1. Introduction
Acute appendicitis remains the most frequent abdominal surgical emergency worldwide, with an estimated annual incidence ranging from 96.5 to 100 cases per 100,000 adults [
1]. Despite its prevalence, the diagnosis of acute appendicitis is often challenging due to the variability in clinical presentation, which can mimic other abdominal pathologies and overlap with other causes of right lower quadrant pain [
2,
3].
Inaccurate diagnosis may result either in delayed treatment or in unnecessary surgical intervention, leading to so-called “white” appendectomies, which historically range from 15% to 45%, both situations increasing patient morbidity. To enhance diagnostic accuracy and reduce unnecessary imaging and surgical exploration, several clinical scoring systems have been developed. Among them, the Alvarado score is the most widely known and used. However, its development based on operated cases or retrospective surgical cohorts introduces selection bias and limits its applicability to patients presenting with initial symptoms of suspected appendicitis [
4,
5].
While modern imaging, particularly computed tomography (CT), has significantly improved diagnostic accuracy, its systematic use increases healthcare costs and exposes patients—often young—to ionizing radiation. In response to these limitations, the François score was proposed [
6].
Developed from a prospective cohort of patients presenting to the emergency department with suspected appendicitis, the François score integrates simple clinical and biological parameters, including age, sex, duration of symptoms, right lower quadrant guarding, leukocytosis, presence of genitourinary symptoms, and palpable right-sided mass on rectal examination. The score stratifies patients into three diagnostic probability groups, helping guide decision-making and potentially reducing the need for imaging or surgical intervention in low-risk cases. Although the François score was validated in a French population, its performance in different healthcare settings remains underexplored [
7].
The present study therefore aims to evaluate the diagnostic performance of the François score in a Tunisian population presenting with suspected acute appendicitis at the General Surgery Department of Habib Bougatfa Hospital in Bizerte.
2. Materials and Methods
We conducted a prospective cohort study evaluating diagnostic performance at the General Surgery Department of Habib Bougatfa Hospital in Bizerte, Tunisia, over a seven-month period from October 1, 2021, to April 30, 2022.
All consecutive patients aged 15 years and older admitted with suspected acute appendicitis were eligible for inclusion. Suspicion was based on the presence of right lower quadrant abdominal pain, with or without fever or biological signs of inflammation. A total of 139 patients meeting these criteria were included.
Exclusion criteria included pregnancy and history of appendectomy, and no additional exclusion criteria were applied.
Upon admission, patients were examined by the on-call surgeon who completed a standardized data collection sheet including demographic characteristics, medical and surgical history, clinical findings, laboratory results, and imaging data (ultrasound or computed tomography when performed). The variables required for the François score were prospectively collected at admission using a standardized form. The score was subsequently calculated by an investigator who was blinded to the final diagnosis.
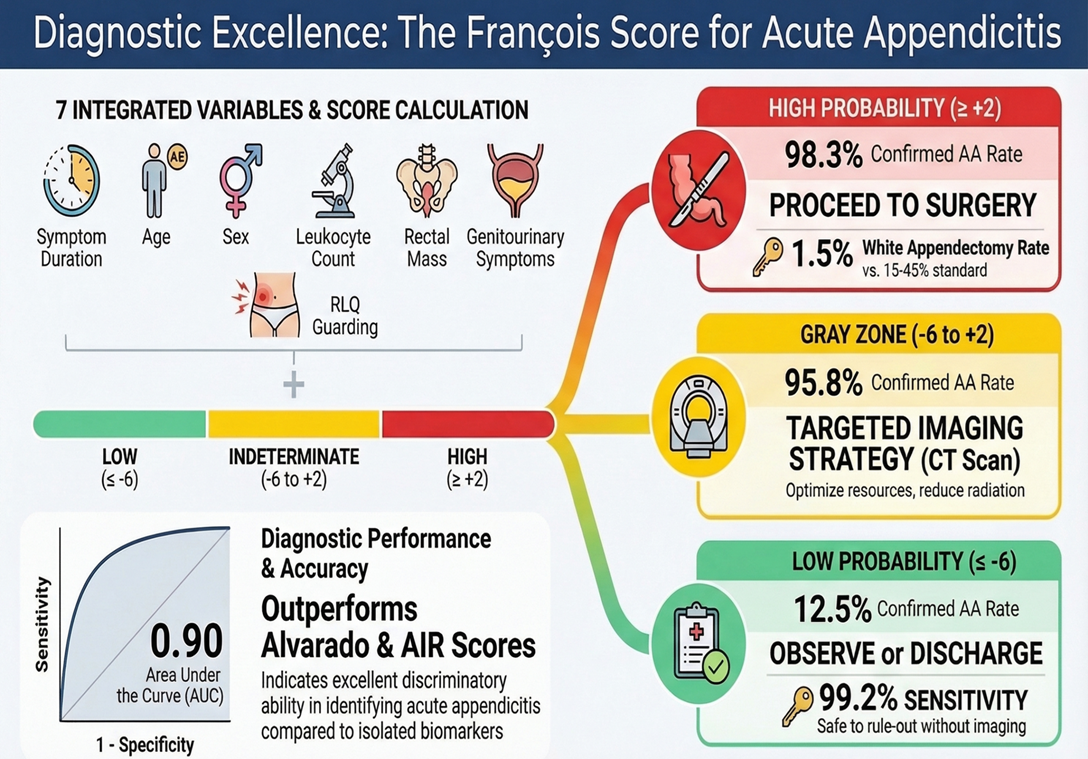
The François score assigns weighted coefficients ranging from −3 to +3 to seven variables: sex, age, duration of symptoms, presence of right lower quadrant guarding, leukocyte count, presence of a right-sided mass on rectal examination, and presence of genitourinary symptoms.
Total scores range from −17 to +13, and patients are stratified into three diagnostic probability groups: high probability (Group 1: score ≥ +2), indeterminate probability (Group 2: score between −6 and +2), and low probability (Group 3: score ≤ −6).
For operated patients, the diagnosis of acute appendicitis was confirmed by histopathological examination of the appendectomy specimen. For non-operated patients, the reference standard relied on concordant imaging findings and clinical follow-up.
Data were analyzed using SPSS software version 26. Continuous variables were expressed as means ± standard deviation (SD) or medians with interquartile ranges (IQR) according to data distribution, while categorical variables were presented as frequencies and percentages.
The diagnostic performance of the François score was assessed by calculating sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV). Receiver operating characteristic (ROC) curves were generated and the area under the curve (AUC) was used to evaluate overall diagnostic performance. Optimal cut-off values were determined using the Youden index. Odds ratios (OR) with 95% confidence intervals (CI) were calculated to evaluate the association between score categories and the final diagnosis. A p-value ≤ 0.05 was considered statistically significant.
Ethical principles were respected throughout the study, all data were anonymized, and no diagnostic or therapeutic interventions were imposed by the study protocol.
3. Results
A total of 139 patients were included in the study. The median age was 27 years (IQR: 19–41) with a male predominance (66.2%; sex ratio M/F = 1.95). Acute appendicitis was confirmed in 128 patients (92.1%), either by histopathology in operated cases or by imaging and clinical follow-up in non-operated patients, while 11 patients (7.9%) were diagnosed with non-appendicular conditions.
The mean François score was 0.58 ± 4.31, with values ranging from −11 to +10. According to predefined thresholds, patients were distributed into three probability groups: high probability (score ≥ +2) representing 42.4% (n=59), indeterminate probability (score between −6 and +2) representing 51.8% (n=72), and low probability (score ≤ −6) representing 5.8% (n=8).
In the high-probability group, appendicitis was confirmed in 58 patients (98.3%), whereas only one patient (1.7%) had a non-appendicular diagnosis. In the indeterminate group, appendicitis was confirmed in 69 patients (95.8%) and excluded in three patients (4.2%). In the low-probability group, appendicitis was confirmed in only one patient (12.5%), while seven patients (87.5%) had no appendicitis, demonstrating a strong inverse correlation between low scores and the presence of appendicitis (p<0.001; OR=0.004; 95% CI: 0–0.046). Conversely, a score greater than or equal to +2 was significantly associated with the diagnosis of appendicitis (p=0.024; OR=8.286; 95% CI: 1.03–66.647). (
Table 1)
Evaluation of diagnostic accuracy at predefined thresholds showed that a cut-off > −6 yielded a sensitivity of 99.2%, specificity of 63.6%, positive predictive value (PPV) of 96.9%, and negative predictive value (NPV) of 87.5%, allowing identification of nearly all cases of appendicitis but with moderate specificity. In contrast, a cut-off ≥ +2 showed lower sensitivity (45.3%) but higher specificity (90.9%), with a high PPV of 98.3% and a low NPV of 12.5%. Considering only the extreme categories of the score (positive score ≥ +2 indicating appendicitis and negative score ≤ −6 indicating absence of appendicitis), the François score demonstrated high diagnostic performance, with sensitivity of 98.3%, specificity of 87.5%, PPV of 98.3%, and NPV of 87.5%, highlighting its ability to correctly identify both patients with appendicitis and those without disease. (
Table 2)
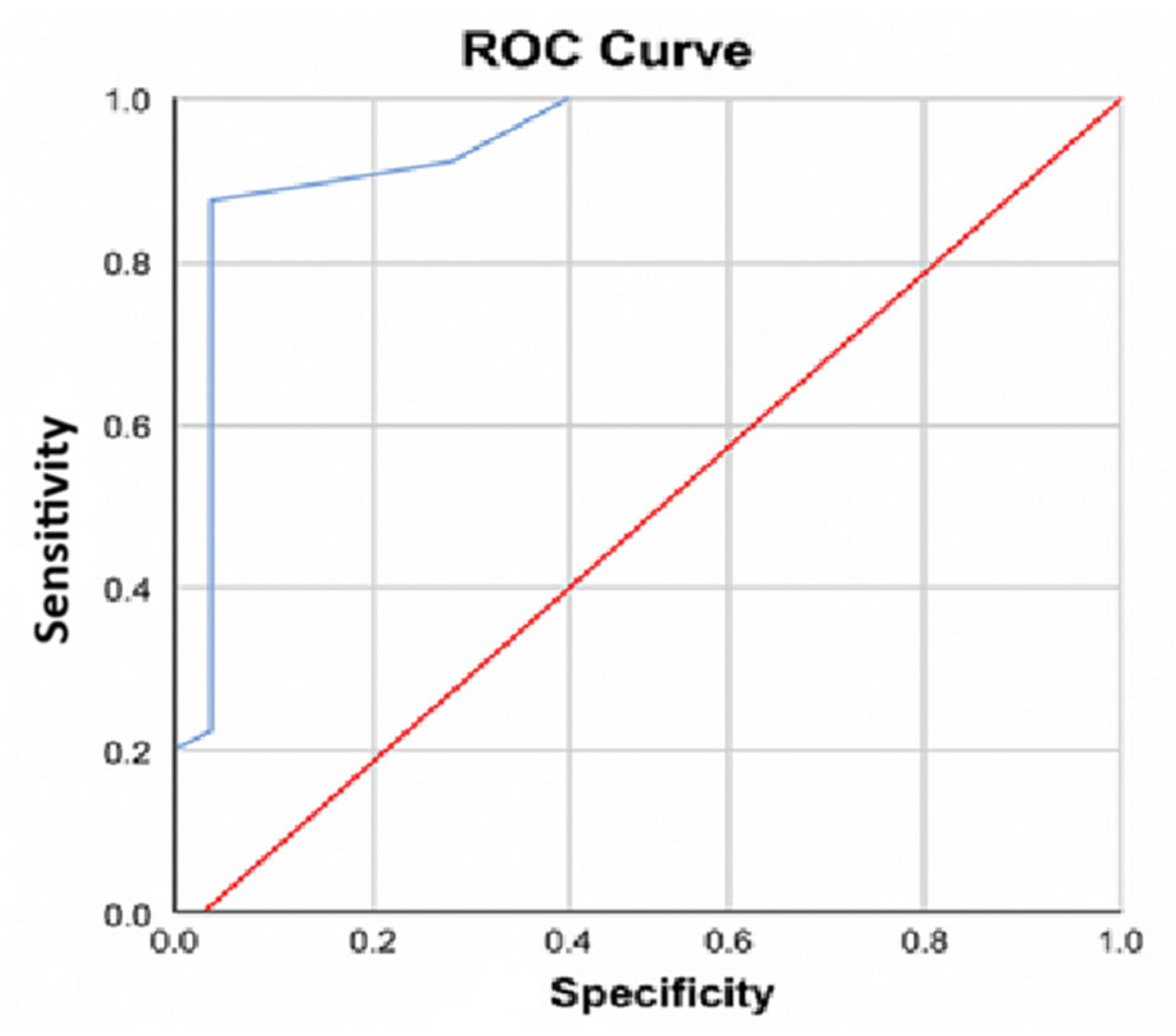
Receiver operating characteristic (ROC) curve analysis further confirmed the strong diagnostic performance of the François score. The area under the curve (AUC) was 0.90 (95% CI: 0.762–1; p<0.001), indicating excellent discriminatory ability. The optimal statistical threshold was −3.5, corresponding to a sensitivity of 88.3% and a specificity of 90.9% for the diagnosis of acute appendicitis. (
Figure 1)
Regarding imaging correlation, ultrasound was performed in 62 patients (44.6%) and was inconclusive in 79% of patients with high François scores. Computed tomography (CT) was performed in 84 patients (60.4%) and confirmed appendicitis in 97.2% of patients with high scores, whereas in patients with low scores CT ruled out appendicitis in 83.3% of cases. These findings suggest that imaging often did not modify the clinical decision already supported by a high pre-test probability provided by the score, while in low-score patients imaging mainly confirmed the absence of disease and might therefore be avoided in selected cases.
In terms of management and outcomes, 130 patients (93.5%) underwent appendectomy, predominantly using a laparoscopic approach (85.4%). Postoperative complications occurred in 7.7% of cases, mainly intra-abdominal collections. The median postoperative hospital stay was 2 days (IQR: 1–3), and one postoperative death was reported (0.8%).
4. Discussion
In this prospective study conducted in a Tunisian surgical department, we evaluated the diagnostic performance of the François score in patients presenting with suspected acute appendicitis. Our findings confirm that this clinical score, originally validated in France, retains good discriminatory power in a North African population and could therefore serve as a reliable clinical triage tool in emergency settings [
7,
8].
The François score demonstrated a strong association with the final diagnosis of appendicitis: a score ≥ +2 was significantly predictive of appendicitis (OR = 8.29, p = 0.024), whereas a score ≤ −6 was strongly associated with the absence of appendicitis (OR = 0.004, p < 0.001). These findings are consistent with the original validation study by François et al., where the score achieved a sensitivity of 94.7% and a negative predictive value (NPV) of 97.4% in a French emergency department cohort [
6,
7,
8].
In our population, the François score showed a sensitivity of 99.2% for scores above −6 and a specificity of 90.9% for scores ≥ +2, allowing clinically meaningful stratification of patients into low-risk, indeterminate, and high-risk groups. Patients with low scores (≤ −6) can often be safely observed or discharged without systematic imaging, whereas patients with high scores (≥ +2) represent strong candidates for early surgical evaluation even in the absence of advanced imaging.
Several clinical scoring systems have been proposed to improve the diagnostic accuracy of acute appendicitis. The Alvarado score remains the most widely used tool; however, it was developed retrospectively from operated cases, which introduces potential selection bias and limits its applicability in early emergency presentations [
9,
10,
11,
12,
13].
Furthermore, it incorporates variables such as pain migration, anorexia, and rebound tenderness, which may be subjectively reported or examiner-dependent. The Andersson Appendicitis Inflammatory Response (AIR) score emphasizes inflammatory biomarkers such as C-reactive protein and leukocyte count and has shown promising diagnostic performance, but its reliance on biochemical parameters may limit its utility in settings where laboratory results are not immediately available [
14,
15,
16,
17].
Other scores, such as the Ohmann and Fenyö-Lindberg scores, have undergone limited validation and have not achieved widespread clinical adoption [
18,
19].
Compared with these systems, the François score represents a simpler and cost-effective alternative because it relies on readily available clinical and biological variables and does not require complex algorithms or systematic imaging. In our study, its diagnostic performance—sensitivity 98.3%, specificity 87.5%, PPV 98.3%, and NPV 87.5%—compares favorably with historical results reported for other scoring systems. (
Table 3)
The François score showed excellent diagnostic performance in our cohort. Although other scoring systems such as the AIR score have demonstrated good accuracy in previous studies, direct comparison was not performed in our study.
An important implication of these findings is the potential of the François score to rationalize the use of imaging investigations. In our cohort, only one out of eight patients with a score ≤ −6 was ultimately diagnosed with appendicitis, supporting the safety of conservative management or observation in this group. Conversely, in the high-score group, computed tomography confirmed appendicitis in 97.2% of cases, indicating that imaging rarely changed the clinical decision already supported by a very high pre-test probability. Among patients with indeterminate scores, imaging remained essential for diagnostic clarification. These results suggest that the François score may help reduce unnecessary imaging studies, particularly CT scans, thereby limiting radiation exposure and healthcare costs while maintaining diagnostic safety [
6,
7,
19]. By implementing a score-guided strategy, CT scans could be reserved for the indeterminate "grey zone" (–6 to +2), where diagnostic uncertainty is highest, while safely avoiding systematic imaging in extreme probability groups [
1,
2,
3,
5,
6,
7,
8].
Our findings also indicate that the François score performs favorably when compared with isolated inflammatory biomarkers such as C-reactive protein (AUC ≈ 0.60) or leukocyte count (AUC ≈ 0.63), which alone provide limited diagnostic discrimination. By integrating clinical and biological parameters into a single weighted model, the score offers a more robust diagnostic framework. In addition, its clinical application may contribute to reducing unnecessary surgical procedures. In our series, the rate of negative appendectomy was particularly low, reflecting the ability of the score to accurately identify patients requiring surgical intervention.
The integration of the François score into surgical decision-making has a direct impact on reducing the negative appendectomy rate (or white appendectomy rate), which was remarkably low at 1.5% in our series. This performance is significantly superior to historical literature standards, which often range from 15% to 45%. The score’s effectiveness is most visible in the high-probability group, where the Positive Predictive Value (PPV) reached 98.3%; notably, no patient classified in this high-score group who underwent surgery was found to have a healthy appendix [
11,
12,
13,
14,
15,
16]. By objectively identifying patients at risk of overdiagnosis—typically those with atypical presentations and low or intermediate scores—the François score enhances the relevance of surgical indications and minimizes the morbidity associated with unnecessary abdominal explorations
Our results therefore confirm that the François score maintains a high level of diagnostic discrimination in a North African population, with an overall area under the ROC curve (AUC) of 0.90. Although the negative predictive value observed in our cohort (87.5%) was slightly lower than that reported in the original Strasbourg validation study (97.4%), the score demonstrated excellent performance in identifying high-risk patients, with a positive predictive value of 98.3% [
15,
16,
17]. This finding is clinically relevant because it supports the use of the score as a reliable rule-in tool for surgical decision-making while maintaining a very low rate of negative appendectomies. The consistency between the original French validation and our Tunisian experience suggests that the François score is robust across different healthcare environments and patient populations. In practical terms, its simplicity and reliance on readily available clinical variables make it particularly valuable for optimizing diagnostic pathways in emergency departments, especially in settings where access to advanced imaging may be limited or where reducing unnecessary CT scans is a priority.
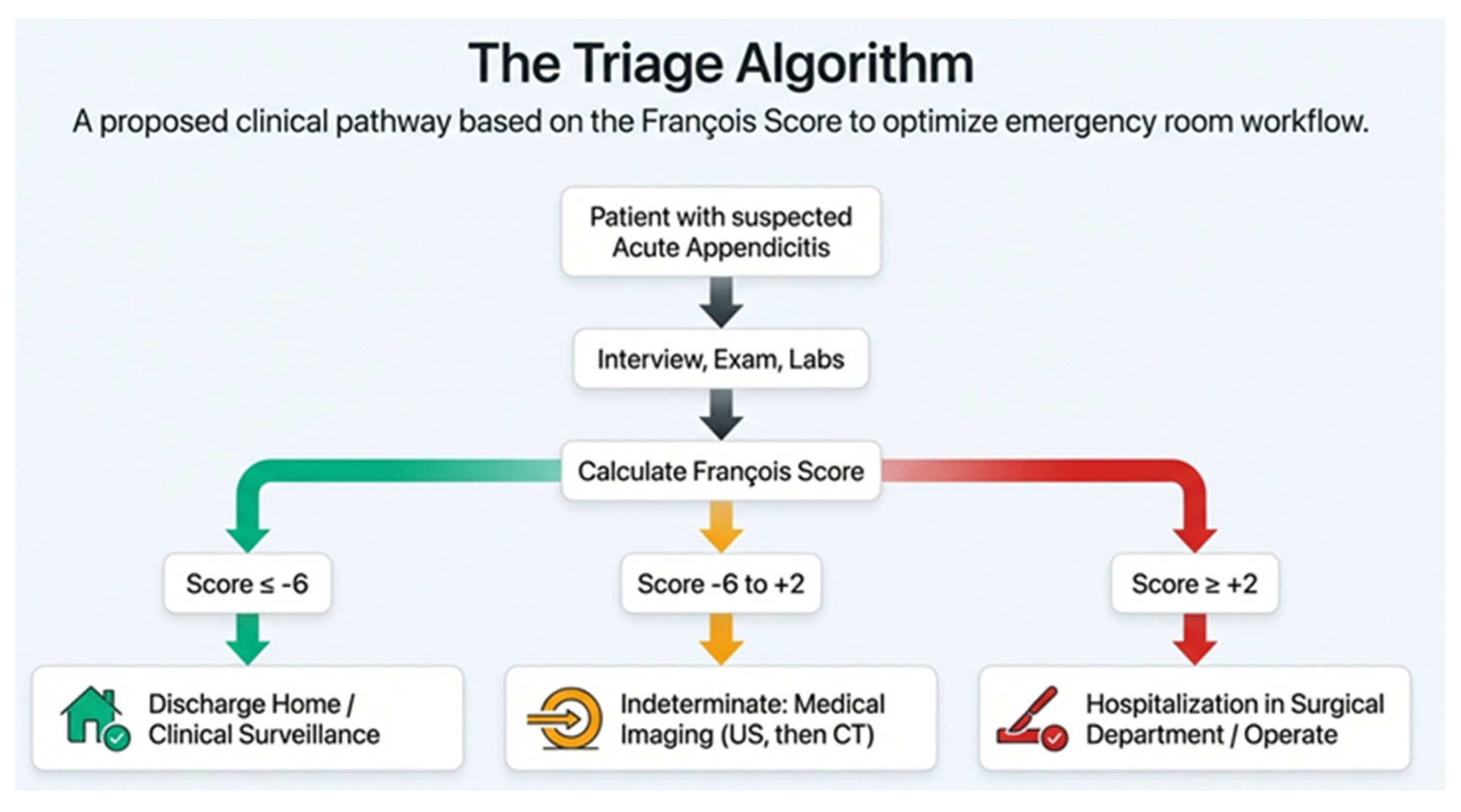
Figure 2.
Proposed diagnostic algorithm based on the François score.
Figure 2.
Proposed diagnostic algorithm based on the François score.
Nevertheless, several limitations must be acknowledged. First, this study was conducted at a single center, which may limit the generalizability of the results to other healthcare systems. Second, although the François score was calculated independently of therapeutic decision-making, surgeons were not blinded to the clinical presentation of the patients, which may introduce observational bias.
Finally, the relatively small number of patients without appendicitis (n = 11) limits the precision of specificity estimates and may widen confidence intervals for some diagnostic indicators. Despite these limitations, the present study represents one of the few external validations of the François score outside Europe. The prospective design, inclusion of both operated and non-operated patients, and systematic data collection strengthen the methodological robustness of our findings and support the clinical utility of this score in routine surgical practice.
5. Conclusions
In this prospective study, the François score demonstrated excellent diagnostic accuracy for patients presenting with suspected acute appendicitis in a tertiary-care setting. The score showed high sensitivity and specificity, confirming its reliability as a clinical decision-support tool for early risk stratification.
Beyond its diagnostic performance, the François score may contribute to optimizing the management of suspected appendicitis by identifying patients with a high probability of disease who could proceed directly to surgery, while reserving imaging for cases with intermediate diagnostic uncertainty. Such an approach may help reduce unnecessary computed tomography use, limit radiation exposure, and improve resource utilization in emergency departments.
These findings support the integration of the François score into structured diagnostic pathways for suspected appendicitis, particularly in healthcare settings where access to advanced imaging may be limited. Further multicenter studies involving larger and more diverse populations are warranted to confirm these results and to better define the role of this score within modern appendicitis diagnostic algorithms.
Funding
“This research received no external funding”.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors on request.
Acknowledgments
The authors have reviewed and edited the output and take full responsibility for the content of this publication.”.
Conflicts of Interest
“The authors declare no conflicts of interest.” “The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results”.
References
- Moris, D; Paulson, EK; Pappas, TN. Diagnosis and management of acute appendicitis in adults: a review. J Am Med Assoc. 2021, 326(22), 2299-311. [Google Scholar] [CrossRef] [PubMed]
- Bergeron, E. Clinical judgment remains of great value in the diagnosis of acute appendicitis. Can J Surg 2006, 49(2), 96–100. [Google Scholar] [PubMed]
- Bhangu, A; Søreide, K; Di Saverio, S; Assarsson, JH; Drake, FT. Acute appendicitis: modern understanding of pathogenesis, diagnosis, and management. Lancet 2015, 386(10000), 1278-87. [Google Scholar] [CrossRef] [PubMed]
- Alvarado, A. A practical score for the early diagnosis of acute appendicitis. Ann Emerg Med 1986, 15(5), 557-64. [Google Scholar] [CrossRef] [PubMed]
- Brigand, C; Steinmetz, JP; Rohr, S. The usefulness of scores in the diagnosis of appendicitis. J Chir. 2009, 146(1), 2-7. [Google Scholar]
- François, Y; Bonvoisin, S; Descos, L; Vignal, J. Prospective study of a predictive scoring system for the diagnosis of appendicitis in patients with right lower quadrant pain. Long-term outcome. GastroenterolClin Biol. 1991, 15(11), 794–9. [Google Scholar]
- Sanchez, F. Valeur d’un score clinico-biologique pour le diagnostic de l’appendicite aiguë de l’adulte. In Étude prospective de 202 cas [thèse: médecine]; Université de Strasbourg: Strasbourg, 2004. [Google Scholar]
- Hodonou, MA; TamouSambo, B; Allode, SA; Gbédolo, RR; Séto, DM; Tobomé, SR; et al. Usefulness of a clinico-biological François’ score in the diagnosis of acute appendicitis: experience of the university hospital center of Parakou, Benin. Surg Sci. 2018, 9(3), 91-6. [Google Scholar] [CrossRef]
- McKay, R; Shepherd, J. The use of the clinical scoring system by alvarado in the decision to perform computed tomography for acute appendicitis in the ED. Am J Emerg Med 2007, 25(5), 489–93. [Google Scholar] [CrossRef] [PubMed]
- Winn, RD; Laura, S; Douglas, C; Davidson, P; Gani, JS. Protocol-based approach to suspected appendicitis, incorporating the alvarado score and outpatient antibiotics. ANZ J Surg 2004, 74(5), 324–9. [Google Scholar] [CrossRef] [PubMed]
- Ohle, R; O’Reilly, F; O’Brien, KK; Fahey, T; Dimitrov, BD. The alvarado score for predicting acute appendicitis: a systematic review. BMC Med. 2011, 9, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Maghrebi, H; Maghraoui, H; Makni, A; Sebei, A; Fredj, SB; Mrabet, A; et al. Role of the alvarado score in the diagnosis of acute appendicitis. Pan Afr Med J. 2018, 29, 1–8. [Google Scholar]
- Zouaghi, W. Validation du score d’Alvarado dans le diagnostic d’appendicite aigüe dans une population tunisienne [thèse: médecine]; Université de Monastir: Monastir, 2014. [Google Scholar]
- Andersson, M; Andersson, RE. The appendicitis inflammatory response score: a tool for the diagnosis of acute appendicitis that outperforms the Alvarado score. World J Surg 2008, 32(8), 1843-9. [Google Scholar] [CrossRef] [PubMed]
- Andersson, RE; Hugander, A; Ravn, H; Offenbartl, K; Ghazi, SH; Nyström, PO; et al. Repeated clinical and laboratory examinations in patients with an equivocal diagnosis of appendicitis. World J Surg 2000, 24(4), 479-85. [Google Scholar] [CrossRef] [PubMed]
- Fenyö, G; Lindberg, G; Blind, P; Enochsson, L; Oberg, A. Diagnostic decision support in suspected acute appendicitis: validation of a simplified scoring system. Eur J Surg. 1997, 163(11), 831-8. [Google Scholar] [PubMed]
- Enochsson, L; Gudbjartsson, T; Hellberg, A; Rudberg, C; Wenner, J; Ringqvist, I; et al. The fenyö-lindberg scoring system for appendicitis increases positive predictive value in fertile women--a prospective study in 455 patients randomized to either laparoscopic or open appendectomy. SurgEndosc 2004, 18(10), 1509-13. [Google Scholar]
- Ohmann, C; Franke, C; Yang, Q. Clinical benefit of a diagnostic score for appendicitis: results of a prospective interventional study. German study group of acute abdominal pain. Arch Surg 1999, 134(9), 993–6. [Google Scholar] [CrossRef] [PubMed]
- Sabbagh, R. La construction d’un score clinico-biologique prédictif d’appendicite aiguë. thèse: médecine Université de Sousse, Sousse, 2018. [Google Scholar]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).