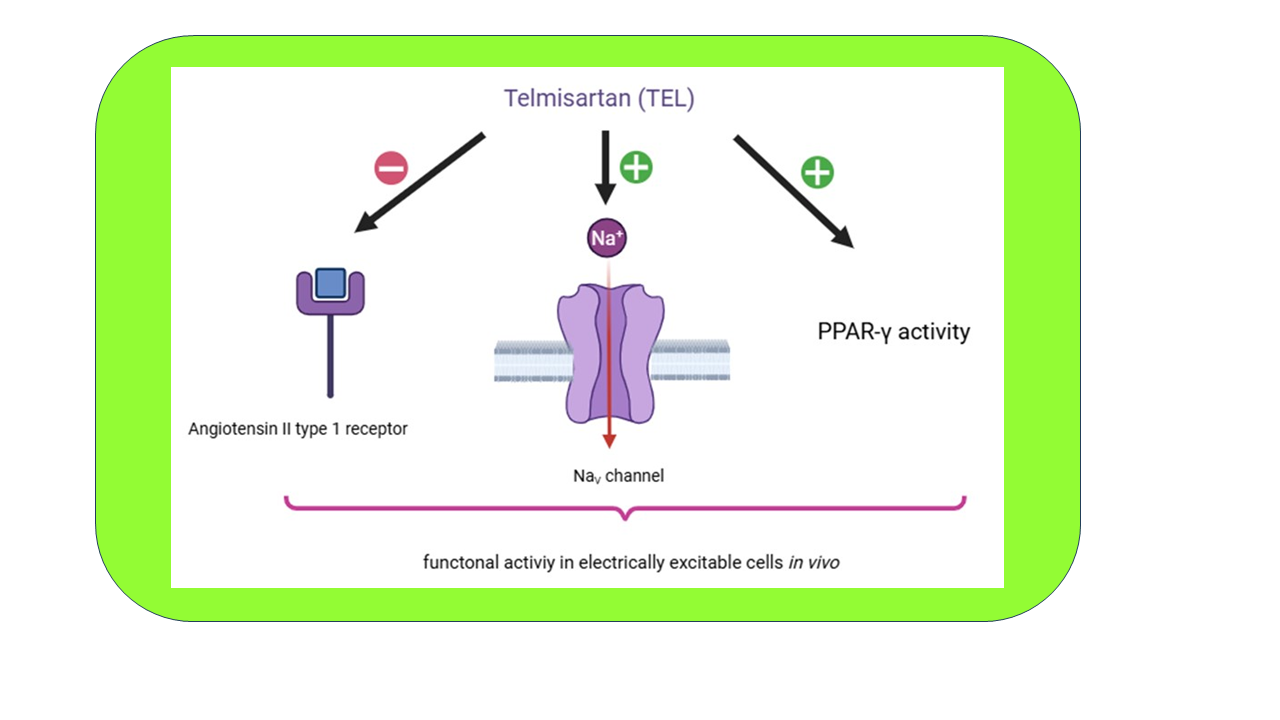
Telmisartan (TEL) is a non-peptide, orally administered antihypertensive agent primarily known as angiotensin II type 1 (AT1) blocker. In this review, we provide a detailed overview of how TEL modulates voltage-gated Na+ current (INa) and affects action potential (AP) firing behavior. TEL exerts differential stimulatory effects on the peak and late components of INa when subjected to brief depolarizing pulses across a range of cell types, such as mHippoE-14 hippocampal neuron, cultured dorsal root ganglion neurons, and HL-1 atrial cardiomyocytes. TEL can augment the inactivating (persistent) INa elicited by ascending long ramp pulse in mHippoE-14 cells. By using a parvalbumin-expressing interneuron-based modeled cell combined with bifurcation analysis, it is possible to predict how applied current influences subthreshold oscillations and the generation of somatic spiking in the presence of TEL. According to the Hodgkin-Huxley model, mimicking the action of TEL—characterized by an increased peak amplitude of INa and a slowed inactivation time course—leads to the emergence of periodic oscillations in membrane potential. Using a Markovian process, a separate model can also be mathematically constructed, showing that changes in certain rate constants can simulate the effect of TEL on INa in cardiac cells. The molecular docking prediction between TEL and the NaV1.7 channel was made by expected formation of hydrophobic interactions as well as hydrogen bonding. Beyond its antagonistic action on AT1 receptor and agonistic activation of peroxisome proliferator-activator-γ, the direct stimulation of INa may also contribute to its modulation of AP firing in various excitable cells. Current evidence supports TEL’s modulatory impact on NaV channel activity and cellular excitability, while also acknowledging that the mechanism—whether direct or indirect—remains under investigation.