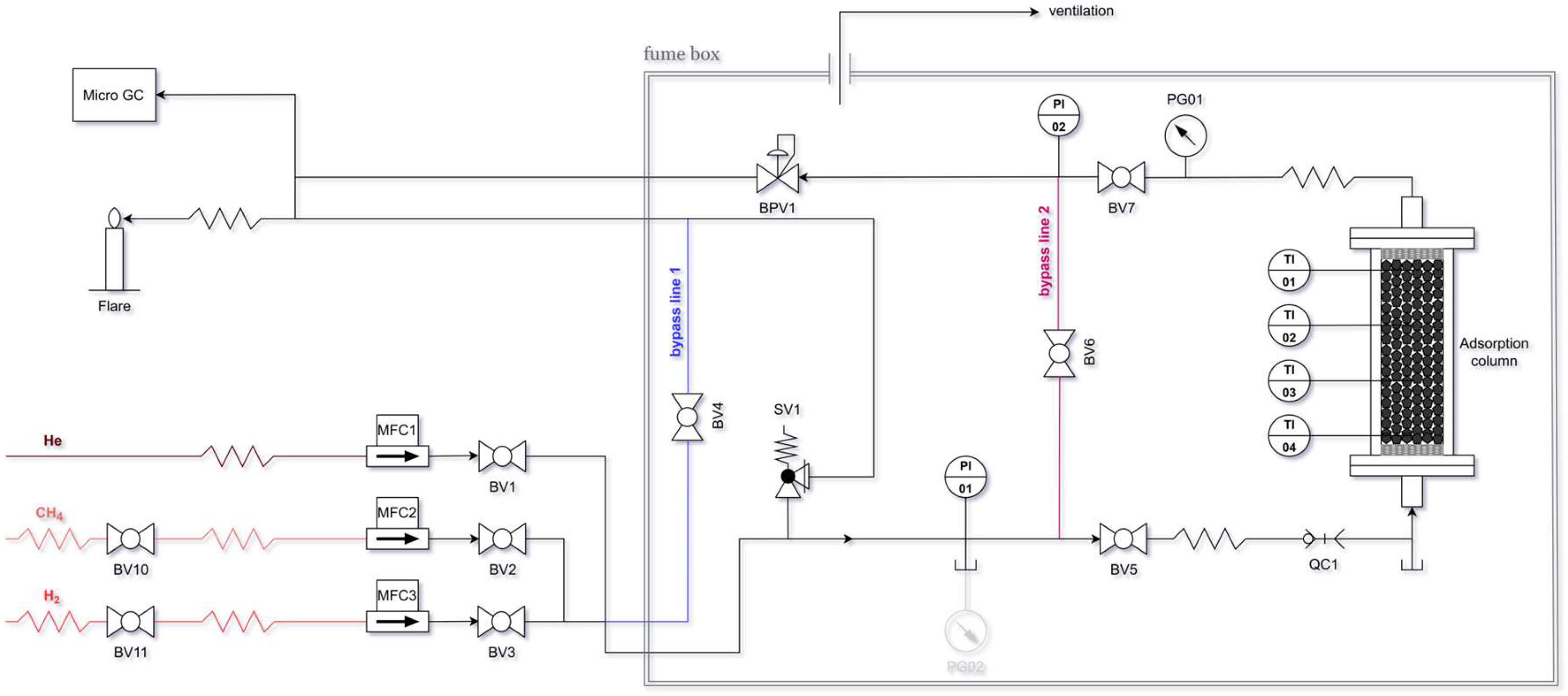
3.1. Textural Properties of the Activated Carbons
The textural characteristics of the activated carbons selected for the adsorption experiments from the previous study are summarized and presented in
Table 2 [
14]. Two biomass-derived activated carbons, prepared from pine and birch and activated at 800 °C using a KOH impregnation ratio of 3:1, were chosen based on their comparatively high surface areas and pore volumes. These materials are compared with a commercial activated carbon used as a benchmark.
The pine- and birch-derived activated carbons exhibit BET surface areas of 1416 and 1398 m2 g−1, respectively, both exceeding that of the commercial material (1023 m2 g−1). Higher total pore volumes are observed for the laboratory-prepared carbons, reaching 0.59 cm3 g−1 for pine and 0.58 cm3 g−1 for birch, compared to 0.44 cm3 g−1 for the commercial sample. The average pore diameters are similar across all three materials, ranging from approximately 0.83–0.85 nm, indicating a predominantly microporous pore structure.
These textural properties provide the structural basis for evaluating methane adsorption and breakthrough behavior under high-pressure PSA conditions, which are discussed in the following sections.
3.2. Methane Breakthrough Behavior Under Different Pressures
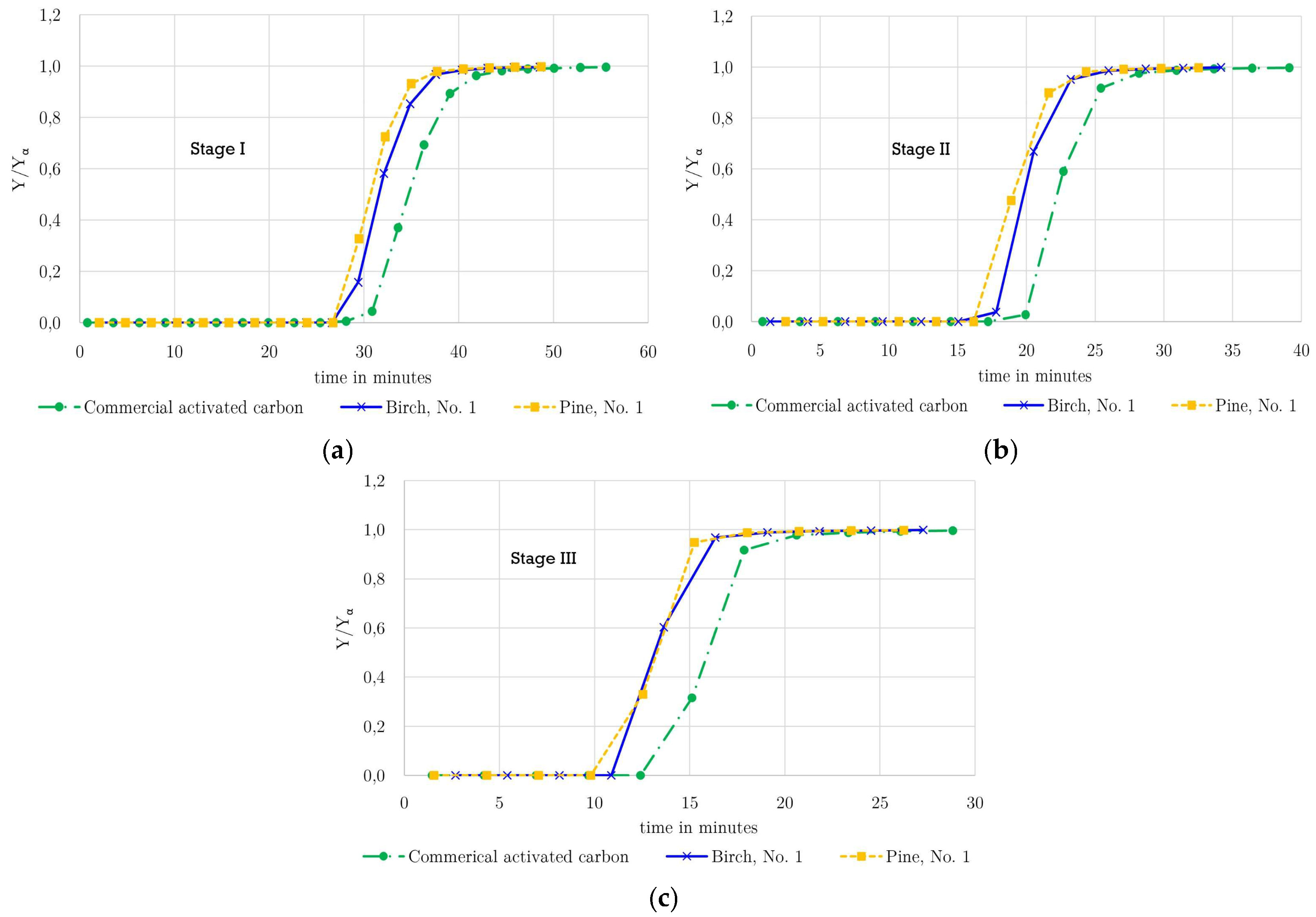
For each operating stage, the breakthrough curves obtained during adsorption are presented in
Figure 2 (a: Stage I, b: Stage II, and c: Stage III). For the birch- and pine-derived activated carbons, one representative breakthrough curve is shown and compared with the corresponding curve of the commercial adsorbent. Repeated experiments demonstrated reproducible results in the adsorption measurements, particularly with respect to the breakthrough profiles, which showed consistent behavior across all runs.
In addition, all data sets were recorded using the same sampling interval of 2.73 min, while their respective time stamps are asynchronous. When adsorption is initiated at a certain time, it is possible that the injection of the sample into the MicroGC’s column takes place within the next 2.73 minutes, which means that the detection by the GC is only a snapshot at a given time. During post-processing, this systematic offset between the actual start of adsorption and the first GC measurement was accounted for. Because of the measurement interval of ~ 2,73 min, the exact breakthrough times t_b cannot be determined, as the real breakthrough occurs somewhere between t_b + t_lag, where .
In total, 16 adsorption experiments were performed. For each pressure stage, one for the commercial adsorbent and two for the birch and pine samples, to ensure comparability and consistency of the data, particularly for the laboratory-prepared activated carbon. The breakthrough curves indicate that as operating pressures increase and methane concentrations in the feed gas decrease, the time it takes for breakthrough to occur also increases. This is expected because the partial pressure of the adsorbate in the feed gas decreases.
The results are presented in
Table 3,
Table 4 and
Table 5. For each sample, the mass of the adsorbent that fits inside the column was weighed, and the bulk density was estimated to occupy approximately 88% of the total reactor volume.
This estimation takes into account that the reactor is equipped with netting and fiberglass wool at both the entrance and exit to prevent any potential entry of activated carbon abrasion into the piping system. These additional components cover the entire inner column diameter of 2.2 cm and have a height of approximately 6 mm each. As a result, the “usable height” of the column is 8.8 cm, as described at the beginning of Chapter 2.3.
As shown in
Figure 2, a clear shift of the breakthrough curves toward longer times is observed with increasing operating pressure from Stage III (25 bar) to Stage I (50 bar). The breakthrough times listed in
Table 6 increase systematically with pressure for all three adsorbents, which is attributed to the decrease in methane concentration in the feed gas at higher operating pressures, resulting in a lower adsorbate partial pressure and delayed breakthrough. Such behavior is consistent with adsorption theory and previously reported studies on high-pressure gas adsorption.
A comparison among the three activated carbons reveals distinct differences in breakthrough performance. At Stage I (50 bar), both pine- and birch-derived activated carbons exhibit slightly longer breakthrough times than the commercial activated carbon, representing acceptable adsorption performance under high-pressure conditions. In contrast, at lower pressures (Stages II and III), the commercial activated carbon shows longer breakthrough times than the biomass-derived materials. This behavior can be partially explained by differences in bulk density. The bulk densities of the pine (0.297 g·cm−3) and birch (0.247 g·cm−3) activated carbons are nearly half that of the commercial activated carbon (0.538 g·cm−3), resulting in a lower mass of adsorbent per unit bed volume.
Among the laboratory-prepared adsorbents, pine-derived activated carbon shows consistently strong breakthrough performance across all pressure stages, which motivated its selection for extended cyclic stability testing and subsequent structural analysis by Raman spectroscopy in section 3.4.4. Despite the lower adsorbent mass of the pine-derived activated carbon in the column, a slightly longer breakthrough time is observed at Stage I, which is consistent with its higher gravimetric adsorption capacity. At Stages II and III, the pine-derived activated carbon exhibits shorter breakthrough times compared to the commercial activated carbon. This does not indicate lower adsorption capacity but rather reflects the combined effects of lower bulk density, reduced adsorbent mass, and earlier bed saturation. It should be noted that, due to the discrete sampling interval of the MicroGC, the breakthrough times are associated with higher uncertainty.
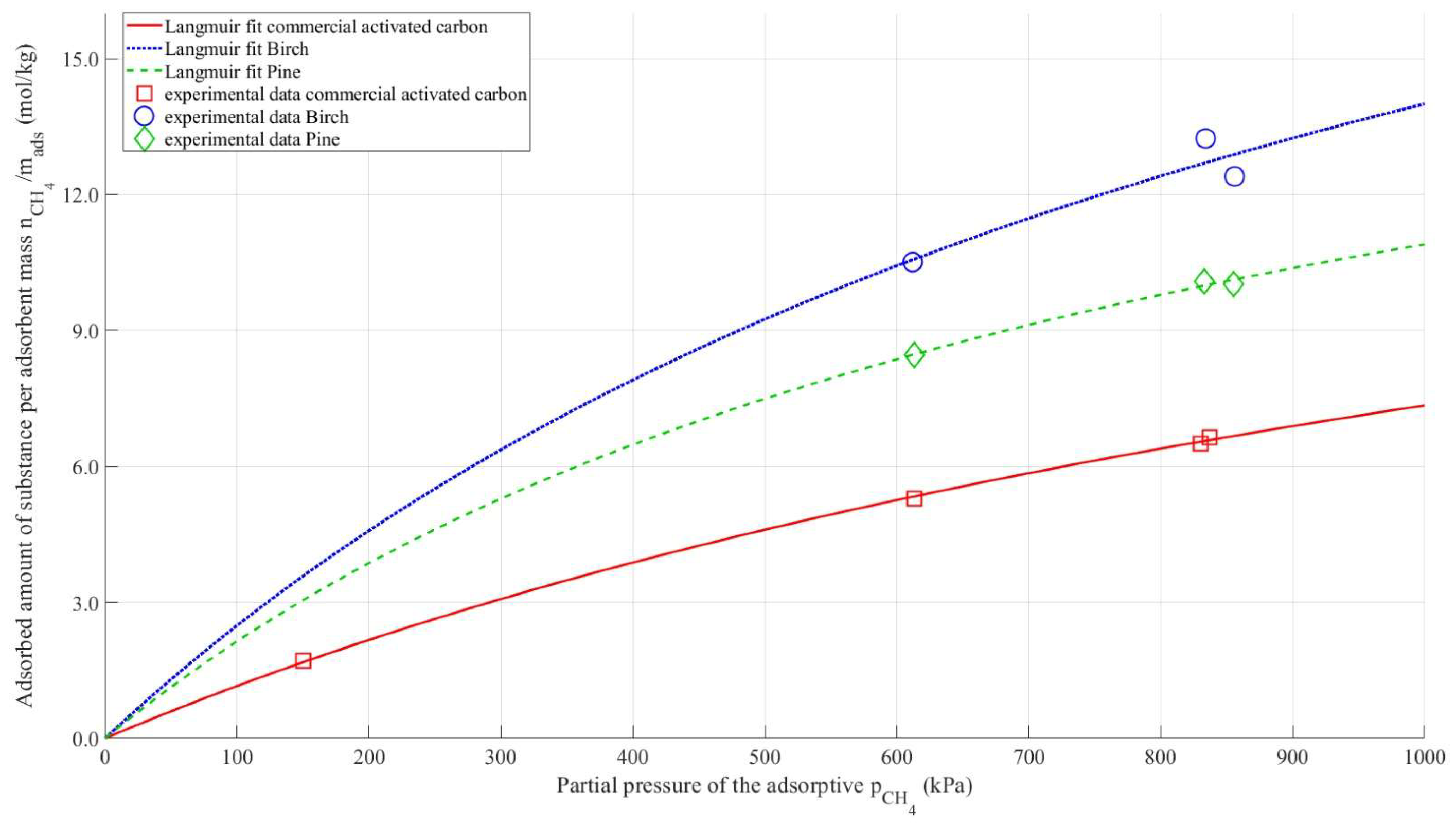
Figure 3 presents the methane adsorption isotherms for commercial, pine-derived, and birch-derived activated carbons, together with the corresponding Langmuir model fits. The adsorbed amount increases steadily with increasing methane partial pressure for all materials, indicating typical physisorption behavior dominated by micropore filling.
At all pressures, the biomass-derived activated carbons show significantly higher gravimetric methane uptake than the commercial activated carbon, which is consistent with their higher BET surface areas and well-developed microporous structures. In contrast, the commercial activated carbon exhibits a lower adsorption capacity over the entire pressure range. The experimental data are well described by the Langmuir model, suggesting that methane adsorption proceeds primarily via monolayer adsorption on energetically similar adsorption sites.
The bulk density of the pine and birch samples is nearly half that of commercial activated carbon. As a result, the amount of substance adsorbed per unit mass of the adsorbent is significantly higher when using these samples compared to the commercial adsorbent. In all experiments, the volume of the column filled with adsorbent was kept constant. Therefore, in
Figure 3, the amount of substance adsorbed is plotted on the y-axis without relating it to the total mass of the adsorbent.
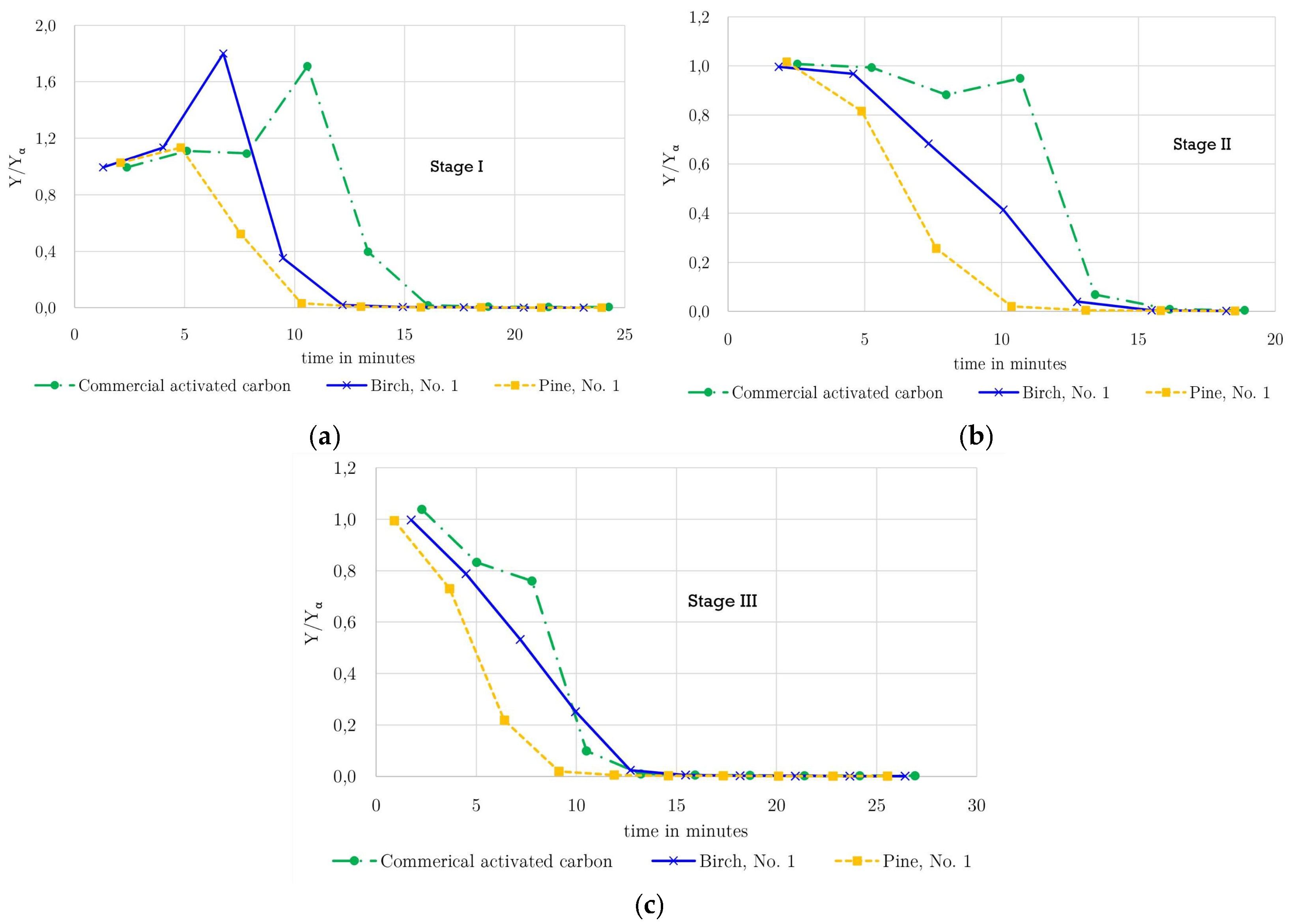
3.3. Desorption
During the desorption step shown in
Figure 4 (a: Stage I, b: Stage II, and c: Stage III), the overall shape of the concentration profiles remained comparable; however, slight variations were observed between individual desorption curves. These differences arise from the manual control of the depressurization step, resulting in small variations in the pressure release rate (Δp/Δt) between experiments.
A comparison among the three activated carbons reveals that methane desorption from the pine-derived activated carbon occurs more rapidly than from the birch-derived and commercial activated carbons. As demonstrated by the adsorption breakthrough times (
Table 6), the pine-derived activated carbon exhibits shorter breakthrough times, particularly at lower pressures, indicating a lower methane loading at the end of the adsorption step. In addition, due to its lower bulk density (0.297 g cm
−3), a smaller mass of pine-derived activated carbon is present in the column compared to the commercial material (0.538 g cm
−3). Consequently, the total amount of methane stored in the adsorber at the onset of desorption is lowest for the pine-derived activated carbon, leading to a steeper decline in Y/Yα and shorter apparent desorption times.
In contrast, the commercial activated carbon retains a larger methane inventory due to its longer breakthrough times and higher bulk density, resulting in more extended desorption profiles. The birch-derived activated carbon exhibits intermediate behavior, consistent with its adsorption capacity and bulk density. Importantly, all investigated materials exhibit nearly complete methane removal during desorption, confirming that adsorption under the applied conditions is predominantly physisorptive and reversible, demonstrating good regenerability of both commercial and biomass-derived activated carbons.
3.4. Post PSA Structural and Surface Characterization
3.4.1. Morphological Stability and Elemental Composition After PSA (SEM-EDX)
Scanning electron microscopy was employed to gain further insight into the surface morphology of the activated carbons before and after exposure to high-pressure PSA adsorption–desorption experiments, as presented in
Figure 5.
The fresh commercial activated carbon exhibits a heterogeneous surface morphology with irregularly shaped particles and a relatively compact morphology, as shown in
Figure 5-a. After PSA operation (
Figure 5-b), the commercial material shows roughening and particle fragmentation. However, the overall particle morphology and structural framework remain intact, indicating no severe structural collapse under the applied operating conditions. The fresh birch-derived activated carbon (
Figure 5-c) exhibits a well-developed porous structure characterized by extensive and interconnected cavities. This reflects the intrinsic cellular structure of the biomass precursor and the effects of chemical activation. Following adsorption–desorption experiments in
Figure 5-d, the porous structure is largely preserved, with no evidence of pore blockage, cracking, or collapse. Minor surface changes are detected, suggesting good resistance to mechanical stress during pressurization and depressurization.
The pine-derived fresh activated carbon (
Figure 5-e), shows a highly open and hierarchical pore structure in its fresh state, characterized by large cavities and thin pore walls. Comparison of the fresh and post-PSA samples (
Figure 5-f), shows that this morphology remains unchanged after high-pressure operation. There is no significant deformation of pores or structural degradation observed, indicating that the open pore structure of pine activated carbon remains mechanically stable under the tested PSA conditions.
SEM observation shows that all three types of activated carbon maintain their morphological stability during PSA operation.
The elemental composition of the activated carbons, both before and after the PSA operation, was analyzed using EDX analysis to complement the morphological observations obtained from SEM. The results indicate that PSA cycling does not lead to significant changes in elemental composition, as presented in
Table 7.
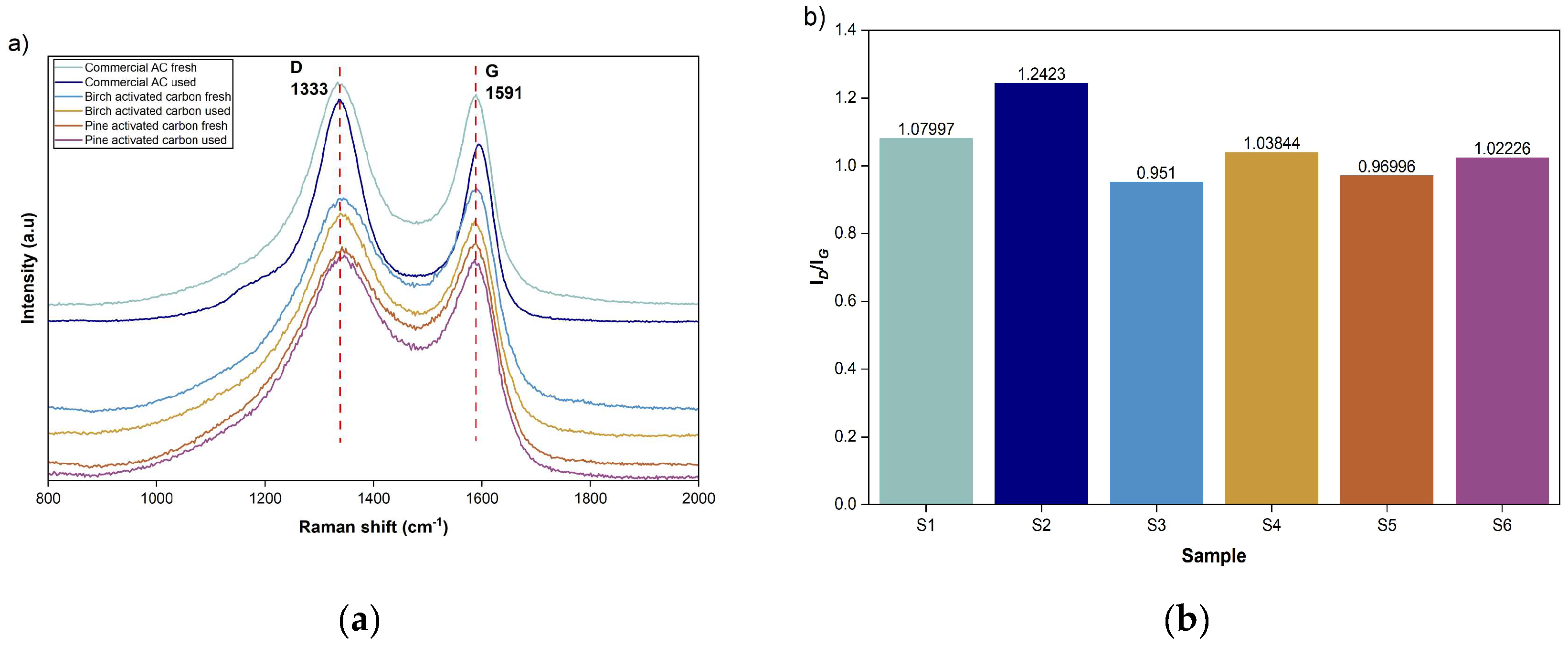
3.4.2. Structural Features of Activated Carbons After PSA (Raman Spectroscopy)
Raman spectroscopy was used to assess the material structure and defect characteristics of the activated carbons before [
14], and after the PSA operation. Typically, structural defects and disordered carbon is presented in
Figure 6-a, displaying the characteristic D band at approximately 1333 cm
−1 , and the G band at approximately 1591 cm
−1, representing the graphitized structure of carbon[
27,
28,
29]. To assess the degree of structural disorder, the intensity ratio I
D/I
G of the D and G bands was calculated and summarized in
Figure 6-b.
The fresh commercial activated carbon (S1) exhibits an I
D/I
G ratio of 1.08, reflecting a predominantly disordered carbon structure typical of activated carbons [
30]. After conducting the PSA cycling experiments with the commercial activated carbon (S2), it was observed that the ratio increased to 1.24, indicating an increase in defect density or a slight reduction in structural order. This change suggests that repeated high-pressure adsorption-desorption cycles may introduce additional lattice distortions or microstructural defects within the commercial carbon.
In contrast, the fresh birch-derived activated carbon (S3) shows a lower ID/IG ratio of 0.95, indicating a comparatively higher degree of structural order. After PSA operation (S4), the ratio increases moderately to 1.04, pointing to a limited increase in disorder. A similar trend is observed for the pine-derived activated carbon, where the ID/IG ratio increases from 0.97 for the fresh sample (S5) to 1.02 after PSA cycling (S6).
The smaller changes observed in the ID/IG ratio of biomass-derived carbons, compared to commercial material, indicate a higher resistance to structural disorder during high-pressure operations, supporting their suitability for PSA conditions.
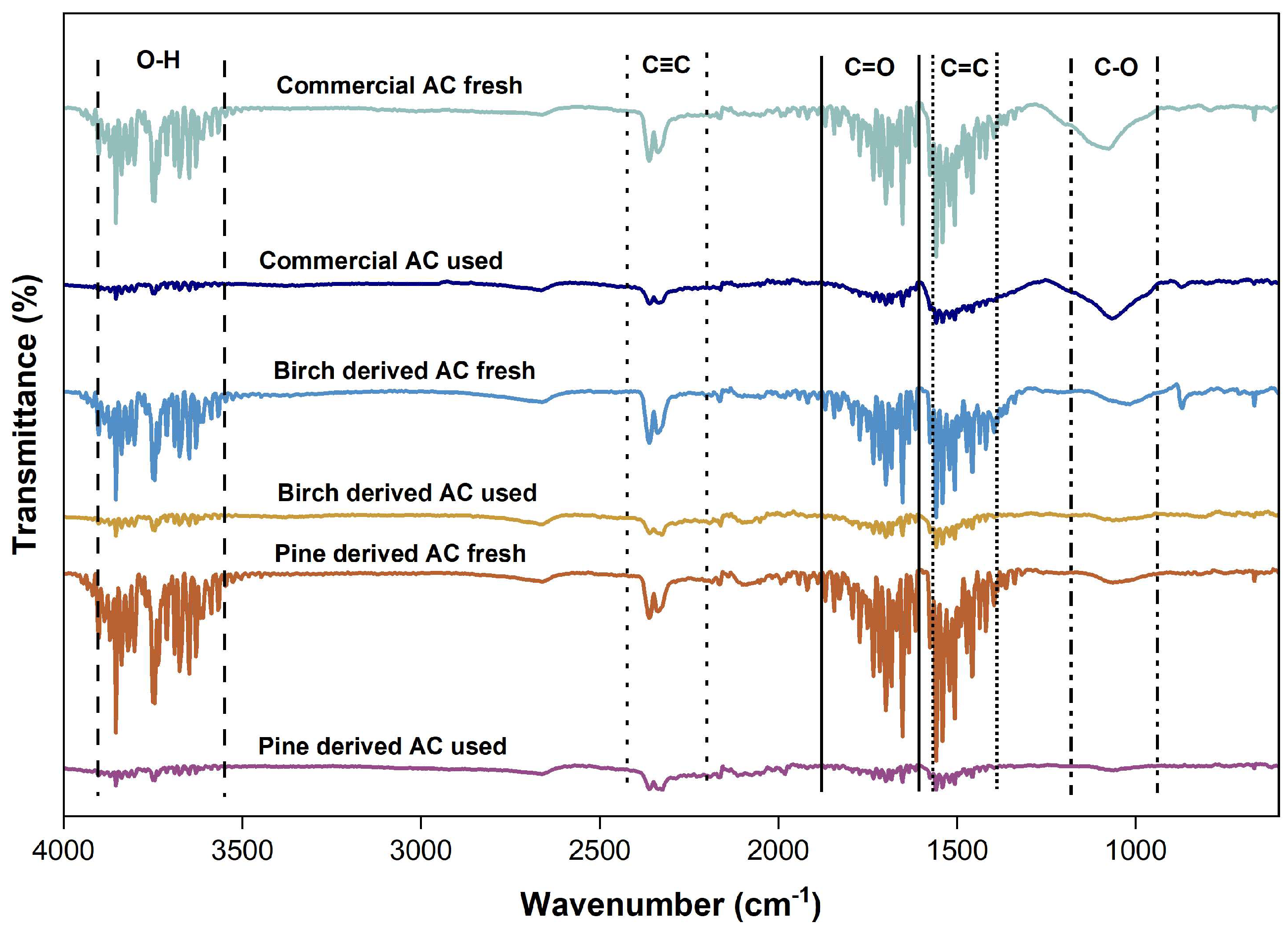
3.4.3. Surface Chemistry Evolution After PSA (FTIR Spectroscopy)
The surface chemistry of the commercial and biomass-derived activated carbons before and after PSA operations was examined by FTIR spectroscopy, as presented in
Figure 7. The analysis provides insight into the chemical stability of the adsorbents under high-pressure adsorption–desorption conditions and complements the structural information obtained from SEM, EDX, and Raman spectroscopy.
All fresh activated carbons exhibit a broad absorption band in the range of 3600–3200 cm
−1, which is attributed to O–H stretching vibrations associated with surface hydroxyl groups and typically adsorbed moisture. This band is typical for activated carbons and reflects the presence of oxygen-containing surface functionalities, which support effective adsorption[31,32]. After the PSA experiments, the intensity of this band decreases slightly across all samples. This suggests that weakly bound surface species are partially removed during the pressurization conditions and helium purging, rather than indicating chemical degradation of the carbon framework. Weak bands observed around 2300–2100 cm
−1 can be attributed to C≡C stretching vibrations or to adsorbed gas species, which are more pronounced in the fresh samples [33]. These features become less distinct after PSA operation, suggesting effective desorption and surface cleaning during the experimental process. In the region between 1700 and 1500 cm
−1, absorption bands corresponding to C=O stretching (carbonyl, carboxyl) and aromatic C=C vibrations are evident for all activated carbons [34]. These bands remain present after PSA experiments, with only minor changes in intensity, indicating that the aromatic carbon structure and oxygen-containing functional groups are largely preserved. Bands in the range of 1200–1000 cm
−1, associated with C–O stretching vibrations of ether, or ester groups, are observed in both fresh and used samples [
20,31]. The persistence of these features after PSA cycling confirms that the surface chemistry of the activated carbons remains stable under the investigated operating conditions.
No new absorption bands appear after PSA operation, and no significant peak shifts are detected, supporting the absence of chemical modification or surface oxidation induced by high-pressure adsorption–desorption cycles.
3.4.4. Cyclic Stability and Structural Evolution of Pine-Derived Activated Carbon Under PSA Operation
Pine-derived activated carbon was chosen for extended cyclic testing due to its textural properties, specifically its highest specific surface area among the activated carbons tested. To evaluate the adsorption and structural stability of the pine-derived activated carbon under high-pressure operation, eight consecutive breakthrough experiments were conducted at 25 bar (Stage IV conditions). These experiments were performed in a cyclic adsorption–desorption manner, and after every two cycles, one sample was withdrawn for Raman spectroscopy in order to monitor potential structural changes.
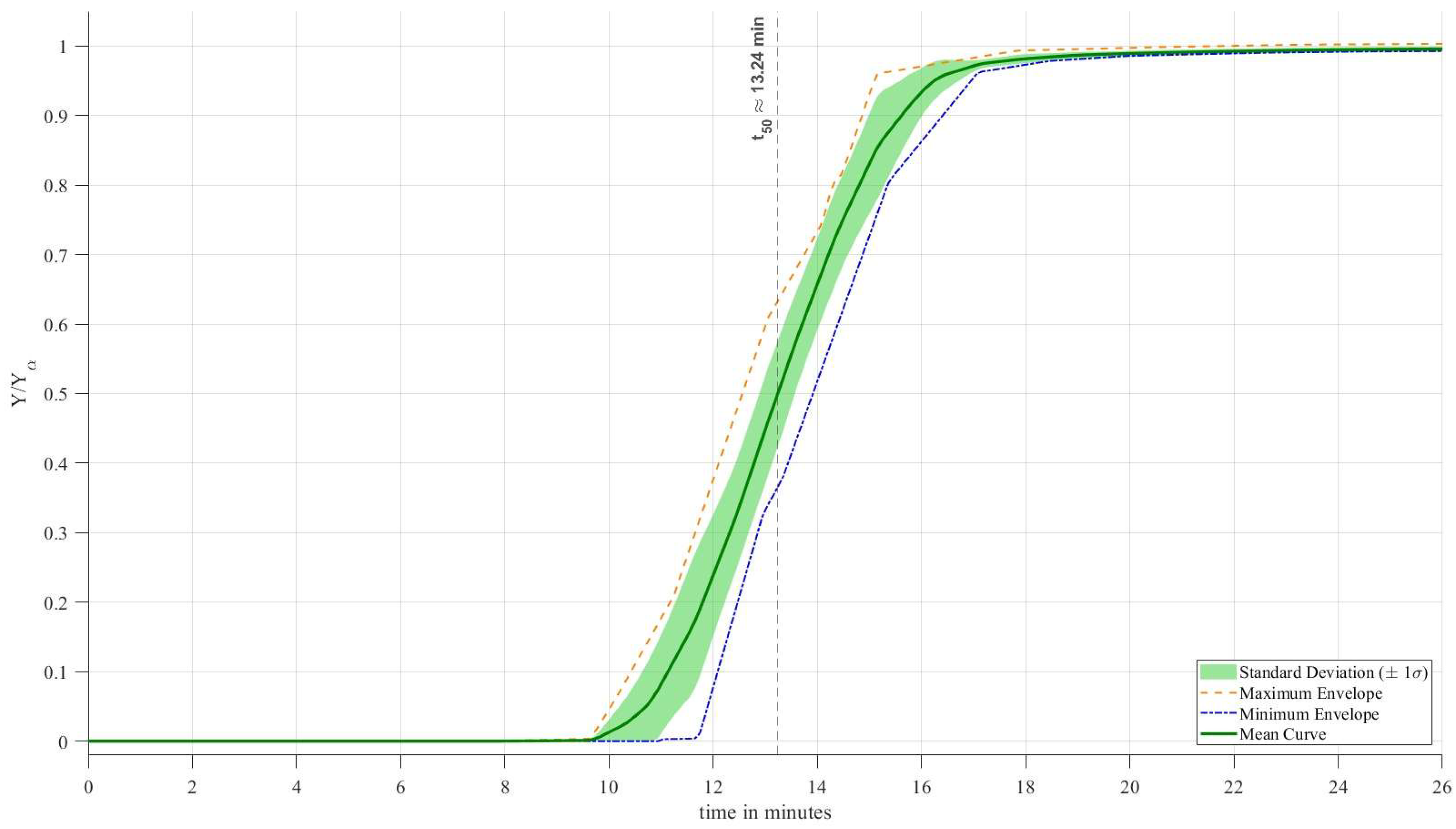
In addition, two breakthrough experiments obtained during the initial screening phase (
Figure 2) were included for comparative purposes in the breakthrough analysis. As a result, a total of ten breakthrough curves were included in the statistical evaluation and presented in
Figure 8, while Raman analysis was conducted on samples obtained from the eight consecutive cyclic experiments as shown in
Figure 9.
To quantitatively assess the reproducibility and stability of the adsorption performance, the ten breakthrough curves were statistically processed using MATLAB. Because the experimental curves were recorded at slightly different time intervals, a synchronization procedure was first applied. A uniform time axis ranging from 0 to 26 minutes was generated, and each individual breakthrough curve was interpolated onto this common grid. For each curve, 300 equally spaced time steps were calculated, compared to the original 9–10 experimentally measured data points within the same time range. This interpolation enabled a direct point-by-point comparison of all ten experiments. At every single time step, the script calculated the mean value between the 10 curves and the standard deviation. The solid line represents the mean breakthrough curve and the shaded region corresponds to ±1 standard deviation (±1σ). The narrow width of the shaded band over the entire breakthrough region demonstrates excellent reproducibility of the adsorption behavior. The low statistical dispersion indicates that no significant performance degradation or progressive loss of adsorption capacity occurred during repeated high-pressure cycling. The script calculated the t_50 point of the mean value curve, t_50 which is the time at which the system reached 50% of its capacity. The calculated value of t50≈13.24 min confirms the consistent adsorption capacity of the pine-derived activated carbon across all ten cycles. The absence of any systematic shift in the breakthrough front suggests that the material maintains stable adsorption and pore accessibility under repeated pressurization and regeneration.
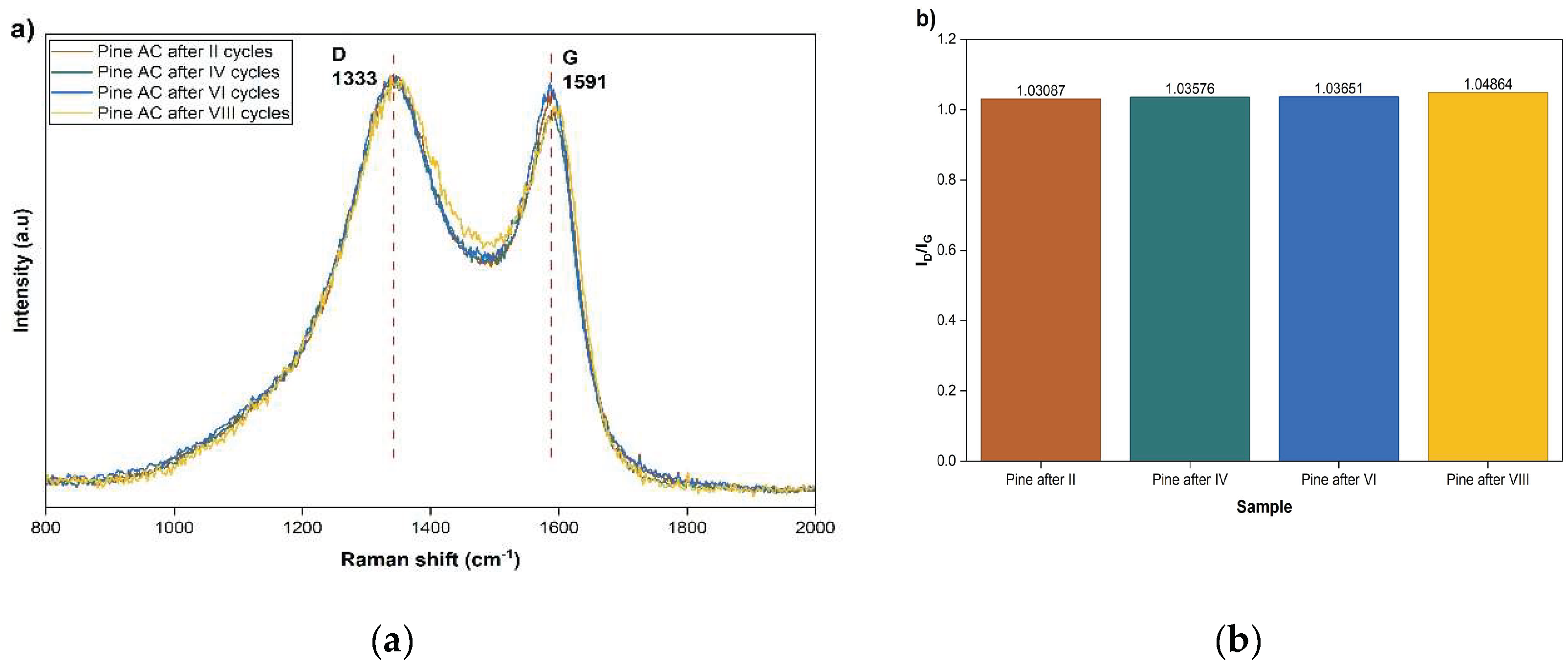
The Raman spectra of all samples exhibit the characteristic D band at approximately 1333 cm
−1 and G band at around 1591 cm
−1, corresponding to disordered sp
2 carbon and graphitic sp
2 domains, as shown in
Figure 9-a.
A direct comparison of the spectra reveals no significant peak shifts, band broadening, indicating that no major graphitization, structural collapse, or formation of highly ordered carbon phases occurred during repeated PSA operation. The evolution of the intensity ratio ID/IG , presented in
Figure 9-b, provides a slight but systematic increase in I
D/I
G from 1.0309 (after II cycles) to 1.0486 (after VIII cycles). The magnitude of this increase is small (< 2% relative change), indicating that the structural modifications are subtle and do not translate into measurable deterioration of adsorption performance, as evidenced by the stable breakthrough curves and constant t50 value. Thus, while Raman spectroscopy detects minor microstructural evolution at the atomic scale, these changes do not significantly affect the macroscopic adsorption behavior.
In summary, the combined breakthrough and Raman analyses demonstrate that pine-derived activated carbon maintains both adsorption capacity and structural integrity under repeated high-pressure PSA cycling. The material exhibits high mechanical and chemical stability, with only negligible increases in structural disorder after multiple adsorption–desorption cycles.