Submitted:
07 March 2026
Posted:
09 March 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Environmental Degradation of Pyrethroids
Microbial Degradation of Pyrethroids
Factors Influencing Persistence of Pyrethroids
Environmental Persistence and Bioavailability of Pyrethroids
Pyrethroid Toxicity to Molluscs
Absorption and Distribution of Pyrethroids in Molluscs
Absorption
Distribution
Pyrethroid Metabolic Pathways in Molluscs
Phase I Metabolism
Phase II Metabolism
Life Stage Dependent Detoxification of Pyrethroids in Molluscs
Effects of Pyrethroids on Molluscan Behaviours
Effects of Pyrethroids on Molluscsan Foraging and Feeding Behaviour
Effects of Pyrethroids on Molluscsan Mating Strategies and Evolutionary Implications
Effects of Pyrethroids on Molluscsan Neurobiology and Behavioural Gene Expressions
Effects of Pyrethroids on Molluscsan Memory and Learning Mechanisms
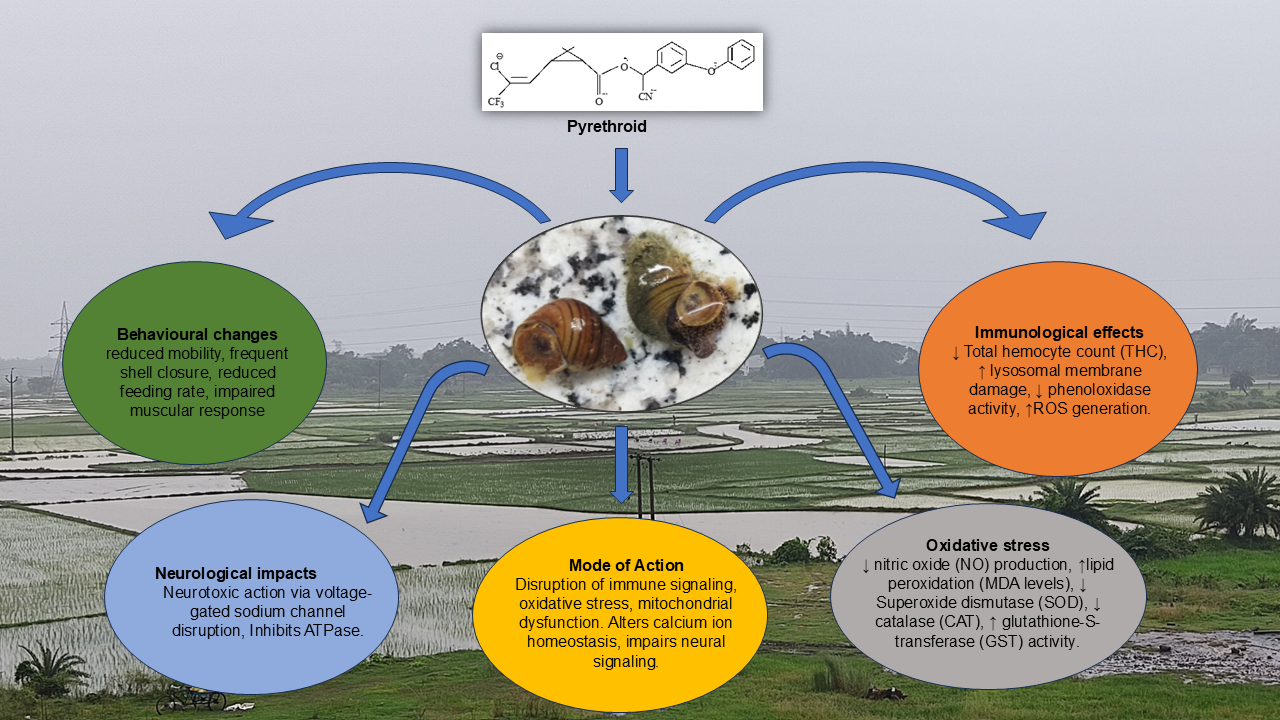
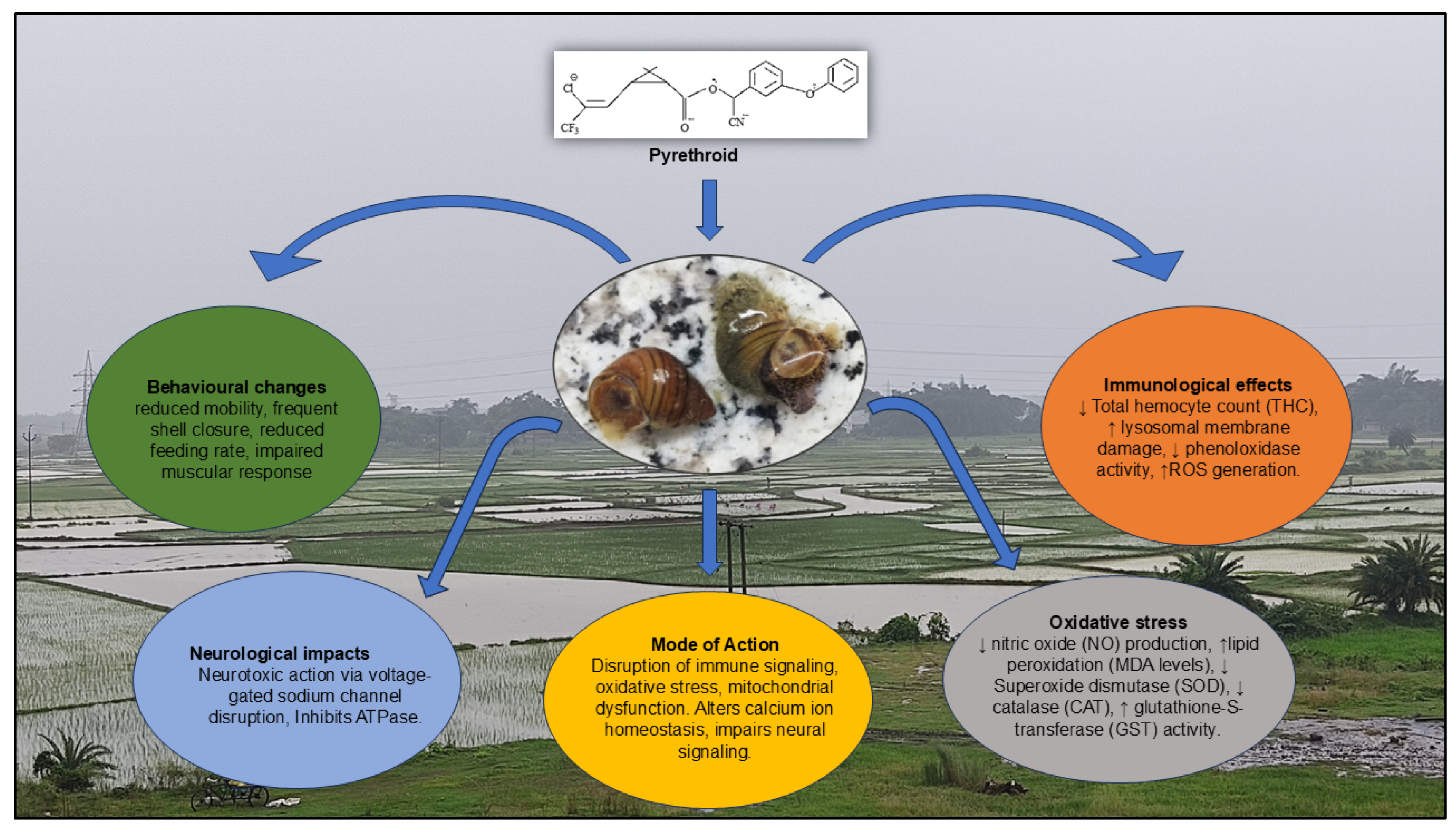
| Pyrethroid Type | Immunological Effects | Behavioral Changes | Mode of Action | Reference |
|---|---|---|---|---|
| Cypermethrin | ↓ Total hemocyte count (THC), ↑ lysosomal membrane damage, ↓ phenoloxidase activity, ↑ROS generation. | Lethargy, reduced mobility, frequent shell closure | Neurotoxic action via voltage-gated sodium channel disruption, oxidative stress-induced hemocyte apoptosis. | San Juan et al., 2020 |
| Deltamethrin | ↓ Phagocytic index, ↓ nitric oxide (NO) production, ↑lipid peroxidation (MDA levels). | Altered locomotion, reduced feeding rate | Disruption of immune signalling, oxidative stress, mitochondrial dysfunction. | Jiang et al., 2021 |
| Permethrin | ↓ Superoxide dismutase (SOD), ↓ catalase (CAT), ↑ glutathione-S-transferase (GST) activity. | Reduced surface activity, impaired muscular response | Generation of oxidative stress, reduced antioxidant defines. | Arslan, 2023 |
| Cyfluthrin | ↓ Hemocyanin level, ↑ pro-inflammatory cytokine expression. | Erratic movements, periodic inactivity | Alters calcium ion homeostasis, impairs neural signalling. | Arman., 2023 |
| Fenvalerate | ↑ Apoptosis of hemocytes, ↑ malondialdehyde (MDA), ↓ total protein content. | Spontaneous shell withdrawal, hyperreactivity to stimuli | Inhibits ATPase, induces ROS-mediated damage to immune cells. | Mukherjee, S., & Mandal, C. ,2023 |
| Esfenvalerate | DNA fragmentation in hemocytes, ↓ immune gene expression. | Disorientation, shell spinning | Genotoxic and epigenetic modulation of immune pathways. | Werner, I., & Moran, K., 2008 |
Molecular Mechanisms Behind Pyrethroid-Induced Changes in Molluscs
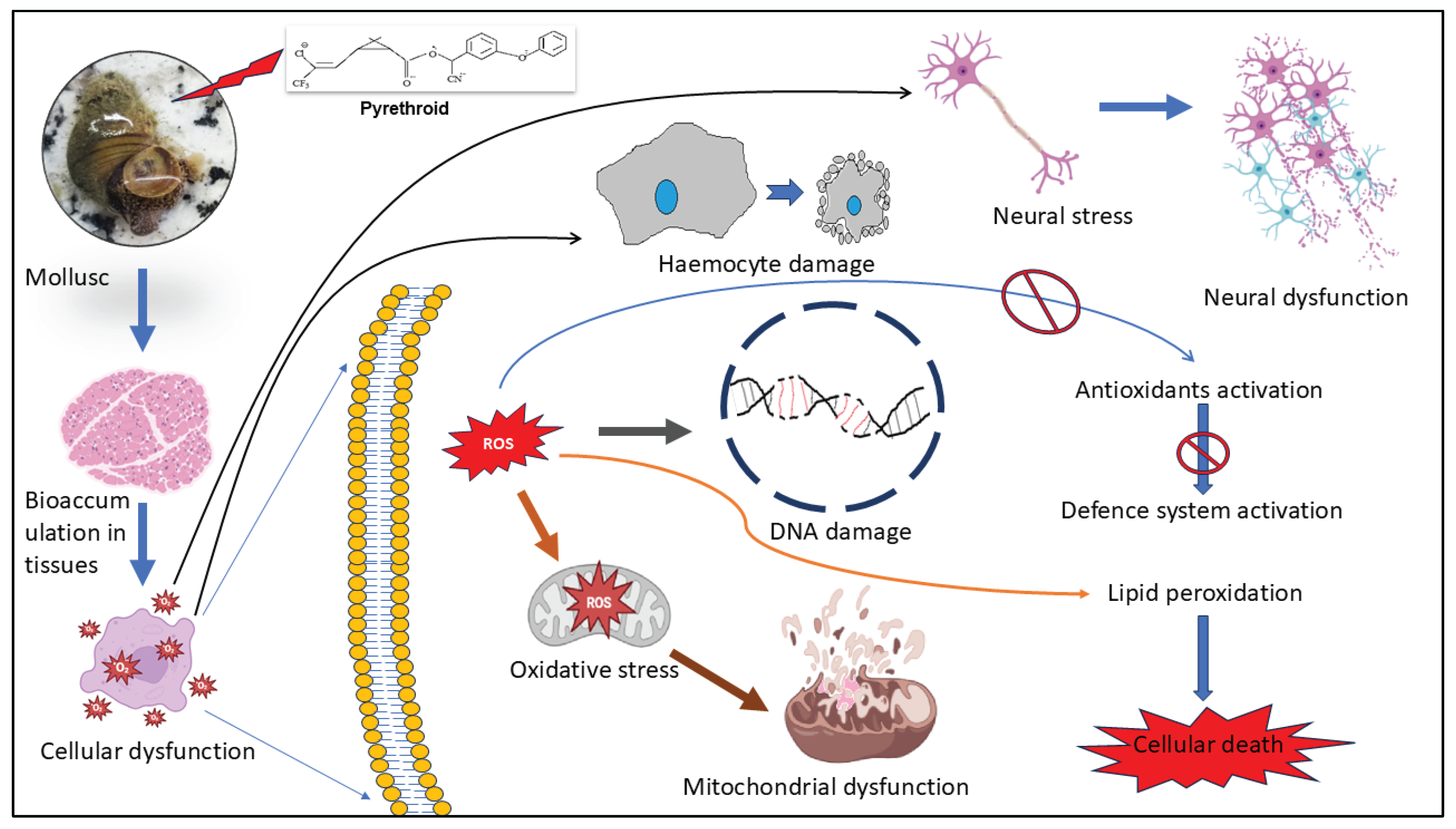
Neurotoxic Mechanisms
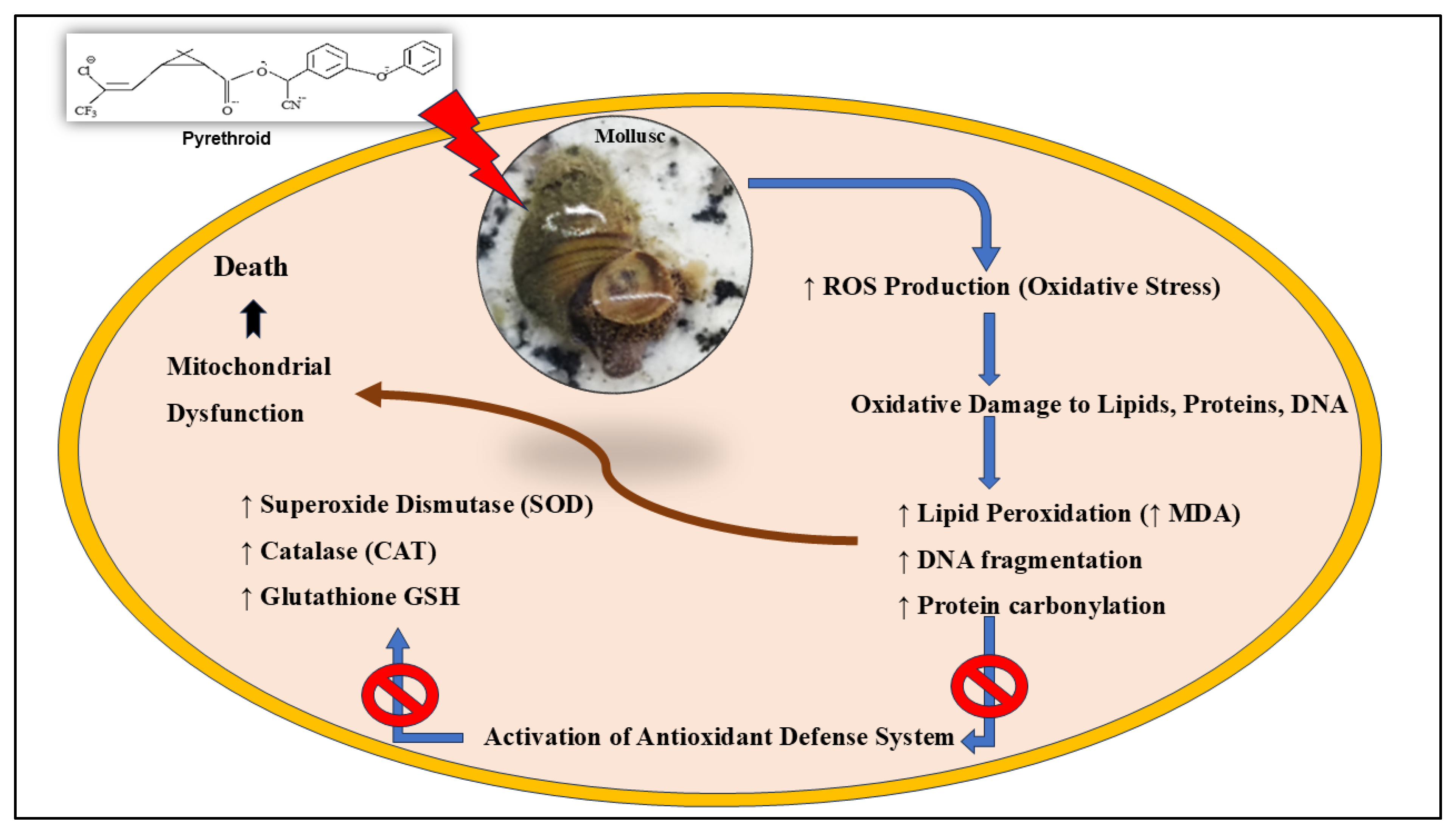
Induce Oxidative Stress
Endocrine Disruption
Pyrethroids and Molluscan Immunity
Disrupt Haemocyte Function
Histopathological Changes
Ecological and Toxicological Implications
Impact on Molluscan Populations
Delayed Growth and Reproduction
Implications for Molluscan Diversity
Impact on Molluscan Communication
Disruption of Pheromone Signalling
Interference with Alarm Cues
Impairment of Olfactory Functions
Pyrethroid Pollution Control
Discussion and Conclusions
Funding
Data and Materials Availability
Conflicts of Interest
Ethical Approval
Code Availability
References
- Ait Ayad, M.; Ait Fdil, M.; Mouabad, A. Effects of cypermethrin (pyrethroid insecticide) on the valve activity behavior, byssal thread formation, and survival in air of the marine mussel Mytilus galloprovincialis. Archives of environmental contamination and toxicology 2011, 60(3), 462–470. [Google Scholar] [CrossRef]
- Atta, S. A.; Fahmy, Z. H.; Selim, E. A.; Aboushousha, T.; Mostafa, R. R. Effect of linex treatment on IFN-γ and IL-4 in mice infected with Trichinella. BMC Infectious Diseases 2024, 24(1), 1360. [Google Scholar] [CrossRef] [PubMed]
- Arman, S. Effects of Single and Combined Exposure to Environmentally Relevant Concentrations of Cyfluthrin and Copper on Digestive Gland Histology of Lymnaea stagnalis. Sakarya University Journal of Science 2023, 27(1), 39–48. [Google Scholar] [CrossRef]
- Arslan, P. Pyrethroid-induced oxidative stress and biochemical changes in the primary mussel cell cultures. Environmental Science and Pollution Research 2023, 30(16), 48484–48490. [Google Scholar] [CrossRef]
- Auguste, M.; Balbi, T.; Ciacci, C.; Canesi, L. Conservation of cell communication systems in invertebrate host–defence mechanisms: possible role in immunity and disease. Biology 2020, 9(8), 234. [Google Scholar] [CrossRef]
- Australian Industrial Chemicals Introduction Scheme (AICIS). Available online: https://services.industrialchemicals.gov.au/search-inventory/.
- Aznar-Alemany, Ò.; Piotrowska-Seget, M.Z. Introduction to pyrethroid insecticides: chemical structures, properties, mode of action and use. In Pyrethroid insecticides (pp. 1-16). Cham: Springer International Publishing. https://doi.org/10.1007/698_2019_435 Cycoń, M.; Piotrowska-Seget, Z. (2016). Pyrethroiddegrading microorganisms and their potential for the bioremediation of contaminated soils: a review. Frontiers in microbiology 2020, 7, 1463. [Google Scholar] [CrossRef]
- Bacqué-Cazenave, J.; Bharatiya, R.; Barrière, G.; Delbecque, J. P.; Bouguiyoud, N.; Di Giovanni, G.; De Deurwaerdère, P. Serotonin in animal cognition and behavior. International journal of molecular sciences 2020, 21(5), 1649. [Google Scholar] [CrossRef]
- Bayne, B. L. Phenotypic flexibility and physiological tradeoffs in the feeding and growth of marine bivalve molluscs. Integrative and Comparative Biology 2004, 44(6), 425–432. [Google Scholar] [CrossRef]
- Benjamin, P. R.; Kemenes, G. Lymnaea learning and memory. Scholarpedia 2010, 5(8), 4247. [Google Scholar] [CrossRef]
- Benjamin, P.; Crossley, M. Gastropod feeding systems: Evolution of neural circuits that generate diverse behaviors. In Oxford Research Encyclopedia of Neuroscience; 2020. [Google Scholar] [CrossRef]
- Bergamini, G.; Sacchi, S.; Ferri, A.; Franchi, N.; Montanari, M.; Ahmad, M.; Malagoli, D. Clodronate liposome-mediated phagocytic hemocyte depletion affects the regeneration of the cephalic tentacle of the invasive snail, Pomacea canaliculata. Biology 2023, 12(7), 992. [Google Scholar] [CrossRef] [PubMed]
- Berthoud, H. R. Mind versus metabolism in the control of food intake and energy balance. Physiology & behavior 2004, 81(5), 781–793. [Google Scholar] [CrossRef]
- Bernal-González, K. G.; Covantes-Rosales, C. E.; Camacho-Pérez, M. R.; Mercado-Salgado, U.; Barajas-Carrillo, V. W.; Girón-Pérez, D. A.; Girón-Pérez, M. I. Organophosphate-pesticide-mediated immune response modulation in invertebrates and vertebrates. International Journal of Molecular Sciences 2023, 24(6), 5360. [Google Scholar] [CrossRef]
- Boutet, I.; Tanguy, A.; Moraga, D. Molecular identification and expression of two non-P450 enzymes, monoamine oxidase A and flavin-containing monooxygenase 2, involved in phase I of xenobiotic biotransformation in the Pacific oyster, Crassostrea gigas. Biochimica et Biophysica Acta (BBA)-Gene Structure and Expression 2004, 1679(1), 29–36. [Google Scholar] [CrossRef]
- Braun, M. H.; Lukowiak, K.; Karnik, V.; Lukowiak, K. Differences in neuronal activity explain differences in memory forming abilities of different populations of Lymnaea stagnalis. Neurobiology of Learning and Memory 2012, 97(1), 173–182. [Google Scholar] [CrossRef]
- Burns, C. J.; Pastoor, T. P. Pyrethroid epidemiology: a quality-based review. Critical reviews in toxicology 2018, 48(4), 297–311. [Google Scholar] [CrossRef]
- Casida, J. E.; Durkin, K. A. Neuroactive insecticides: targets, selectivity, resistance, and secondary effects. Annual review of entomology 2013, 58(1), 99–117. [Google Scholar] [CrossRef]
- Chen, C. H. Phase II Enzymes. In Activation and Detoxification Enzymes: Functions and Implications; Springer New York: New York, NY, 2011; pp. 37–48. [Google Scholar] [CrossRef]
- Cycoń, M.; Piotrowska-Seget, Z. Pyrethroid-degrading microorganisms and their potential for the bioremediation of contaminated soils: a review. Frontiers in microbiology 2016, 7, 1463. [Google Scholar] [CrossRef] [PubMed]
- Deidda, I.; Russo, R.; Bonaventura, R.; Costa, C.; Zito, F.; Lampiasi, N. Neurotoxicity in marine invertebrates: an update. Biology 2021, 10(2), 161. [Google Scholar] [CrossRef] [PubMed]
- De la Ballina, N. R.; Maresca, F.; Cao, A.; Villalba, A. Bivalve haemocyte subpopulations: a review. Frontiers in immunology 2022, 13, 826255. [Google Scholar] [CrossRef] [PubMed]
- Dhiman, V.; Pant, D. Environmental biomonitoring by snails. Biomarkers 2021, 26(3), 221–239. [Google Scholar] [CrossRef]
- Di Cosmo, A.; Winlow, W.; Farag, M. R.; Alagawany, M.; Bilal, R. M.; Gewida, A. G.; Dhama, K.; Abdel-Latif, H. M.; Naiel, M. A.; Neuroecology and neuroethology in Molluscs The interface between behaviour and environment (pp. 1-239). NOVA PUBLISHER. An overview on the potential hazards of pyrethroid insecticides in fish, with special emphasis on cypermethrin toxicity. Animals 2014, 11(7), 1880. [Google Scholar] [CrossRef]
- Dutta, S. M.; Mustafi, S. B.; Raha, S.; Chakraborty, S. K. Assessment of thermal stress adaptation by monitoring Hsp70 and MnSOD in the freshwater gastropod, Bellamya bengalensis (Lamark 1882). Environmental monitoring and assessment 2014, 186(12), 8961–8967. [Google Scholar] [CrossRef]
- Fernández San Juan, M. R.; Cortelezzi, A.; Albornoz, C. B.; Landro, S. M.; Arrighetti, F.; Najle, R.; Lavarias, S. M. L. Ecotoxicology and environmental safety toxicity of pyrethroid cypermethrin on the freshwater snail Chilina parchappii: Lethal and sublethal effects. 2020. [Google Scholar] [CrossRef]
- Gillette, R.; Brown, J. W. The sea slug, Pleurobranchaea californica: a signpost species in the evolution of complex nervous systems and behavior. Integrative and Comparative Biology 2015, 55(6), 1058–1069. [Google Scholar] [CrossRef] [PubMed]
- Gökoğlu, N. Molluscan shellfish. In Shellfish processing and preservation; Springer International Publishing: Cham, 2021; pp. 129–250. [Google Scholar] [CrossRef]
- Griscom, S. B.; Fisher, N. S. Bioavailability of sediment-bound metals to marine bivalve molluscs: an overview. Estuaries 2004, 27(5), 826–838. [Google Scholar] [CrossRef]
- Hazardous Substances Data Bank (HSDB). Available online: https://pubchem.ncbi.nlm.nih.gov/source/hsdb/1511.
- Hossain, M. A.; Sarker, T. R.; Sutradhar, L.; Hussain, M.; Iqbal, M. M. Toxic effects of chlorpyrifos on the growth, hemocytes counts, and vital organ’s histopathology of freshwater mussel, Lamellidens marginalis. Journal of King Saud University-Science 2023, 35(2), 102482. [Google Scholar] [CrossRef]
- ILO-WHO International Chemical Safety Cards (ICSCs). Available online: https://www.ilo.org/dyn/icsc/showcard.display?p_version=2&p_card_id=0212.
- Impellitteri, F.; Curpăn, A. S.; Plăvan, G.; Ciobica, A.; Faggio, C. Hemocytes: a useful tool for assessing the toxicity of microplastics, heavy metals, and pesticides on aquatic invertebrates. International journal of environmental research and public health 2022, 19(24), 16830. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, H. H.; Qadir, A.; Ahmad, S. R.; Riaz, M. A.; Riaz, A.; Shahid, N.; Arslan, M. Residual assessment of emerging pesticides in aquatic sinks of Lahore, Pakistan. Sustainability 2024, 16(21), 9257. [Google Scholar] [CrossRef]
- Jaggi, R. C.; Aulakh, M. S.; Sharma, R. Impacts of elemental S applied under various temperature and moisture regimes on pH and available P in acidic, neutral and alkaline soils. Biology and Fertility of Soils 2005, 41(1), 52–58. [Google Scholar] [CrossRef]
- Jiang, Q.; Ao, S.; Ji, P.; Zhou, Y.; Tang, H.; Zhou, L.; Zhang, X. Assessment of deltamethrin toxicity in Macrobrachium nipponense based on histopathology, oxidative stress and immunity damage. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology 2021, 246, 109040. [Google Scholar] [CrossRef]
- Jordaens, K.; Dillen, L.; Backeljau, T. Effects of mating, breeding system and parasites on reproduction in hermaphrodites: pulmonate gastropods (Mollusca). Animal Biology 2007, 57(2), 137–195. [Google Scholar] [CrossRef]
- Jordaens, K.; Dillen, L.; Backeljau, T. Shell shape and mating behaviour in pulmonate gastropods (Mollusca). Biological Journal of the Linnean Society 2009, 96(2), 306–321. [Google Scholar] [CrossRef]
- Kamrin, M. A.; Katagi, T.; Pesticide profiles: toxicity; environmental impact; and fate. Photodegradation of pesticides on plant and soil surfaces. In Reviews of environmental contamination and toxicology: Continuation of residue reviews; CRC press, 1997; pp. 1–78. [Google Scholar] [CrossRef]
- Kaneko, H. Pyrethroid chemistry and metabolism. In Hayes’ handbook of pesticide toxicology; Academic press, 2010; pp. 1635–1663. [Google Scholar] [CrossRef]
- Katagi, T. Bioconcentration, bioaccumulation, and metabolism of pesticides in aquatic organisms. Reviews of environmental contamination and toxicology 2009, 1–132. [Google Scholar] [CrossRef]
- Kiss, T. Do terrestrial gastropods use olfactory cues to locate and select food actively? Invertebrate Neuroscience 2017, 17(3), 9. [Google Scholar] [CrossRef]
- Koene, J. M. Neuro-endocrine control of reproduction in hermaphroditic freshwater snails: mechanisms and evolution. Frontiers in behavioral neuroscience 2010, 4, 167. [Google Scholar] [CrossRef]
- Kotsyuba, E.; Dyachuk, V. Role of the neuroendocrine system of marine bivalves in their response to hypoxia. International Journal of Molecular Sciences 2023, 24(2), 1202. [Google Scholar] [CrossRef]
- Kowall, C. A.; Batabyal, A.; Lukowiak, K.; Phillips, I. D. Agricultural use of insecticides alters homeostatic behaviors and cognitive ability in Lymnaea stagnalis. Environmental Toxicology and Chemistry 2023, 42(11), 2466–2477. [Google Scholar] [CrossRef]
- Landrum, P. F.; Robbins, J. A. Bioavailability of sediment-associated contaminants to benthic invertebrates. In Sediments; CRC Press, 2020; pp. 237–263. [Google Scholar]
- Langston, W. J.; Pope, N. D. Molluscs. In Ecotoxicology of Antifouling biocides; Springer Japan: Tokyo, 2009; pp. 271–289. [Google Scholar] [CrossRef]
- Leung, K. M.; Grist, E. P.; Morley, N. J.; Morritt, D.; Crane, M. Chronic toxicity of tributyltin to development and reproduction of the European freshwater snail Lymnaea stagnalis (L.). Chemosphere 2007, 66(7), 1358–1366. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Zhao, X.; Kong, L.; Yu, H. J. I. S. J. Transcriptomic response to stress in marine bivalves. Invertebrate survival journal 2013, 10(1), 84–93. [Google Scholar]
- Li, M.; Wang, M.; Wang, W.; Wang, L.; Liu, Z.; Sun, J.; Song, L. The immunomodulatory function of invertebrate specific neuropeptide FMRFamide in oyster Crassostrea gigas. Fish & shellfish immunology 2019, 88, 480–488. [Google Scholar] [CrossRef]
- Lodi, M.; Koene, J. M. The love-darts of land snails: integrating physiology, morphology and behaviour. Journal of Molluscan Studies 2016, 82(1), 1–10. [Google Scholar] [CrossRef]
- López-Pedrouso, M.; Lorenzo, J. M.; Varela, Z.; Fernández, J. Á.; Franco, D. Finding biomarkers in antioxidant molecular mechanisms for ensuring food safety of bivalves threatened by marine pollution. Antioxidants 2022, 11(2), 369. [Google Scholar] [CrossRef]
- Major, K. M.; Brander, S. M. The ecological and evolutionary implications of pyrethroid exposure: a new perspective on aquatic ecotoxicity. In Pyrethroid insecticides; Springer International Publishing: Cham, 2020; pp. 109–148. [Google Scholar] [CrossRef]
- Matthews, R. W.; Matthews, J. R. Foraging and feeding. In Insect Behavior: 2nd Edition; Springer Netherlands: Dordrecht, 2009; pp. 131–184. [Google Scholar] [CrossRef]
- Méjanelle, L.; Jara, B.; Dachs, J. Fate of pyrethroids in freshwater and marine environments. In Pyrethroid insecticides; Springer International Publishing: Cham, 2020; pp. 81–107. [Google Scholar] [CrossRef]
- Middlebrooks, M. L.; Pierce, S. K.; Bell, S. S. Foraging behavior under starvation conditions is altered via photosynthesis by the marine gastropod, Elysia clarki. PLoS One 2011, 6(7), e22162. [Google Scholar] [CrossRef] [PubMed]
- Miller, M. W. Dopamine as a multifunctional neurotransmitter in gastropod molluscs: an evolutionary hypothesis. The Biological Bulletin 2020, 239(3), 189–208. [Google Scholar] [CrossRef] [PubMed]
- Morley, N. J. Interactive effects of infectious diseases and pollution in aquatic molluscs. Aquatic toxicology 2010, 96(1), 27–36. [Google Scholar] [CrossRef]
- Mukherjee, S.; Mandal, C.; Ngandjui, Y. A. T.; Kereeditse, T. T.; Kamika, I.; Madikizela, L. M.; Msagati, T. A. M.; Phagocytic response and phenoloxidase activity of the hemocytes of Bellamya bengalensis exposed to synthetic fenvalerate. Nutraceutical and medicinal importance of marine molluscs. Journal of Stress Physiology & Biochemistry;Marine Drugs 2023, 19(3) 22(5), 143–151 201. [Google Scholar] [CrossRef]
- Ng, T. P.; Saltin, S. H.; Davies, M. S.; Johannesson, K.; Stafford, R.; Williams, G. A. Snails and their trails: the multiple functions of trail-following in gastropods. Biological Reviews 2013, 88(3), 683–700. [Google Scholar] [CrossRef]
- Nkya, T. E.; Akhouayri, I.; Kisinza, W.; David, J. P. Impact of environment on mosquito response to pyrethroid insecticides: facts, evidences and prospects. Insect biochemistry and molecular biology 2013, 43(4), 407–416. [Google Scholar] [CrossRef]
- Peshin, R.; Zhang, W. Integrated pest management and pesticide use. In Integrated Pest Management: Pesticide Problems; Springer Netherlands: Dordrecht, 2014; Vol. 3, pp. 1–46. [Google Scholar] [CrossRef]
- Petushok, N.; Gabryelak, T.; Pałecz, D.; Zavodnik, L.; Varga, I. S.; Deér, K. A. Comparative study of the xenobiotic metabolising system in the digestive gland of the bivalve molluscs in different aquatic ecosystems and in aquaria experiments. Aquatic toxicology 2002, 61(1-2), 65–72. [Google Scholar] [CrossRef]
- PubChem. Available online: https://pubchem.ncbi.nlm.nih.gov/.
- Quiroz-Barroso, S. A.; Guerrero-Arenas, R.; García-Barrera, P.; Sánchez-Beristain, F. Mollusca: Bivalvia and Gastropoda. In Past Environments of Mexico: Unveiling the Past Environments of a Megadiverse Country Through its Fossil Record; Springer Nature Switzerland: Cham, 2024; pp. 63–97. [Google Scholar] [CrossRef]
- Ray, M.; Bhunia, A. S.; Bhunia, N. S.; Ray, S. Density shift, morphological damage, lysosomal fragility and apoptosis of hemocytes of Indian molluscs exposed to pyrethroid pesticides. Fish & shellfish immunology 2013, 35(2), 499–512. [Google Scholar] [CrossRef]
- Romano, R. A. Feeding Biology of the Aeolid Nudibranch Flabellina verrucosa; University of New Hampshire, 2022. [Google Scholar]
- Romero, A.; Novoa, B.; Figueras, A. The complexity of apoptotic cell death in mollusks: An update. Fish & Shellfish Immunology 2015, 46(1), 79–87. [Google Scholar] [CrossRef]
- Russo, J.; Lagadic, L. Effects of parasitism and pesticide exposure on characteristics and functions of hemocyte populations in the freshwater snail Lymnaea palustris (Gastropoda, Pulmonata). Cell biology and toxicology 2000, 16(1), 15–30. [Google Scholar] [CrossRef]
- Saha, R.; Dutta, S. M. Pesticides’ mode of action on aquatic life. Toxicology Reports 2024, 13, 101780. [Google Scholar] [CrossRef]
- Saha, R.; Dutta, S. M. Pyrethroids have become a barrier to the daily existence of molluscs. Journal of Hazardous Materials Letters 2025, 100144. [Google Scholar] [CrossRef]
- Sakakibara, M. Cellular and molecular aspects of short-term and long-term memory from molluscan systems. In Novel Trends in Brain Science: Brain Imaging, Learning and Memory, Stress and Fear, and Pain; Springer Japan: Tokyo, 2008; pp. 131–148. [Google Scholar] [CrossRef]
- Salice, C. J.; Kimberly, D. A. Environmentally relevant concentrations of a common insecticide increase predation risk in a freshwater gastropod. Ecotoxicology 2013, 22(1), 42–49. [Google Scholar] [CrossRef] [PubMed]
- Samia, B.; Socorro, J.; Durand, A.; Quivet, E.; Wortham, H. Photolytic degradation of commonly used pesticides adsorbed on silica particles. Science of the Total Environment 2024, 949, 174964. [Google Scholar] [CrossRef]
- San Juan, M. F.; Cortelezzi, A.; Albornoz, C. B.; Landro, S. M.; Arrighetti, F.; Najle, R.; Lavarías, S. M. L. Toxicity of pyrethroid cypermethrin on the freshwater snail Chilina parchappii: Lethal and sublethal effects. Ecotoxicology and environmental safety 2020, 196, 110565. [Google Scholar] [CrossRef]
- Shan, Y.; Yan, S.; Hong, X.; Zha, J.; Qin, J. Effect of imidacloprid on the behavior, antioxidant system, multixenobiotic resistance, and histopathology of Asian freshwater clams (Corbicula fluminea). Aquatic Toxicology 2020, 218, 105333. [Google Scholar] [CrossRef] [PubMed]
- Sogorb, M. A.; Vilanova, E. Enzymes involved in the detoxification of organophosphorus, carbamate and pyrethroid insecticides through hydrolysis. Toxicology letters 2002, 128(1-3), 215–228. [Google Scholar] [CrossRef]
- Sternberg, R. M.; Gooding, M. P.; Hotchkiss, A. K.; LeBlanc, G. A. Environmental-endocrine control of reproductive maturation in gastropods: implications for the mechanism of tributyltin-induced imposex in prosobranchs. Ecotoxicology 2010, 19(1), 4–23. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Teng, J.; Wang, D.; Zhao, J.; Shan, E.; Wang, Q. The adverse impact of microplastics and their attached pathogen on hemocyte function and antioxidative response in the mussel Mytilus galloprovincialis. Chemosphere 2023, 325, 138381. [Google Scholar] [CrossRef]
- Tamvacakis, A. N.; Senatore, A.; Katz, P. S. Single neuron serotonin receptor subtype gene expression correlates with behaviour within and across three molluscan species. Proceedings of the Royal Society B 2018, 285(1885), 20180791. [Google Scholar] [CrossRef]
- Tripathi, P. K.; Singh, A. Carbaryl induced alterations in the reproduction and metabolism of freshwater snail Lymnaea acuminata. Pesticide biochemistry and physiology 2004, 79(1), 1–9. [Google Scholar] [CrossRef]
- Toxin and Toxin Target Database (T3DB). Available online: http://www.t3db.ca/toxins/T3D1843.
- USEPA/Office of Pesticide Programs. Reregistration Eligibility Decision for Allethrins p.5 EPA 738-R-07001 (June 2007). 7 June 2008. Available online: https://www.epa.gov/pesticides/reregistration/status.htm.
- Vaughn, C. C.; Hoellein, T. J. Bivalve impacts in freshwater and marine ecosystems. Annual review of ecology, evolution, and systematics 2018, 49(1), 183–208. [Google Scholar] [CrossRef]
- Wei, S.; Xu, T.; Jiang, T.; Yin, D. Chemosensory dysfunction induced by environmental pollutants and its potential as a novel neurotoxicological indicator: a review. Environmental science & technology 2021, 55(16), 10911–10922. [Google Scholar] [CrossRef]
- Werner, I.; Moran, K. Effects of pyrethroid insecticides on aquatic organisms. Synthetic pyrethroids: Occurrence and behavior in aquatic environments 2008, 991, 310–335. [Google Scholar]
- Wu, M.; Tang, X.; Sun, C.; Miao, J.; Wang, Q.; Pan, L. Kinetics of uptake and depuration of synthetic pyrethroid insecticides in manila clam (Ruditapes philippinarum). Environmental Science and Pollution Research 2023, 30(30), 76246–76252. [Google Scholar] [CrossRef]
- Wyeth, R. C.; Woodward, O. M.; Willows, A. D. Orientation and navigation relative to water flow, prey, conspecifics, and predators by the nudibranch mollusc Tritonia diomedea. The Biological Bulletin 2006, 210(2), 97–108. [Google Scholar] [CrossRef]
- Yu, Z. L.; Yang, M. J.; Song, H.; Zhang, T.; Yuan, X. T. Gastropod chemoreception behaviors—Mechanisms underlying the perception and location of targets and implications for shellfish fishery development in aquatic environments. Frontiers in Marine Science 2023, 9, 1042962. [Google Scholar] [CrossRef]
- Zha, C.; Sossin, W. S. The molecular diversity of plasticity mechanisms underlying memory: An evolutionary perspective. Journal of neurochemistry 2022, 163(6), 444–460. [Google Scholar] [CrossRef]
| Pesticide | Molecular formula and molecular weight | xLogP3 | log Kow | Solubility and density | Applications | EPA Toxicity |
| Allethrin | C19H26O3 ; 302.4 g/mol |
4.8 | 4.78 | Solubility in water: none Relative density (water = 1): 1.01 |
Household and agricultural insecticide | LD50 Rat female oral 685 mg/kg; LD50 Mallard duck, oral <2000 mg/kg |
| Bifenthrin | C23H22ClF3O2 ; 422.9 g/mol |
6 | 6.00 | water <1.0X10-3 mg/L/ at 20 °C Density: 1.2 g/cu m at 125 °C |
Household and agricultural insecticide | LD50 Rat oral 54.5 mg/kg; LD50 Quail oral 18 mg/kg |
| Permethrin | C21H20Cl2O3 ; 391.3 g/mol | 6.5 | 6.5 | water, 6.00X10-3 mg/L (pH 7, 20 °C); cis-isomers 0.20 mg/L (25 °C); trans-isomers 0.13 mg/L (25 °C); density: 1.19–1.27 at 20 °C |
Household insecticide | LC50 Brook Trout 2.3; LC50 Channel Catfish 7.2 |
| Phenothrin | C23H26O3 ; 350.4 g/mol | 6.2 | 6.01 | Density: 1.06 at 20 °C; In methanol, >5.0 g/mL; hexane, >4.96 g/mL at 25 °C; In water, <9.7 ug/L at 25 °C |
Agricultural insecticide | LD50 Rat >10mg/kg; LD50 Quail >2150 mg/kg |
| Resmethrin | C22H26O3 ; 338.4 g/mol | 6.1 | 5.43 | Density:0.958-0.968 at 20 °C; In water, 0.0379 mg/L at 25 °C |
Veterinary pesticide and household insecticide | LD50: > 5000 mg/kg (Oral, Rat); |
| Tefluthrin | C17H14ClF7O2 ; 418.7 g/mol | 5.4 | 6.4 | Density: 1.48 g/mL (25 °C); In acetone, hexane, toluene, dichloromethane, ethylacetate: >500; methanol 263 (all in g/L, 21 °C); In water, 0.02 mg/L at 20 °C |
Agricultural insecticide | LD50: 22 mg/kg (Oral, Rat); LD50: Duck oral 4190 mg/kg |
| Tetramethrin | C19H25NO4 ; 331.4 g/mol | 4.7 | 4.73 | Density: 1.108 at 20 °C/20 °C; Methanol (53 g/kg), hexane (20 g/kg), xylene (1 g/kg), acetone, toluene. ; In water, 1.83 mg/L at 25 °C. |
Agricultural insecticide and household insecticide | LD50: 5000 mg/kg (Oral, Rat); |
| Metofluthrin | C18H20F4O3 ; 360.3 g/mol | 3.7 | 4.97-5.03 at 25 °C | Density: 1.21 at 20 °C; Solvent solubility (20 to 25 °C; g/L): acetone, methanol, ethyl acetate, toluene, n-hexanes, dichloromethane, n-octanol, isopropyl alcohol; In water, 0.73 mg/L at pH 7, 20 °C) |
Insecticides, mosquito control. | LD50 Rat oral > 2,000 mg/kg |
| Transfluthrin | C15H12Cl2F4O2 ; 371.2 g/mol | 5.0 | 5.46 at 20 °C | Density:1.51g/cm3(23C); In water is less than 1.13 mg/L at 20 °C. In organic solvents such as heptane, p-xylene, 1,2-dichloroethane,methanol, acetone, and ethyl acetate. |
Household insecticide | LC50 Rainbow Trout 0.71 PPB; LC50 Water flea 0.0686 PPB; LC50 Bob white quail 4909 PPM |
| Cyfluthrin | C22H18Cl2FNO3 ; 434.3 g/mol | 6.2 | 5.95 | >1000 g/L dichloromethane; In water, 3.0X10-3 mg/L at 20 °C; Density: 1.34 g/cu cm at 22 °C |
Agricultural insecticide | LD50: 869-1271 mg/kg (Oral, Rat); LD50: >5000 mg/kg (Quail) |
| Cyhalothrin | C23H19ClF3NO3 ; 449.8 g/mol | 6.1 | 6.8 | Acetone, dichloromethane, methanol, diethyl ether, ethyl acetate, hexane, toluene, In water, 5.0X10-3 mg/L, temp not specified; Density: 1.25 at 25 °C |
Agricultural insecticide | LD50: 144-243 mg/kg (Oral, Rat); LD50: >5000 mg/kg for Duck. |
| Cypermethrin | C22H19Cl2NO3 ; 416.3 g/mol | 6.0 | 6.60 | Acetone, chloroform, cyclohexanone, xylene, ethanol, hexane; In water, 4X10-3 mg/L at 20 °C; Density: 1.25 g/cu cm at 20 °C |
Agricultural insecticide and household insecticide | LD50: 250-300 mg/kg (Oral, Mouse); LD50: 400 mg/kg for hamster |
| Deltamethrin | C22H19Br2NO3 ; 505.2 g/mol | 6.2 | 6.20 | Cyclohexanone, dichloromethane, acetone, benzene, dimethyl sulphoxide, xylene, isopropanol; In water, <0.002 mg/L at 25 °C Relative density (water = 1): 0.5 |
Agricultural insecticide and household insecticide | LD50: 4123 mg/kg (Oral, Rat); >4640 mg/kg for Duck |
| Fenpropathrin | C22H23NO3 ; 349.4 g/mol | 5.7 | 6.0 at 20 °C | Xylene, cyclohexanone, Methanol; In water, 1.41X10-2 mg/l at 25 °C. Density: 1.15 at 25 °C |
Veterinary pesticides | LD50: 70.6 mg/kg (Oral, Rat); 1089 mg/kg for duck |
| Fenvalerate | C25H22ClNO3 ; 419.9 g/mol | 6.2 | 6.20 | Solubility at 20 °C (g/L): acetone >450; chloroform >450; methanol >450; hexane 77; In water, 2.4X10-2 mg/L at 22 °C (seawater); Density: 1.17 at 23 °C/4 °C |
Agricultural insecticide | LD50: 70.2 mg/kg (Oral, Rat); >4000 mg/kg for Quail. |
| Flumethrin | C28H22Cl2FNO3 ; 510.4 g/mol | 7.6 | 6.2 | solubility in pure water reported as 0.1 µg/L for one isomer and 0.2 µg/L for the sum of both isomers; in acetone, xylene, methanol, acetonitrile, ethylacetate, and dimethylsulfoxide; density: 1.342 g/cm³ |
Veterinary pesticide | LD50: > 100 mg/kg (Oral, Rat); |
| Fluvalinate | C26H22ClF3N2O3 ; 502.9 g/mol | 7.7 | 7.02 | Solubility in water, 0.012 mg/L at 25 °C ; density: 1.29 g/cu cm at 25 °C |
Agricultural insecticide and Veterinary pesticide | LD50: 261-282 mg/kg (Oral, Rat); >2510 mg/kg for quail |
| Tralomethrin | C22H19Br4NO3 ; 665.0 g/mol | 8.0 | approximately 5 at 25 °C | In water, 0.8 mg/L; In acetone, dichloromethane, toluene, xylene >1000; dimethyl sulfoxide >500; ethanol >180 (all in g/L); density: 1.70 at 20 °C |
Agricultural insecticide | LD50: 1250 mg/kg (Oral, Rat); >2150 mg/kg for quail |
| Esfenvalerate | C25H22ClNO3; 419.9 g/mol | 6.2 | 6.22 | xylene, acetone, chloroform, ethyl acetate, dimethylformamide, dimethyl sulfoxide, hexane, methanol; In water, 2.0X10-3 mg/L at 25 °C; density: 1.26 (4-26 °C) |
Agricultural insecticide and Veterinary pesticide | LD50: 70.2 mg/kg (Oral, Rat); |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




