Submitted:
04 March 2026
Posted:
06 March 2026
You are already at the latest version
Abstract
Keywords:
Introduction
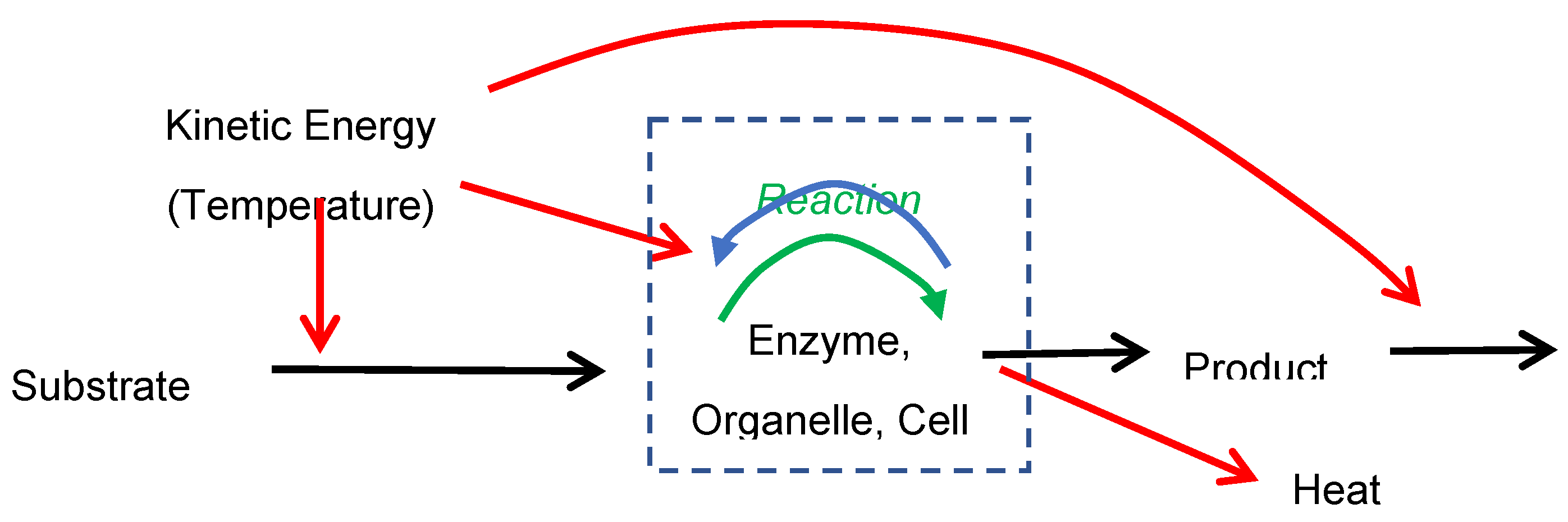
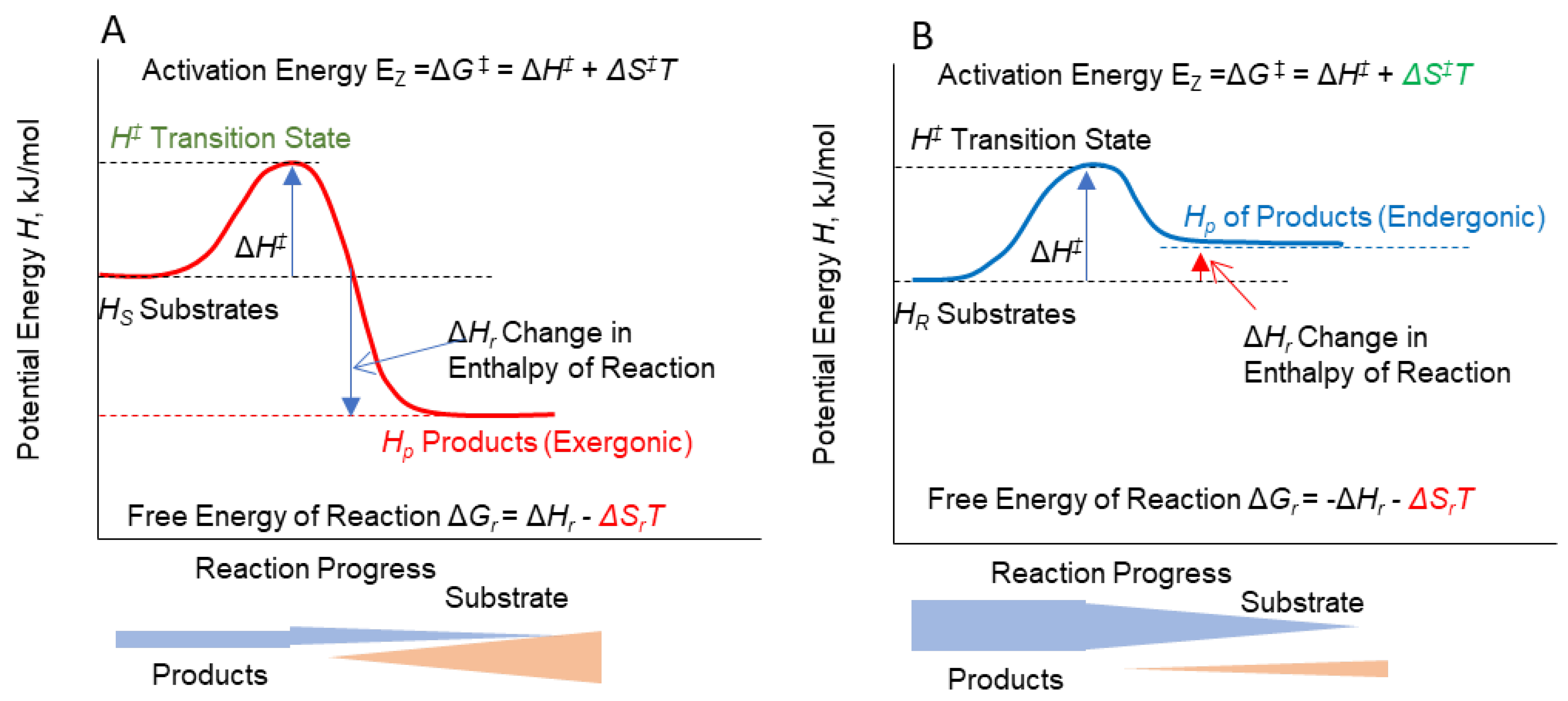
Reaction Rates
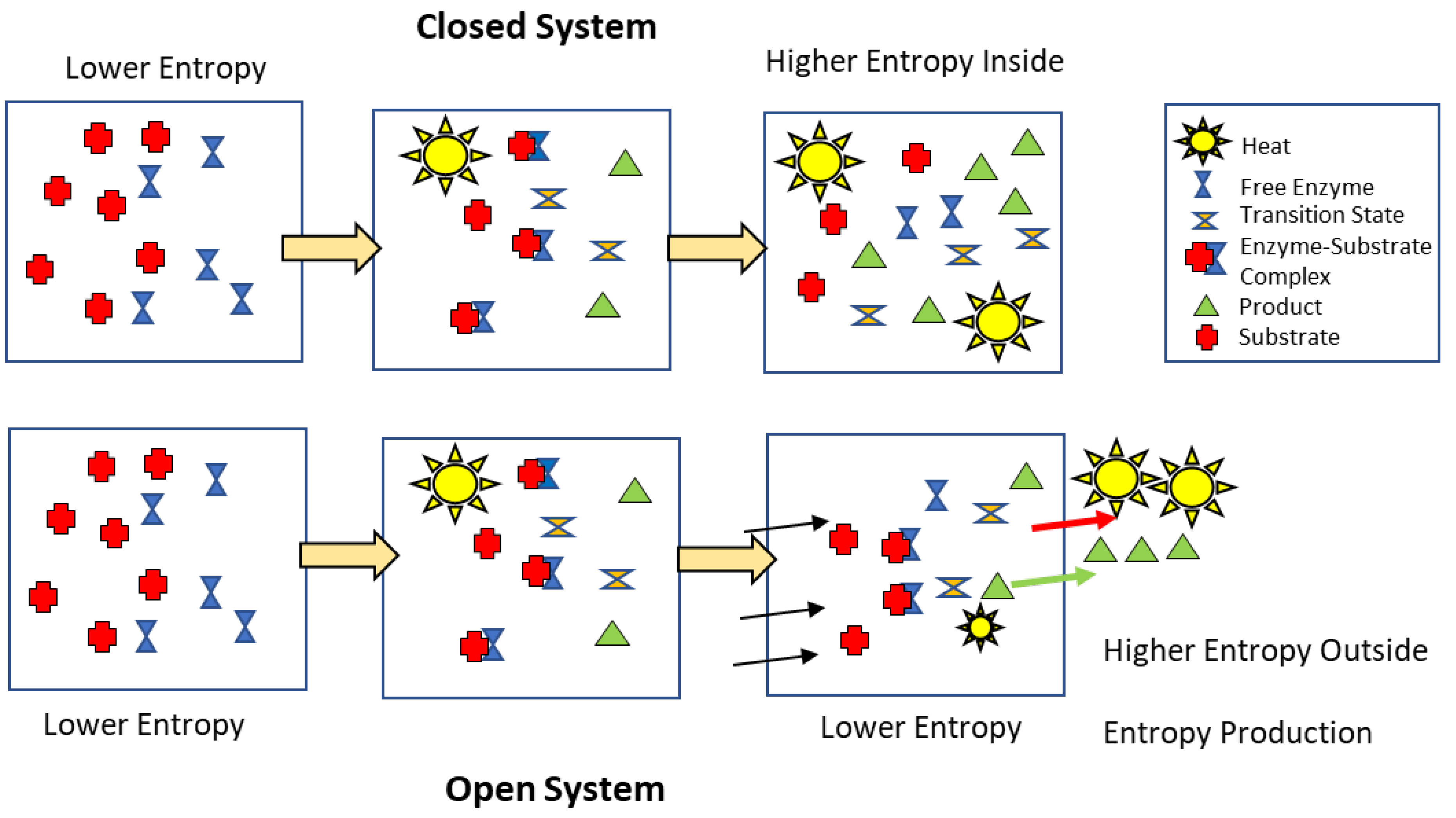
Entropy Production and Power
Entropy and Steady-State
Metabolic Goals and Testable Predictions
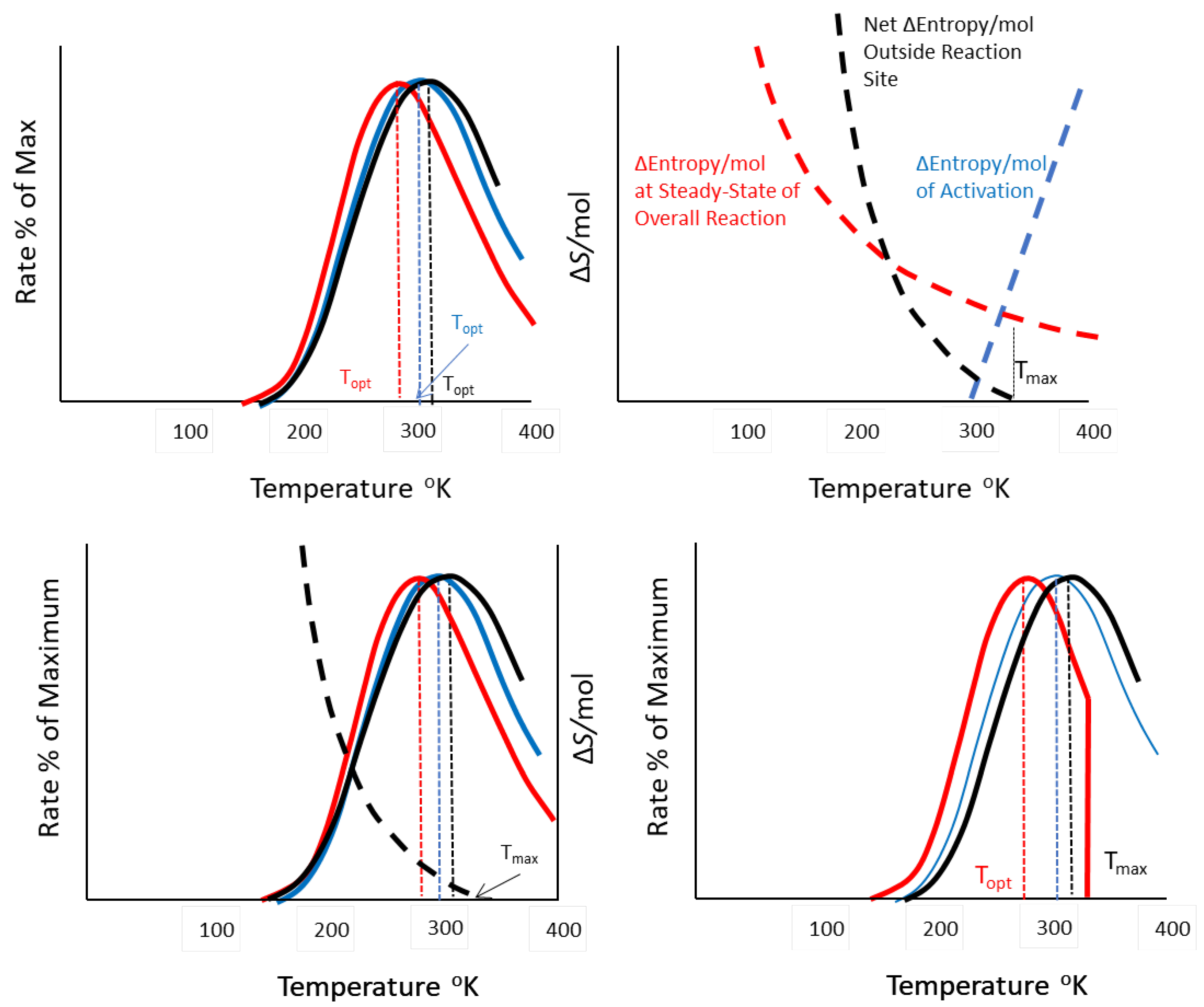
| Effect on Optimal Temperature | |||
| Parameter | Variable | Transition State | Entropy Production |
| Activation Energy | ΔHŧ | + | + |
| Change in Heat Capacity | ΔCP | - | NA |
| Reaction Constant | k | NA | - |
| Enzyme Concentration | Z | NA | - |
| Reaction Favorability | Keq | NA | + |
| Environmental Substrate | Ao | NA | + |
| Environmental Product | Po | NA | - |
| Diffusivity | D | NA | + |

Conclusions
References
- Alster, C.J.; van de Laar, A.; Goodrich, J.P.; et al. Quantifying thermal adaptation of soil microbial respiration. Nature Communications 2023, 14, 5459. [Google Scholar] [CrossRef] [PubMed]
- Åqvist, J.; van der Ent, F. Calculation of Heat Capacity Changes in Enzyme Catalysis and Ligand Binding. Journal of Chemical Theory and Computation 2022, 18, 6345–6353. [Google Scholar] [CrossRef] [PubMed]
- Arcus, V. L.; Mulholland, A. J. Temperature, dynamics, and enzyme-catalyzed reaction rates. Annual Review of Biophysics and Bioengineering 2020, 49, 163–180. [Google Scholar] [CrossRef] [PubMed]
- Arcus, V. L.; Prentice, E. J.; Hobbs, J. K.; Mulholland, A. J.; Van der Kamp, M. W.; Pudney, C. R.; Parker, E. J.; Schipper, L. A. On the temperature dependence of enzyme-catalyzed rates. Biochemistry 2016, 55, 1681–1688. [Google Scholar] [CrossRef]
- Arroyo, J. I.; Diez, B.; Kempes, C. P.; Marquet, P. A. A general theory for temperature dependence in biology. Proceedings of the National Academy of Sciences 2022, 112, e2119872119. [Google Scholar] [CrossRef]
- Brown, J. H.; Gillooly, J. F.; Allen, A. P.; Savage, V. M.; West, G. Toward a metabolic theory of ecology; Ecology, 2004; pp. 1771–1789. [Google Scholar]
- Chapin, F. S., III; Vitosek, P. M.; Van Cleve, K. The nature of nutrient limitation in plant communities. American Naturalist 1986, 127, 48–58. [Google Scholar] [CrossRef]
- Charnov, E. L. Trade-off invariant rules for evolutionary stable life histories. Nature 1997, 387, 393–394. [Google Scholar] [CrossRef]
- Claunch, N. M.; Nix, E.; Royal, A. E.; Burgos, L. P.; Corn, M.; DuBois, P. M.; Ivey, K. N.; King, E. C.; Rucker, K. A.; Shea, T. K.; Stepanek, J.; Vansdadia, S.; Taylor, E. N. Body size impacts critical thermal maximum measurements in lizards. Journal of Experimental Zoology Part A: Ecological and Integrative Physiology 2021, 335, 96–107. [Google Scholar] [CrossRef]
- Daniel, R. M.; Danson, M. J. A new understanding of how temperature affects the catalytic activity of enzymes. Trends in Biochemical Sciences 2010, 35, 584–591. [Google Scholar] [CrossRef]
- Daniel, R. M.; Dines, M.; Petach, H. H. The denaturation and degradation of stable enzymes at high temperatures. Biochem J 1996, 317 Pt 1, 1–11. [Google Scholar] [CrossRef]
- Dean, A. M.; Thornton, J. W. N. R. G. Mechanistic approaches to the study of evolution: The functional synthesis. Nature Reviews Genetics 2007, 8, 675–688. [Google Scholar] [CrossRef] [PubMed]
- Dell, A. I.; Pawar, S.; Savage, V. M. Systematic variation in the temperature dependence of physiological and ecological traits. Proceedings of the National Academy of Sciences 2011, 108, 10591–10596. [Google Scholar] [CrossRef]
- DeLong, J. P.; Bachman, G.; Gibert, J. P.; Luhring, T. M.; Montooth, K. L.; Neyer, A.; Reed, B. Habitat, latitude and body mass influence the temperature dependence of metabolic rate. Biol Lett 2018, 14. [Google Scholar] [CrossRef]
- DeLong, J. P.; Gibert, J. P.; Luhring, T. M.; Bachman, G.; Reed, B.; Neyer, A.; Montooth, K. L. The combined effects of reactant kinetics and enzyme stability explain the temperature dependence of metabolic rates. Ecology and Evolution 2017, 7, 3940–3950. [Google Scholar] [CrossRef] [PubMed]
- Dewar, R. Information theory explanation of the fluctuation theorem, maximum entropy production and self-organized criticality in non-equilibrium stationary states. Journal of Physics a-Mathematical and General 2003, 36, 631–641. [Google Scholar] [CrossRef]
- England, J.L. Statistical physics of self-replication. Journal of Chemical Physics 2013, 139(12), 121923. [Google Scholar] [CrossRef]
- Evans, M. G.; Polanyi, M. Some applications of the transition state method to the calculation of reaction velocities, especially in solution. Transactions of the Faraday Society 1935, 31, 875–894. [Google Scholar] [CrossRef]
- Fridley, J. D. Plant energetics and the synthesis of population and ecosystem ecology. Journal of Ecology 2017, 105, 95–110. [Google Scholar] [CrossRef]
- Fujita, H.; Kishimoto, A.; Matsumoto, K. Concentration and temperature dependence of diffusion coefficients for systems polymethyl acrylate and n-alkyl acetates. Transactions of the Faraday Society 1960, 56, 424–437. [Google Scholar] [CrossRef]
- Hall, C. A. S. Maximum Power: The ideas and applications of H.T.Odum; Colorado University Press: Boulder CO, 1995. [Google Scholar]
- Hall, C. A. S.; McWhirter, T. Maximum power in evolution, ecology and economics. Philos Trans A Math Phys Eng Sci 2023, 381, 20220290. [Google Scholar] [CrossRef] [PubMed]
- Harte, J. Maximum entropy and ecology; Oxford University Press: Oxford UK, 2011. [Google Scholar]
- Haynie, D. T. Biological thermodynamics; Cambridge University Press: Cambridge UK, 2008. [Google Scholar]
- Hill, T. L.; Simmons, R. M. Free energy levels and entropy production associated with biochemical kinetic diagrams. Proceedings of the National Academy of Sciences 1976, 73, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Hirshfield, M. F.; Tinkle, D. W. Natural selection and the evolution of reproductive effort. Proceedings of the National Academy of Sciences 1975, 72, 2227–2231. [Google Scholar] [CrossRef]
- Hobbs, J. K.; Jiao, W.; Easter, A. D.; Parker, E. J.; Schipper, L. A.; Arcus, V. L. Change in heat capacity for enzyme catalysis determines temperature dependence of enzyme catalyzed rates. ACS Chemical Biology 2013, 8, 2388–2393. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, S. E.; Nielsen, S. N.; Mejer, H. Emergy, envirion, exergy and ecological modeling. Ecological Modeling 1995, 77, 99–109. [Google Scholar] [CrossRef]
- Jorgensen, S. E.; Svirezhev, Y. M. Towards a thermodynamic theory for ecological systems; Elsevier: Amsterdam, 2004. [Google Scholar]
- Jorgensen, S. E.; Svirezhev, Y. M. Towards a thermodynamic theory for ecological systems; Elsevier: New York, 2007. [Google Scholar]
- Knapp, B. D.; Huang, K. C. The effects of temperature on cellular physiology. Annual Review of Biophysics 2022, 51, 499–526. [Google Scholar] [CrossRef]
- Kurzynski, M. The thermodynamic machinery of life; Springer: Berlin, 2006. [Google Scholar]
- Leiva, F. P.; Calosi, P.; Verberk, W. C. E. P. Scaling of thermal tolerance with body mass and genome size in ectotherms: a comparison between water- and air-breathers. Philosophical Transactions of the Royal Society B: Biological Sciences 2019, 374, 20190035. [Google Scholar] [CrossRef]
- Lotka, A. J. Contribution to the energetics of evolution. Proceedings of the National Academy of Sciences 1922, 8, 147–151. [Google Scholar] [CrossRef]
- Martyushev, L. M. Entropy and entropy production: old misconceptions and new breakthroughs. Entropy 2013, 15, 1152–1170. [Google Scholar] [CrossRef]
- Niven, R. K. Steady state of a dissipative flow-controlled system and the maximum entropy production principle. Physical Review E 2009, 80, 0211131–02111315. [Google Scholar] [CrossRef]
- Odum, H. T. Systems ecology: ecological and general systems; Wiley: New York, 1993. [Google Scholar]
- Pänke, O.; Rumberg, B. Energy and entropy balance of ATP synthesis. Biochimica et Biophysica Acta (BBA) - Bioenergetics 1997, 1322, 183–194. [Google Scholar] [CrossRef]
- Schoolfield, R. M.; Sharpe, P. J. H.; Magnuson, C. E. Non-linear regression of biological temperature-dependent rate models based on absolute reaction-rate theory. Journal of Theoretical Biology 1981, 88, 719–731. [Google Scholar] [CrossRef]
- Sharpe, P. J. H.; DeMichele, D. W. Reaction kinetics of poikilotherm development. Journal of Theoretical Biology 1977, 64, 649–670. [Google Scholar] [CrossRef]
- Tang, J.; Riley, W. J. A chemical kinetics theory for interpreting the non-monotonic temperature dependence of enzymatic reactions. Biogeosciences 2024, 21, 1061–1070. [Google Scholar] [CrossRef]
- Tateno, M.; Chapin, F. S., III. The logic of carbon and nitrogen interactions in terrestrial ecosystems. American Naturalist 1997, 149, 723–744. [Google Scholar] [CrossRef]
- West, J. B. History of respiratory gas exchange. Comprehensive Physiology 2011, 1, 1509–1523. [Google Scholar] [CrossRef]
- Wynne-Jones, W. F. K.; Eyring, H. The absolute rate of reactions in condensed phases. Journal of Chemical Physics 1935, 3, 492–502. [Google Scholar] [CrossRef]
| Variable | Name | Units | Source | Range of values |
| R | gas contant | kJ mol-1 oK-1 | standard constant | 0.00832 |
| T | temperature | oK | N/A | variable |
| ln(1/a) | 1/chemical activity | dimensionless | Ritchie (2018) | -4 to 12* |
| ∆GŦ | Gibbs energy of activation | kJ/mol | Fig. 2, Gillooly et al (2001) | -60 |
| ED | T sensitivity of diffusion | kJ/mol | Ritchie (2018) | -30 |
| k0 | reaction constant | time-1 | Wynne-Jones & Eyring (1935) | 1010 |
| T0 | reference T | oK | Commonly used | 298 |
| ∆S0Ŧ | Standardized molar entropy change of activation at T = 298oK | kJ mol-1 oK-1 | (Pänke and Rumberg 1997) for ATP hydrolysis | 11 |
| ΔS‡ | change in entropy of activation | kJ mol-1 oK-1 | Hobbs et al (2013) | 0-5 |
| ΔH‡ | change in enthalpy of activation | kJ/mol | Gillooly et al (2001) | -60 |
| ΔSr | change in entropy of overall reaction | kJ mol-1 oK-1 | Haynie (2008) | variable |
| ΔHr | change in enthalpy of overall reaction | kJ/mol | Haynie (2008) | variable |
| σ | entropy production | kJ oK-1 time-1 | N/A | variable |
| r | reaction rate | mol time-1 | N/A | variable |
| J | power | kJ time-1 | N/A | variable |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




