille E, Lledo G, Flesh M, Mery-Mignard D, et al. FOLFIRI followed by FOLFOX6 or the reverse sequence in advanced colorectal cancer: a randomized GERCOR study. J Clin Oncol. 2004;22(2):229–37. [CrossRef]
- Gupta S, Takebe N, Lorusso P. Targeting the Hedgehog pathway in cancer. Ther Adv Med Oncol. 2010;2(4):237–50. [CrossRef]
- Benson AB, Ajani JA, Catalano RB, Engelking C, Kornblau SM, Martenson JA, et al. Recommended guidelines for the treatment of cancer treatment-induced diarrhea. J Clin Oncol. 2004;22(14):2918–26. [CrossRef]
- Glimelius B. Benefit-risk assessment of irinotecan in advanced colorectal cancer. Drug Saf. 2005;28(5):417–33. [CrossRef]
- Gallois C, Artru P, Lièvre A, Auclin E, Lecomte T, Locher C, et al. Evaluation of two nutritional scores’ association with systemic treatment toxicity and survival in metastatic colorectal cancer: an AGEO prospective multicentre study. Eur J Cancer. 2019 Sep 1;119:35–43. [CrossRef]
- Ræder H, Henriksen C, Bøhn SK, O’de Fey Vilbo AR, Henriksen HB, Kværner AS, et al. Agreement between PG-SGA category and fat-free mass in colorectal cancer patients. Clin Nutr ESPEN. 2018 Oct 1;27:24–31. [CrossRef]
- da Silva Couto A, Gonzalez MC, Martucci RB, Feijó PM, Rodrigues VD, de Pinho NB, et al. Predictive validity of GLIM malnutrition diagnosis in patients with colorectal cancer. JPEN J Parenter Enteral Nutr. 2023 Mar 1;47(3):420–8. [CrossRef]
- Bauer J, Capra S, Ferguson M. Use of the scored Patient-Generated Subjective Global Assessment (PG-SGA) as a nutrition assessment tool in patients with cancer. Eur J Clin Nutr. 2002;56(8):779–85. [CrossRef]
- Barao K, Abe Vicente Cavagnari M, Silva Fucuta P, Manoukian Forones N. Association Between Nutrition Status and Survival in Elderly Patients With Colorectal Cancer. Nutr Clin Pract. 2017 Oct 1;32(5):658–63. [CrossRef]
- Okada S, Yamazaki S, Kaiga T, Funada T, Kochi M, Takayama T. Impact of nutritional status in the era of FOLFOX/FIRI-based chemotherapy. World J Surg Oncol. 2017 Aug 24;15(1). [CrossRef]
- Sánchez-Diestro LE, Macias-Montero R, Ramalho-Galhanas AI, Aguiar-Frias AM, Paniagua-Vivas MS, Guerrero-Martín J. Nutritional Status Assessment Using the Patient-Generated Subjective Global Assessment (PG-SGA) in Individuals with Colorectal Cancer Undergoing Chemotherapy Regimens. J Clin Med. 2025 Sep 1;14(18). [CrossRef]
- Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, et al. ESPEN guidelines on nutrition in cancer patients. Clinical Nutrition. 2017 Feb 1;36(1):11–48. [CrossRef]
- Charney P, Charney D. Nutrition Support in Renal Failure. Nutrition in Clinical Practice. 2002 Aug 1;17(4):226–36. [CrossRef]
- Prado CM, Lieffers JR, McCargar LJ, Reiman T, Sawyer MB, Martin L, et al. Prevalence and clinical implications of sarcopenic obesity in patients with solid tumours of the respiratory and gastrointestinal tracts: a population-based study. Lancet Oncol. 2008 Jul 1;9(7):629–35. [CrossRef]
- Lang J, Shao Y, Liao J, Chen J, Zhou X, Deng R, et al. Patient-Generated Subjective Global Assessment (PG-SGA) predicts length of hospital stay in lung adenocarcinoma patients. Br J Nutr. 2021 May 28;127(10):1543. [CrossRef]
- Martin L, Birdsell L, MacDonald N, Reiman T, Clandinin MT, McCargar LJ, et al. Cancer cachexia in the age of obesity: Skeletal muscle depletion is a powerful prognostic factor, independent of body mass index. Journal of Clinical Oncology. 2013 Apr 20;31(12):1539–47. [CrossRef]
- Barret M, Antoun S, Dalban C, Malka D, Mansourbakht T, Zaanan A, et al. Sarcopenia is linked to treatment toxicity in patients with metastatic colorectal cancer. Nutr Cancer. 2014 May 19;66(4):583–9. [CrossRef]
- Shachar SS, Deal AM, Weinberg M, Nyrop KA, Williams GR, Nishijima TF, et al. Skeletal muscle measures as predictors of toxicity, hospitalization, and survival in patients with metastatic breast cancer receiving taxane-based chemotherapy. Clinical Cancer Research. 2017 Feb 1;23(3):658–65.
- Yang X, Hu Z, Chan S, Chan E, Goh B, Duan W, et al. Novel Agents that Potentially Inhibit Irinotecan-Induced Diarrhea. Curr Med Chem. 2005 May 28;12(11):1343–58. [CrossRef]
- Miya T, Fujikawa R, Fukushima JI, Nogami H, Koshiishi Y, Goya T. Bradycardia Induced by Irinotecan: A Case Report. Jpn J Clin Oncol. 1998;28(11):709–11. [CrossRef]
- Seok YK, Jae HH, Kwang JL, Choi JH, Jung IP, Hyoung IK, et al. Low Expression of Bax Predicts Poor Prognosis in Patients with Locally Advanced Esophageal Cancer Treated with Definitive Chemoradiotherapy. Clinical Cancer Research. 2007 Jul 15;13(14):4146–53. [CrossRef]
- Chemama S, Raynard B, Antoun S. [Impact of cancer muscle mass loss on anticancer treatment toxicities]. Bull Cancer. 2016 Sep 1;103(9):786–93. [CrossRef]
- Blauwhoff-Buskermolen S, Versteeg KS, De Van Der Schueren MAE, Den Braver NR, Berkhof J, Langius JAE, et al. Loss of Muscle Mass During Chemotherapy Is Predictive for Poor Survival of Patients With Metastatic Colorectal Cancer. J Clin Oncol. 2016 Apr 20;34(12):1339–44. [CrossRef]
- Aparicio T, Canouï-Poitrine F, Caillet P, François E, Cudennec T, Carola E, et al. Treatment guidelines of metastatic colorectal cancer in older patients from the French Society of Geriatric Oncology (SoFOG). Digestive and Liver Disease. 2020 May 1;52(5):493–505. [CrossRef]
- Comella P, Massidda B, Filippelli G, Natale D, Farris A, Buzzi F, et al. Safety and Efficacy of Irinotecan plus High-Dose Leucovorin and Intravenous Bolus 5-Fluorouracil for Metastatic Colorectal Cancer: Pooled Analysis of Two Consecutive Southern Italy Cooperative Oncology Group Trials. Clin Colorectal Cancer. 2005 Sep 1;5(3):203–10. [CrossRef]
- Takenami T, Tsujinaka S, Miyakura Y, Kakizawa N, Maemoto R, Machida E, et al. Impact of sarcopenia on surgical and oncologic outcomes of laparoscopic surgery for colorectal cancer. Asian J Surg. 2022 Dec 1;45(12):2686–90. [CrossRef]
- Ravasco P, Monteiro-Grillo I, Vidal PM, Camilo ME. Dietary counseling improves patient outcomes: a prospective, randomized, controlled trial in colorectal cancer patients undergoing radiotherapy. J Clin Oncol. 2005;23(7):1431–8. [CrossRef]
- Reid J, Blair C, Dempster M, McKeaveney C, Slee A, Fitzsimons D. Multimodal interventions for cachexia management. Cochrane Database Syst Rev. 2023 Jun 27;2023(6):CD015749. [CrossRef]
- de van der Schueren MAE, Laviano A, Blanchard H, Jourdan M, Arends J, Baracos VE. Systematic review and meta-analysis of the evidence for oral nutritional intervention on nutritional and clinical outcomes during chemo(radio)therapy: current evidence and guidance for design of future trials. Annals of Oncology. 2018 May 1;29(5):1141. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).