1. Introduction
Measles is a highly contagious viral infection caused by
Measles morbillivirus, a member of the genus
Morbillivirus within the family
Paramyxoviridae [
15,
16]. Despite the availability of an effective vaccine, measles has resurged globally, driven by declining vaccination coverage, vaccine hesitancy, and migration [
1,
2,
14]. In 2023 alone, the World Health Organization reported a 20% increase in measles cases and a 43% increase in deaths compared with the previous year, with 57 countries experiencing large or disruptive outbreaks [
1,
14]. In the Americas, the Pan American Health Organization issued an epidemiological alert in 2025 documenting renewed measles transmission after the region had briefly regained elimination status [
32].
Respiratory complications remain the primary cause of measles-associated morbidity and mortality, with pneumonia accounting for the majority of hospitalizations and deaths [
3,
15]. However, structural complications involving the air leak syndrome spectrum—pneumomediastinum, subcutaneous emphysema, pneumothorax, and pneumoperitoneum—are rarely reported and may be underdiagnosed in epidemic contexts [
4,
26,
27].
Spontaneous pneumomediastinum is defined as the presence of free air within the mediastinum in the absence of trauma, mechanical ventilation, or invasive procedures [
21]. The accepted pathophysiological mechanism is the Macklin effect: alveolar rupture secondary to increased intraalveolar pressure gradients, followed by air dissection along bronchovascular sheaths toward the mediastinum [
5,
6,
23]. In the setting of measles, intense cough and bronchioloalveolar inflammation serve as plausible precipitating factors [
16].
The literature on air leak complications in measles is limited, consisting primarily of isolated case reports and small historical series dating back to the 1960s [
7,
8,
26,
28]. Recent reports have renewed attention to this entity in the context of current outbreaks [
4,
29,
30].
We present a case of extensive air leak syndrome—including pneumomediastinum, subcutaneous emphysema, left pneumothorax, and pneumoperitoneum—in a pediatric patient with RT-PCR-confirmed measles, managed conservatively with favorable outcome. This report was prepared in accordance with the CARE (CAse REport) guidelines [
13].
2. Case Presentation
2.1. Clinical History
A 9-year-old previously healthy girl with no reported or documentable measles–rubella vaccination presented to the pediatric emergency department of a tertiary referral hospital.
Ten days prior to admission, she developed generalized abdominal pain, followed by odynophagia and unquantified intermittent fever responsive to paracetamol. A maculopapular exanthem with cephalocaudal distribution appeared on illness day 5. Persistent cough developed subsequently. The mother reported vigorous coughing episodes accompanied by wheezing suggestive of bronchospasm, prompting evaluation by a local physician who prescribed unspecified nebulized therapy without clinical improvement. The epidemiological investigation identified travel to Mexico City 12 days before exanthem onset (within the 21-day incubation period) and contact with a symptomatic household member.
Two days before admission, the patient developed sudden subcutaneous swelling of the thorax and face, accompanied by pain in both shoulders and the posterior thoracic region, exacerbated by breathing. She was evaluated at two external facilities before being referred to our institution with suspected subcutaneous emphysema.
2.2. Admission Findings
At presentation, the patient was hemodynamically stable. Vital signs were: heart rate 120 bpm, respiratory rate 35 breaths/min, temperature 37.0 °C, blood pressure 90/60 mmHg, and oxygen saturation 95% on room air. Weight was 26 kg and height 126 cm.
Physical examination revealed an alert, reactive child with increased respiratory effort and tachypnea. Palpable crepitation was present from the skull base to the nipple line bilaterally, consistent with extensive subcutaneous emphysema. Auscultation demonstrated preserved vesicular breath sounds with crepitant rales in the left hemithorax. The remainder of the examination, including cardiovascular, abdominal, and neurological assessments, was unremarkable. Residual Koplik spots were noted on the buccal mucosa, consistent with the late exanthematous phase of measles.
2.3. Diagnostic Workup
2.3.1. Laboratory Findings
Initial laboratory results are summarized in
Table 1. The complete blood count showed a normal leukocyte count (4.94 × 10
3/
L) with unremarkable differential except for an elevated basophil percentage (9.70%, reference 0–2.5%). Hepatic function tests were notably abnormal: gamma-glutamyl transferase (GGT) 109 IU/L (reference 8–30), alanine aminotransferase (ALT) 56 U/L (reference 10–40), aspartate aminotransferase (AST) 64 IU/L (reference 10–50), alkaline phosphatase (ALP) 201 IU/L (reference 53–128), and lactate dehydrogenase (LDH) 734 U/L (reference 91–190). D-dimer was elevated at 1159 ng/mL (reference <386). Electrolytes, renal function, total bilirubin, albumin, and coagulation parameters (prothrombin time 13.6 s, INR 1.24, activated partial thromboplastin time 32.3 s, fibrinogen 297 mg/dL) were within normal limits or near-normal ranges.
2.3.2. Molecular and Serological Confirmation
On hospital day 1, an oropharyngeal swab was collected, placed in viral universal transport medium, and transported at 2–8 °C under cold-chain conditions to the State Public Health Laboratory. Magnetic bead–based automated nucleic acid extraction was performed, followed by InDRE-validated real-time RT-PCR targeting the measles virus
N gene and the rubella virus
E1 gene, with human RNase P as an internal control [
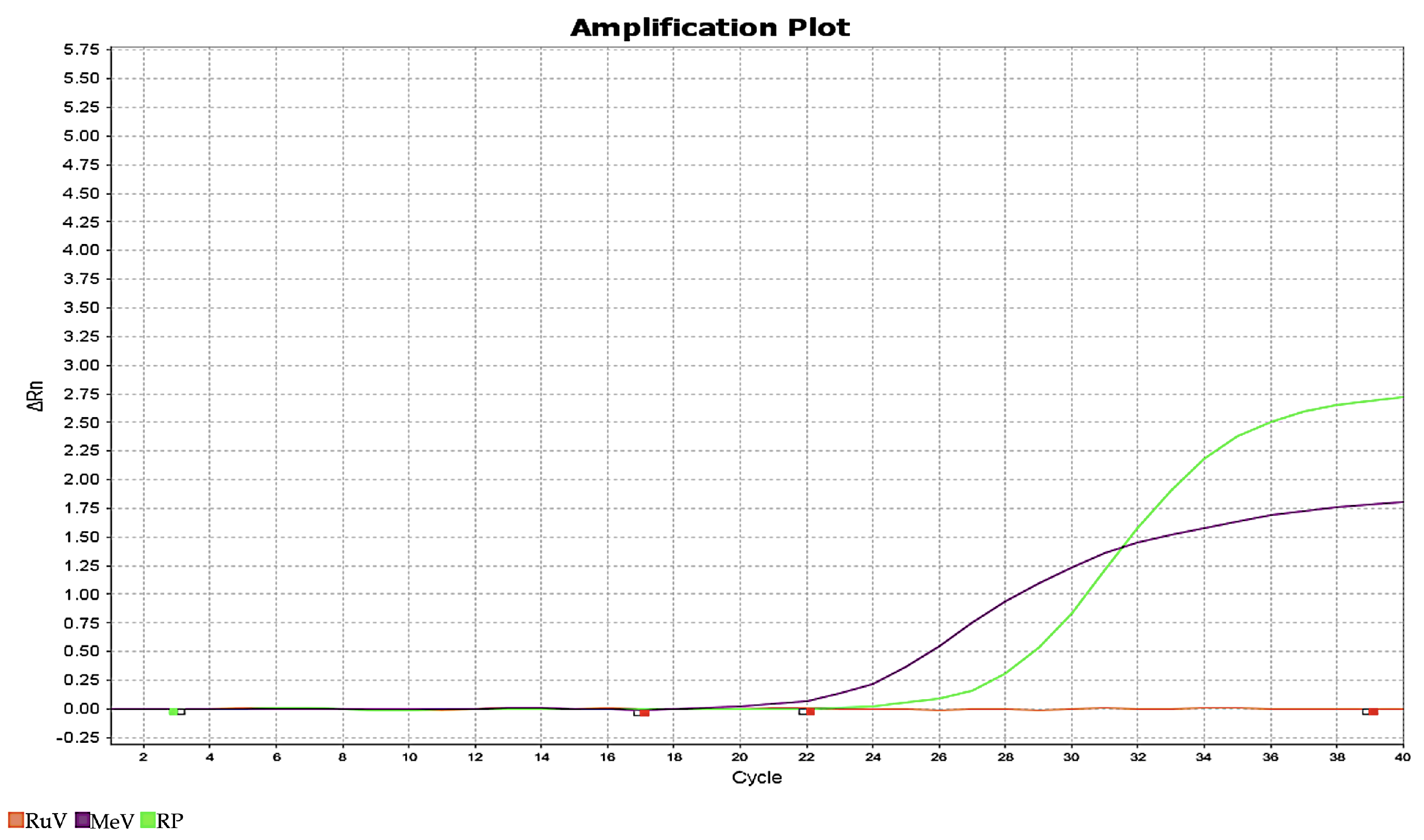
12]. Measles virus RNA was detected with a quantification cycle (Cq) value of 24 (positive: ≤40); rubella virus RNA was not detected; and RNase P amplified at Cq 28, confirming specimen adequacy (Figure 2). Serum immunoglobulin M (IgM) enzyme-linked immunosorbent assay (ELISA) was positive with a ratio of 5.401 (positive: >1.100). Rubella virus infection was excluded by both RT-PCR and rubella IgM ELISA.
2.3.3. Imaging
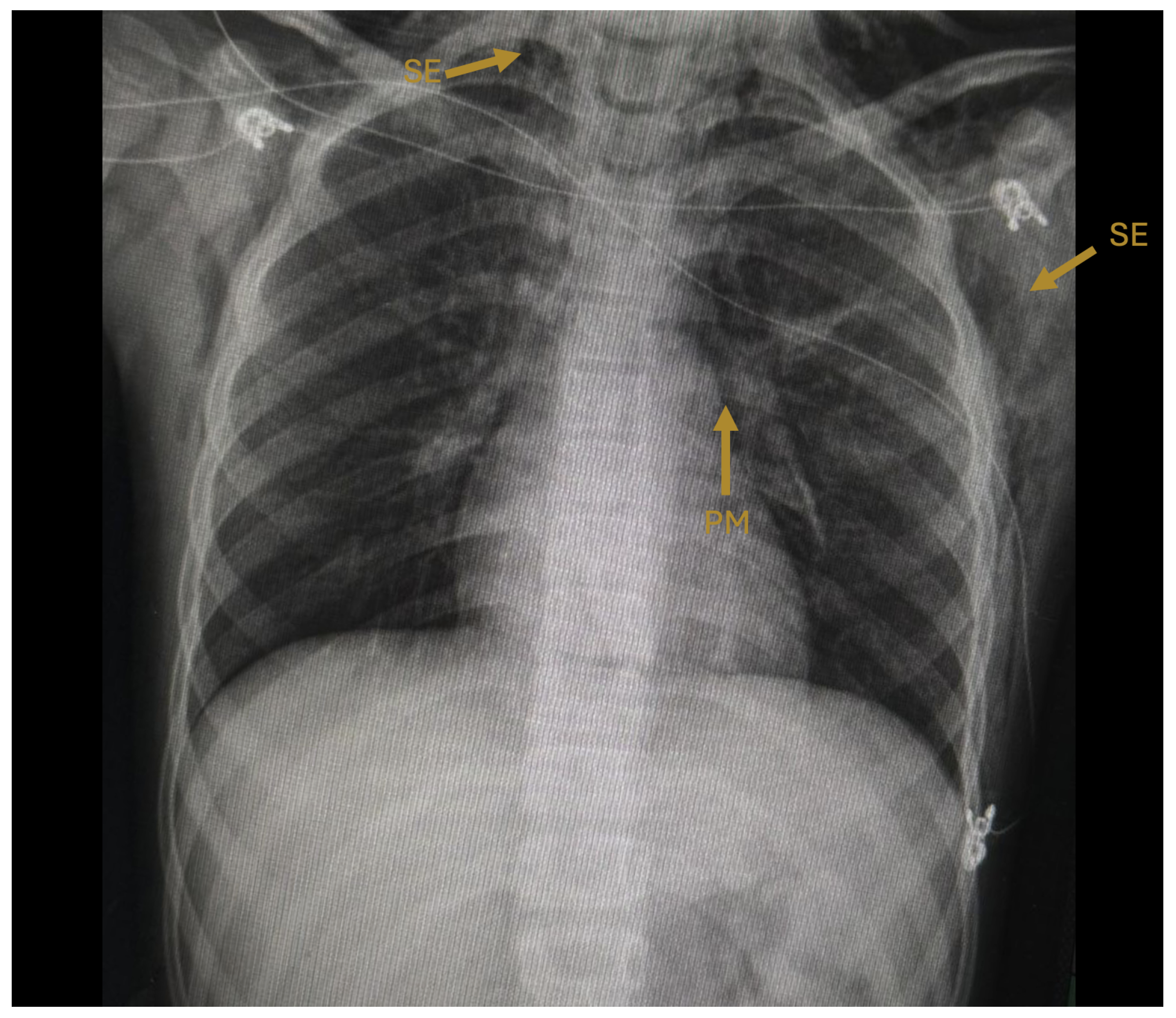
The initial chest radiograph demonstrated pneumomediastinum and extensive subcutaneous emphysema extending from the skull base to the mid-thorax, with a central trachea and normal pulmonary parenchyma (
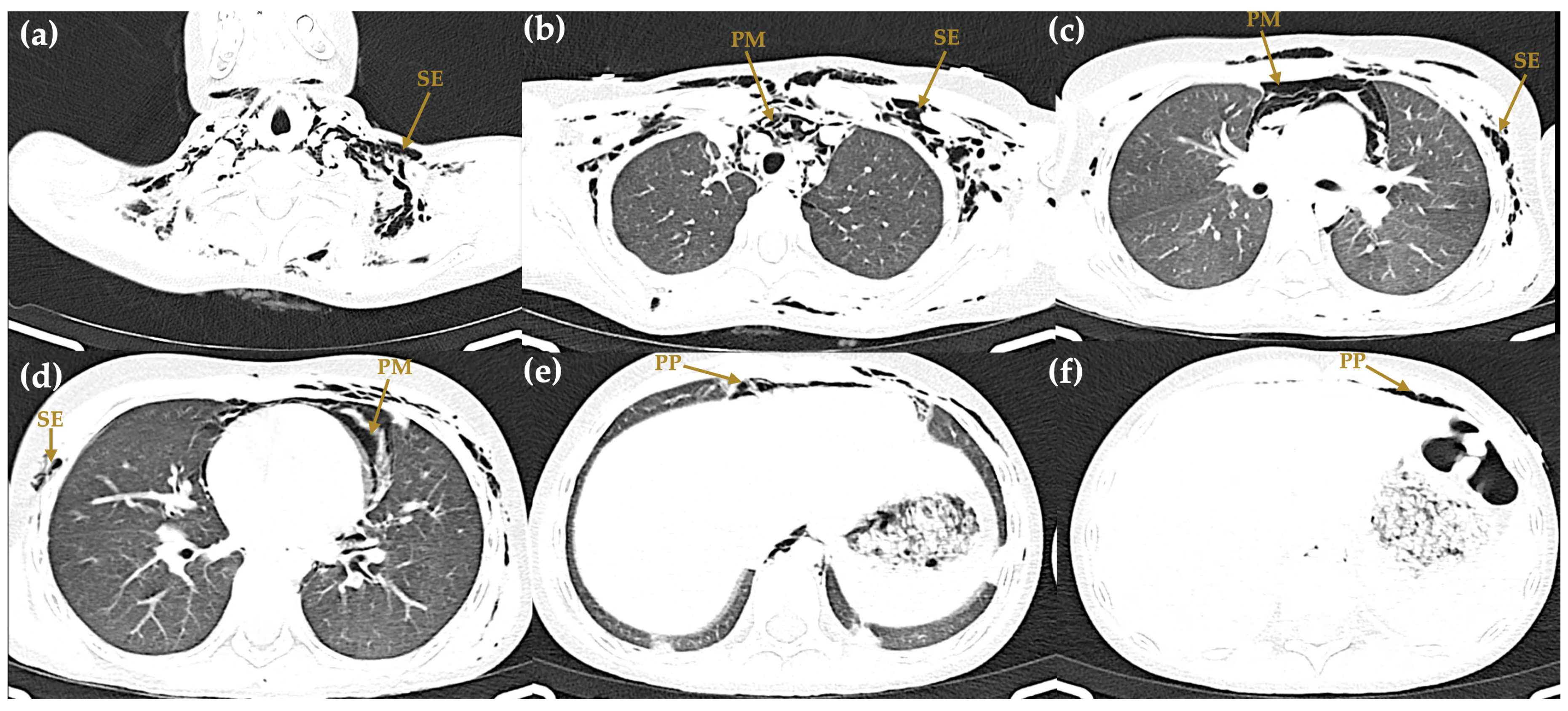
Figure 1). On hospital day 2, computed tomography (CT) of the chest confirmed pneumomediastinum with air dissecting along the esophagus, trachea, and pericardium, as well as a left-sided pneumothorax, pneumoperitoneum, and subcutaneous emphysema extending through the cervical, shoulder, and pectoral musculature (
Figure 4).
Figure 1.
Initial posteroanterior chest radiograph (hospital day 1). Arrows indicate pneumomediastinum (PM), visible as a radiolucent halo outlining the cardiac silhouette, and subcutaneous emphysema (SE) dissecting through the cervical and pectoral soft tissues bilaterally. The trachea is midline and pulmonary parenchyma appears without consolidation.
Figure 1.
Initial posteroanterior chest radiograph (hospital day 1). Arrows indicate pneumomediastinum (PM), visible as a radiolucent halo outlining the cardiac silhouette, and subcutaneous emphysema (SE) dissecting through the cervical and pectoral soft tissues bilaterally. The trachea is midline and pulmonary parenchyma appears without consolidation.
Figure 2.
Real-time RT-PCR amplification plot (Rn versus cycle number). The purple curve (MeV) corresponds to the measles virus N gene (Cq = 24), the green curve (RP) represents the human RNase P internal control (Cq = 28), and the orange curve (RuV) shows the rubella virus E1 gene target (not detected). The positive amplification of the measles N gene confirms active viral infection, while RNase P amplification validates specimen adequacy.
Figure 2.
Real-time RT-PCR amplification plot (Rn versus cycle number). The purple curve (MeV) corresponds to the measles virus N gene (Cq = 24), the green curve (RP) represents the human RNase P internal control (Cq = 28), and the orange curve (RuV) shows the rubella virus E1 gene target (not detected). The positive amplification of the measles N gene confirms active viral infection, while RNase P amplification validates specimen adequacy.
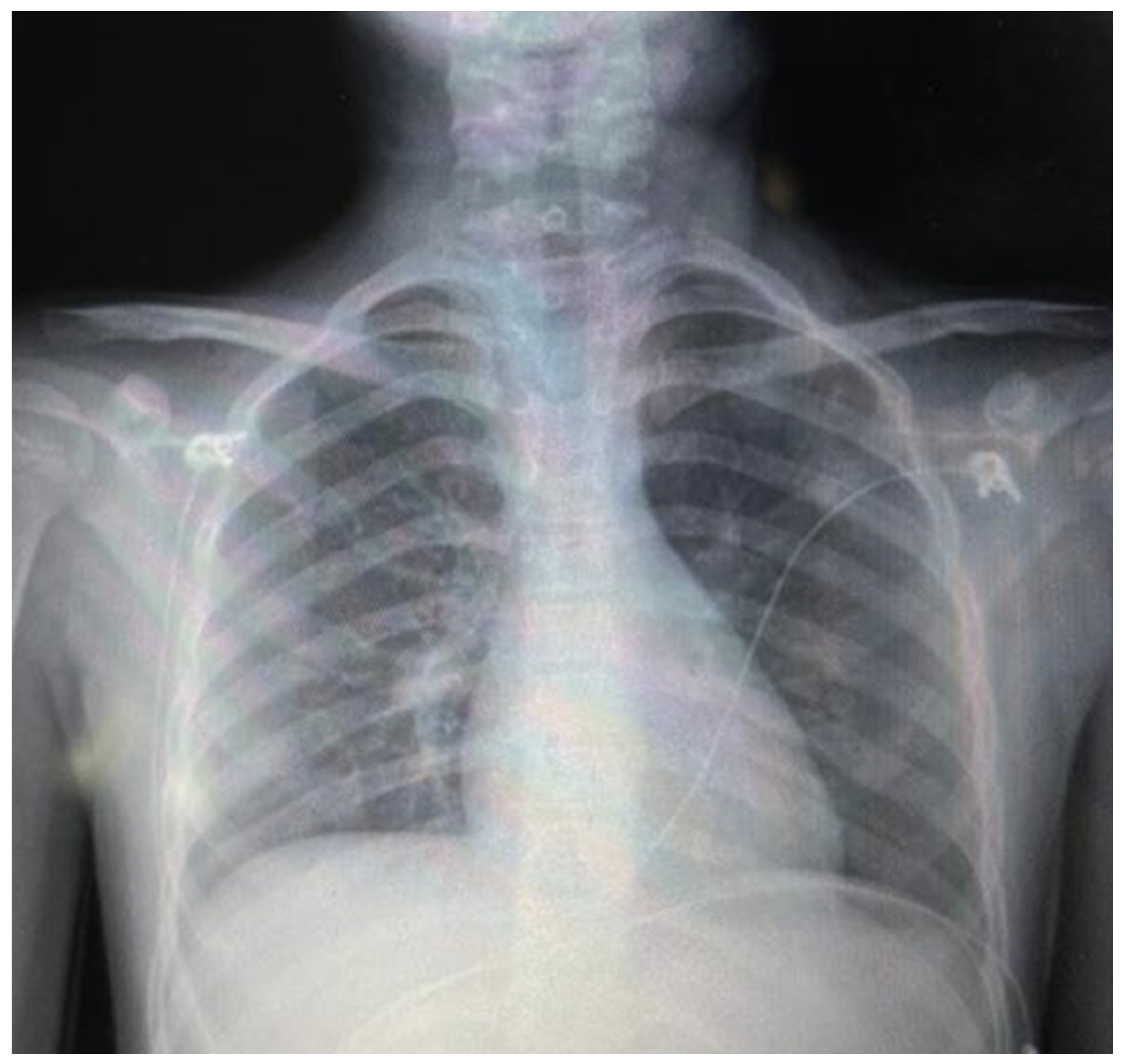
Figure 3.
Follow-up posteroanterior chest radiograph (hospital day 3, pre-discharge) demonstrating near-complete resolution of subcutaneous emphysema and pneumomediastinum compared with the admission study (
Figure 1). The cardiac and mediastinal contours are now clearly delineated without surrounding radiolucent halos.
Figure 3.
Follow-up posteroanterior chest radiograph (hospital day 3, pre-discharge) demonstrating near-complete resolution of subcutaneous emphysema and pneumomediastinum compared with the admission study (
Figure 1). The cardiac and mediastinal contours are now clearly delineated without surrounding radiolucent halos.
2.3.4. Endoscopic Evaluation
Flexible bronchoscopy performed on hospital day 1 under sedation revealed no tracheobronchial lesions or mucosal abnormalities. Upper gastrointestinal endoscopy, performed subsequently to exclude esophageal perforation, was similarly normal.
2.4. Management and Clinical Course
Diagnosis of air leak syndrome (pneumomediastinum, subcutaneous emphysema, left pneumothorax, and pneumoperitoneum) secondary to measles infection was established after exclusion of structural airway and esophageal injury.
Conservative management was instituted with supplemental oxygen (initially via nasal cannula at 1 L/min, subsequently escalated to a non-rebreather mask at 10 L/min on recommendation from pediatric surgery to promote nitrogen washout and reabsorption of extrapulmonary air). Intravenous crystalloid maintenance fluids were administered initially and discontinued once oral intake was adequate. No antibiotics or antiviral agents were administered throughout hospitalization. Pediatric surgery and pediatric infectious disease services were consulted. Multiple reassessments by pediatric surgery confirmed no indication for surgical intervention.
The patient remained hemodynamically stable and afebrile throughout hospitalization. Supplemental oxygen was gradually weaned. A follow-up chest radiograph on hospital day 3 demonstrated near-complete resolution of subcutaneous emphysema and pneumomediastinum compared with admission (
Figure 3). The patient was discharged on hospital day 3 (February 7, 2026), asymptomatic, tolerating a complete oral diet, and maintaining oxygen saturation above 94% on room air.
Figure 4.
Axial computed tomography of the chest and abdomen (hospital day 2). (a) Cervical level (lung window) showing subcutaneous emphysema (SE) dissecting through the deep cervical soft tissues. (b) Upper thorax (lung window) demonstrating pneumomediastinum (PM) with air surrounding the great vessels, and bilateral subcutaneous emphysema (SE) in the pectoral musculature. (c) Mid-thorax (lung window) confirming extensive pneumomediastinum (PM) and subcutaneous emphysema (SE). (d) Lower thorax (lung window) showing persistent pneumomediastinum (PM) and subcutaneous emphysema (SE) at the level of the pulmonary hila. (e) Upper abdomen (soft-tissue window) revealing pneumoperitoneum (PP) as free air anterior to the liver. (f) Upper abdomen (soft-tissue window) with additional evidence of pneumoperitoneum (PP) and residual air dissection.
Figure 4.
Axial computed tomography of the chest and abdomen (hospital day 2). (a) Cervical level (lung window) showing subcutaneous emphysema (SE) dissecting through the deep cervical soft tissues. (b) Upper thorax (lung window) demonstrating pneumomediastinum (PM) with air surrounding the great vessels, and bilateral subcutaneous emphysema (SE) in the pectoral musculature. (c) Mid-thorax (lung window) confirming extensive pneumomediastinum (PM) and subcutaneous emphysema (SE). (d) Lower thorax (lung window) showing persistent pneumomediastinum (PM) and subcutaneous emphysema (SE) at the level of the pulmonary hila. (e) Upper abdomen (soft-tissue window) revealing pneumoperitoneum (PP) as free air anterior to the liver. (f) Upper abdomen (soft-tissue window) with additional evidence of pneumoperitoneum (PP) and residual air dissection.
3. Discussion
This case illustrates an extensive air leak syndrome—encompassing pneumomediastinum, subcutaneous emphysema, left pneumothorax, and pneumoperitoneum—as a complication of RT-PCR-confirmed measles in a pediatric patient. Several aspects of this presentation deserve discussion.
3.1. Pathophysiology: The Macklin Effect in Measles
The pathogenesis of spontaneous pneumomediastinum is best explained by the Macklin effect [
5,
23]. This mechanism involves alveolar rupture due to a sudden increase in the intraalveolar pressure gradient—typically provoked by vigorous coughing, bronchospasm, or Valsalva maneuvers—followed by centripetal air dissection along bronchovascular sheaths toward the mediastinum [
6,
24]. From the mediastinum, air may further dissect into the subcutaneous tissues of the neck and chest, the pleural space (producing pneumothorax), and the retroperitoneal and peritoneal spaces (producing pneumoperitoneum) [
6,
11,
24,
25].
In measles, the combination of intense, persistent cough and diffuse bronchioloalveolar inflammation creates favorable conditions for alveolar rupture [
7,
16,
28]. The inflammatory process weakens alveolar wall integrity while paroxysmal coughing generates the pressure gradient necessary for rupture. The presence of all four air leak compartments (mediastinum, subcutaneous tissue, pleural space, and peritoneal cavity) in our patient reflects the extensive and contiguous nature of air dissection along fascial planes.
3.2. Epidemiological Context
This case occurred during the ongoing global resurgence of measles, attributed to declining vaccination coverage [
1,
14,
32]. The epidemiological investigation identified an unvaccinated child with a plausible exposure (travel to a metropolitan area during an active outbreak period and contact with a symptomatic household member). The absence of any documented or reported measles–rubella vaccination in a school-age child underscores the consequences of suboptimal immunization coverage. Indeed, measles virus infection has been shown to diminish preexisting antibodies against other pathogens, amplifying the individual and population-level consequences of each missed vaccination opportunity [
17].
Data from previous outbreaks suggest that subcutaneous emphysema in measles may not be as rare as traditionally believed. Kaba et al. reported frequencies ranging from 0.59% to 3.8% in outbreak settings, reaching 15.4% among children under five years in a refugee camp [
4]. These figures suggest that air leak complications may be underdiagnosed, particularly in resource-limited settings where imaging is not routinely available.
3.3. Hepatic Involvement
A notable finding in our patient was the significant elevation of hepatic enzymes (GGT 3.6× ULN, ALT 1.4× ULN, AST 1.3× ULN, ALP 1.6× ULN) and LDH (3.9× ULN) without hyperbilirubinemia. Measles-associated hepatitis is a recognized but underreported complication, documented in up to 80% of measles cases when systematically assessed [
2,
3,
18,
19]. The hepatic involvement is attributed to direct viral cytopathic effects and immune-mediated injury. In our patient, preserved synthetic function (normal albumin, near-normal coagulation parameters) suggests a transient, self-limited hepatic insult consistent with measles hepatitis rather than severe hepatocellular injury.
The elevated D-dimer (1159 ng/mL, 3× ULN) likely reflects the systemic inflammatory state associated with measles and the tissue damage from air leak rather than a thromboembolic event, as the patient showed no clinical signs of venous thromboembolism. Ohnishi and Kato demonstrated that intravascular coagulation activation and fibrinolysis occur during the acute febrile phase of measles, providing a pathophysiological basis for D-dimer elevation in this context [
20].
3.4. Diagnostic Approach
The diagnostic strategy in our case followed a systematic approach to exclude potentially life-threatening etiologies. The differential diagnosis for pneumomediastinum in a pediatric patient includes tracheobronchial rupture, esophageal perforation (Boerhaave syndrome), barotrauma, and foreign body aspiration [
11,
21]. Flexible bronchoscopy and upper gastrointestinal endoscopy were performed to rule out structural airway and esophageal injury; both were normal, supporting the diagnosis of spontaneous air leak.
Of note, the pneumomediastinum was identified on the initial chest radiograph at admission, before any clinical deterioration, consistent with the observation by Bloch and Vardy that this complication may be radiographically apparent before it is clinically suspected [
7]. This finding underscores the importance of careful radiographic interpretation in patients presenting with measles and respiratory symptoms.
3.5. Conservative Management
Our management approach—high-flow supplemental oxygen therapy and close surveillance—aligns with current evidence supporting conservative treatment of spontaneous pneumomediastinum in hemodynamically stable patients [
9,
10,
22]. High-flow oxygen promotes nitrogen washout from extrapulmonary air collections, accelerating reabsorption [
11,
31]. In our patient, the escalation from nasal cannula (1 L/min) to a non-rebreather mask (10 L/min) was recommended by the pediatric surgery team specifically for this purpose.
No antibiotics or antiviral agents were administered during hospitalization. The patient remained afebrile throughout, and the absence of leukocytosis or radiographic consolidation argued against bacterial superinfection. Resolution was achieved within three hospital days, consistent with the benign, self-limited course described in pediatric series of spontaneous pneumomediastinum [
8,
9,
21,
22].
Takada et al. have argued against routine invasive evaluation in clinically stable patients with spontaneous pneumomediastinum, noting that many patients undergo unnecessary procedures without altering management [
9]. In our case, the extensive subcutaneous emphysema and the context of an active infectious process warranted endoscopic evaluation to exclude structural injury. Once this was excluded, conservative management was both justified and successful.
3.6. Comparison with Published Cases
Our case shares similarities with the recent report by Kaba et al. [
4], who described a 12-year-old unvaccinated male with measles-associated pneumomediastinum and subcutaneous emphysema, confirmed by IgM and PCR, managed conservatively with resolution over 13 days. Our case adds several elements: (1) documentation of pneumoperitoneum and left pneumothorax in addition to pneumomediastinum, illustrating the full spectrum of air dissection; (2) normal bronchoscopy and upper gastrointestinal endoscopy formally excluding structural injury; (3) characterization of hepatic enzyme elevations as a concurrent finding; and (4) a shorter hospital course (3 days vs. PICU admission with respiratory support).
Mastroianni et al. recently described a 19-year-old unvaccinated female with pneumomediastinum, subcutaneous emphysema, pneumopericardium, and pneumothorax complicating measles; their literature review found no previous reports with all four air leak manifestations combined [
29]. Our case adds pneumoperitoneum as a fifth compartment, potentially representing the most extensive air leak syndrome documented in measles to date. Janmohamed et al. reported pneumomediastinum complicating measles in a vaccinated adult, demonstrating that this complication may occur regardless of prior vaccination status [
30].
The historical cases described by Bloch and Vardy [
7] in 1968, the pediatric series by McSweeney and Stempel [
8] in 1973, and the Nigerian series by Odita and Akamaguna [
26] in 1984 established the association between measles and air leak syndrome. Moons and Thallinger reported a high incidence of subcutaneous emphysema during a measles outbreak in a Somali refugee camp, suggesting this complication may be more common than previously recognized in epidemic settings [
27]. Piastra et al. documented measles-induced air leak and acute respiratory distress syndrome in the pediatric intensive care unit during the 2006–2007 Rome outbreak [
28]. Our case updates this association under contemporary diagnostic standards, including molecular confirmation by RT-PCR, CT characterization, and endoscopic exclusion of structural injury.
3.7. Limitations
As a single case report, this study cannot establish a causal relationship between measles and the air leak syndrome. However, the temporal association, the compatible pathophysiological mechanism, and the systematic exclusion of alternative etiologies strongly support this link. Additionally, serial hepatic enzyme measurements were not obtained to document the time course of hepatic recovery.
4. Conclusions
Spontaneous pneumomediastinum, subcutaneous emphysema, pneumothorax, and pneumoperitoneum may occur as complications of measles through the Macklin effect, driven by intense cough and bronchioloalveolar inflammation. Early radiographic recognition, systematic exclusion of structural airway and esophageal injury, and conservative management with high-flow oxygen therapy constitute a safe and effective approach in hemodynamically stable pediatric patients. In the context of the global resurgence of measles, clinicians should maintain awareness of these rare but clinically significant air leak complications to avoid unnecessary invasive interventions and optimize patient outcomes.
Author Contributions
Conceptualization, R.M.D.-N. and J.B.-R.; investigation, R.M.D.-N., R.G.C.-A., A.D.-P., K.A.R.-G. and B.D.G.-F.; data curation, R.M.D.-N., A.D.-P. and K.A.R.-G.; writing—original draft preparation, R.M.D.-N. and J.C.D.A.-J.; writing—review and editing, J.B.-R., J.C.D.A.-J., R.G.C.-A. and B.D.G.-F.; visualization, R.G.C.-A., A.D.-P. and K.A.R.-G.; supervision, J.B.-R. and J.C.D.A.-J. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Ethical review and approval were waived for this study (protocol CUTLAJO/DS/CEICS/06/26, approved on 22 February 2026), as it constitutes a case report based on routine clinical care without experimental intervention, in accordance with institutional guidelines and the Declaration of Helsinki.
Informed Consent Statement
Written informed consent was obtained from the patient’s legal guardian on 6 February 2026 for the publication of this case report and accompanying clinical images, ensuring confidentiality and anonymization of personal data.
Data Availability Statement
Data supporting the findings of this case report are contained within the article. Additional clinical data are not publicly available due to patient privacy restrictions.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| ALP |
Alkaline phosphatase |
| ALT |
Alanine aminotransferase |
| aPTT |
Activated partial thromboplastin time |
| AST |
Aspartate aminotransferase |
| CARE |
CAse REport |
| Cq |
Quantification cycle |
| CT |
Computed tomography |
| ELISA |
Enzyme-linked immunosorbent assay |
| GGT |
Gamma-glutamyl transferase |
| IgM |
Immunoglobulin M |
| INR |
International normalized ratio |
| LDH |
Lactate dehydrogenase |
| RT-PCR |
Reverse transcription polymerase chain reaction |
| ULN |
Upper limit of normal |
| WHO |
World Health Organization |
References
- World Health Organization. Measles. Available online: https://www.who.int/news-room/fact-sheets/detail/measles (accessed on 1 March 2026).
- Moss, W.J.; Griffin, D.E. Measles. Lancet 2012, 379, 153–164. [Google Scholar] [CrossRef] [PubMed]
- Perry, R.T.; Halsey, N.A. The Clinical Significance of Measles: A Review. J. Infect. Dis. 2004, 189 (Suppl. 1), S4–S16. [Google Scholar] [CrossRef]
- Kaba, Ö.; Karaman, K.; Öztürk, E. A Rare and Forgotten Complication of Measles: A Case Report of Subcutaneous Emphysema and Pneumomediastinum and Literature Review. Pediatr. Infect. Dis. J. 2026, 45, e109. [Google Scholar] [CrossRef]
- Macklin, M.T.; Macklin, C.C. Malignant Interstitial Emphysema of the Lungs and Mediastinum as an Important Occult Complication in Many Respiratory Diseases and Other Conditions: An Interpretation of the Clinical Literature in the Light of Laboratory Experiment. Medicine 1944, 23, 281–358. [Google Scholar] [CrossRef]
- Murayama, S.; Gibo, S. Spontaneous Pneumomediastinum and Macklin Effect: Overview and Appearance on Computed Tomography. World J. Radiol. 2014, 6, 850–854. [Google Scholar] [CrossRef]
- Bloch, A.; Vardy, P.A. Pneumomediastinum and Subcutaneous Emphysema in Measles. Clin. Pediatr. 1968, 7, 7–10. [Google Scholar] [CrossRef]
- McSweeney, W.J.; Stempel, D.A. Non-Iatrogenic Pneumomediastinum in Infancy and Childhood. Pediatr. Radiol. 1973, 1, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Takada, K.; Matsumoto, S.; Hiramatsu, T.; Kojima, E.; Watanabe, H.; Sizu, M.; Okachi, S.; Ninomiya, K. Management of Spontaneous Pneumomediastinum Based on Clinical Experience of 25 Cases. Respir. Med. 2008, 102, 1329–1334. [Google Scholar] [CrossRef] [PubMed]
- Iyer, V.N.; Joshi, A.Y.; Ryu, J.H. Spontaneous Pneumomediastinum: Analysis of 62 Consecutive Adult Patients. Mayo Clin. Proc. 2009, 84, 417–421. [Google Scholar] [CrossRef]
- Iteen, A.J.; Bianchi, W.; Sharman, T. Pneumomediastinum. [Updated 2023 May 1]. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2025. Available online: https://www.ncbi.nlm.nih.gov/books/NBK557440/ (accessed on 4 March 2026).
- Instituto de Diagnóstico y Referencia Epidemiológicos (InDRE); Dirección General de Epidemiología; Secretaría de Salud (Mexico). Lineamientos para la Vigilancia Epidemiológica de la Enfermedad Febril Exantemática por Laboratorio (LVL_EFE_4T). Secretaría de Salud: Ciudad de México, México, 2018. Available online: https://www.gob.mx/cms/uploads/attachment/file/487585/LVL_EFE_4T.pdf (accessed on 4 March 2026).
- Gagnier, J.J.; Kienle, G.; Altman, D.G.; Moher, D.; Sox, H.; Riley, D. The CARE Guidelines: Consensus-Based Clinical Case Reporting Guideline Development. J. Med. Case Rep. 2013, 7, 223. [Google Scholar] [CrossRef]
- Minta, A.A.; Ferrari, M.; Antoni, S.; Lambert, B.; Sayi, T.S.; Hsu, C.H.; Steulet, C.; Gacic-Dobo, M.; Rota, P.A.; Mulders, M.N.; et al. Progress Toward Measles Elimination — Worldwide, 2000–2023. MMWR Morb. Mortal. Wkly. Rep. 2024, 73, 1036–1042. [Google Scholar] [CrossRef]
- Moss, W.J. Measles. Lancet 2017, 390, 2490–2502. [Google Scholar] [CrossRef]
- Laksono, B.M.; de Vries, R.D.; McQuaid, S.; Duprex, W.P.; de Swart, R.L. Measles Virus Host Invasion and Pathogenesis. Viruses 2016, 8, 210. [Google Scholar] [CrossRef]
- Mina, M.J.; Kula, T.; Leng, Y.; Li, M.; de Vries, R.D.; Knip, M.; Siljander, H.; Rewers, M.; Choy, D.F.; Wilson, M.S.; et al. Measles Virus Infection Diminishes Preexisting Antibodies That Offer Protection from Other Pathogens. Science 2019, 366, 599–606. [Google Scholar] [CrossRef]
- Khatib, R.; Siddique, M.S.; Abbass, M. Measles Associated Hepatobiliary Disease: An Overview. Infection 1993, 21, 112–114. [Google Scholar] [CrossRef]
- Bîrluţiu, V.; Bîrluţiu, R.-M. Measles—Clinical and Biological Manifestations in Adult Patients, Including a Focus on the Hepatic Involvement: Results from a Single-Center Observational Cohort Study from Romania. J. Clin. Med. 2024, 13, 5535. [Google Scholar] [CrossRef] [PubMed]
- Ohnishi, K.; Kato, Y. Circulating D-Dimer and Thrombomodulin Levels in Acute Febrile Phase of Measles. J. Infect. 2002, 45, 180–183. [Google Scholar] [CrossRef] [PubMed]
- Chalumeau, M.; Le Clainche, L.; Sayeg, N.; Sannier, N.; Michel, J.L.; Marianowski, R.; Jouvet, P.; Scheinmann, P.; de Blic, J. Spontaneous Pneumomediastinum in Children. Pediatr. Pulmonol. 2001, 31, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Fitzwater, J.W.; Silva, N.N.; Knight, C.G.; Malvezzi, L.; Ramos-Irizarry, C.; Burnweit, C.A. Management of Spontaneous Pneumomediastinum in Children. J. Pediatr. Surg. 2015, 50, 983–986. [Google Scholar] [CrossRef]
- Wintermark, M.; Schnyder, P. The Macklin Effect: A Frequent Etiology for Pneumomediastinum in Severe Blunt Chest Trauma. Chest 2001, 120, 543–547. [Google Scholar] [CrossRef]
- Maunder, R.J.; Pierson, D.J.; Hudson, L.D. Subcutaneous and Mediastinal Emphysema: Pathophysiology, Diagnosis, and Management. Arch. Intern. Med. 1984, 144, 1447–1453. [Google Scholar] [CrossRef] [PubMed]
- Mularski, R.A.; Sippel, J.M.; Osborne, M.L. Pneumoperitoneum: A Review of Nonsurgical Causes. Crit. Care Med. 2000, 28, 2638–2644. [Google Scholar] [CrossRef] [PubMed]
- Odita, J.C.; Akamaguna, A.I. Mediastinal and Subcutaneous Emphysema Associated with Childhood Measles. Eur. J. Pediatr. 1984, 142, 33–36. [Google Scholar] [CrossRef]
- Moons, P.; Thallinger, M. High Incidence of Subcutaneous Emphysema in Children in a Somali Refugee Camp during Measles Outbreak. Pediatr. Infect. Dis. J. 2014, 33, 96–98. [Google Scholar] [CrossRef] [PubMed]
- Piastra, M.; Onesimo, R.; De Luca, D.; Lancella, L.; Marzano, L.; De Rosa, G.; Pietrini, D.; Valentini, P.; Conti, G. Measles-Induced Respiratory Distress, Air-Leak and ARDS. Eur. J. Clin. Microbiol. Infect. Dis. 2010, 29, 181–185. [Google Scholar] [CrossRef]
- Mastroianni, A.; Mauro, M.V.; Manfredi, R.; Greco, S. Spontaneous Pneumomediastinum Associated with Subcutaneous Emphysema, Pneumopericardium and Pneumothorax: A Rare Association Complicating Measles. Recenti Prog. Med. 2024, 115, 25–29. [Google Scholar] [CrossRef]
- Janmohamed, A.; Abbara, A.; Ghani, R.; Kinderlerer, A.; Sritharan, T.; Hatcher, J. Pneumomediastinum Complicating Adult-Onset Measles. Clin. Med. 2018, 18, 403–405. [Google Scholar] [CrossRef]
- Park, C.B.; Moon, M.H.; Jeon, H.W.; Cho, D.G.; Song, S.W.; Won, Y.D.; Kim, Y.H.; Kim, Y.D.; Jeong, S.C.; Kim, K.S.; et al. Does Oxygen Therapy Increase the Resolution Rate of Primary Spontaneous Pneumothorax? J. Thorac. Dis. 2017, 9, 5239–5243. [Google Scholar] [CrossRef]
- Pan American Health Organization/World Health Organization. Epidemiological Alert: Measles in the Americas Region, 28 February 2025. PAHO/WHO: Washington, DC, USA, 2025. Available online: https://www.paho.org/sites/default/files/2025-03/2025-feb-28-phe-epialert-measles-eng-final.pdf (accessed on 4 March 2026).
Table 1.
Laboratory findings on admission (hospital day 1). Abnormal values are highlighted in bold.
Table 1.
Laboratory findings on admission (hospital day 1). Abnormal values are highlighted in bold.
| Parameter |
Result |
Reference Range |
Units |
| Complete blood count |
| White blood cells |
4.94 |
4.60–10.20 |
×103/L |
| Neutrophils |
50.63 |
37.0–80.0 |
% |
| Lymphocytes |
23.14 |
10.0–50.0 |
% |
| Basophils
|
9.70 |
0.00–2.50 |
% |
| Hemoglobin |
14.31 |
12.20–18.10 |
g/dL |
| Platelets |
238.5 |
142.0–424.0 |
×103/L |
| Metabolic panel |
| Glucose |
84 |
60–125 |
mg/dL |
| Creatinine |
0.5 |
0.5–1.2 |
mg/dL |
| Sodium |
136 |
135–145 |
mmol/L |
| Potassium |
3.90 |
3.50–5.10 |
mmol/L |
| Hepatic function |
| GGT
|
109 |
8–30 |
IU/L |
| ALT
|
56 |
10–40 |
U/L |
| AST
|
64 |
10–50 |
IU/L |
| ALP
|
201 |
53–128 |
IU/L |
| LDH
|
734 |
91–190 |
U/L |
| Total bilirubin |
0.4 |
0.2–1.2 |
mg/dL |
| Albumin |
4.40 |
3.50–5.00 |
g/dL |
| Coagulation |
| Prothrombin time |
13.6 |
9–13 |
s |
| INR |
1.24 |
— |
— |
| aPTT |
32.3 |
25–35 |
s |
| Fibrinogen |
297 |
200–400 |
mg/dL |
| Inflammatory marker |
| D-dimer
|
1159 |
<386 |
ng/mL |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).