1. Introduction
Chronic lymphocytic leukemia (CLL) is the most prevalent leukemia of adults in Europe and North America and remains a biologically heterogeneous malignancy characterized by the progressive accumulation of mature, antigen-experienced B lymphocytes accompanied by profound immune dysregulation. Over the past decade, the therapeutic landscape of CLL has experienced a fundamental change, shifting from chemoimmunotherapy (CIT) to highly effective targeted agents, especially Bruton’s tyrosine kinase inhibitors (BTKi) and BCL2 inhibitors (BCL2i), which now form the core of international treatment recommendations. Despite these advances, CLL remains incurable, and therapeutic resistance, treatment intolerance, and cumulative toxicity continue to be major factors driving disease progression and unmet clinical needs, prompting intense ongoing research by the scientific community. [
1,
2,
3,
4,
5].
Historically, immunotherapy with anti-CD20 monoclonal antibodies has been a key part of CLL management. Regimens such as fludarabine, cyclophosphamide, and rituximab (FCR) have shown durable responses in certain patient groups [
6,
7]. However, it has become increasingly clear that CIT-based approaches are linked to significant toxicity and lower effectiveness in biologically high-risk disease, especially in patients with TP53 mutations or 17p deletion. These genetic abnormalities, now recognized as dominant factors in treatment failure, confer resistance to DNA-damaging therapies and are strongly linked to aggressive disease biology and reduced survival [
2,
8]. Moreover, therapy-related immunosuppression further worsened infectious complications, secondary malignancies, and hematologic toxicity, reducing the therapeutic window of conventional therapies CIT.
The introduction of mechanism-driven targeted therapies has fundamentally changed outcomes for this high-risk population. Both BTKi and venetoclax-based regimens have shown substantial clinical effectiveness in TP53-aberrant CLL and are now consistently recommended across major international guidelines, including those of iwCLL, ESMO, NCCN, and local guidelines [
1,
2,
3,
4,
9,
10]. Nevertheless, these therapies are not exempt from limitations. Treatment-emergent toxicities, cardiovascular complications, infectious events, and acquired resistance mechanisms remain clinically relevant, while double-refractory disease has emerged as an increasingly challenging therapeutic scenario [
11,
12,
13,
14,
15,
16].
These observations highlight a key biological principle: CLL is not just a malignancy of B-cell proliferation but also a disorder of immune system balance. Significant dysfunction of both adaptive and innate immunity — including T-cell exhaustion, impaired immune synapse formation, and dysregulated cytokine signaling — contributes to disease persistence, immune escape, and resistance to therapy [
17,
18]. Therefore, strategies that can restore or redirect effective antitumor immunity may be crucial for overcoming resistance mechanisms and achieving lasting disease control. In this context, immunotherapy becomes increasingly important, not just as an adjunctive approach but as a fundamentally different strategy that targets the immune–tumor interface.
Recent advances have substantially expanded the immunotherapeutic armamentarium in CLL beyond classical anti-CD20 antibodies. Novel monoclonal antibodies targeting disease-specific targets, bispecific antibodies that engage T-cell cytotoxicity, chimeric antigen receptor (CAR)-T cell therapies, and emerging natural killer (NK) cell-based platforms collectively represent a new generation of immune-directed strategies [
19,
20,
21]. These approaches show particular promise in high-risk and treatment-resistant diseases, where pathway inhibition alone might be inadequate. However, their best integration into current treatment protocols still needs to be determined and calls for careful assessment of efficacy, safety, and sequencing.
In this review, we discuss the evolving role of immunotherapy in CLL, focusing on its biological rationale, clinical evidence, and future therapeutic directions, with particular emphasis on strategies to overcome high-risk disease biology, resistance, and immune escape.
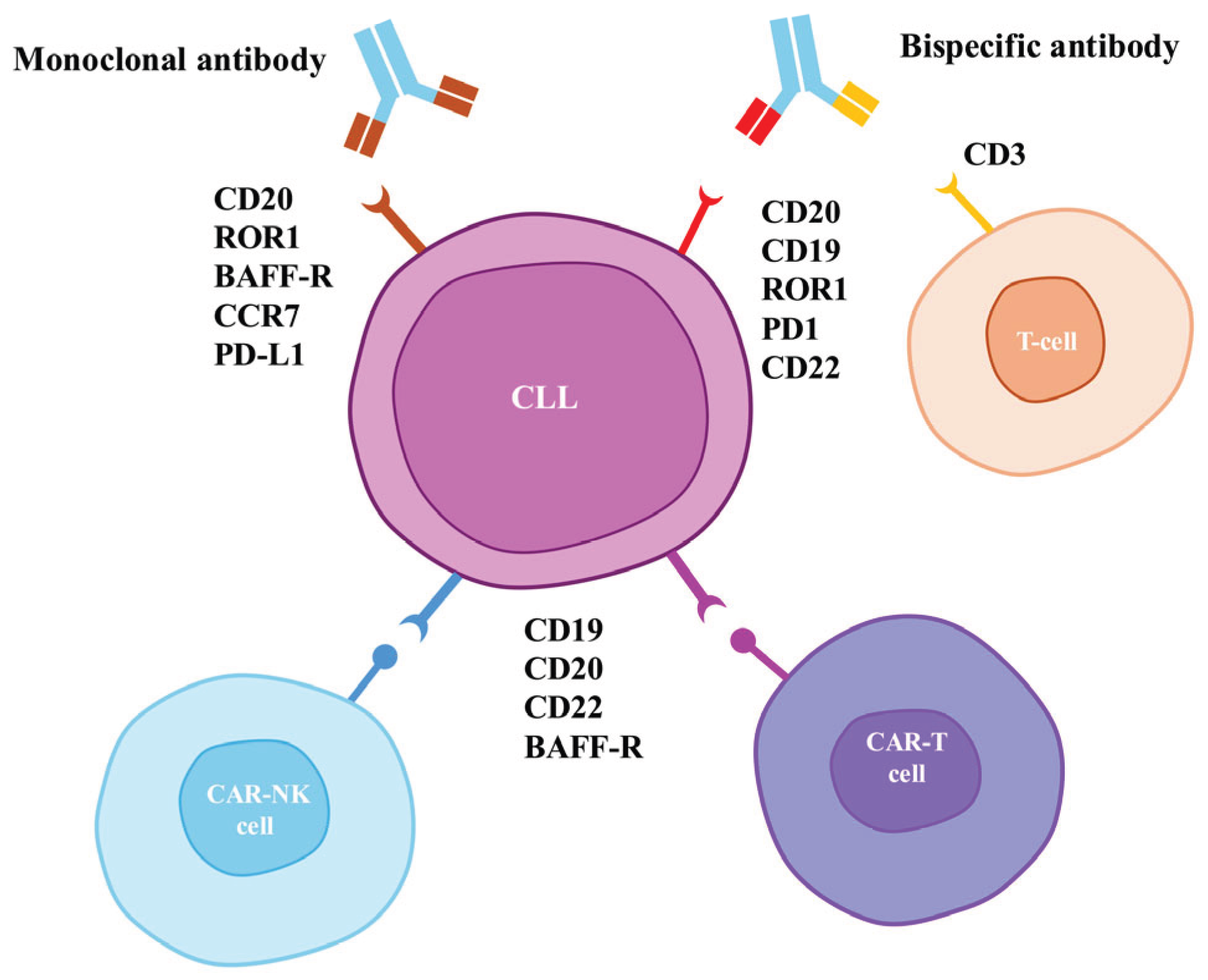
Figure 1.
Therapeutic targets of immunotherapy in chronic lymphocytic leukemia. CLL—chronic lymphocytic leukemia; ROR1—receptor tyrosine kinase-like orphan receptor 1; CD—Cluster of Differentiation; BAFF-R—B-cell activating factor receptor; PD-1—programmed cell death protein 1; PD-L1—programmed death-ligand 1; CCR7—human chemokine receptor 7.
Figure 1.
Therapeutic targets of immunotherapy in chronic lymphocytic leukemia. CLL—chronic lymphocytic leukemia; ROR1—receptor tyrosine kinase-like orphan receptor 1; CD—Cluster of Differentiation; BAFF-R—B-cell activating factor receptor; PD-1—programmed cell death protein 1; PD-L1—programmed death-ligand 1; CCR7—human chemokine receptor 7.
2. Novel Monoclonal Antibodies
The introduction of anti-CD20 monoclonal antibodies was the first breakthrough in CLL immunotherapy. Monoclonal antibodies have revolutionized treatment of CLL and have been used since then as monotherapy or in combination with other agents [
22]. Development of novel antibodies targeting new molecular targets is underway (
Table 1).
2.1. Ianalumab
Ianalumab is a fully human monoclonal antibody targeting B-cell activating factor receptor (BAFF-R) and is being widely studied in autoimmune diseases, immune thrombocytopenia, and CLL. Its mechanism of action includes B-cell depletion, antibody-dependent cellular cytotoxicity (ADCC), interruption of BAFF-R-mediated signaling, and activation of NK and T cells. In a phase Ib trial, 39 patients with CLL who had failed to achieve a complete response (CR) or had developed resistance mutations after at least 1 year of ibrutinib therapy were enrolled. Patients received intravenous ianalumab in combination with ibrutinib for up to six 28-day cycles. Upon achieving CR after 6 cycles, the experimental drug was discontinued, and ibrutinib was continued for an additional two cycles. In patients who achieved undetectable minimal residual disease (uMRD) at the start of cycle 9, discontinuation of ibrutinib was permitted at the investigator’s discretion. Among evaluable patients, the CR/CRi rate on day 1 of cycle 9 was 38%. uMRD in peripheral blood was achieved in 13 patients, and 8 patients achieved uMRD in both blood and bone marrow. Of these, 6 patients were selected to discontinue ibrutinib and remained off therapy at the time of data cutoff [
23].
2.2. CAP-100
CAP-100 is a monoclonal antibody targeting human chemokine receptor 7 (CCR7), and neutralizing its ligand-binding site. Chemokines are a family of cytokines that mediate leukocyte trafficking. Among them, CCR7 plays a crucial role in cell migration, entry, and prolonged residency within the lymph nodes. The expression of CCR7 in CLL is abnormally high compared to the corresponding normal CD5+ B-cell population or pan-B cells [
24]. In R/R CLL, ibrutinib may downregulate CCR7 expression, whereas current data suggest that venetoclax treatment does not significantly alter the CCR7 expression pattern or CAP-100’s mechanism of action [
25]. The ongoing phase Ia/Ib trial (#NCT04704323) aims to evaluate the safety and preliminary clinical benefit of CAP-100 monotherapy in patients with R/R disease who have received at least two prior standard systemic regimens. The results of this study have not yet been published.
2.3. Belimumab
Belimumab is a human monoclonal antibody directed against soluble B-cell activating factor (BAFF), also known as BLyS or TNFSF13B. By neutralising circulating BAFF, a B-cell survival factor, belimumab prevents BAFF from binding its receptors on lymphocytes. The agent is approved for the treatment of systemic lupus erythematosus (SLE). Preclinical studies, including those by Tandler
et al., have demonstrated that BAFF impairs the therapeutic efficacy of small-molecule inhibitors such as idelalisib, ibrutinib, and venetoclax, as well as monoclonal antibodies such as rituximab, in patients with CLL. These findings suggest that therapeutic BAFF blockade with belimumab may restore or enhance CLL cells’ sensitivity to these agents [
26]. An ongoing phase II clinical trial is evaluating the efficacy of adding belimumab to a rituximab/venetoclax regimen in patients with relapsed or refractory CLL, with MRD negativity designated as the primary endpoint (#NCT05069051). The estimated primary completion date is July 2026 [
26].
2.4. Tislelizumab
Tislelizumab, a PD-1 monoclonal antibody approved for the treatment of advanced esophageal squamous cell carcinoma, is also under investigation in hemato-oncology, particularly in combination with zanubrutinib for patients with Richter’s transformation. The combination of tislelizumab plus zanubrutinib was evaluated in an open-label, phase II study among patients with Richter’s transformation (RT) who had received up to one prior RT-directed therapy. A total of 48 out of 59 patients who received at least two cycles of study treatment, including at least one administered dose in cycle three, were included in the analysis. Patients received tislelizumab intravenously at a fixed dose of 200 mg on day 1 of each 21-day cycle in combination with zanubrutinib at a fixed dose of 160 mg twice daily from day 1 onwards. The induction therapy consisted of six treatment cycles, followed by six consolidation cycles. The primary endpoint — ORR at interim staging (after induction therapy) was 58.3%, including 9 (18.8%) patients with CR and 19 (39.6%) with PR. The median PFS was 10 months, with a 12-month rate of 46.9%; the median overall survival was not reached (12-month overall survival rate, 74.7%). The most common side effects included hematological toxicities, gastrointestinal disorders, and infections [
27].
2.5. Pembrolizumab
Pembrolizumab, an anti–PD-1 inhibitor, demonstrated modest activity in combination with dinaciclib (CDK2/5/9 Inhibitor) in patients with R/R CLL in the phase Ib KEYNOTE-155 trial. Although the treatment was well tolerated, with any-grade adverse events (AEs) in 54 patients (predominantly fatigue, nausea, and anaemia), the ORR was 29.4%, and the median duration of response (DOR) was 10.3 months in R/R CLL. By the data cutoff, all patients had discontinued treatment, primarily (52.8%) due to progressive disease (PD). In conclusion, the observed ORR and DOR in the R/R CLL cohort demonstrated limited antitumor activity of pembrolizumab plus dinaciclib [
28,
29].
2.6. Atezolizumab
Atezolizumab is a PD-L1 immune checkpoint inhibitor that was investigated in a phase II trial of treatment-naïve CLL patients (NCT02846623). Between July 2019 and February 2023, 37 patients were enrolled and received combination therapy with obinutuzumab, venetoclax, and atezolizumab. A total of 31 patients completed 14 cycles of treatment, of whom 30 (97%) were bone marrow uMRD4. The median follow-up was 40 months. The 4-year PFS and OS estimates were 89% and 94%, respectively. Atezolizumab was discontinued in 3 patients due to immune-related adverse events [
30].
2.7. ABBV-319
The antibody-drug conjugate (ADC) ABBV-319 comprises a glucocorticoid receptor modulator (GRM) payload conjugated to a CD19 antibody. The structure of this drug enables three distinct mechanisms of action that drive antitumor activity in B cells: delivery of the GRM payload
via CD19 to induce apoptotic cell death, inhibition of downstream CD19 signaling, and enhanced ADCC
via afucosylation of the antibody backbone. In contrast to systemic glucocorticoids, ABBV-319 does not adversely affect NK cell function and may be associated with a lower risk of glucocorticoid-related adverse effects [
31]. Collectively, these encouraging preclinical findings provide the rationale for the ongoing phase I clinical trial (#NCT05512390) evaluating the safety, tolerability, and preliminary efficacy of ABBV-319 [
32].
2.8. Zilovertamab
Zilovertamab (Zilo), also known as cirmtuzumab, is a humanized monoclonal antibody directed against receptor tyrosine kinase-like orphan receptor 1 (ROR1). ROR1, an oncofetal surface antigen, is absent in normal adult tissues but is aberrantly expressed on a variety of aggressive hematologic malignancies and solid tumor cells. Zilo, in combination with ibrutinib, was evaluated in a phase I/II clinical trial (#NCT03088878). Among patients with CLL, the ORR was 91.2%, and at a median follow-up of 31.4 months, the median PFS was not reached. In part 3 of the study, the CLL patients were randomized 2:1 to receive Zilo with ibrutinib (Zilo+Ibr) or ibrutinib (Ibr) as monotherapy. At median follow-up of 21.1 months in Part 3, mPFS was not reached for both Zilo+Ibr and Ibr arms. The data from CLL patients compare favorably with those from Ibr monotherapy. Notably, in patients with
TP53 mutations treated in the Zilo+Ibr arm, the 30-month landmark PFS reached 100%, which is highly encouraging and supports further investigation of this combination in this high-risk patient population [
33].
In the ongoing single-center phase II trial (#NCT04501939), investigators are evaluating the efficacy of cirmtuzumab in combination with venetoclax as consolidation therapy in patients with CLL/SLL who have received venetoclax for at least 12 months and have detectable residual disease (>0.01% leukemia cells). The primary endpoint is the uMRD rate after 6 months of combination therapy. The study is expected to be completed in July 2026.
2.9. Zilovertamab Vedotin
Zilovertamab vedotin (ZV) is an innovative ADC consisting of zilovertamab, conjugated to the anti-microtubule cytotoxin monomethyl auristatin E (MMAE)
via a protease-cleavable maleimidocaproyl-valine-citrulline-para-aminobenzoate linker. This compound is currently being investigated in several clinical trials involving patients with B-cell lymphomas. Results from the phase I WAVELINE-001 trial demonstrated that ZV exhibits a manageable safety profile and encouraging antitumor activity in heavily pretreated patients with diffuse large B-cell lymphoma (DLBCL), mantle cell lymphoma (MCL), and Richter transformation (RT). A total of 56 patients were enrolled in Schedule 1; after 14 months of follow-up, the ORR was 29%, 53%, and 57% for DLBCL, MCL, and RT, respectively. Treatment-related AEs were reported in 41 patients (73%), with grade 3/4 AEs observed in 27 patients (48%), the most common being cytopenias. Although the results in the RT cohort are very encouraging, it should be noted that this cohort consisted of 7 patients [
34]. In the ongoing phase II WAVELINE-006 trial, ZV is being evaluated either as monotherapy or in combination with nemtabrutinib in patients with R/R B-cell malignancies. Currently, only preliminary data from Cohort C, which enrolled 28 patients with R/R MCL, have been reported. The median duration from first dose to data cutoff was 7.3 months. In addition to the expected hematologic toxicities, 19 patients (68%) developed peripheral neuropathy-related events, which included symptoms described as gait disturbance, muscle weakness, peripheral neuropathy, paresthesia, peripheral sensory neuropathy, and polyneuropathy. Grade 3/4 neuropathic events occurred in 3 patients (11%). The ORR was 64% with CR and PR observed in 9 patients (32%) each [
35]. Data for patients with R/R CLL and follicular lymphoma (FL) (Cohort D) are currently maturing and have not yet been reported.
Table 1.
Clinical trials evaluating the combination therapies with monoclonal antibodies in B-cell malignancies.
Table 1.
Clinical trials evaluating the combination therapies with monoclonal antibodies in B-cell malignancies.
| Agent |
Phase |
Antibody target |
No. of participants |
ORR |
CR/CRi |
PFS |
OS |
Most Common
Grade ≥ 3 AEs [%] |
Ref. |
| Ianalumab + ibrutinib |
Ib |
BAFF-R |
39 (expansion n=24) |
N/A |
37,5% |
N/A |
N/A |
Gr. ≥3 TEAEs 41.0%
TRAEs: 23.1% |
[23] |
| Tislelizumab + zanubrutinib |
II |
PD-1 |
59 (enrolled); 48 (comprised in full analysis set) |
58,3% |
18,8% |
Median:
10 months
46.9% at 1 year |
Median: not reached
74,7% at 12 months |
Gr. ≥1 TEAEs
98.2% (n=57)
Gastrointestinal disorders: 56.1%
Infections: 78.9% with UTI 21.1%
Pyrexia: 19.3%
Peripheral edema: 17.5%
Anaemia: 19.3%
Neutropenia: 21.1%
Thrombocytopenia: 19.3% |
[27] |
| Pembrolizumab + dinaciclib |
Ib |
PD-1 |
72 (enrolled); 17 (R/R CLL), 38 (R/R DLBCL), 17 (R/R MM) |
29.4% (R/R CLL) |
N/A |
Median: 5.2 months
(R/R CLL) |
Median: 21.7 months (R/R CLL) |
Gr. ≥3 TEAEs:
32% (n= 12);
Lymphopenia:13%; Neutropenia: 11%; Thrombocytopenia: 8%;
Leukopenia: 8%;
Laboratory TLS: 5% |
[28,29] |
| Atezolizumab + obinutuzumab |
II
|
PD-L1 |
37 (enrolled) |
N/A |
N/A |
94% at 2 years;
89% at 4 years
|
94% at 2 years
94% at 4 years |
Gr. ≥3 TEAEs:
Neutropenia: 59%; Thrombocytopenia: 32%
|
[30] |
| Zilovertamab + ibrutinib |
I/II |
ROR1 |
70 (enrolled), 34 (CLL),
26 (R/R MCL) |
91.2% |
8.8% |
Median: not reached |
N/A |
Gr. ≥3 TEAEs
Hypertension:10.6%;
Pneumonia: 7.1%; Neutropenia: 5.9%;
Atrial fibrillation: 5.9%;
Fatigue: 5.9%
TEAEs due to Zilo: 23.5% (CLL) |
[33] |
| Zilovertamab vedotin + nemtabrutinib |
I |
ROR1 |
56 (enrolled);
17 (DLBCL);
17 (MCL);
7 (RT) |
57% (RT)
|
14% (RT) |
Median: 4.7 months (RT) |
Median: 19.4 months (RT) |
Gr. ≥3 TEAEs:
48% (n=27);
Neutropenia: 32%;
Thrombocytopenia: 11% |
[34] |
3. Bispecific Antibodies
A relatively new class of agents that could potentially be highly effective in CLL therapy are T-cell engagers (TCEs) [
36]. In this group, bispecific antibodies (BsAbs), already widely used in other hematological malignancies (especially in relapsed or refractory cases) such as multiple myeloma (talquetamab, teclistamab, elranatamab), follicular lymphoma (mosunetuzumab), and DLBCL (epcoritamab, glofitamab), are involved in several promising CLL clinical trials [
37]. BsAbs (also trispecific and tetraspecific antibodies) function by redirecting the physiological function of polyclonal cytotoxic T lymphocytes toward malignant B-cells
via artificial connections between these two subgroups of white blood cells. The main advantage of BsAbs over CAR-T therapies is the avoidance of
ex vivo procedures—they are based on antibody structure, synthetically manufactured, and available off-the-shelf. Their components allow them to bind multiple antigens and influence other metabolic pathways, which could be beneficial in the context of double-refractory CLL [
38]. Numerous BsAbs are being explored as the potential CLL therapeutic option (
Table 2).
3.1. Epcoritamab
To date, the strongest evidence on the efficacy and safety of BsAbs in CLL therapy originates from epcoritamab clinical trials. In the multicenter, open-label, phase Ib/II EPCORE CLL-1 trial (#NCT04623541), the compound was administered to 42 patients with RT [
39]. Thirty-two were male, with a median age of 69 years. The median time from disease onset to Richter transformation was 7.6 years. Epcoritamab, a full-length CD20×CD3 IgG1 TCE, was administered subcutaneously in a step-up dosing regimen. In 50% of cases, epcoritamab was a first-line treatment. At a median follow-up of 22.9 months, the overall response rate (ORR) was 47.6%. In the first-line population and subsequent lines of therapy, the ORR was 57.1% and 38.1% respectively, while in patients with baseline
TP53 aberration and/or del(17p), it reached 40%. The most frequent grade 3 or 4 adverse events (AEs) according to CTCAE were neutropenia (45%), anemia (38%), thrombocytopenia (38%), infection (21%), pneumonia (10%), and COVID-19 (5%). Cytokine release syndrome (CRS) was observed in 86% of patients, with only 7% of grade 3. Immune effector cell-associated neurotoxicity syndrome (ICANS) of grade 1-2 occurred in 5 patients (12%), and only 2 developed clinical tumor lysis syndrome (TLS). Although three fatal AEs were reported, investigators stated that none were considered related to the study treatment.
In the EPCORE CLL-1 clinical trial, the efficacy of epcoritamab in combination with lenalidomide (Arm 2B), and epcoritamab in combination with R-CHOP (Arm 2C) in patients with RT was investigated [
40]. Arm 2B involved 11 patients – 6 male (55%) and 5 female (45%), and the median age was 74 years. Prior therapy for CLL or SLL was administered in 6 patients (55%). With a median follow-up of 12.6 months, ORR was 82%, and the CR rate was 73%. The estimated median PFS was 5.7 months while median OS was not reached. Frequent AEs were CRS (100%, grade 3/4 in 2 patients), neutropenia (82%), thrombocytopenia (73%), anemia, and hypokalemia (45% each). ICANS occurred in 2 patients (grade 1/2) and fatal AEs in 1 patient. Arm 2C involved 30 patients – 22 male (73%) and 8 female (27%), and the median age was 72 years. Prior therapy for CLL or SLL was administered in 17 patients (56%). With a median follow-up of 10.1 months, ORR was 73%, and the CR rate was 60%. The estimated median PFS and OS were 9.9 months and 16.4 months, respectively. Frequent AEs included CRS (60%), anemia (60%), neutropenia (60%), diarrhea (33%), and febrile neutropenia (30%). ICANS occurred in 4 patients (grade 1/2/3), and fatal AEs occurred in 3 patients.
In the same clinical trial, epcoritamab was also administered to 40 patients diagnosed with relapsed or refractory CLL [
41]. The expansion cohort consisted of 23 patients, and the optimization cohort of 17 patients – an additional cohort was created to mitigate CRS by introducing a step-up dose. The median follow-up was 22.8 months in the expansion cohort and 2.9 months in the optimization group. The median age was 71.5 years. Prior to epcoritamab administration, a median of 4 lines of therapy were administered (range, 2-10). All participants had previously received a BTKi; 88% had received chemoimmunotherapy, and 85% were double-exposed to a BTKi and a BCL2i. TP53 aberrations were confirmed in 63% of participants and unmutated IGHV (uIGHV) status was present in 70%. To date, only data from the expansion cohort have been presented. Epcoritamab therapy induced an ORR of 61% and a CR rate of 39%, with a median time to response of 2.0 months and a median time to CR of 5.6 months. There were no significant differences in the outcomes among the subgroups of double-exposed patients, those with TP53 aberrations, or those with uIGHV. The median PFS was 12.8 months, while the median OS was not reached. The most common nonhematologic AEs included CRS (96%, 17% grade 3; none led to therapy discontinuation), peripheral edema (48%), diarrhea (48%), fatigue (43%), and injection-site reactions (43%). Cytopenias were frequent, although most patients already had baseline anemia and thrombocytopenia. The four reported fatal AEs included cases of pneumonia, sepsis, and squamous cell carcinoma.
3.2. NVG-111
Another BsAb that has been extensively evaluated in recent years is NVG-111 – a first-in-class, humanized, tandem scFv, ROR1×CD3 bispecific T-cell engager administered as a continuous intravenous infusion over 21 days, followed by 7 days off drug. It is administered either in combination with ibrutinib or as monotherapy. In the open-label, dose-escalation study (#NCT04763083), 11 patients diagnosed with CLL—82% of whom had high-risk molecular features, including
TP53,
ATM, and
NOTCH1 mutations—and 3 patients with mantle cell lymphoma (MCL) received time-limited NVG-111 treatment. Among 12 patients evaluated for efficacy, 67% (n=8) experienced an objective response, including 33% (n=4) who achieved CR. Deeper responses were noted in patients who received additional cycles. Among all participants, the median PFS was 26.9 months. Among 13 patients evaluated for safety, adverse events occurred in all cases, most commonly during the first week. The most frequent were nausea (grade 1/2), headache, and fatigue. CRS of grade 1-2 was observed in 57% of patients, and ICANS (grade 2/3) in 14%. Grade 4 neutropenia was also observed in one patient [
42].
3.3. Mosunetuzumab
The next promising molecule is mosunetuzumab – a CD20×CD3 BsAb approved by the FDA for the treatment of R/R follicular lymphoma in adult patients who have undergone 2 or more lines of systemic therapy. There are three ongoing clinical trials examining mosunetuzumab as a potential treatment for patients with relapsed or refractory CLL (both as monotherapy and combined with venetoclax), for patients with RT (as a first-line therapy with CHOP), and for CLL with MRD clearance (both as monotherapy and combined with BTKi). None of the trial results have been published yet.
3.4. Glofitamab
Glofitamab is a CD20×CD3 BsAB with a novel 2:1 tumor-to-T-cell binding ratio, successfully redirecting a patient's own T cells to eradicate malignant B cells. Glofitamab therapy was tested effective for DLBCL [
43]. In the study by Carlo-Stella
et al., 11 patients with RT with a median age of 71 years old were treated with the glofitamab as monotherapy. ORR and CR were 63.6% and 45.5%, respectively. CRS occurred in 72.7% of patients (mostly gr. 1–2). ICANS-like neurologic AEs affected 5 patients (mostly gr 1). There were no fatal AEs or treatment discontinuations linked to glofitamab [
44]. Phase II study of glofitamab as monotherapy or in combination with polatuzumab vedotin, pirtobrutinib, or atezolizumab in RT is currently recruiting (#NCT06043674).
3.5. IGLV3-21R110-Directed Bispecific Antibody (R110-bsAB)
IGLV3-21R110 is a specific genetic point mutation in the B-cell receptor (BCR) of a high-risk, aggressive subset of patients with CLL. The R110-BsAb targeting the IGLV3-21R110 mutation and CD3 specifically eliminated both primary CLL cells and engineered cell lines overexpressing this neoepitope, utilizing T cells obtained from healthy donors and patients with CLL as effectors.
In vitro, R110-bsAb successfully preserved healthy polyclonal B cells and CD34+ hematopoietic stem cells.
In vivo, the R110-BsAb selectively eliminated CLL cells and cell lines expressing IGLV3-21R110, sparing normal peripheral blood mononuclear cells [
45].
Many other agents are currently being investigated in CLL therapy, including GB261 (a CD20×CD3 BiTE), surovatamig/AZD0486 (a CD19×CD3 BiTE) and JNJ-75348780 (a CD3×CD22 BiTE).
Table 2.
An overview of clinical trials examining the safety and efficacy of bispecific antibodies in CLL.
Table 2.
An overview of clinical trials examining the safety and efficacy of bispecific antibodies in CLL.
Agent
&
Target |
Agent characteristics |
Clinical Trial Number |
Trial characteristics |
No. of participants |
| Epcoritamab |
BsAb CD20×CD3 approved for DLBCL, FL and high-grade B-cell Lymphoma |
NCT04623541
(EPCORE CLL-1) |
Phase I/II study on epcoritamab monotherapy in R/R CLL. ORR: 61%, CR: 39%, mPFS: 12.8 months, mOS: not reached [41]. |
23 |
| Phase I/II study on epcoritamab monotherapy in RT. ORR: 47.6%, CR: 40%, mPFS: 3.0 months, mOS: 13.0 months [39]. |
42 |
| Phase I/II study on epcoritamab with lenalidomide in RT. ORR: 82%, CR: 73%, mPFS: 5.7 months, mOS: not reached [40]. |
11 |
| Phase I/II study on epcoritamab with R-CHOP in RT ORR: 61%, CR: 39%, mPFS: 12.8 months, OS: not reached [40]. |
30 |
| NCT07108998 |
Phase II study oCLL/SLL
(consolidation therapy for 2nd generation BTKi +/- obinutuzumab) |
22 (estimated) |
| NCT05791409 |
Phase I/II study on epcoritamab with venetoclax in R/R CLL/SLL. Estimated study completion in 2032. |
112 (estimated) |
| NCT06676033 |
Phase I study on epcoritamab in CLL and RT. Estimated study completion in 2027. |
5 |
NCT07218510
(LonGEVity Trial) |
Phase II study on epicoritamab as a consolidation therapy for venetoclax and obinutuzumab in previously untreated CLL/SLL. Estimated study completion in 2029. |
33 (estimated) |
| Mosunetuzumab |
humanized BsAb CD3×CD20 approved for R/R FL |
NCT05091424 |
Phase I study of mosunetuzumab alone or in combination with venetoclax in R/R CLL. Estimated study completion in 2030. |
137 (estimated) |
| NCT06926205 |
Phase II study of mosunetuzumab with CHOP as a first line in RT. Estimated study completion in 2028. |
34 (estimated) |
| NCT07052695 |
Phase I/II study of mosunetuzumab alone or in combination with BTKi in CLL/SLL. Estimated study completion in 2032. |
40 (estimated) |
| NCT02500407 |
Phase I/II study on mosunetuzumab monotherapy and with atezolizumab in R/RCLL and B-cell NHL. Trial completed. |
713 |
GB261
CD20×CD3 |
BsAb CD20×CD3 computationally designed to maintain Fc effector function |
NCT04923048* |
Phase I/II study in CLL and R/R B-cell NHL. Unknown status. |
460 (estimated) |
AZD0486
(Surovatamig) |
IgG4 fully human BsAb CD19×CD3 |
NCT06564038 |
Phase I/II study on AZD0486
as monotherapy or in combination with other anticancer agents in R/R CLL/SLL and other mature B-Cell malignancies. Estimated study completion 2028. |
276 (estimated) |
NVG111
ROR1×CD3 |
first in class, humanized, tandem scFv, ROR1xCD3 BsAb |
NCT04763083* |
Phase I study in R/R ROR1+ malignancies. Unknown status of completion. |
90 (estimated) |
ONO4685
PD1×CD3 |
First in class PD1×CD3 BsAb |
NCT06547528 |
Phase I study in CLL/SLL and T-cell lymphoma. Estimated completion 2029. |
108 (estimated) |
JNJ 75348780
CD22×CD3 |
CD22×CD3 human BsAb |
NCT04540796 |
Phase I study in R/R CLL NHL completed in 2025. |
147 |
4. Cell Therapies
In the past decade, CAR-T cell therapies have been effectively integrated into the therapeutic landscape for several hematologic malignancies, including B-cell non-Hodgkin lymphomas (NHLs) [
46,
47,
48,
49,
50,
51] and B-acute lymphoblastic leukemia [
52,
53]. The use of CAR-T cells in CLL has proven to be challenging. However, recent studies suggest that they could provide a successful therapeutic option for patients with relapsed or refractory disease (
Table 3).
4.1. Challenges
The application of CAR-T cell therapy in CLL has faced significant challenges. Immunosuppressive tumor microenvironment and T cell exhaustion play key roles [
54,
55,
56]. In CLL, chronic antigen exposure drives T cells into exhaustion, leading to reduced expansion, persistence, and functional capacity. Riches
et al. showed that T-cells from patients with CLL exhibit elevated levels of exhaustion markers, including CD244, CD160, and PD-1. These T cells exhibited impaired proliferation and cytotoxicity, with the cytolytic deficit linked to defective granzyme loading into vesicles and disorganized, nonpolarized degranulation [
54]. Patients with CLL who exhibit higher levels of exhaustion markers, including PD-1, TIM-3, and LAG-3, tend to have reduced responses to CAR-T cell therapy [
57]. The tumor microenvironment (TME) supports the growth and survival of CLL leukemic cells. It provides a supportive niche, and its interactions with CLL cells drive disease progression and reduce treatment efficacy. The TME includes stromal cells that maintain cell-to-cell communication and support leukemia cell growth, as well as immune-suppressive cells such as nurse-like cells, myeloid-derived suppressor cells, and regulatory T cells [
58]. Nurse-like cells interact with CLL cells, reducing CLL cells’ SDF-1 receptors and thereby protecting them from spontaneous apoptosis [
59]. Indoleamine 2,3-dioxygenase-secreting myeloid-derived suppressor cells have been shown to inhibit T cell activity and promote the development of regulatory T cells [
60]. Resistance mechanisms contribute to CLL refractoriness. Therefore, new CAR-T cell therapies should be designed to overcome them. Efforts to utilize CAR-T cell therapy in CLL include combining therapeutic agents, CAR design, and using allogenic CAR-T cells.
4.2. Autologous CD19 CAR-T Cells
4.2.1. Lisocabtagene Maraleucel
Lisocabtagene maraleucel (liso-cel) is an autologous CD19-targeted CAR-T cell therapy composed of defined and equal doses of CD8⁺ and CD4⁺ CAR-positive T cells with a 4-1BB costimulatory domain [
61]. In March 2024, the FDA approved liso-cel for patients who have progressed after at least two lines of therapy, including a BTKi and BCL-2 inhibitor. The approval was based on encouraging data from the TRANSCEND CLL 004 trial (phase I/II) [
21]. One hundred and seventeen CLL patients with BTKi failure, including 70 with venetoclax failure, received liso-cel. Patients were treated with liso-cel at two dose levels: 50×10⁶ CAR-T cells or 100×10⁶ CAR-T cells. Among the efficacy-evaluable population (n = 96), the ORR was 48%, with a CR/CRi observed in 18% patients. Among patients who progressed on a BTKi and did not respond to venetoclax
, response rates were similar: ORR of 43% and CR rate of 18%. In the efficacy-evaluable population, the DOR was approximately 35 months, and the median PFS was 11.9 months. Among patients previously treated with both a BTKi and venetoclax, the median DOR was also 35 months, while the median PFS was 12 months. In the double-refractory patients, uMRD was achieved in 64% of cases in peripheral blood and 59% in bone marrow (at 10
-5 sensitivity). Ten patients (9%) had grade 3 CRS; none had grade 4 or 5 CRS. Twenty-one patients (18%) experienced grade 3 neurotoxicity, and one patient (1%) experienced grade 4 neurotoxicity. At 24 months of follow-up, disease responses remained durable with consistently high rates of uMRD and no new safety concerns observed [
62].
4.2.2. Tisagenlecleucel
Tisagenlecleucel (Tisa-Cel), a second-generation anti-CD19 CAR-T cell therapy featuring a 4-1BB costimulatory domain and CD8α hinge, was the first CD19-targeting CAR-T cell therapy to demonstrate early success in B-cell malignancies [
63]. In the trial, 14 patients with R/R CLL received tisa-cel. At a median follow-up of 19 months, the ORR was 57%, with 4 CRs and 4 PRs. Among the 14 treated and evaluable patients, median OS was 29 months, with an 18-month OS rate reaching 71%. The median PFS was 7 months, and the 18-month PFS was 28.6%. The infusions were generally well tolerated. Neutropenia-related complications, such as fever, and delayed CRS were the most common treatment-related AEs. A recent follow-up confirmed that two of the patients remain in MRD-negative remission ten years after treatment, with detectable circulating CAR-T cells [
64].
4.2.3. Axicabtagene Ciloleucel
Axicabtagene ciloleucel (axi-cel) is another autologous anti-CD19 CAR-T cell therapy. It also utilizes the CD28 domain but does not involve 4-1BB, in contrast to liso-cel and tisa-cel. Cappell
et al. reported long-term results of axi-cel therapy in seven heavily pretreated CLL patients. They observed an ORR of 88% and a CR rate of 63%. The median duration of sustained response was 82 months, with 50% of the patients maintaining a response for more than three years [
65].
4.2.4. Brexucabtagene Autoleucel
Brexucabtagene autoleucel (brexu-cel) has the same structure as axi-cel, however it undergoes an additional T cell selection process during manufacturing to remove leukemic cell contamination [
49]. In the ZUMA-8 trial, 15 enrolled patients were divided into four cohorts: cohorts 1 and 2 received brexu-cel at doses of 1×10⁶ and 2×10⁶ cells/kg, respectively; cohort 3 included patients with low tumour burden, and cohort 4 included patients with ibrutinib as their last line of therapy. With a median follow-up of 24.3 months, one dose-limiting toxicity (grade 4 CRS) was reported in cohort 3. Grade ≥3 neurologic events occurred in 3 patients (20%). The ORR reached 47%, including one CR (7%). All cohort 3 patients responded. CAR-T cell expansion occurred in 4 patients (27%). CAR-T cell expansion and responses were observed in patients with low tumour burden. Due to suboptimal CAR-T cell expansion in most patients, the ZUMA-8 trial was terminated early [
66].
4.2.5. GLPG5201
GLPG5201 is a second-generation CAR-T cell therapy targeting CD19 with a 4-1BB co-stimulatory domain, given as a one-time fixed intravenous dose. It was evaluated in EUPLAGIA-1 — a Phase I/II, open-label, multicenter trial involving patients with R/R CLL, R/R SLL, and RT. In December 2024, the initial safety and efficacy results were presented. All 15 enrolled patients had R/R CLL, and 9 also had RT. Of 15 efficacy-evaluable patients, 13 responded (86.7% ORR) and 10 achieved CR (66.7%). At a median follow-up of 6 months, 8/10 patients who achieved CR, (80%) had an ongoing CR. GLPG5201 demonstrated a favourable safety profile, with the majority of grade ≥3 treatment-related AEs being hematological. No grade ≥3 CRS or any-grade ICANS were observed [
67].
4.2.6. huCART19-IL18
huCART19-IL18, an armoured CAR-T cell therapy engineered to target CD19 and secrete interleukin-18 to enhance antitumor effects, was evaluated in 21 patients with R/R NHLs. The safety profile was manageable: CRS occurred in 62% of patients, mostly mild or moderate (47% grade 1–2), and ICANS was reported in 14%, all grade 1–2. Importantly, no unexpected AEs were observed. CAR-T cell expansion was robust across all dose levels. At 3 months post-infusion, ORR was 81%, with 52% achieving a CR. After a median follow-up of 17.5 months, the median DOR was 9.6 months [
68].
4.2.7. Varnimcabtagene Autoleucel
Varnimcabtagene autoleucel (ARI-0001) is an autologous second-generation CAR-T cell product targeting CD19 with 4-1BB costimulation, developed entirely at Hospital Clínic in Barcelona, Spain. It was approved in Spain for the treatment of R/R B-cell ALL. Between 2017 and 2024, 13 patients with R/R CLL and 16 with RT were treated with ARI-0001. CRS was observed in 90% of patients; however, grade ≥3 cases were observed only in 3.4%. Only 2 cases (6.9%) of ICANS were reported, all grade ≤2. In the CLL cohort, the ORR and CR rates were both 84.6%. With a median follow-up of 1.3 years, the median DOR, PFS, and OS were not reached. The median duration of circulating CAR-T cells was 3 years [
69].
4.2.8. JCAR014
JCAR014 — another CD19 CAR-T cell therapy with a 1:1 ratio of CD8+: CD4+ was tested in a phase I/II clinical trial, which enrolled 47 R/R CLL patients, including 9 with prior or current RT. 94% of patients had high-risk cytogenetics. Median follow-up was 79.6 months. Median DOR was 18.9 months. Median PFS was 8.9 months, and the 6-year PFS was 17.8%. By day 28, OR and CR rates were 70% and 17%, respectively. The 6-year DOR and OS were 26% and 31%, respectively. CRS and neurotoxicity occurred in 82% (14% grade ≥3) and 33% (27% grade ≥3) of patients, respectively [
70].
4.2.9. HD-CAR-1
HD-CAR-1 is a third-generation autologous CD19-directed CAR-T cell product featuring two costimulatory domains CD28 and 4-1BB. It was tested in the phase I/II trial in patients with double R/R CLL. By day 90, six patients (67%) achieved a CR, with five of them (83%) showing uMRD. With a median follow-up of 27 months, 2-year PFS and OS were 30% and 69%, respectively. In non-responders, there was a marked enrichment of effector memory-like CD8+ T cells with high expression of CD39 and/or CD197. Treatment-specific toxicity was exceptionally low, with only one case of grade 3 CRS and no neurotoxicity [
71].
4.2.10. BAFF-R CAR T-Cell Therapy
Another potential target is the BAFF-R, which serves as a specific marker involved in B-lymphocyte development and the survival of mature B cells [
72]. BAFF-R is especially significant in CLL because it is highly expressed in the clonally expanded population of mature B cells [
73]. BAFF-R CAR T-cell therapy demonstrated cytotoxic activity against both CLL cell lines and primary B-cells obtained from CLL patients. Moreover, these CAR-T cells were effective against CD19-knockout CLL cells that are resistant to CD19 CAR-T cell therapy [
74]. Two phase I clinical trials are currently recruiting patients to evaluate BAFF-R CAR-T cell therapy for the treatment of R/R CLL (#NCT06191887, #NCT06916767).
4.3. Dual-Target CAR-T Cells
The development of dual-targeting methods represents a significant advancement in preventing antigen escape and enhancing therapeutic results. The CD19/CD22-targeting CAR-T cells were tested in two patients who developed DLBCL-RT 2 years after CLL diagnosis. Both patients remained in CR (17- and 7-months post-infusion) with good tolerability and no severe infections. Both experienced grade 1 CRS. Patient 1 showed rapid CAR-T cell expansion, and patient 2 showed moderate CAR-T cell expansion [
75]. A Phase I/II study of CD19/CD22-targeting CAR-T cells for the treatment of R/R CD19/CD22-positive B-ALL and indolent and aggressive B-cell lymphomas is currently recruiting (#NCT06834529).
Safety and outcomes of lentiviral bispecific anti-CD20/anti-CD19 (LV20.19) CAR-T cells were evaluated in 14 patients with R/R RT and CLL. All developed CRS and 93% required tocilizumab. Sixty-four percent of patients developed immune effector cell–associated hemophagocytic lymphohistiocytosis-like syndrome (IEC-HS), a hyperinflammatory complication of immune effector cell-based therapies characterized by macrophage-activation features such as cytopenias, hyperferritinemia, and coagulopathy. Among these, two patients with CLL experienced grade 3 and grade 4 IEC-HS, respectively. At day 28, the ORR was 92% among evaluable patients (n=13). All responders (n=12) had disease-negative bone marrow. With a median follow-up of 11 months, only one RT patient relapsed. The median DOR was not reached, and the median OS was 15 months. Although LV20.19 CAR-T cells showed efficacy in CLL and RT, the high incidence of IEC-HS in CLL patients raises concerns [
76].
Other dual-targeting CAR-T cell therapies are currently under investigation, including CD19/BAFF-R dual-targeted CAR-T cells. [
77].
4.4. Triple-Target CAR-T Cell Therapy
Targeting three B-cell antigens simultaneously is a new approach to prevent antigen-negative relapse. The safety of administering CAR-T cells targeting CD19, CD20, and CD22 (TriCAR19.20.22 T cells) will be assessed in patients with R/R CLL, with or without RT. CAR-T cells will be infused after an immunosuppressive conditioning regimen with fludarabine and cyclophosphamide (#NCT07166419).
4.5. Allogeneic CAR-T Cells
Allogeneic CAR-T cells can serve as an “off-the-shelf” alternative to autologous CAR-T cell therapy. Instead of being made from a patient’s own T-cells, they are prepared in advance from healthy donor cells. This method enables immediate treatment, which is vital for patients with rapidly advancing leukemia, and removes the risk of leukemic cell contamination that can occur when using a patient’s own cells [
78].
CARCIK-CD19 is a donor-derived, CD19-targeted, cytokine-induced CAR-T cell therapy engineered using the non-viral Sleeping Beauty transposon system [
79]. A Phase I/II clinical trial evaluating its safety and clinical activity in patients with CLL is currently recruiting (#NCT05869279). The ARDENT trial is evaluating SC291, an “off-the-shelf”, hypoimmune-modified, CD19-directed allogeneic CAR-T therapy for R/R B-cell malignancies, including CLL. The CAR-T cells feature HLA depletion and CD47 overexpression to prevent rejection (#NCT05878184). Another investigational allogeneic CAR-T therapy for CLL is ALLO-501A. It involves disrupting the TCRα constant gene to reduce the risk of GvHD and editing the CD52 gene, enabling the use of a humanized anti-CD52 mAb to selectively deplete host T cells while sparing the donor CAR-T cells [
80]. Safety and efficacy of ALLO-501A in patients with R/R CLL are assessed in the ALPHA-2 trial (#NCT04416984).
CTX112 is an allogeneic, CRISPR-Cas9-engineered CAR-T cell therapy that features five precise edits: site-specific insertion of an anti-CD19 CAR into the TRAC locus and targeted disruption of the TRAC, B2M, TGFBR2, and ZC3H12A genes to prevent GvHD, evade immune rejection, resist immunosuppression, and enhance persistence [
81]. The Phase I/II open-label, multicenter trial is evaluating the safety and effectiveness of CTX112 in patients with relapsed or refractory (R/R) B-cell malignancies, including CLL.
4.6. Combination Therapies with CAR-T Cells
4.6.1. Ibrutinib
Combining CAR-T cell therapy with ibrutinib is gaining attention for enhancing efficacy and minimizing adverse effects. Ibrutinib improves activated T cell survival, lowers Treg/CD4⁺ ratios, and inhibits CLL-mediated immune suppression through both BTK inhibitor-related and unrelated mechanisms [
82]. Simultaneous administration of ibrutinib improves both engraftment and therapeutic efficacy of anti-CD19 CAR-T cells in murine models of CLL [
83]. Ibrutinib also decreases CRS caused by CAR-T cells [
84]. In a study by Gauthier et al., 19 CLL patients received ibrutinib ≥2 weeks before leukapheresis and continued it for at least 3 months post–CAR-T cell therapy. At 4 weeks, the iwCLL 2018 ORR was 83%, with 61% achieving MRD-negative marrow by IGH sequencing. Concurrently, ibrutinib reduced CRS severity and cytokine levels compared to CAR-T–only treatment [
85]. Liso-cel combined with ibrutinib for R/R CLL/SLL was also assessed in the dose-escalation cohort of the phase I/II TRANSCEND CLL 004 study. Among 19 patients with ≥1-month follow-up, 95% responded. All responses occurred by day 30, and 89% remained ongoing at ≥6 months. Eighty-nine percent of patients achieved MRD negativity in peripheral blood samples
via flow cytometry, and 79% did in bone marrow by NGS. No dose-limiting toxicities occurred. Ibrutinib-related AEs were observed in 79% of patients, with 37% ≥ grade 3. AEs led to dose reductions in 2 patients and discontinuations in 4. CRS occurred in 74% of patients (1 grade 3), and 32% experienced neurological events (3 ≥ grade 3). Tocilizumab and/or corticosteroids for the management of its management were required in 42% of patients [
86]. Gill
et al. conducted a single-center phase II trial adding autologous huCART-19 cells to ibrutinib in CLL patients who did not achieve CR after ≥6 months of ibrutinib. Primary endpoints were safety, feasibility, and CR within 3 months. Of 20 enrolled patients, 19 received huCART-19. Median follow-up was 41 months. CRS occurred in 18 patients (grades 1–2 in 15, grade 3 in 2, grade 4 in 1), and 5 experienced neurotoxicity (grades 1–2 in 4, grade 4 in 1). The 3-month CR rate was 44%, and the 12-month MRD negativity was 72%. The estimated 48-month OS and PFS were 84% and 70%, respectively. Of 15 patients with early MRD negativity, 13 remained in ongoing CR. In CLL patients who did not achieve CR with ibrutinib alone, huCART-19 induces deep and durable remissions [
87].
4.6.2. Lenalidomide
An immunomodulatory agent, lenalidomide, is also being studied in combination therapies. When used with CD23-targeted CAR-T cells, lenalidomide maintained CAR-T cell cytotoxicity, cytokine production, proliferation, and the formation of immune synapses between CAR-T and CLL cells in vitro. In a xenograft model of CLL, this combination improved CAR-T cell migration to leukemic sites and slowed disease progression [
88].
4.6.3. PI3K Inhibition
PI3K inhibitors can cause T cell–mediated autoimmunity, and it is hypothesized that temporarily inhibiting PI3K during CAR-T cell production could improve their effectiveness in patients with CLL. The impact of adding the dual PI3Kδ/γ inhibitor duvelisib during CAR-T cell manufacturing was evaluated. Duvelisib-treated CAR-T therapy (Duv-CAR-T) normalized CD4/CD8 ratios, increased the number of T-stem cell memory, naïve, and memory cells, and boosted cytotoxicity against CD19⁺ CLL. In CLL xenografts, Duv-CAR-T cells expanded more, eliminated leukemia faster, persisted longer, and improved survival compared to conventional CAR-T cells [
89].
4.7. Allogeneic CAR-NK Cells
There is increasing enthusiasm for using NK cells in CAR engineering, as they provide several benefits, including ADCC, their ability to mediate GvL activity without causing GvHD, and the option to source them from multiple origins, such as healthy donor blood or cord blood, enabling an “off-the-shelf” approach [
90,
91,
92]. Liu et al. demonstrated that CD19-targeted CAR-NK cells effectively eliminated CD19-positive leukemia cells in vitro and significantly extended survival in a xenograft mouse model [
93]. In the dose-escalation phase of the phase I/II trial, the anti-CD19 CAR-NK cells derived from cord blood were administered to 11 patients with R/R NHL or R/R CLL. None of the patients developed CRS, neurotoxicity or GVHD. Among the 11 patients treated, 73% responded to therapy, with 64% achieving CR [
94]. The expansion phase included 26 additional patients. In total 37 patients were treated. No significant toxicities, including CRS, neurotoxicity, or GvHD were reported. The ORR at days 30 and 100 was 49% each, while the 1-year OS and PFS rates were 68% and 32%, respectively. Patients who achieved a response showed greater CAR-NK cell expansion and longer persistence [
95].
Table 3.
Clinical trials evaluating selected cell therapies for CLL patients.
Table 3.
Clinical trials evaluating selected cell therapies for CLL patients.
| Agent |
Phase |
Target |
No. of Parti-cipants |
ORR |
CR / CRi |
PFS rate |
OS rate |
Safety |
Ref. |
| Lisocabtagene maraleucel (liso-cel) |
I / II |
CD19 |
117 |
47% |
18% |
Median: 11.9 months |
Median 30.3 months |
CRS gr. ≥3: 9%; NT gr ≥3: 19% |
[21] |
| Tisagenlecleucel (tisa-cel) |
I |
CD19 |
14 |
57% |
29% |
Median: 7 months |
Median: 29 months |
Neutropenia fever, delayed CRS |
[63] |
| Axicabtagene ciloleucel (axi-cel) |
I/II |
CD19 |
7 |
88% |
63% |
N/A |
Median OS not reached |
N/A |
[65] |
| Brexucabtagene autoleucel (brexu-cel) |
I |
CD19 |
15 |
47% |
13% |
N/A |
N/A |
CRS gr. 4: 7%; NT gr ≥3: 20% |
[66] |
| GLPG5201 |
I / II |
CD19 |
15 |
93% |
66.7% |
N/A |
N/A |
No grade ≥3 CRS or any-grade ICANS were observed |
[67] |
| huCART19-IL18 |
I |
CD19 |
21 (R/R NHL) |
81% at 3 months |
52% |
N/A |
N/A |
CRS: 62% (gr. ≥3: 15%); ICANS: 14% (all gr. 1-2) |
[68] |
| Varnimcabtagene autoleucel (ARI-0001) |
I |
CD19 |
13 (CLL cohort) |
84.6% |
84.6% |
60% at 4 years |
Not reached |
CRS: 90% (gr ≥3: 3.4%); ICANS gr.2: 6.9% |
[69] |
| JCAR014 |
I / II |
CD19 |
47 |
70% |
17% |
Median: 8.9 months |
Median: 25 months |
CRS: 82% (Gr ≥3: 14%); NT: 33% (Gr ≥3: 27%) |
[70] |
| HD-CAR-1 |
I / II |
CD19 |
9 |
67% |
67% |
30% at 2 years |
69% at 2 years |
CRS gr. 3: 9%; no NT |
[71] |
| LV20.19 |
I |
CD20 / CD19 |
14 (RT and CLL) |
92% |
46% |
N/A |
Median: 15 months |
CRS: 100% (gr. ≥3: 14.3%); ICANS: 21% (gr. 3: 14.3%); IEC-HS: 64% (Gr ≥3: 14.3%) |
[76] |
| Liso-cel + Ibrutinib |
I |
CD19 |
19 |
95% |
63% |
N/A |
N/A |
CRS: 74% (Gr ≥3: 5%); NT: 32% (gr ≥3: 16%) |
[86] |
| huCART-19 + Ibrutinib |
II |
CD19 |
19 |
83% |
43% at 3 months |
84% at 4 years |
70% at 4 years |
CRS: 94.7% (gr. ≥3: 15.8%); NT: 26.3% (gr. 4: 5.3%) |
[87] |
| Anti-CD19 CAR-NK |
I / II |
CD19 |
37 |
49% |
37.8% |
32% at 1 year |
68% at 1 year |
No significant CRS, NT, or GvHD |
[95] |
5. Conclusions
The therapeutic paradigm of CLL has undergone a fundamental transformation, evolving from chemotherapy to chemoimmunotherapy and now to highly effective, mechanism-driven targeted agents. The widespread adoption of BTKi and BCL2i has markedly reduced reliance on classical anti-CD20 monoclonal antibodies, whose role is now largely confined to selected clinical contexts. Nevertheless, monoclonal antibodies remain a biologically compelling platform, and the identification of novel targets such as ROR1, BAFF-R, and other disease-specific antigens underscores their continued relevance. Next-generation antibodies may yet redefine combination strategies, particularly in overcoming therapeutic resistance and refining immune-mediated tumor control.
More importantly, the future of CLL therapy is increasingly likely to be defined by immunotherapeutic approaches that actively engage cellular cytotoxicity. Bispecific antibodies capable of redirecting endogenous T cells have demonstrated the ability to induce deep responses independent of conventional resistance pathways in CLL and RT patients, representing a highly promising and scalable strategy. Similarly, CAR-T cell therapies offer the prospect of profound and potentially durable disease eradication, challenging the traditional concept of continuous treatment in CLL. Advances in CAR design, toxicity mitigation, and manufacturing platforms are expected to broaden their applicability and improve therapeutic indices, making them a potential „off-the-shelf” therapy as well.
In parallel, natural killer (NK) cell-based therapies are emerging as a compelling complementary modality. NK cells provide innate, MHC-independent antitumor activity with a potentially more favorable safety profile, offering opportunities for off-the-shelf cellular therapies and innovative combinatorial approaches. The convergence of NK-cell platforms with antibody engineering and cellular redirection technologies may represent a critical next step in immunotherapy development.
Despite their transformative potential, major questions remain unresolved. The optimal sequencing and integration of BsAb, CAR-T cells, and NK-cell therapies in comparison to established targeted agents are unknown, especially regarding long-term efficacy and safety. Determining which patients derive the greatest benefit, how to balance efficacy against immune-related toxicities, and whether these modalities can deliver sustained treatment-free remissions requires rigorous prospective evaluation. Future progress will depend on biomarker-driven trial designs, deeper understanding of immune escape mechanisms, and rational combination strategies. In conclusion, while small-molecule inhibitors currently anchor CLL management, the trajectory of innovation clearly points toward cellularly engaged immunotherapies. Bispecific antibodies, CAR-T cells, and NK-cell–based strategies are poised not merely to complement existing treatments but to potentially redefine therapeutic objectives. Establishing their optimal use now represents one of the most critical priorities in contemporary CLL research.
Author Contributions
BP designed the study. All authors contributed to writing and reviewing the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data availability statement
Not applicable.
Conflicts of interest
The authors declare no conflicts of interest.
References
- Hallek, M.; Cheson, B.D.; Catovsky, D.; Caligaris-Cappio, F.; Dighiero, G.; Döhner, H.; Hillmen, P.; Keating, M.; Montserrat, E.; Chiorazzi, N.; et al. iwCLL guidelines for diagnosis, indications for treatment, response assessment, and supportive management of CLL. Blood 2018, 131, 2745–2760. [Google Scholar] [CrossRef]
- Eichhorst, B.; Robak, T.; Montserrat, E.; Ghia, P.; Niemann, C.U.; Kater, A.P.; Gregor, M.; Cymbalista, F.; Buske, C.; Hillmen, P.; et al. Chronic lymphocytic leukaemia: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2021, 32, 23–33. [Google Scholar] [CrossRef]
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma Version 2025; National Comprehensive Cancer Network (NCCN), 2025. [Google Scholar]
- Hallek, M. Chronic Lymphocytic Leukemia: 2025 Update on the Epidemiology, Pathogenesis, Diagnosis, and Therapy. Am J Hematol 2025, 100, 450–480. [Google Scholar] [CrossRef]
- Robak, T.; Iskierka-Jażdżewska, E.; Puła, B. XXI International Workshop on Chronic Lymphocytic Leukemia in Kraków 2025 — conference report. Acta Haematologica Polonica 2025, 56, 477–482. [Google Scholar] [CrossRef]
- Hallek, M.; Fischer, K.; Fingerle-Rowson, G.; Fink, A.M.; Busch, R.; Mayer, J.; Hensel, M.; Hopfinger, G.; Hess, G.; von Grünhagen, U.; et al. Addition of rituximab to fludarabine and cyclophosphamide in patients with chronic lymphocytic leukaemia: a randomised, open-label, phase 3 trial. Lancet 2010, 376, 1164–1174. [Google Scholar] [CrossRef]
- Fischer, K.; Bahlo, J.; Fink, A.M.; Goede, V.; Herling, C.D.; Cramer, P.; Langerbeins, P.; von Tresckow, J.; Engelke, A.; Maurer, C.; et al. Long-term remissions after FCR chemoimmunotherapy in previously untreated patients with CLL: updated results of the CLL8 trial. Blood 2016, 127, 208–215. [Google Scholar] [CrossRef]
- Stilgenbauer, S.; Zenz, T. Understanding and managing ultra high-risk chronic lymphocytic leukemia. Hematology Am Soc Hematol Educ Program 2010, 2010, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Hus, I.; Giannopoulos, K.; Jamroziak, K.; Wolowiec, D.; Roliński, J.; Robak, T. Diagnostic and therapeutic recommendations of the Polish Society of Haematologists and Transfusiologists and Polish Adult Leukemia Group-CLL for chronic lymphocytic leukemia in 2025. Acta Haematologica Polonica 2025, 56, 143–172. [Google Scholar] [CrossRef]
- Wolska-Washer, A.E.; Robak, T. Acalabrutinib in treatment of patients with chronic lymphocytic leukemia including those at high genetic risk. Acta Haematologica Polonica 2025, 56, 172–186. [Google Scholar] [CrossRef]
- Sharman, J.P.; Egyed, M.; Jurczak, W.; Skarbnik, A.; Pagel, J.M.; Flinn, I.W.; Kamdar, M.; Munir, T.; Walewska, R.; Corbett, G.; et al. Acalabrutinib with or without obinutuzumab versus chlorambucil and obinutuzmab for treatment-naive chronic lymphocytic leukaemia (ELEVATE TN): a randomised, controlled, phase 3 trial. Lancet 2020, 395, 1278–1291. [Google Scholar] [CrossRef] [PubMed]
- Seymour, J.F.; Kipps, T.J.; Eichhorst, B.; Hillmen, P.; D'Rozario, J.; Assouline, S.; Owen, C.; Gerecitano, J.; Robak, T.; De la Serna, J.; et al. Venetoclax-Rituximab in Relapsed or Refractory Chronic Lymphocytic Leukemia. N Engl J Med 2018, 378, 1107–1120. [Google Scholar] [CrossRef] [PubMed]
- Quartermaine, C.; Ghazi, S.M.; Yasin, A.; Awan, F.T.; Fradley, M.; Wiczer, T.; Kalathoor, S.; Ferdousi, M.; Krishan, S.; Habib, A.; et al. Cardiovascular Toxicities of BTK Inhibitors in Chronic Lymphocytic Leukemia: JACC: CardioOncology State-of-the-Art Review. JACC CardioOncol 2023, 5, 570–590. [Google Scholar] [CrossRef] [PubMed]
- Zygmunciak, P.; Dancewicz, H.; Stróżna, K.; Błażowska, O.; Bieliński, K.; Robak, T.; Puła, B. Double Strike in Chronic Lymphocytic Leukemia-The Combination of BTK and BCL2 Inhibitors in Actual and Future Clinical Practice. Int J Mol Sci 2025, 26. [Google Scholar] [CrossRef]
- Zygmunciak, P.; Robak, T.; Puła, B. Treatment of Double-Refractory Chronic Lymphocytic Leukemia-An Unmet Clinical Need. Int J Mol Sci 2024, 25. [Google Scholar] [CrossRef]
- Robak, T.; Iskierka-Jażdżewska, E.; Puła, A.; Robak, P.; Puła, B. The Development of Novel Therapies for Chronic Lymphocytic Leukaemia in the Era of Targeted Drugs. J Clin Med 2025, 14. [Google Scholar] [CrossRef]
- Ramsay, A.G.; Gribben, J.G. Immune dysfunction in chronic lymphocytic leukemia T cells and lenalidomide as an immunomodulatory drug. Haematologica 2009, 94, 1198–1202. [Google Scholar] [CrossRef]
- Riches, J.C.; Davies, J.K.; McClanahan, F.; Fatah, R.; Iqbal, S.; Agrawal, S.; Ramsay, A.G.; Gribben, J.G. T cells from CLL patients exhibit features of T-cell exhaustion but retain capacity for cytokine production. Blood 2013, 121, 1612–1621. [Google Scholar] [CrossRef]
- Kipps, T.J. ROR1: an orphan becomes apparent. Blood 2022, 140, 1583–1591. [Google Scholar] [CrossRef]
- Choi, M.Y.; Widhopf, G.F., 2nd; Ghia, E.M.; Kidwell, R.L.; Hasan, M.K.; Yu, J.; Rassenti, L.Z.; Chen, L.; Chen, Y.; Pittman, E.; et al. Phase I Trial: Cirmtuzumab Inhibits ROR1 Signaling and Stemness Signatures in Patients with Chronic Lymphocytic Leukemia. Cell Stem Cell 2018, 22, 951–959.e953. [Google Scholar] [CrossRef]
- Siddiqi, T.; Maloney, D.G.; Kenderian, S.S.; Brander, D.M.; Dorritie, K.; Soumerai, J.; Riedell, P.A.; Shah, N.N.; Nath, R.; Fakhri, B.; et al. Lisocabtagene maraleucel in chronic lymphocytic leukaemia and small lymphocytic lymphoma (TRANSCEND CLL 004): a multicentre, open-label, single-arm, phase 1–2 study. The Lancet 2023, 402, 641–654. [Google Scholar] [CrossRef] [PubMed]
- Casan, J.M.L.; Wong, J.; Northcott, M.J.; Opat, S. Anti-CD20 monoclonal antibodies: reviewing a revolution. Human Vaccines & Immunotherapeutics 2018, 14, 2820–2841. [Google Scholar] [CrossRef]
- Rogers, K.A.; Yan, P.; Flinn, I.W.; Stephens, D.M.; Kipps, T.J.; Larson, S.M.; Martz, L.; Chen, X.; Wang, H.; Hopping, E.; et al. Addition of Ianalumab (VAY736) to Ibrutinib in Patients with Chronic Lymphocytic Leukemia on Ibrutinib Therapy: Results from a Phase Ib Study. Clin Cancer Res 2025, 31, 5145–5158. [Google Scholar] [CrossRef]
- Klein, U.; Tu, Y.; Stolovitzky, G.A.; Mattioli, M.; Cattoretti, G.; Husson, H.; Freedman, A.; Inghirami, G.; Cro, L.; Baldini, L.; et al. Gene expression profiling of B cell chronic lymphocytic leukemia reveals a homogeneous phenotype related to memory B cells. J Exp Med 2001, 194, 1625–1638. [Google Scholar] [CrossRef]
- Mateu-Albero, T.; Marcos-Jimenez, A.; Delgado-Wicke, P.; Terrón, F.; Loscertales, J.; López-Matencio, J.M.S.; Muñoz-Calleja, C.; Cuesta-Mateos, C. Evaluation of the novel therapeutic anti-CCR7 antibody CAP-100 as an add-on therapy in chronic lymphocytic leukemia patients receiving venetoclax. Hematol Oncol 2023, 41, 869–876. [Google Scholar] [CrossRef] [PubMed]
- Tandler, C.; Schmidt, M.; Heitmann, J.S.; Hierold, J.; Schmidt, J.; Schneider, P.; Dörfel, D.; Walz, J.; Salih, H.R. Neutralization of B-Cell Activating Factor (BAFF) by Belimumab Reinforces Small Molecule Inhibitor Treatment in Chronic Lymphocytic Leukemia. Cancers (Basel) 2020, 12. [Google Scholar] [CrossRef]
- Al-Sawaf, O.; Ligtvoet, R.; Robrecht, S.; Stumpf, J.; Fink, A.M.; Tausch, E.; Schneider, C.; Boettcher, S.; Mikusko, M.; Ritgen, M.; et al. Tislelizumab plus zanubrutinib for Richter transformation: the phase 2 RT1 trial. Nat Med 2024, 30, 240–248. [Google Scholar] [CrossRef]
- Gregory, G.P.; Kumar, S.; Wang, D.; Mahadevan, D.; Walker, P.; Wagner-Johnston, N.; Escobar, C.; Bannerji, R.; Bhutani, D.; Chang, J.; et al. Pembrolizumab plus dinaciclib in patients with hematologic malignancies: the phase 1b KEYNOTE-155 study. Blood Adv 2022, 6, 1232–1242. [Google Scholar] [CrossRef]
- Gregory, G.; Walker, P.; Mahadevan, D.; Wang, D.; Chang, J.; Hernandez-Ilizaliturri, F.; Klein, A.; Rybka, W.; Wagner-Johnston, N.; Escobar, C.; et al. ANTITUMOR ACTIVITY OF PEMBROLIZUMAB PLUS DINACICLIB IN PATIENTS WITH DIFFUSE LARGE B CELL LYMPHOMA: THE PHASE 1B KEYNOTE-155 STUDY. Hematological Oncology 2019, 37, 328–329. [Google Scholar] [CrossRef]
- Reville, P.K.; Sajedi, S.; Ferrajoli, A.; Burger, J.A.; Keating, M.J.; Garg, N.; Deng, Q.; Green, M.R.; Abbas, H.A.; Wierda, W.G.; et al. Atezolizumab Combined with Venetoclax and Obinutuzumab for Frontline CLL. Blood 2024, 144, 1014. [Google Scholar] [CrossRef]
- Rose, A.L.; Smith, B.E.; Maloney, D.G. Glucocorticoids and rituximab in vitro: synergistic direct antiproliferative and apoptotic effects. Blood 2002, 100, 1765–1773. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.A.; Emberley, E.; D'Souza, A.L.; Zhao, W.; Cosgrove, C.; Parrish, K.; Mitra, D.; Payson, E.; Oleksijew, A.; Ellis, P.; et al. ABBV-319: a CD19-targeting glucocorticoid receptor modulator antibody-drug conjugate therapy for B-cell malignancies. Blood 2024, 144, 757–770. [Google Scholar] [CrossRef]
- Lee, H.J.; Choi, M.Y.; Siddiqi, T.; Rhodes, J.M.; Wierda, W.G.; Isufi, I.; Tuscano, J.M.; Lamanna, N.; Subbiah, S.; Koff, J.L.; et al. Phase 1/2 study of zilovertamab and ibrutinib in mantle cell lymphoma (MCL) or chronic lymphocytic leukemia (CLL). Journal of Clinical Oncology 2022, 40, 7520–7520. [Google Scholar] [CrossRef]
- Wang, M.L.; Mei, M.; Barr, P.M.; Barrientos, J.C.; de Vos, S.; Furman, R.R.; Patel, K.; Wierda, W.G.; Choi, M.Y.; Kallam, A.; et al. Zilovertamab vedotin (MK-2140) in relapsed or refractory (R/R) non-Hodgkin lymphoma (NHL): 14-month follow-up of the phase 1 waveLINE-001 study. Hematological Oncology 2023, 41, 571–572. [Google Scholar] [CrossRef]
- Paszkiewicz-Kozik, E.; Moreira, C.; Turgut, M.; Garrido, M.; Glimelius, I.; Lee, S.T.; Sawalha, Y.; Ren, Y.; Ryland, K.; Ogbu, U.C.; et al. Zilovertamab Vedotin in Combination with Nemtabrutinib for Patients with Relapsed or Refractory Mantle Cell Lymphoma: Cohort C of the Open-Label, Phase 2 Waveline-006 Study. Blood 2024, 144, 3025–3025. [Google Scholar] [CrossRef]
- Nayyar, M.; Menezes, R.C.B.; Ailawadhi, S.; Parrondo, R.D. Chronic Lymphocytic Leukemia: Novel Therapeutic Targets Under Investigation. Cancers (Basel) 2025, 17. [Google Scholar] [CrossRef] [PubMed]
- Shahzad, M.; Basharat, A.; Irfan, S.; Sadiq, M.H.; Amin, M.K.; Jaglal, M.V. Future Landscapes of Bispecific Antibodies in Chronic Lymphocytic Leukemia (CLL). Systematic Review of Ongoing Trials. Blood 2024, 144, 6804–6804. [Google Scholar] [CrossRef]
- Iskierka-Jażdżewska, E.; Puła, B.; Jamroziak, K.; Robak, T. Chronic lymphocytic leukemia: what clinical progress have we seen in the last five years? Expert Opin Investig Drugs 2025, 34, 267–285. [Google Scholar] [CrossRef] [PubMed]
- Kater, A.P.; Janssens, A.; Eradat, H.; Offner, F.; Sandoval-Sus, J.D.; Shadman, M.; Poulsen, C.B.; Christensen, J.H.; Thompson, M.C.; Guan, M.; et al. Epcoritamab monotherapy for Richter transformation (EPCORE CLL-1): findings from a single-arm, multicentre, open-label, phase 1b/2 trial. Lancet Haematol 2026, 13, e8–e21. [Google Scholar] [CrossRef]
- Thompson, P.; Tadmor, T.; Bentzen, H.H.; Cordoba, R.; Snauwaert, S.; Goldschmidt, N.; Simkovic, M.; Christensen, J.; Lopez Jimenez, J.; Benjamini, O.; et al. Epcoritamab combinations demonstrate promising efficacy in patients (pts) with Richter transformation (RT): First results from arms 2B (epcor + lenalidomide [LEN]) and 2C (epcor + R-CHOP) of the phase 1b/2 EPCORE CLL-1 trial. Blood 2025, 146, 1015. [Google Scholar] [CrossRef]
- Danilov, A.; Fakhri, B.; Awan, F.T.; Bentzen, H.H.; Eradat, H.A.; Niemann, C.U.; Offner, F.; Poulsen, C.B.; Hoeyer, T.; Bellido, M.; et al. Epcoritamab Monotherapy in Patients (Pts) with Relapsed or Refractory (R/R) Chronic Lymphocytic Leukemia (CLL): Results from CLL Expansion and Optimization Cohorts of Epcore CLL-1. Blood 2024, 144, 883–883. [Google Scholar] [CrossRef]
- Townsend, W.; Leong, S.; Shah, M.; Batten, T.; Tucker, D.; Pottinger, B.; Paneesha, S.; El-Sharkawi, D.; Granger, D.; O'Donovan, K.; et al. Encouraging long-term efficacy and safety from first-in-human phase 1 study of time-limited NVG-111 in relapsed/refractory CLL and MCL. Blood 2025, 146, 2125–2125. [Google Scholar] [CrossRef]
- Dickinson, M.J.; Carlo-Stella, C.; Morschhauser, F.; Bachy, E.; Corradini, P.; Iacoboni, G.; Khan, C.; Wróbel, T.; Offner, F.; Trněný, M.; et al. Glofitamab for Relapsed or Refractory Diffuse Large B-Cell Lymphoma. New England Journal of Medicine 2022, 387, 2220–2231. [Google Scholar] [CrossRef]
- Carlo-Stella, C.; Hutchings, M.; Offner, F.; Mulvihill, E.; Relf, J.; Byrne, B.; Lundberg, L.; Dickinson, M. GLOFITAMAB MONOTHERAPY INDUCES DURABLE COMPLETE REMISSIONS AND HAS A MANAGEABLE SAFETY PROFILE IN PATIENTS WITH RICHTER’S TRANSFORMATION. Hematological Oncology 2023, 41, 63–65. [Google Scholar] [CrossRef]
- Fischer, C.; Chen, S.-S.; Nimmerfroh, J.; Eugster, A.; Stücheli, S.; Schultheiß, C.; Widmer, C.; Heim, D.; Kasenda, B.; Passweg, J.; et al. IGLV3-21R110-directed bispecific antibodies activate T cells and promote killing in a high-risk subset of chronic lymphocytic leukemia. Haematologica 2026, 111, 572–582. [Google Scholar] [CrossRef]
- Neelapu, S.S.; Locke, F.L.; Bartlett, N.L.; Lekakis, L.J.; Miklos, D.B.; Jacobson, C.A.; Braunschweig, I.; Oluwole, O.O.; Siddiqi, T.; Lin, Y.; et al. Axicabtagene Ciloleucel CAR T-Cell Therapy in Refractory Large B-Cell Lymphoma. New England Journal of Medicine 2017, 377, 2531–2544. [Google Scholar] [CrossRef] [PubMed]
- Schuster, S.J.; Bishop, M.R.; Tam, C.S.; Waller, E.K.; Borchmann, P.; McGuirk, J.P.; Jäger, U.; Jaglowski, S.; Andreadis, C.; Westin, J.R.; et al. Tisagenlecleucel in Adult Relapsed or Refractory Diffuse Large B-Cell Lymphoma. New England Journal of Medicine 2019, 380, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, C.A.; Chavez, J.C.; Sehgal, A.R.; William, B.M.; Munoz, J.; Salles, G.; Munshi, P.N.; Casulo, C.; Maloney, D.G.; de Vos, S.; et al. Axicabtagene ciloleucel in relapsed or refractory indolent non-Hodgkin lymphoma (ZUMA-5): a single-arm, multicentre, phase 2 trial. The Lancet Oncology 2022, 23, 91–103. [Google Scholar] [CrossRef]
- Wang, M.; Munoz, J.; Goy, A.; Locke, F.L.; Jacobson, C.A.; Hill, B.T.; Timmerman, J.M.; Holmes, H.; Jaglowski, S.; Flinn, I.W.; et al. KTE-X19 CAR T-Cell Therapy in Relapsed or Refractory Mantle-Cell Lymphoma. New England Journal of Medicine 2020, 382, 1331–1342. [Google Scholar] [CrossRef]
- Abramson, J.S.; Palomba, M.L.; Gordon, L.I.; Lunning, M.; Wang, M.; Arnason, J.; Purev, E.; Maloney, D.G.; Andreadis, C.; Sehgal, A.; et al. Two-year follow-up of lisocabtagene maraleucel in relapsed or refractory large B-cell lymphoma in TRANSCEND NHL 001. Blood 2024, 143, 404–416. [Google Scholar] [CrossRef]
- Fowler, N.H.; Dickinson, M.; Dreyling, M.; Martinez-Lopez, J.; Kolstad, A.; Butler, J.; Ghosh, M.; Popplewell, L.; Chavez, J.C.; Bachy, E.; et al. Tisagenlecleucel in adult relapsed or refractory follicular lymphoma: the phase 2 ELARA trial. Nature Medicine 2022, 28, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Maude, S.L.; Laetsch, T.W.; Buechner, J.; Rives, S.; Boyer, M.; Bittencourt, H.; Bader, P.; Verneris, M.R.; Stefanski, H.E.; Myers, G.D.; et al. Tisagenlecleucel in Children and Young Adults with B-Cell Lymphoblastic Leukemia. New England Journal of Medicine 2018, 378, 439–448. [Google Scholar] [CrossRef]
- Shah, B.D.; Ghobadi, A.; Oluwole, O.O.; Logan, A.C.; Boissel, N.; Cassaday, R.D.; Leguay, T.; Bishop, M.R.; Topp, M.S.; Tzachanis, D.; et al. KTE-X19 for relapsed or refractory adult B-cell acute lymphoblastic leukaemia: phase 2 results of the single-arm, open-label, multicentre ZUMA-3 study. The Lancet 2021, 398, 491–502. [Google Scholar] [CrossRef]
- Riches, J.C.; Davies, J.K.; McClanahan, F.; Fatah, R.; Iqbal, S.; Agrawal, S.; Ramsay, A.G.; Gribben, J.G. T cells from CLL patients exhibit features of T-cell exhaustion but retain capacity for cytokine production. Blood 2013, 121, 1612–1621. [Google Scholar] [CrossRef]
- Roessner, P.M.; Seiffert, M. T-cells in chronic lymphocytic leukemia: Guardians or drivers of disease? Leukemia 2020, 34, 2012–2024. [Google Scholar] [CrossRef] [PubMed]
- Borogovac, A.; Siddiqi, T. Advancing CAR T-cell therapy for chronic lymphocytic leukemia: exploring resistance mechanisms and the innovative strategies to overcome them. Cancer Drug Resist 2024, 7, 18. [Google Scholar] [CrossRef] [PubMed]
- Fraietta, J.A.; Lacey, S.F.; Orlando, E.J.; Pruteanu-Malinici, I.; Gohil, M.; Lundh, S.; Boesteanu, A.C.; Wang, Y.; O’Connor, R.S.; Hwang, W.-T.; et al. Determinants of response and resistance to CD19 chimeric antigen receptor (CAR) T cell therapy of chronic lymphocytic leukemia. Nature Medicine 2018, 24, 563–571. [Google Scholar] [CrossRef] [PubMed]
- Taghiloo, S.; Asgarian-Omran, H. Cross-talk between leukemic and immune cells at the tumor microenvironment in chronic lymphocytic leukemia: An update review. European Journal of Haematology 2024, 113, 4–15. [Google Scholar] [CrossRef]
- Burger, J.A.; Tsukada, N.; Burger, M.; Zvaifler, N.J.; Dell'Aquila, M.; Kipps, T.J. Blood-derived nurse-like cells protect chronic lymphocytic leukemia B cells from spontaneous apoptosis through stromal cell–derived factor-1. Blood 2000, 96, 2655–2663. [Google Scholar] [CrossRef]
- Jitschin, R.; Braun, M.; Büttner, M.; Dettmer-Wilde, K.; Bricks, J.; Berger, J.; Eckart, M.J.; Krause, S.W.; Oefner, P.J.; Le Blanc, K.; et al. CLL-cells induce IDOhi CD14+HLA-DRlo myeloid-derived suppressor cells that inhibit T-cell responses and promote TRegs. Blood 2014, 124, 750–760. [Google Scholar] [CrossRef]
- Teoh, J.; Brown, L.F. Developing lisocabtagene maraleucel chimeric antigen receptor T-cell manufacturing for improved process, product quality and consistency across CD19+ hematologic indications. Cytotherapy 2022, 24, 962–973. [Google Scholar] [CrossRef]
- Siddiqi, T.; Maloney, D.G.; Kenderian, S.S.; Brander, D.M.; Dorritie, K.; Soumerai, J.; Riedell, P.A.; Shah, N.N.; Nath, R.; Fakhri, B.; et al. Lisocabtagene Maraleucel (liso-cel) in R/R CLL/SLL: 24-Month Median Follow-up of TRANSCEND CLL 004. Blood 2023, 142, 330–330. [Google Scholar] [CrossRef]
- Porter, D.L.; Hwang, W.-T.; Frey, N.V.; Lacey, S.F.; Shaw, P.A.; Loren, A.W.; Bagg, A.; Marcucci, K.T.; Shen, A.; Gonzalez, V.; et al. Chimeric antigen receptor T cells persist and induce sustained remissions in relapsed refractory chronic lymphocytic leukemia. Science Translational Medicine 2015, 7, 303ra139–303ra139. [Google Scholar] [CrossRef] [PubMed]
- Melenhorst, J.J.; Chen, G.M.; Wang, M.; Porter, D.L.; Chen, C.; Collins, M.A.; Gao, P.; Bandyopadhyay, S.; Sun, H.; Zhao, Z.; et al. Decade-long leukaemia remissions with persistence of CD4(+) CAR T cells. Nature 2022, 602, 503–509. [Google Scholar] [CrossRef]
- Cappell, K.M.; Sherry, R.M.; Yang, J.C.; Goff, S.L.; Vanasse, D.A.; McIntyre, L.; Rosenberg, S.A.; Kochenderfer, J.N. Long-Term Follow-Up of Anti-CD19 Chimeric Antigen Receptor T-Cell Therapy. Journal of Clinical Oncology 2020, 38, 3805–3815. [Google Scholar] [CrossRef]
- Davids, M.S.; Kenderian, S.S.; Flinn, I.; Hill, B.T.; Maris, M.; Ghia, P.; Byrne, M.; Bartlett, N.L.; Pagel, J.M.; Zheng, Y.; et al. ZUMA-8: a phase 1 study of brexucabtagene autoleucel in patients with relapsed/refractory chronic lymphocytic leukemia. Blood 2025, 146, 938–943. [Google Scholar] [CrossRef]
- Ortiz-Maldonado, V.; Martínez-Cibrián, N.; Alserawan, L.; Betriu, S.; Triguero, A.; Blum, S.; Faes, M.; Liefaard, M.C.; Pont, M.J.; Spoon, M.; et al. Euplagia-1: A Phase 1/2 Trial of GLPG5201, a Fresh Stem-like Early Memory CD19 CAR T-Cell Therapy with a 7-Day Vein-to-Vein Time, in Patients with Relapsed/Refractory CLL and RT. Blood 2024, 144, 3452–3452. [Google Scholar] [CrossRef]
- Svoboda, J.; Landsburg, D.J.; Gerson, J.; Nasta, S.D.; Barta, S.K.; Chong, E.A.; Cook, M.; Frey, N.V.; Shea, J.; Cervini, A.; et al. Enhanced CAR T-Cell Therapy for Lymphoma after Previous Failure. N Engl J Med 2025, 392, 1824–1835. [Google Scholar] [CrossRef]
- Albiol N., M.-C.N.; Franch, A; Navarro, A.; África Martín-López, A. 1905 - Academic CD19-directed CAR-T ARI-0001 (varnimcabtagene autoleucel) for patients with relapsed/refractory chronic lymphocytic leukemia and Richter’s transformation. In Proceedings of the XXII International Workshop on CLL, Kraków, 2025. [Google Scholar]
- Liang, E.C.; Albittar, A.; Huang, J.J.; Hirayama, A.V.; Kimble, E.L.; Portuguese, A.J.; Chapuis, A.; Shadman, M.; Till, B.G.; Cassaday, R.D.; et al. Factors associated with long-term outcomes of CD19 CAR T-cell therapy for relapsed/refractory CLL. Blood Advances 2023, 7, 6990–7005. [Google Scholar] [CrossRef]
- Derigs, P.; Schubert, M.-L.; Dreger, P.; Schmitt, A.; Yousefian, S.; Haas, S.; Röthemeier, C.; Neuber, B.; Hückelhoven-Krauss, A.; Brüggemann, M.; et al. Third-generation anti-CD19 CAR T cells for relapsed/refractory chronic lymphocytic leukemia: a phase 1/2 study. Leukemia 2024, 38, 2419–2428. [Google Scholar] [CrossRef]
- Thompson, J.S.; Bixler, S.A.; Qian, F.; Vora, K.; Scott, M.L.; Cachero, T.G.; Hession, C.; Schneider, P.; Sizing, I.D.; Mullen, C.; et al. BAFF-R, a Newly Identified TNF Receptor That Specifically Interacts with BAFF. Science 2001, 293, 2108–2111. [Google Scholar] [CrossRef] [PubMed]
- Rodig, S.J.; Shahsafaei, A.; Li, B.; Mackay, C.R.; Dorfman, D.M. BAFF-R, the major B cell–activating factor receptor, is expressed on most mature B cells and B-cell lymphoproliferative disorders. Human Pathology 2005, 36, 1113–1119. [Google Scholar] [CrossRef]
- Qie, Y.; Gadd, M.E.; Shao, Q.; To, T.; Liu, A.; Li, S.; Rivera-Valentin, R.; Yassine, F.; Murthy, H.S.; Dronca, R.; et al. Targeting chronic lymphocytic leukemia with B-cell activating factor receptor CAR T cells. MedComm (2020) 2024, 5, e716. [Google Scholar] [CrossRef]
- Hug, S.; Phely, L.; Riedel, A.; Killinger, S.; Hensen, L.; Faul, C.; Bethge, W.A.; Lengerke, C. Dual CD19/CD22-Targeting CAR-T Cells for Richter Transformation: A Case Series. Blood 2024, 144, 6544–6544. [Google Scholar] [CrossRef]
- Shah, N.N.; Atallah, E.L.; Abedin, S.; Murthy, G.S.G.; Runaas, L.; Michaelis, L.C.; Longo, W.; Furqan, F.; Bucklan, D.; Szabo, A.; et al. Phase 1 Trial of LV20.19 CAR T-Cells for Relapsed, Refractory CLL and Richter's Transformation. Transplantation and Cellular Therapy 2024, 30, S35–S36. [Google Scholar] [CrossRef]
- Wang, X.; Dong, Z.; Awuah, D.; Chang, W.C.; Cheng, W.A.; Vyas, V.; Cha, S.C.; Anderson, A.J.; Zhang, T.; Wang, Z.; et al. CD19/BAFF-R dual-targeted CAR T cells for the treatment of mixed antigen-negative variants of acute lymphoblastic leukemia. Leukemia 2022, 36, 1015–1024. [Google Scholar] [CrossRef] [PubMed]
- Mansoori, S.; Noei, A.; Maali, A.; Seyed-Motahari, S.S.; Sharifzadeh, Z. Recent updates on allogeneic CAR-T cells in hematological malignancies. Cancer Cell International 2024, 24, 304. [Google Scholar] [CrossRef]
- Lussana, F.; Magnani, C.F.; Galimberti, S.; Gritti, G.; Gaipa, G.; Belotti, D.; Cabiati, B.; Napolitano, S.; Ferrari, S.; Moretti, A.; et al. Donor-derived CARCIK-CD19 cells engineered with Sleeping Beauty transposon in acute lymphoblastic leukemia relapsed after allogeneic transplantation. Blood Cancer J 2025, 15, 54. [Google Scholar] [CrossRef] [PubMed]
- Lekakis, L.J.; Locke, F.L.; Tees, M.; Neelapu, S.S.; Malik, S.A.; Hamadani, M.; Frank, M.J.; Popplewell, L.L.; Abramson, J.S.; de Vos, S.; et al. ALPHA2 Study: ALLO-501A Allogeneic CAR T in LBCL, Updated Results Continue to Show Encouraging Safety and Efficacy with Consolidation Dosing. Blood 2021, 138, 649. [Google Scholar] [CrossRef]
- Ghobadi, A.; McGuirk, J.P.; Shaughnessy, P.; Tam, C.S.; Allen, M.; Pan, C.; Li, Z.; Stevens, W.N.; Weaver, A.; Cheah, C.Y. CTX112, a Next-Generation Allogeneic CRISPR-Cas9 Engineered CD19 CAR T Cell with Novel Potency Edits: Data from Phase 1 Dose Escalation Study in Patients with Relapsed or Refractory B-Cell Malignancies. Blood 2024, 144, 4829–4829. [Google Scholar] [CrossRef]
- Long, M.; Beckwith, K.; Do, P.; Mundy, B.L.; Gordon, A.; Lehman, A.M.; Maddocks, K.J.; Cheney, C.; Jones, J.A.; Flynn, J.M.; et al. Ibrutinib treatment improves T cell number and function in CLL patients. The Journal of Clinical Investigation 2017, 127, 3052–3064. [Google Scholar] [CrossRef]
- Fraietta, J.A.; Beckwith, K.A.; Patel, P.R.; Ruella, M.; Zheng, Z.; Barrett, D.M.; Lacey, S.F.; Melenhorst, J.J.; McGettigan, S.E.; Cook, D.R.; et al. Ibrutinib enhances chimeric antigen receptor T-cell engraftment and efficacy in leukemia. Blood 2016, 127, 1117–1127. [Google Scholar] [CrossRef]
- Ruella, M.; Kenderian, S.S.; Shestova, O.; Klichinsky, M.; Melenhorst, J.J.; Wasik, M.A.; Lacey, S.F.; June, C.H.; Gill, S. Kinase inhibitor ibrutinib to prevent cytokine-release syndrome after anti-CD19 chimeric antigen receptor T cells for B-cell neoplasms. Leukemia 2017, 31, 246–248. [Google Scholar] [CrossRef]
- Gauthier, J.; Hirayama, A.V.; Purushe, J.; Hay, K.A.; Lymp, J.; Li, D.H.; Yeung, C.C.S.; Sheih, A.; Pender, B.S.; Hawkins, R.M.; et al. Feasibility and efficacy of CD19-targeted CAR T cells with concurrent ibrutinib for CLL after ibrutinib failure. Blood 2020, 135, 1650–1660. [Google Scholar] [CrossRef]
- Wierda, W.G.; Dorritie, K.A.; Munoz, J.; Stephens, D.M.; Solomon, S.; Gillenwater, H.H.; Gong, L.; Yang, L.; Ogasawara, K.; Thorpe, J.; et al. TRANSCEND CLL 004: PHASE 1 COHORT OF LISOCABTAGENE MARALEUCEL (LISO-CEL) COMBINED WITH IBRUTINIB (IBR) FOR PATIENTS (PTS) WITH R/R CLL/SLL. Hematological Oncology 2021, 39. [Google Scholar] [CrossRef]
- Gill, S.; Vides, V.; Frey, N.V.; Hexner, E.O.; Metzger, S.; O'Brien, M.; Hwang, W.T.; Brogdon, J.L.; Davis, M.M.; Fraietta, J.A.; et al. Anti-CD19 CAR T cells in combination with ibrutinib for the treatment of chronic lymphocytic leukemia. Blood Adv 2022, 6, 5774–5785. [Google Scholar] [CrossRef]
- Tettamanti, S.; Rotiroti, M.C.; Giordano Attianese, G.M.P.; Arcangeli, S.; Zhang, R.; Banerjee, P.; Galletti, G.; McManus, S.; Mazza, M.; Nicolini, F.; et al. Lenalidomide enhances CD23.CAR T cell therapy in chronic lymphocytic leukemia. Leuk Lymphoma 2022, 63, 1566–1579. [Google Scholar] [CrossRef]
- Funk, C.R.; Wang, S.; Chen, K.Z.; Waller, A.; Sharma, A.; Edgar, C.L.; Gupta, V.A.; Chandrakasan, S.; Zoine, J.T.; Fedanov, A.; et al. PI3Kδ/γ inhibition promotes human CART cell epigenetic and metabolic reprogramming to enhance antitumor cytotoxicity. Blood 2022, 139, 523–537. [Google Scholar] [CrossRef]
- Sportoletti, P.; De Falco, F.; Del Papa, B.; Baldoni, S.; Guarente, V.; Marra, A.; Dorillo, E.; Rompietti, C.; Adamo, F.M.; Ruggeri, L.; et al. NK Cells in Chronic Lymphocytic Leukemia and Their Therapeutic Implications. Int J Mol Sci 2021, 22. [Google Scholar] [CrossRef]
- Herrera, L.; Santos, S.; Vesga, M.A.; Anguita, J.; Martin-Ruiz, I.; Carrascosa, T.; Juan, M.; Eguizabal, C. Adult peripheral blood and umbilical cord blood NK cells are good sources for effective CAR therapy against CD19 positive leukemic cells. Scientific Reports 2019, 9, 18729. [Google Scholar] [CrossRef] [PubMed]
- Olson, J.A.; Leveson-Gower, D.B.; Gill, S.; Baker, J.; Beilhack, A.; Negrin, R.S. NK cells mediate reduction of GVHD by inhibiting activated, alloreactive T cells while retaining GVT effects. Blood 2010, 115, 4293–4301. [Google Scholar] [CrossRef]
- Liu, E.; Tong, Y.; Dotti, G.; Shaim, H.; Savoldo, B.; Mukherjee, M.; Orange, J.; Wan, X.; Lu, X.; Reynolds, A.; et al. Cord blood NK cells engineered to express IL-15 and a CD19-targeted CAR show long-term persistence and potent antitumor activity. Leukemia 2018, 32, 520–531. [Google Scholar] [CrossRef]
- Liu, E.; Marin, D.; Banerjee, P.; Macapinlac, H.A.; Thompson, P.; Basar, R.; Nassif Kerbauy, L.; Overman, B.; Thall, P.; Kaplan, M.; et al. Use of CAR-Transduced Natural Killer Cells in CD19-Positive Lymphoid Tumors. N Engl J Med 2020, 382, 545–553. [Google Scholar] [CrossRef]
- Marin, D.; Li, Y.; Basar, R.; Rafei, H.; Daher, M.; Dou, J.; Mohanty, V.; Dede, M.; Nieto, Y.; Uprety, N.; et al. Safety, efficacy and determinants of response of allogeneic CD19-specific CAR-NK cells in CD19(+) B cell tumors: a phase 1/2 trial. Nat Med 2024, 30, 772–784. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).