1. Introduction
Pertussis, or whooping cough, is a highly contagious respiratory infection caused by the gram-negative bacterium
Bordetella pertussis. It is characterized by prolonged paroxysmal coughing, inspiratory “whoop,” and post-tussive vomiting, often lasting several weeks [
1]. Despite being vaccine-preventable, pertussis has re-emerged globally since the 1980s [
2], even in countries with high vaccination coverage such as the United States [
3], Canada [
4], Australia [
5], and Brazil [
6,
7] a phenomenon referred to as pertussis re-emergence.
According to the World Health Organization (WHO), pertussis affects approximately 24 million people and causes 160,700 deaths annually [
8], though underreporting likely results in higher true figures. The disease is most prevalent in regions with limited vaccination coverage, such as sub-Saharan Africa and Southeast Asia, but increasing outbreaks in Europe and the Americas indicate that even robust immunization programs face challenges from waning immunity and pathogen evolution [
9].
In Brazil, pertussis has been a notifiable disease since 1975, monitored by the National Notifiable Diseases Information System (SINAN). In 2019, 6,012 cases were recorded. Between 2020 and 2024, case numbers fluctuated, with notable increases among children aged 1–9 years. In 2024 alone, 2,663 cases were reported, with the 10–14 age group most affected [
10,
11].
The National Immunization Program (PNI) recommends the DTP/Hib/HB vaccine at 2, 4, and 6 months, with booster doses at 15 months and 4 years. Despite these measures, declining vaccine coverage and possible waning immunity have been observed, particularly following disruptions during the COVID-19 pandemic [
12,
13].
Given these concerns, this study sought to evaluate the persistence of vaccine-induced humoral immunity in a pediatric population in Duque de Caxias, a densely populated urban area in Rio de Janeiro. By quantifying serum IgG levels against pertussis toxin (PTx) and B. pertussis antigens, we aimed to assess the real-world effectiveness of vaccination and identify potential immunity gaps that may contribute to pertussis resurgence.
2. Materials and Methods
2.1. Study Population and Serum Samples
This cross-sectional sero-epidemiological study analyzed 220 serum samples collected between January and December 2022 from children vaccinated with DTP/Hib/HB with aged 1–14 years at public health units in Duque de Caxias, Rio de Janeiro, Brazil. Samples were grouped as follows: 1–4 years ( 110 samples), 5–9 years (68 samples), 10–14 yearsm (44 samples). This stratification enabled evaluation of immunity at different post-vaccination intervals.
2.2. ELISA Assays
Commercial ELISA kits (SERION ELISA classic, ESR1201G, Brazil) were used to quantify IgG antibodies against B. pertussis and B. parapertussis antigens, including pertussis toxin (PTx) and filamentous hemagglutinin (FHA). The SERION ELISA classic IgG B. pertussis Toxin I kit was used for PTx-specific detection. Results were expressed in IU/mL, with values >40 IU/mL classified as positive, 40–100 IU/mL as borderline, and <40 IU/mL as negative, following manufacturer recommendations.
2.3. Statistical Analysis
Data were analyzed using GraphPad Prism 9. The Kruskal–Wallis test, followed by Dunn’s multiple comparisons, was applied to evaluate differences in antibody levels among age groups. A p-value < 0.05 was considered statistically significant.
3. Results
3.1. Anti-PTx IgG Levels
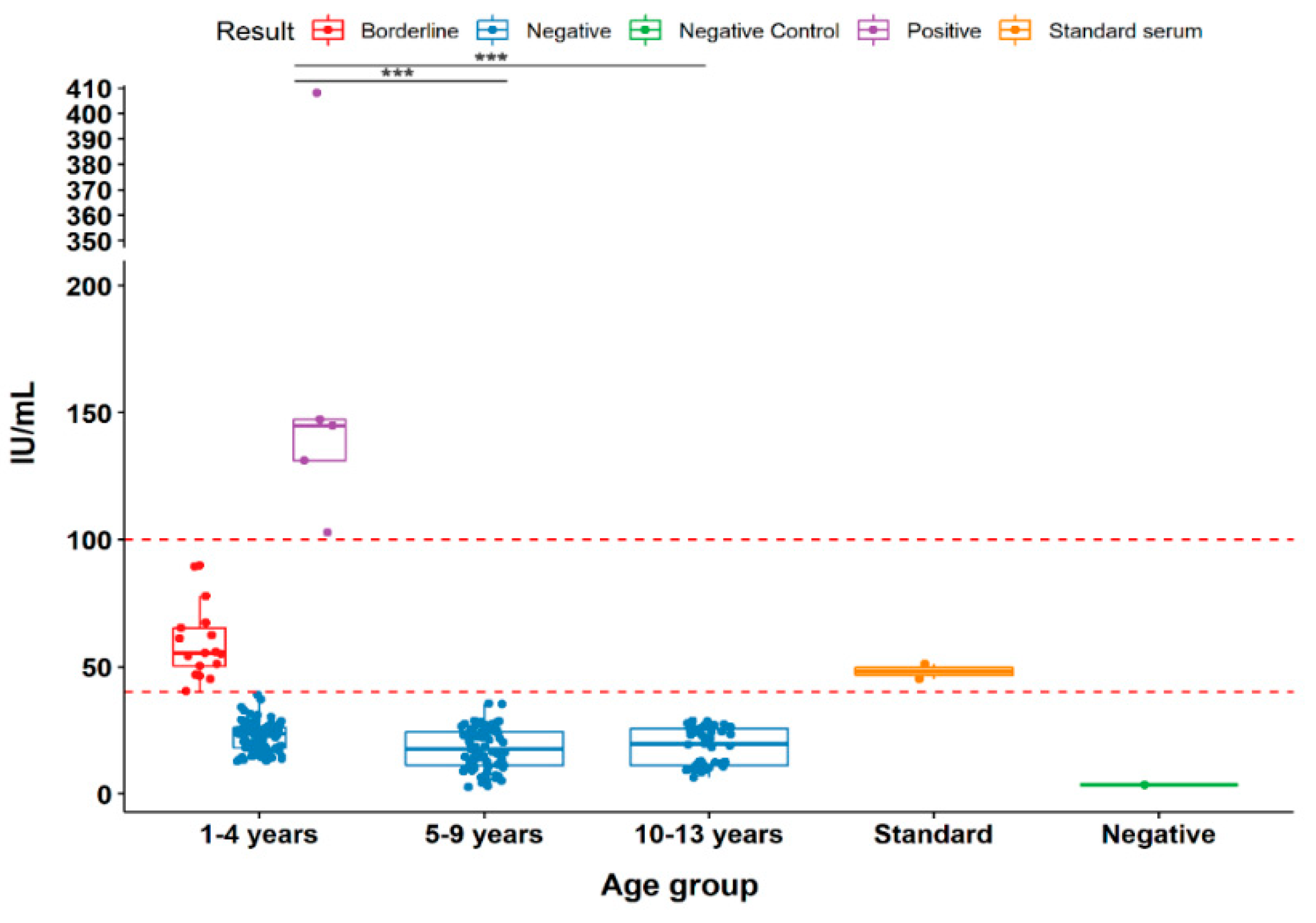
Analysis of anti-PTx IgG levels revealed age-related declines in antibody titers. Among children aged 5–9 years (n = 68) and 10–14 years (n = 44), all sera were below the protective threshold (<40 IU/mL). The 1–4-year group displayed greater variability, with titers ranging from 12.7 to 408 IU/mL. Within this group, 5 samples (4.5%) were positive, 17 (15.4%) borderline, and 87 (79.1%) negative. Overall, only 5 of 220 samples (2.17%) exhibited protective levels (>40 IU/mL). The youngest group showed significantly higher antibody levels than older groups (p < 0.05).
Figure 1.
Antibody response to Bordetella pertussis toxin in children aged 1–13 years. Serum antibody titers (IU/mL) were measured by ELISA in 220 children and are shown as boxplots with individual data points overlaid. Samples were stratified into three age groups (1–4, 5–9, and 10–13 years). Standard positive and negative sera were provided by the ELISA kit. Statistical analysis was performed using the Kruskal–Wallis test followed by Dunn’s multiple comparisons test. Data are presented as median and interquartile range. Statistical significance was set at p < 0.05; **p < 0.001.
Figure 1.
Antibody response to Bordetella pertussis toxin in children aged 1–13 years. Serum antibody titers (IU/mL) were measured by ELISA in 220 children and are shown as boxplots with individual data points overlaid. Samples were stratified into three age groups (1–4, 5–9, and 10–13 years). Standard positive and negative sera were provided by the ELISA kit. Statistical analysis was performed using the Kruskal–Wallis test followed by Dunn’s multiple comparisons test. Data are presented as median and interquartile range. Statistical significance was set at p < 0.05; **p < 0.001.
3.2. IgG Reactivity to B. pertussis Antigens
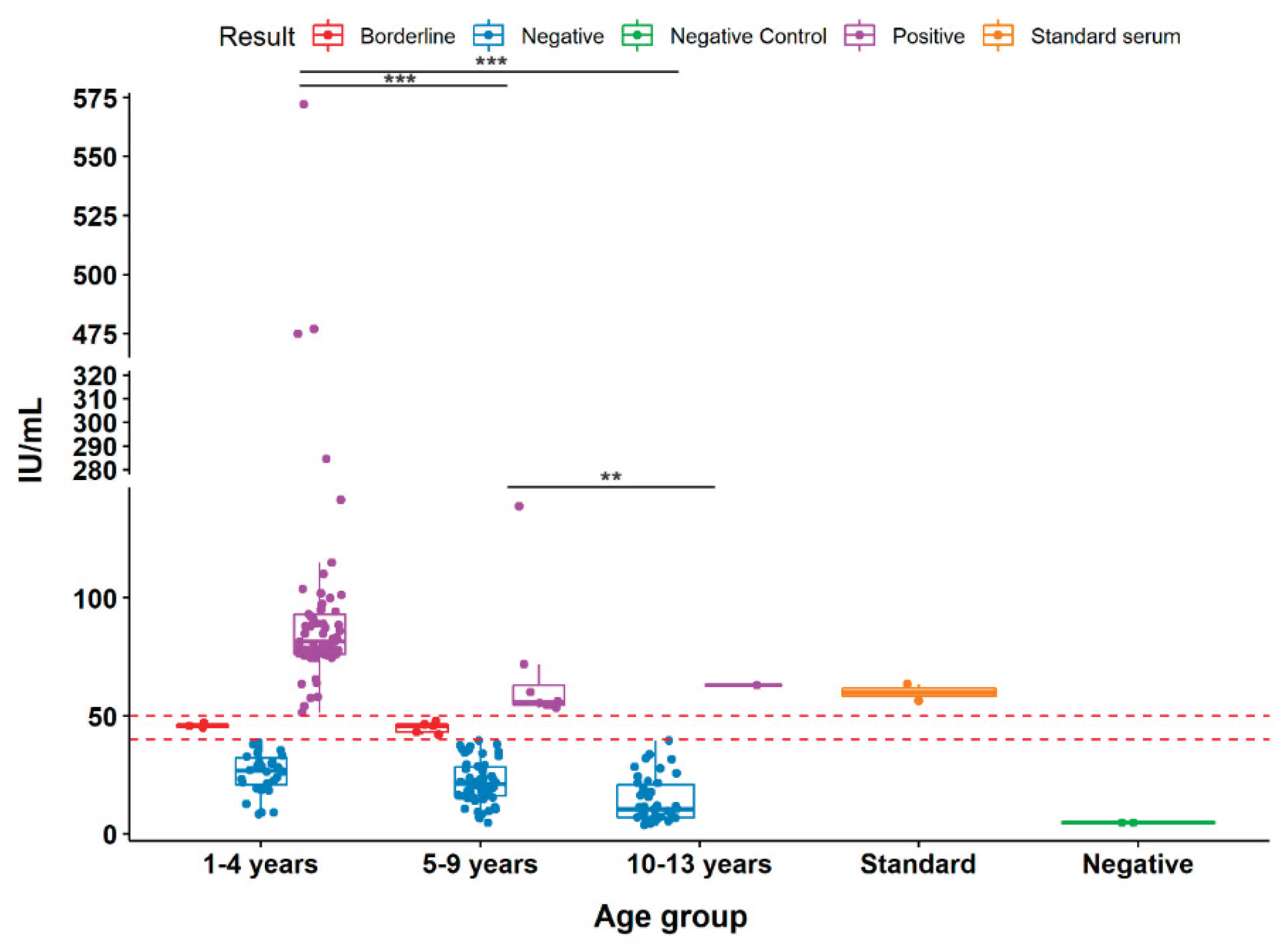
When tested with B. pertussis antigens (PTx + FHA), seropositivity increased to 36.7% (78/220). The mean antibody levels declined with age: 1–4 years: 100.7 IU/mL (n = 69), 5–9 years: 68.2 IU/mL (n = 8), 10–14 years: 63.1 IU/mL (n = 1)
A significant downward trend in titers across age groups (p < 0.05) indicated waning immunity over time. The majority of samples (63.2%) were negative, suggesting inadequate long-term protection.
Figure 2.
Antibody responses to filamentous hemagglutinin (FHA) and pertussis toxin (Ptx) in children aged 1–13 years. Serum antibody titers (IU/mL) were measured by ELISA in 220 children and are shown as boxplots with individual data points overlaid. Samples were stratified into three age groups (1–4, 5–9, and 10–13 years). Standard positive and negative sera were provided by the ELISA kit. Statistical analysis was performed using the Kruskal–Wallis test followed by Dunn’s multiple comparisons test. Data are presented as median and interquartile range. Statistical significance was set at p < 0.05; *p < 0.01, **p < 0.001.
Figure 2.
Antibody responses to filamentous hemagglutinin (FHA) and pertussis toxin (Ptx) in children aged 1–13 years. Serum antibody titers (IU/mL) were measured by ELISA in 220 children and are shown as boxplots with individual data points overlaid. Samples were stratified into three age groups (1–4, 5–9, and 10–13 years). Standard positive and negative sera were provided by the ELISA kit. Statistical analysis was performed using the Kruskal–Wallis test followed by Dunn’s multiple comparisons test. Data are presented as median and interquartile range. Statistical significance was set at p < 0.05; *p < 0.01, **p < 0.001.
4. Discussion
The resurgence of pertussis, even in highly vaccinated populations, underscores the complex interplay between vaccine-induced immunity, pathogen evolution, and public health practices. Brazil’s National Immunization Program (PNI), historically successful in achieving >90% coverage, has seen declines in recent years dropping to 77,2% for DTP/Hib/HB in 2022. The consequences of this reduction are now reflected in rising pertussis incidence.[
14].
Our study revealed that fewer than 3% of vaccinated children exhibited protective anti-PTx antibody levels. Although 36.7% showed broader reactivity to
B. pertussis antigens, the low toxin-specific response highlights insufficient or waning immunity consistent with reports from China, Sri Lanka, and Malaysia, where post-vaccination antibody decline was also observed among older children and adolescents [
15,
16,
17,
18].
In Brazil, all pediatric vaccines currently distributed by PNI are whole-cell (wP) formulations. While effective in controlling disease transmission, wP vaccines can elicit variable immune durability and higher reactogenicity compared with acellular vaccines (aP). The low anti-PTx IgG levels observed in this study suggest that protection derived from wP vaccination may wane faster than expected, emphasizing the need for booster doses beyond early childhood. International experience supports this approach. Countries that introduced adolescent boosters or maternal immunization (using dTpa) have seen reduced pertussis incidence in infants and adolescents. In Brazil, similar policies could mitigate waning immunity and protect vulnerable age groups. Although, this study was limited by single-municipality design and absence of clinical or genetic confirmation of infection, the results provide valuable insight into real-world immunity patterns in an urban Brazilian population and reinforce the need for broader surveillance [
19,
20].
5. Conclusions
The low prevalence of protective antibodies against pertussis toxin among vaccinated children in Duque de Caxias indicates waning immunity and incomplete protection under the current vaccination scheme. Revising the national schedule to include adolescent boosters and incorporating acellular vaccine options are essential steps toward long-term pertussis control in Brazil.
Author Contributions
Conceptualization, FRdS and SGD-S; methodology, FRdS, PN-P, SVC, GCL, JPRdSC, and RCdST; validation, GCL and LRG; writing-original draft, FRdS; writing-review & editing, SGD-S and FRdS; fund raising, SGD-S. All authors read and agreed to the published version of the manuscript.
Funding
This work did not receive funding. SGD-S received support from the National Council for Scientific Research (CNPq No. 30515-2020-5) and the Carlos Chagas Filho Foundation for Research Support of the State of Rio de Janeiro (FAPERJ No. 200.960-2022).
Acknowledgments
The execution of this work was made possible thanks to the support of Coordination for the Improvement of Higher Education Personnel (CAPES), with scholarships for João Pedro R. da S. Carvalho and Rafael C. de S. Tapajóz. Paloma Napoleão-Pêgo, Guilherme C. Lechuga, and Larissa R. Gomes were scholarship holders from FIOCRUZ and FAPERJ, respectively.
Conflicts of Interest
The authors declare no conflicts of interest.
Ethics Statement
The study was approved by the Research Ethics Committee of Unigranrio University (CAAE: 248566119.0.0000.5283). All procedures followed national and international ethical standards for human research.
Consent
All authors give their consent for publication of the present manuscript.
Disclosure
All authors read and approved the final manuscript.
References
- Kuchar, E.; Karlikowska-Skwarnik, M.; Han, S. Pertussis: History of the disease and current prevention failure. Adv Exp Med Biol 2016, 934, 77–82. [Google Scholar] [CrossRef] [PubMed]
- GBD 2021 Causes of Death Collaborators. Global burden of 288 causes of death and life expectancy decomposition in 204 countries and territories and 811 subnational locations, 1990-2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet Erratum: Lancet 2024, 403, 1988.. 2024, 403, 2100–2132. [Google Scholar] [CrossRef] [PubMed]
- Christie, C.D.C. Resurgence of pertussis: whopping the '100-day cough'. Curr Opin Pediatr 2025, 37, 508–516. [Google Scholar] [CrossRef] [PubMed]
- Bhagat, D.; Saboui, M.; Huang, G. Pertussis epidemiology in Canada, 2005-2019. Can Commun Dis Rep 2023, 49, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Marshall, K.S.; Quinn, H.E.; Pillsbury, A.J. Australian vaccine preventable disease epidemiological review series: Pertussis, 2013-2018. Commun Dis Intell 2022, 46. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y. Pertussis epidemic status and vaccine research progress. Occup Health. 2018, 34, 1005–1009. [Google Scholar]
- Haitão, H.; Zhigang, G. Research progress on the resurgence of pertussis and its coping strategies. Med Rev 2015, 21, 1630–1632. [Google Scholar]
- Bhagat, D.; Saboui, M.; Huang, G. Pertussis epidemiology in Canada, 2005-2019. Can Commun Dis Rep 2023, 49, 21–28. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Pertussis (whooping cough); WHO: Western Pacific Region, 2025; Available online: https://www.who.int/westernpacific/.
- Domingues, C.M.; Maranhão, A.G.; Teixeira, A.M. 46 anos do Programa Nacional de Imunizações: uma história repleta de conquistas e desafios a serem superados. Cadernos de Saude Publica English, Portuguese. 2020, 236, e00222919. [Google Scholar] [CrossRef] [PubMed]
- De Barros, E.N.C.; Nunes, A.A.; Abreu, A.D.J.L.D. Pertussis epidemiological pattern and disease burden in Brazil: an analysis of national public health surveillance data. Hum Vaccin Immunother 2020, 16, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Domingues, C.M.A.S.; Teixeira, A.M.S.; Moraes, J.C. Vaccination coverage in children in the period before and during the COVID-19 pandemic in Brazil: a time series analysis and literature review. J Pediatr (Rio J) 2023, 99, S12–S21. [Google Scholar] [CrossRef] [PubMed]
- Locht, C. Pertussis before, during and after Covid-19. EMBO Mol Med. 2025, 17, 594–598. [Google Scholar] [CrossRef] [PubMed]
- BRASIL; Ministério da Saúde. Cobertura Vacinal 2022-2023. Imunizações: Coberturas Vacinais Brasil: Brasília; MS, 2024; Available online: https://www.gov.br/saude/pt-br/vacinacao/publicacoes/.
- Xu, Y.; Xu, E.; Liu, S. Seroepidemiology of pertussis in Hangzhou, China, during 2009-2017. Human Vaccine Immunotherapy 2019, 15, 2564–2570. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Zhu, Y.; Jin, M. The decline in immunity and circulation of pertussis among Chinese population during the COVID-19 pandemic: A cross-sectional sero-epidemiological study. Vaccine 2022, 40, 6956–6962. [Google Scholar] [CrossRef] [PubMed]
- Binti Md Bahar, A.N.; Muhamad, A.N.; C.S.J. The. Prevalence of Bordetella pertussis among children aged 4 years and above presenting with acute respiratory tract infection: a cross-sectional study. Pathog Glob Health 2024, 118, 582–586. [Google Scholar] [CrossRef] [PubMed]
- Jog, P.; Memon, I.A.; Thisyakorn, U. Participants of the 2019 GPI Thailand meeting. Pertussis in Asia: Recent country-specific data and recommendations. Vaccine 2022, 40, 1170–1179. [Google Scholar] [CrossRef] [PubMed]
- Barkoff, A-M.; Knuutila, A.; He, J.; Mertsola, Q. Evaluation of anti-PT antibody response after pertussis vaccination and infection: The importance of both quantity and quality. Toxins 2021, 13, 508. [Google Scholar] [CrossRef] [PubMed]
- Silva, E. P.; Trentini, M; Rodriguez, D. Seroprevalence study reveals pertussis underreporting in Brazil and calls for adolescent/young adult boosting: mouse model demonstrates immunity restoration. Front Immunol. 2024, 15, 1472157. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).