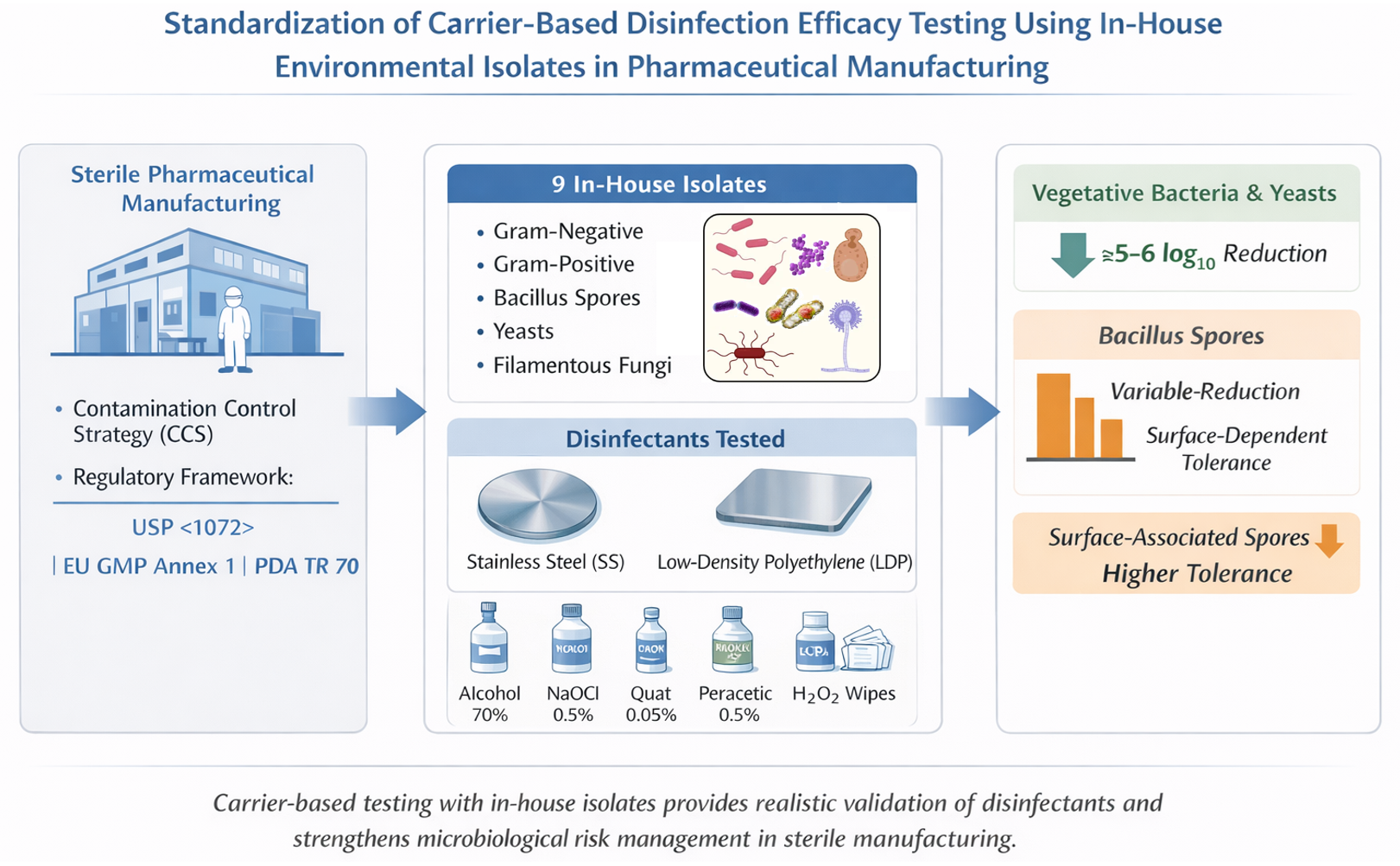
Inadequate surface sanitization represents a significant risk to sterility assurance and regulatory compliance. Therefore an effective cleaning and disinfection programme is a critical component of contamination control strategies in pharmaceutical facilities manufacturing sterile medicinal products. This study aimed to standardize a carrier-based methodology for evaluating the efficacy of disinfectants against in-house environmental isolates recovered from a pharmaceutical industry facility. Nine representative strains (six bacteria and three fungi), selected based on historical environmental monitoring data (2012–2022), were characterized using matrix-assisted laser desorption/ionization - time-of-flight / mass spectrometry (MALDI-TOF MS) and molecular sequencing (16S rRNA or D2 LSU rDNA). Disinfectant efficacy was assessed on stainless-steel and low-density polyethylene surfaces using NF T 72-281:2014 with adaptations, testing alcohol 70%, sodium hypochlorite 0.5%, quaternary ammonium 0.05%, peracetic acid 0.5%, and accelerated hydrogen peroxide wipes. All agents demonstrated ≥5 log₁₀ reductions against vegetative bacteria and fungi on both surfaces. However, variable sporicidal performance was observed, particularly for one Bacillus cereus group strain (B1342/15), which showed limited reduction on stainless-steel. These findings highlight inter-strain variability and the greater tolerance of surface-associated spores. The study reinforces the importance of carrier-based testing using in-house isolates to ensure realistic validation of disinfectants and to strengthen microbiological risk management within pharmaceutical contamination control strategies.