Submitted:
28 February 2026
Posted:
04 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
3. Results
4. Discussion
4.1. KD and MIS Etiology Model
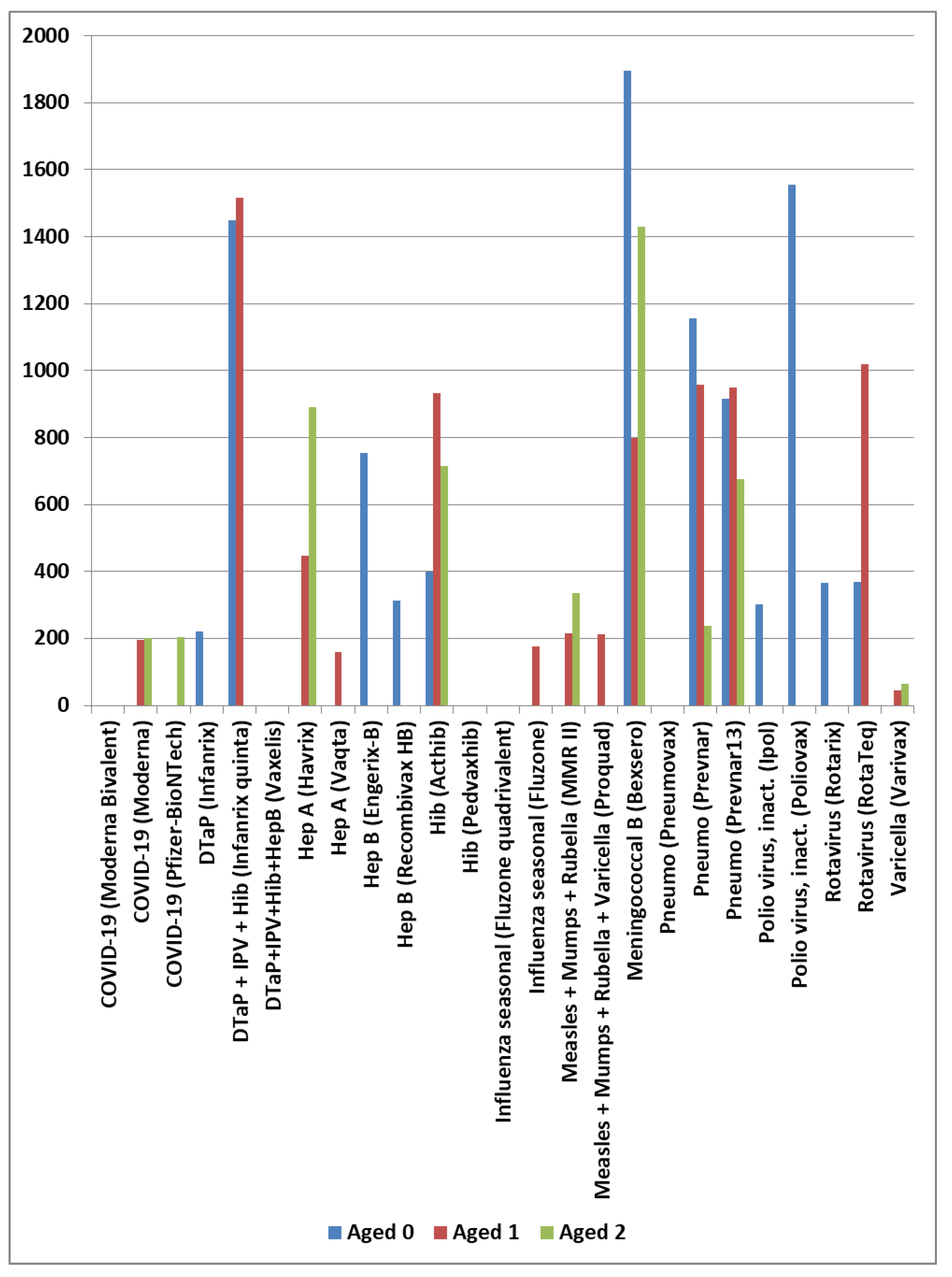
4.2. Age-Related Risk Patterns
4.3. Cardiac Adverse Events and Acquired Heart Disease
4.4. KD and MIS Delayed Onset
4.5. KD Genetics
4.6. IVIG Treatment and IVIG Resistance
4.7. MIS Differences from KD
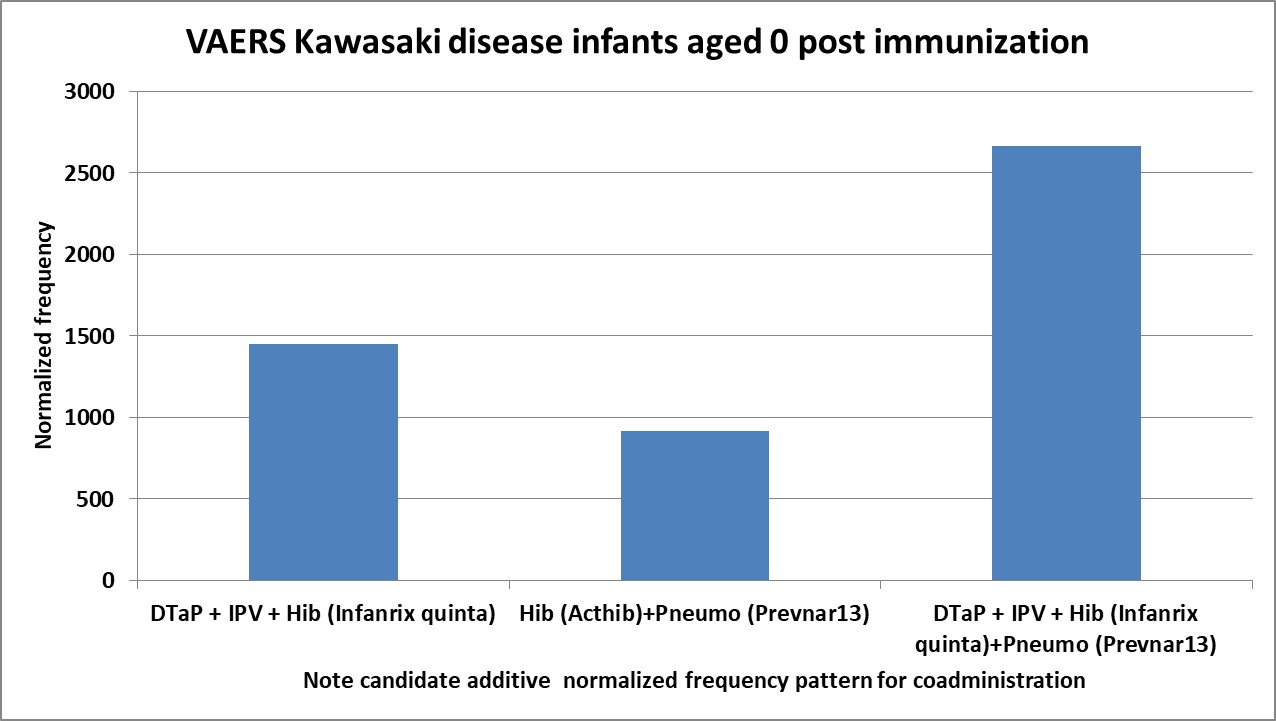
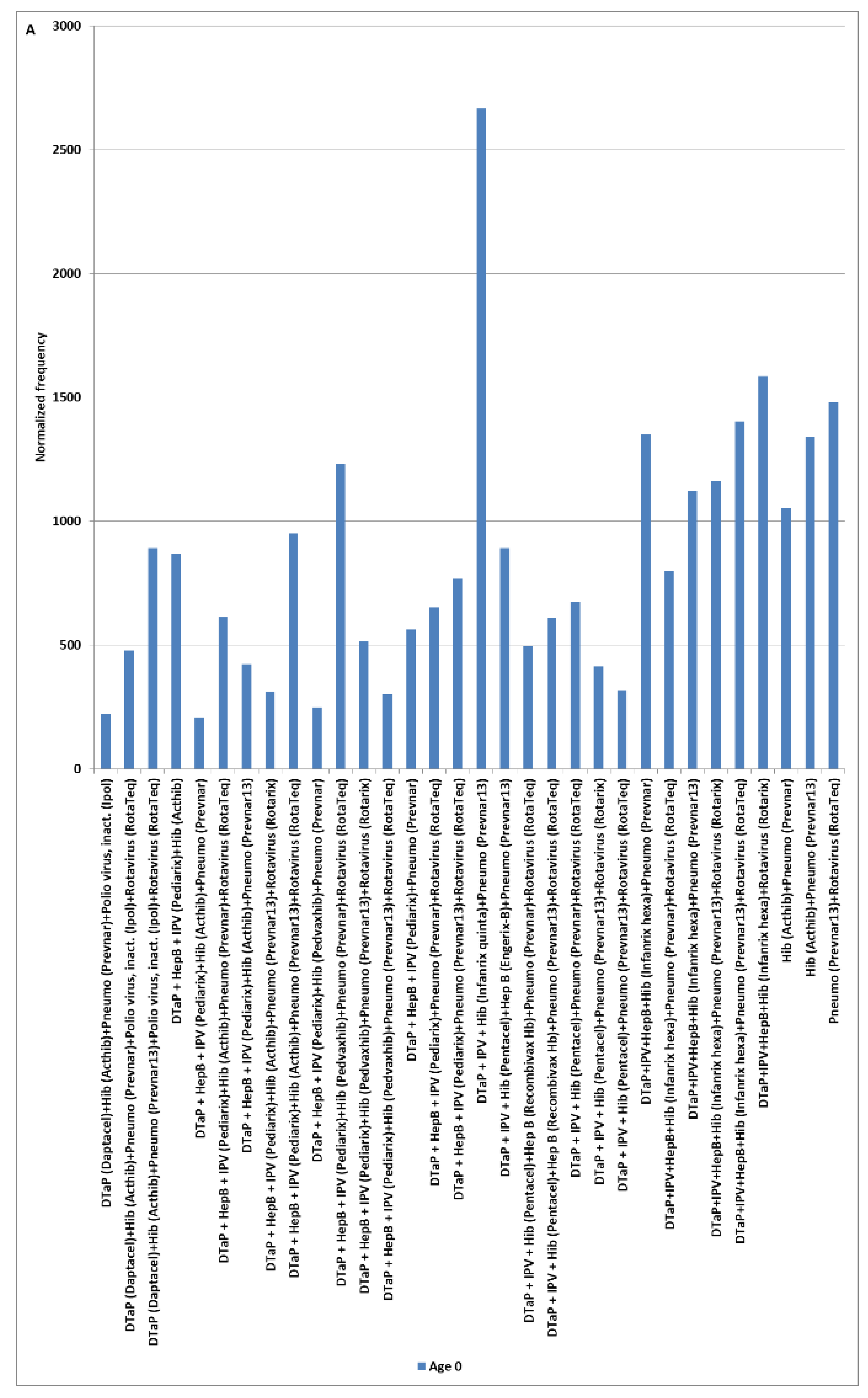
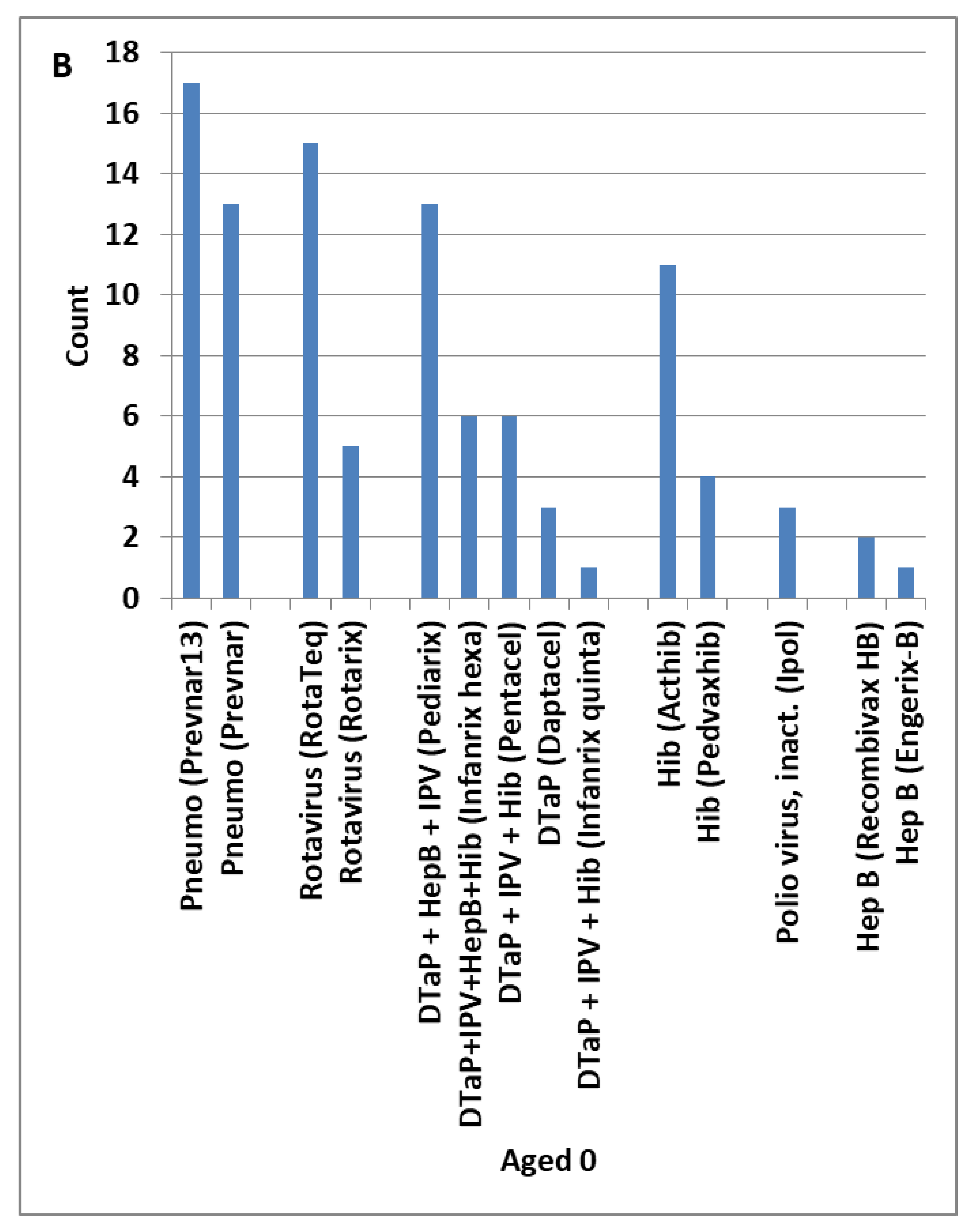
4.8. Candidate Adjunctive Treatments
4.9. Study Limitations
4.10. Study Recommendations
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACE2 | Angiotensin-converting enzyme 2 |
| ADE | Antibody-dependent enhancement |
| AE | Adverse event |
| B-cell | Immune B lymphocyte |
| BLK | B-cell lymphoid tyrosine kinase |
| CAA | Coronary artery aneurysm |
| CALs | Coronary artery lesion |
| CASP3 | Caspase 3 |
| CD14 | Cluster of differentiation 14 |
| CD147 | Cluster of differentiation 147, also known as EMMPRIN (Extracellular Matrix Metalloproteinase Inducer), or Basigin |
| CD40 | Cluster of differentiation 40 |
| CD40L | Cluster of differentiation 40 ligand |
| CO | Carbon monoxide |
| COVID-19 | Coronavirus disease 2019 |
| DTaP | Diphtheria, tetanus, and pertussis (whooping cough) vaccine |
| Fc | fragment crystallizable region of antibody |
| FcγR | Fc gamma receptor |
| FG | plasma fibrinogen |
| GI | gastrointestinal |
| Hep B | hepatitis B |
| Hib | Haemophilus influenzae type b vaccine |
| HIT | histamine intolerance |
| HLA | human leukocyte antigen |
| IgG | immunoglobulin G |
| IGHV | immunoglobulin heavy variable gene |
| IPV | inactivated poliovirus vaccine |
| ITPKC | inositol 1,4,5-trisphosphate 3-kinase C |
| IVIG | intravenous immunoglobulin |
| KCNN2 | Potassium Calcium-Activated Channel Subfamily N Member 2 |
| KD | Kawasaki’s disease |
| KD-MAS | Kawasaki disease complicated with macrophage activation syndrome |
| KD-N | Kawasaki’s disease in neonates |
| KDSS | Kawasaki disease shock/toxic-shock syndrome |
| KD-V | Kawasaki’s disease associated with vaccination |
| LAL | Limulus Amebocyte Lysate |
| LER | Low endotoxin recovery |
| LPS | Lipopolysaccharide |
| MAMPS | Microbe-associated molecular patterns |
| matAbs | Maternally transferred antibodies |
| MCAS | Mast cell activation syndromes |
| MIS | Multisystem inflammatory syndrome |
| MIS-A | Multisystem inflammatory syndrome in adults |
| MIS-C | Multisystem inflammatory syndrome in children |
| MIS-N | Multisystem inflammatory syndrome in neonates |
| MIS-V | Multisystem inflammatory syndrome after COVID-19 vaccination |
| MMR | Measles, mumps, and rubella vaccine |
| MPAs | Monocyte‒platelet aggregates |
| mRNA | Messenger ribonucleic acid |
| MYH14 | Myosin heavy chain 14 |
| NA1 | Neutrophil antigen 1 |
| NO | Nitric oxide |
| NO2 | Nitric dioxide |
| NOx | Nitrogen oxide |
| O3 | Ozone |
| ORAI1 | calcium release-activated calcium modulator 1 |
| ORF | Open reading frame |
| PAMPS | pathogen-associated molecular patterns |
| PCT | Plateletcrit |
| PIMS-TS | Pediatric Inflammatory Syndrome temporally associated with SARS-CoV-2 infection |
| PM10 | Inhalable particulate matter 10 micrometers or smaller |
| PM25 | fine inhalable particles less than or equal to 2.5 micrometers in diameter |
| RBP3 | retinol-binding protein 3 |
| SAE | Serious adverse event |
| SARS-CoV-2 | Severe acute respiratory syndrome coronavirus 2 |
| sCD14 | Soluble CD14 protein |
| SMAD3 | Mothers against decapentaplegic homolog 3 |
| SMAD5 | Mothers against decapentaplegic homolog 5 |
| SO2 | Sulfur dioxide |
| T-cell | T lymphocyte |
| TCRBV2 | T-cell receptor Beta-chain V2 |
| TCRBV6 | T-cell receptor Beta-chain V6 |
| TGF-beta | Transforming growth factor-beta |
| TGFBR2 | Transforming growth factor-beta receptor type 2 |
| TLR | Toll-like receptor |
| VAERS | Vaccine Adverse Event Reporting System |
References
- McCrindle, B.W.; Rowley, A.H.; Newburger, J.W.; Burns, J.C.; Bolger, A.F.; Gewitz, M.; Baker, A.L.; Jackson, M.A.; Takahashi, M.; Shah, P.B.; et al. Diagnosis, Treatment, and Long-Term Management of Kawasaki Disease: A Scientific Statement for Health Professionals from the American Heart Association. Circulation 2017, 135, e927–e999. [Google Scholar] [CrossRef]
- Rife, E.; Gedalia, A. Kawasaki Disease: an Update. Curr. Rheumatol. Rep. 2020, 22, 1–10. [Google Scholar] [CrossRef]
- Bayer. 2025. [PubMed]
- Lamrani, L.; Manlhiot, C.; Elias, M.D.; Choueiter, N.F.; Dionne, A.; Harahsheh, A.S.; Portman, M.A.; McCrindle, B.W.; Dahdah, N. Kawasaki Disease Shock Syndrome vs Classical Kawasaki Disease: A Meta-analysis and Comparison With SARS-CoV-2 Multisystem Inflammatory Syndrome. Can. J. Cardiol. 2021, 37, 1619–1628. [Google Scholar] [CrossRef] [PubMed]
- Embil, J.A.; McFarlane, E.S.; Murphy, D.M.; Krause, V.W.; Stwart, H.B. Adenovirus type 2 isolated from a patient with fatal Kawasaki disease. Can. Med. Assoc. J. 1985, 132, 1400. [Google Scholar]
- Chang, L.-Y.; Lu, C.-Y.; Shao, P.-L.; Lee, P.-I.; Lin, M.-T.; Fan, T.-Y.; Cheng, A.-L.; Lee, W.-L.; Hu, J.-J.; Yeh, S.-J.; et al. Viral infections associated with Kawasaki disease. J. Formos. Med. Assoc. 2014, 113, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Catalano-Pons, C.; Giraud, C.; Rozenberg, F.; Meritet, J.-F.; Lebon, P.; Gendrel, D. Detection of human bocavirus in children with Kawasaki disease. Clin. Microbiol. Infect. 2007, 13, 1220–1222. [Google Scholar] [CrossRef]
- Shirato, K.; Imada, Y.; Kawase, M.; Nakagaki, K.; Matsuyama, S.; Taguchi, F. Possible involvement of infection with human coronavirus 229E, but not NL63, in Kawasaki disease. J. Med Virol. 2014, 86, 2146–2153. [Google Scholar] [CrossRef]
- Esper, F.; Weibel, C.; Ferguson, D.; Landry, M.L.; Kahn, J.S. Evidence of a Novel Human Coronavirus That Is Associated with Respiratory Tract Disease in Infants and Young Children. J. Infect. Dis. 2005, 191, 492–498. [Google Scholar] [CrossRef]
- Catalano-Pons, C.; Quartier, P.; Leruez-Ville, M.; Kaguelidou, F.; Gendrel, D.; Lenoir, G.; Casanova, J.-L.; Bonnet, D. Primary Cytomegalovirus Infection, Atypical Kawasaki Disease, and Coronary Aneurysms in 2 Infants. Clin. Infect. Dis. 2005, 41, e53–e56. [Google Scholar] [CrossRef]
- Jagadeesh, A.; Krishnamurthy, S.; Mahadevan, S. Kawasaki Disease in a 2-year-old Child with Dengue Fever. Indian J. Pediatr. 2015, 83, 602–603. [Google Scholar] [CrossRef]
- Sopontammarak, S.; Promphan, W.; Roymanee, S.; Phetpisan, S. Positive Serology for Dengue Viral Infection in Pediatric Patients With Kawasaki Disease in Southern Thailand. Circ. J. 2008, 72, 1492–1494. [Google Scholar] [CrossRef]
- Weng, K.-P.; Wei, J.C.-C.; Hung, Y.-M.; Huang, S.-H.; Chien, K.-J.; Lin, C.-C.; Huang, S.-M.; Lin, C.-L.; Cheng, M.-F. Enterovirus Infection and Subsequent Risk of Kawasaki Disease: A Population-based Cohort Study. Pediatr. Infect. Dis. J. 2018, 37, 310–315. [Google Scholar] [CrossRef]
- Kikuta, H.; Nakanishi, M.; Ishikawa, N.; Konno, M.; Matsumoto, S. Detection of Epstein-Barr virus sequences in patients with Kawasaki disease by means of the polymerase chain reaction. Intervirology 1992, 33, 1–5. [Google Scholar] [CrossRef]
- Okano, M.; Luka, J.; Thiele, G.M.; Sakiyama, Y.; Matsumoto, S.; Purtilo, D.T. Human herpesvirus 6 infection and Kawasaki disease. J. Clin. Microbiol. 1989, 27, 2379–2380. [Google Scholar] [CrossRef]
- Okano, M. Kawasaki Disease and Human Lymphotropic Virus Infection. Curr. Med Res. Opin. 1999, 15, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Joshi, A.V.; Jones, K.D.; Buckley, A.; E Coren, M.; Kampmann, B. Kawasaki disease coincident with influenza A H1N1/09 infection. Pediatr. Int. 2011, 53, e1–e2. [Google Scholar] [CrossRef] [PubMed]
- Whitby, D.; Hoad, J.G.; Tizard, E.J.; Dillon, M.J.; Weber, J.N.; A Weiss, R.; Schulz, T.F. Isolation of measles virus from child with Kawasaki disease. Lancet 1991, 338, 1215–1215. [Google Scholar] [CrossRef]
- Holm, J.M.; Hansen, L.K.; Oxhøj, H. Kawasaki disease associated with parvovirus B19 infection. Eur. J. Pediatr. 1995, 154, 633–634. [Google Scholar] [CrossRef] [PubMed]
- Nigro, G.; Krzysztofiak, A.; Porcaro, M.; Mango, T.; Zerbini, M.; Gentilomi, G.; Musiani, M. Active or recent parvovirus B19 infection in children with Kawasaki disease. Lancet 1994, 343, 1260–1261. [Google Scholar] [CrossRef]
- Keim, D.; Keller, E.; Hirsch, M. MUCOCUTANEOUS LYMPH-NODE SYNDROME AND PARAINFLUENZA 2 VIRUS INFECTION. Lancet 1977, 310, 303. [Google Scholar] [CrossRef]
- Kim, G.B.; Park, S.; Kwon, B.S.; Han, J.W.; Park, Y.W.; Hong, Y.M. Evaluation of the Temporal Association between Kawasaki Disease and Viral Infections in South Korea. Korean Circ. J. 2014, 44, 250–254. [Google Scholar] [CrossRef]
- Matsuno, S.; Utagawa, E.; Sugiura, A. Association of Rotavirus Infection with Kawasaki Syndrome. J. Infect. Dis. 1983, 148, 177–177. [Google Scholar] [CrossRef] [PubMed]
- Ogboli, M.; Parslew, R.; Verbov, J.; Smyth, R. Kawasaki disease associated with varicella: a rare association. Br. J. Dermatol. 1999, 141, 1136–1152. [Google Scholar] [CrossRef] [PubMed]
- Kossiva, L.; Papadopoulos, M.; Lagona, E.; Papadopoulos, G.; Athanassaki, C. Myocardial infarction in a 35-day-old infant with incomplete Kawasaki disease and chicken pox. Cardiol. Young- 2010, 20, 567–570. [Google Scholar] [CrossRef]
- Thissen, J.B.; Isshiki, M.; Jaing, C.; Nagao, Y.; Aldea, D.L.; E Allen, J.; Izui, M.; Slezak, T.R.; Ishida, T.; Sano, T. A novel variant of torque teno virus 7 identified in patients with Kawasaki disease. PLOS ONE 2018, 13, e0209683. [Google Scholar] [CrossRef]
- Hall, M.; Schlievert, P.M.; Hoyt, L.; Ferrieri, P.; Jenson, H.B. Kawasaki Syndrome-Like Illness Associated with Infection Caused by Enterotoxin B-Secreting Staphylococcus aureus. Clin. Infect. Dis. 1999, 29, 586–589. [Google Scholar] [CrossRef] [PubMed]
- Shinomiya, N.; Takeda, T.; Kuratsuji, T.; Takagi, K.; Kosaka, T.; Tatsuzawa, O.; Tsurumizu, T.; Hashimoto, T.; Kobayashi, N. Variant Streptococcus sanguis as an etiological agent of Kawasaki disease. Prog. Clin. Biol. Res. 1987, 250, 571–2. [Google Scholar]
- Leung, D.; Kotzin, B.; Meissner, H.; Fulton, R.; Murray, D.; Schlievert, P. Toxic shock syndrome toxin-secreting Staphylococcus aureus in Kawasaki syndrome. Lancet 1993, 342, 1385–1388. [Google Scholar] [CrossRef]
- Matsubara, K.; Fukaya, T.; Miwa, K.; Shibayama, N.; Nigami, H.; Harigaya, H.; Nozaki, H.; Hirata, T.; Baba, K.; Suzuki, T.; et al. Development of serum IgM antibodies against superantigens ofStaphylococcus aureusandStreptococcus pyogenesin Kawasaki disease. Clin. Exp. Immunol. 2006, 143, 427–434. [Google Scholar] [CrossRef]
- Yoshioka, T.; Matsutani, T.; Iwagami, S.; Maeda, T.; Yutsudo, T.; Tsuruta, Y.; Suzuki, H.; Uemura, S.; Takeuchi, T.; Koike, M. Polyclonal expansion of TCRBV2- and TCRBV6-bearing T cells in patients with Kawasaki disease. Immunology 1999, 96, 465–472. [Google Scholar] [CrossRef]
- Kusuda, T.; Nakashima, Y.; Murata, K.; Kanno, S.; Nishio, H.; Saito, M.; Tanaka, T.; Yamamura, K.; Sakai, Y.; Takada, H.; et al. Kawasaki Disease-Specific Molecules in the Sera Are Linked to Microbe-Associated Molecular Patterns in the Biofilms. PLOS ONE 2014, 9, e113054–e113054. [Google Scholar] [CrossRef]
- Kang, J.-M.; Jung, J.; Kim, Y.-E.; Huh, K.; Hong, J.; Kim, D.W.; Kim, M.Y.; Jung, S.Y.; Kim, J.-H.; Ahn, J.G. Temporal Correlation Between Kawasaki Disease and Infectious Diseases in South Korea. JAMA Netw. Open 2022, 5, e2147363–e2147363. [Google Scholar] [CrossRef]
- Dean, A.G.; Melish, M.E.; Hicks, R.; Palumbo, N.E. An epidemic of Kawasaki syndrome in Hawaii. J. Pediatr. 1982, 100, 552–557. [Google Scholar] [CrossRef] [PubMed]
- Han, X.-Y.; Qi, H.-R. Pyroptosis in Kawasaki disease: from mechanisms to targeted interventions. Front. Immunol. 2025, 16, 1566985. [Google Scholar] [CrossRef] [PubMed]
- Jia, C.; Zhang, J.; Chen, H.; Zhuge, Y.; Chen, H.; Qian, F.; Zhou, K.; Niu, C.; Wang, F.; Qiu, H.; et al. Endothelial cell pyroptosis plays an important role in Kawasaki disease via HMGB1/RAGE/cathespin B signaling pathway and NLRP3 inflammasome activation. Cell Death Dis. 2019, 10, 1–16. [Google Scholar] [CrossRef] [PubMed]
- DeHaan, L.L.; Copeland, C.D.; Burney, J.A.; Nakamura, Y.; Yashiro, M.; Shimizu, C.; Miyata, K.; Burns, J.C.; Cayan, D.R. Age-Dependent Variations in Kawasaki Disease Incidence in Japan. JAMA Netw. Open 2024, 7, e2355001–e2355001. [Google Scholar] [CrossRef]
- Ae, R.; Shibata, Y.; Kosami, K.; Nakamura, Y.; Hamada, H. Kawasaki Disease and Pediatric Infectious Diseases During the Coronavirus Disease 2019 Pandemic. J. Pediatr. 2021, 239, 50–58.e2. [Google Scholar] [CrossRef]
- Burney, J.A.; Roberts, S.C.; DeHaan, L.L.; Shimizu, C.; Bainto, E.V.; Newburger, J.W.; Dominguez, S.; Jone, P.-N.; Jaggi, P.; Szmuszkovicz, J.R.; et al. Epidemiological and Clinical Features of Kawasaki Disease During the COVID-19 Pandemic in the United States. JAMA Netw. Open 2022, 5, e2217436–e2217436. [Google Scholar] [CrossRef]
- Schmöeller, D.; Keiserman, M.W.; Staub, H.L.M.; Velho, F.P.; Grohe, M.d.F. Yellow Fever Vaccination and Kawasaki Disease. Pediatr. Infect. Dis. J. 2009, 28, 1037–1038. [Google Scholar] [CrossRef]
- Ece, I; Akbayram, S; Demiroren, K; et al. Is Kawasaki Disease a Side Effect of Vaccination as Well? J Vaccines Vaccin n.d., 5, 234. [Google Scholar]
- Banday, A.Z.; Patra, P.K.; Jindal, A.K. Kawasaki disease – when Bacillus Calmette–Guérin (BCG) lymphadenitis blooms again and the vaccination site peels off! Int. J. Dermatol. 2020, 60, E233–E234. [Google Scholar] [CrossRef]
- Alsager, K.; Vadlamudi, N.K.; Jadavji, T.; Bettinger, J.A.; Constantinescu, C.; Vaudry, W.; Tan, B.; Sauvé, L.; Sadarangani, M.; Halperin, S.A.; et al. Kawasaki disease following immunization reported to the Canadian Immunization Monitoring Program ACTive (IMPACT) from 2013 to 2018. Hum. Vaccines Immunother. 2022, 18, 2088215. [Google Scholar] [CrossRef]
- Hall, G.C.; Tulloh, R.M.; E Tulloh, L. The incidence of Kawasaki disease after vaccination within the UK pre-school National Immunisation Programme: an observational THIN database study. Pharmacoepidemiol. Drug Saf. 2016, 25, 1331–1336. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.; Islam, S. Kawasaki disease and vasculitis associated with immunization. Pediatr. Int. 2018, 60, 613–617. [Google Scholar] [CrossRef] [PubMed]
- Miron, D.; Fink, D.; Hashkes, P.J. Kawasaki disease in an infant following immunisation with hepatitis B vaccine. Clin. Rheumatol. 2003, 22, 461–463. [Google Scholar] [CrossRef]
- Jeong, S.W.; Kim, D.H.; Han, M.Y.; Cha, S.; Yoon, K.L. An infant presenting with Kawasaki disease following immunization for influenza: A case report. Biomed. Rep. 2018, 8, 301–303. [Google Scholar] [CrossRef] [PubMed]
- Kraszewska-Głomba, B.; Kuchar, E.; Szenborn, L. Three episodes of Kawasaki disease including one after the Pneumo 23 vaccine in a child with a family history of Kawasaki disease. J. Formos. Med Assoc. 2016, 115, 885–886. [Google Scholar] [CrossRef]
- Shimada, S.; Watanabe, T.; Sato, S. A Patient with Kawasaki Disease Following Influenza Vaccinations. Pediatr. Infect. Dis. J. 2015, 34, 913. [Google Scholar] [CrossRef]
- Yin, S.; Liubao, P.; Chongqing, T.; Xiaomin, W. The first case of Kawasaki disease in a 20-month old baby following immunization with rotavirus vaccine and hepatitis A vaccine in China: A case report. Hum. Vaccines Immunother. 2015, 11, 2740–2743. [Google Scholar] [CrossRef]
- Matsubara, D.; Minami, T.; Seki, M.; Tamura, D.; Yamagata, T. Occurrence of Kawasaki disease after simultaneous immunization. Pediatr. Int. 2019, 61, 1171–1173. [Google Scholar] [CrossRef]
- Huang, W.-T.; Juan, Y.-C.; Liu, C.-H.; Yang, Y.-Y.; Chan, K.A. Intussusception and Kawasaki disease after rotavirus vaccination in Taiwanese infants. Vaccine 2020, 38, 6299–6303. [Google Scholar] [CrossRef]
- Showers, C.R.; Maurer, J.M.; Khakshour, D.; Shukla, M. Case of adult-onset Kawasaki disease and multisystem inflammatory syndrome following SARS-CoV-2 vaccination. BMJ Case Rep. 2022, 15, e249094. [Google Scholar] [CrossRef] [PubMed]
- Peralta-Amaro, A.L.; Tejada-Ruiz, M.I.; Rivera-Alvarado, K.L.; Cobos-Quevedo, O.d.J.; Romero-Hernández, P.; Macías-Arroyo, W.; Avendaño-Ponce, A.; Hurtado-Díaz, J.; Vera-Lastra, O.; Lucas-Hernández, A. Atypical Kawasaki Disease after COVID-19 Vaccination: A New Form of Adverse Event Following Immunization. Vaccines 2022, 10, 126. [Google Scholar] [CrossRef] [PubMed]
- Kinumaki, A.; Sekizuka, T.; Hamada, H.; Kato, K.; Yamashita, A.; Kuroda, M. Characterization of the gut microbiota of Kawasaki disease patients by metagenomic analysis. Front. Microbiol. 2015, 6, 824. [Google Scholar] [CrossRef]
- Esposito, S.; Polinori, I.; Rigante, D. The Gut Microbiota-Host Partnership as a Potential Driver of Kawasaki Syndrome. Front. Pediatr. 2019, 7, 124. [Google Scholar] [CrossRef]
- Burney, J.A.; DeHaan, L.L.; Shimizu, C.; Bainto, E.V.; Newburger, J.W.; DeBiasi, R.L.; Dominguez, S.R.; Portman, M.A.; Melish, M.; Bratincsak, A.; et al. Temporal clustering of Kawasaki disease cases around the world. Sci. Rep. 2021, 11, 1–10. [Google Scholar] [CrossRef]
- Rodó, X.; Curcoll, R.; Robinson, M.; Ballester, J.; Burns, J.C.; Cayan, D.R.; Lipkin, W.I.; Williams, B.L.; Couto-Rodriguez, M.; Nakamura, Y.; et al. Tropospheric winds from northeastern China carry the etiologic agent of Kawasaki disease from its source to Japan. Proc. Natl. Acad. Sci. USA 2014, 111, 7952–7957. [Google Scholar] [CrossRef]
- Kuo, N.-C.; Lin, C.-H.; Lin, M.-C. Prenatal and early life exposure to air pollution and the incidence of Kawasaki disease. Sci. Rep. 2022, 12, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Jung, C.-R.; Chen, W.-T.; Lin, Y.-T.; Hwang, B.-F. Ambient Air Pollutant Exposures and Hospitalization for Kawasaki Disease in Taiwan: A Case-Crossover Study (2000–2010). Environ. Health Perspect. 2017, 125, 670–676. [Google Scholar] [CrossRef]
- Kwon, D.; Choe, Y.J.; Kim, S.; Chun, B.C.; Choe, S. Ambient Air Pollution and Kawasaki Disease in Korean Children: A Study of the National Health Insurance Claim Data. J. Am. Hear. Assoc. 2022, 11, e024092. [Google Scholar] [CrossRef] [PubMed]
- Yoneda, K.; Shinjo, D.; Takahashi, N.; Fushimi, K. Spatiotemporal analysis of the association between Kawasaki disease incidence and PM2.5exposure: a nationwide database study in Japan. BMJ Paediatr. Open 2024, 8, e002887. [Google Scholar] [CrossRef]
- Seposo, A.K.C.; Madaniyazi, L.; Yoshida, L.M.; Toizumi, M. Incidence and Seasonality of Kawasaki Disease in Children in the Philippines, and its Association with Ambient Air Temperature. Isee Conf. Abstr. 2023. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, Y.; Zheng, F.; Guo, Y.; Wang, X.; Meng, H.; Min, L.; Hu, X. Research on the Influence of Air Pollutants and Meteorological Factors on Kawasaki Disease. Aerosol Air Qual. Res. 2025, 25. [Google Scholar] [CrossRef]
- Yang, P.; Zhang, J.; Zhang, K.; Zhang, D.; Liu, Y.; Wu, J.; Wei, Y.; Feng, S.; Yi, Q. Prenatal and Postnatal Ambient Air Pollution and Kawasaki Disease. JACC: Adv. 2025, 4, 101651. [Google Scholar] [CrossRef] [PubMed]
- Elouardi, Y.; Rebahi, H.; Zarrouki, Y.; Ziadi, A.; Younous, S.; Samkaoui, M. COVID-19 associated Kawasaki-like multisystem inflammatory syndrome in an adult. Rev. Esp. Anestesiol. Reanim. Engl. Ed. 2022, 69, 43–47. [Google Scholar] [CrossRef]
- Sharma, C.; Ganigara, M.; Galeotti, C.; Burns, J.; Berganza, F.M.; Hayes, D.A.; Singh-Grewal, D.; Bharath, S.; Sajjan, S.; Bayry, J. Multisystem inflammatory syndrome in children and Kawasaki disease: a critical comparison. Nat. Rev. Rheumatol. 2021, 17, 731–748. [Google Scholar] [CrossRef]
- on behalf of the Rheumatology Study Group of the Italian Pediatric Society; Cattalini, M.; Della Paolera, S.; Zunica, F.; Bracaglia, C.; Giangreco, M.; Verdoni, L.; Meini, A.; Sottile, R.; Caorsi, R.; et al. Defining Kawasaki disease and pediatric inflammatory multisystem syndrome-temporally associated to SARS-CoV-2 infection during SARS-CoV-2 epidemic in Italy: results from a national, multicenter survey. Pediatr. Rheumatol. 2021, 19, 1–11. [Google Scholar] [CrossRef]
- Loke, Y.-H.; Berul, C.I.; Harahsheh, A.S. Multisystem inflammatory syndrome in children: Is there a linkage to Kawasaki disease? Trends Cardiovasc. Med. 2020, 30, 389–396. [Google Scholar] [CrossRef]
- Darby, J.B.; Jackson, J.M. Kawasaki Disease and Multisystem Inflammatory Syndrome in Children: An Overview and Comparison. 2021, 104, 244–252. [Google Scholar]
- Cannon, L.; Campbell, M.J.; Wu, E.Y. Multisystem Inflammatory Syndrome in Children and Kawasaki Disease: Parallels in Pathogenesis and Treatment. Curr. Allergy Asthma Rep. 2023, 23, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Walton, M.; Raghuveer, G.; Harahsheh, A.; Portman, M.A.; Lee, S.; Khoury, M.; Dahdah, N.; Fabi, M.; Dionne, A.; Harris, T.H.; et al. Cardiac Biomarkers Aid in Differentiation of Kawasaki Disease from Multisystem Inflammatory Syndrome in Children Associated with COVID-19. Pediatr. Cardiol. 2023, 46, 116–126. [Google Scholar] [CrossRef]
- Rivas, M.N.; Arditi, M. Kawasaki Disease and Multisystem Inflammatory Syndrome in Children. Rheum. Dis. Clin. North Am. 2023, 49, 647–659. [Google Scholar] [CrossRef]
- Suzuki, J.; Abe, K.; Matsui, T.; Honda, T.; Yasukawa, K.; Takanashi, J.-I.; Hamada, H. Kawasaki Disease Shock Syndrome in Japan and Comparison With Multisystem Inflammatory Syndrome in Children in European countries. Front. Pediatr. 2021, 9, 625456. [Google Scholar] [CrossRef]
- Lee, S.; Harahsheh, A.S.; Raghuveer, G.; Portman, M.A.; Sabati, A.A.; Khoury, M.; Dahdah, N.; Fabi, M.; Jain, S.S.; Dionne, A.; et al. Spectrum of Coronary Artery Involvement With Multisystem Inflammatory Syndrome in Children Versus Kawasaki Disease. J. Am. Hear. Assoc. 2025, 14, e037761. [Google Scholar] [CrossRef]
- Weiler, C.R.; Austen, K.F.; Akin, C.; Barkoff, M.S.; Bernstein, J.A.; Bonadonna, P.; Butterfield, J.H.; Carter, M.; Fox, C.C.; Maitland, A.; et al. AAAAI Mast Cell Disorders Committee Work Group Report: Mast cell activation syndrome (MCAS) diagnosis and management. J. Allergy Clin. Immunol. 2019, 144, 883–896. [Google Scholar] [CrossRef] [PubMed]
- Ricke, D.O. Vaccine-associated Kawasaki disease in children. Microbes Immun. 2025, 025200044. [Google Scholar] [CrossRef]
- Vignesh, P.; Rawat, A.; Shandilya, J.K.; Sachdeva, M.U.S.; Ahluwalia, J.; Singh, S. Monocyte platelet aggregates in children with Kawasaki disease- a preliminary study from a tertiary care centre in North-West India. Pediatr. Rheumatol. 2021, 19, 1–7. [Google Scholar] [CrossRef]
- Kim, S.H.; Hwang, I.J.; Cho, Y.K. Platelet Indices as Diagnostic Marker for Kawasaki Disease. Chonnam Med J. 2022, 58, 110–118. [Google Scholar] [CrossRef]
- Souni, G.; Ayad, G.; Elouali, A.; Rkain, M.; Babakhouya, A. Severe Kawasaki Disease and Thrombocytopenia: A Case Report. Cureus 2023, 15, e42916. [Google Scholar] [CrossRef]
- Beken, B.; Ünal, Ş.; Çetin, M.; Gümrük, F. The Relationship Between Hematological Findings and Coronary Artery Aneurysm in Kawasaki Disease. Turk. J. Hematol. 2014, 31, 199–200. [Google Scholar] [CrossRef] [PubMed]
- Kocatürk, B.; Lee, Y.; Nosaka, N.; Abe, M.; Martinon, D.; Lane, M.E.; Moreira, D.; Chen, S.; Fishbein, M.C.; Porritt, R.A.; et al. Platelets exacerbate cardiovascular inflammation in a murine model of Kawasaki disease vasculitis. J. Clin. Investig. 2023, 8. [Google Scholar] [CrossRef]
- Ricke, D.O.; Smith, N. VAERS Vasculitis Adverse Events Retrospective Study: Etiology Model of Immune Complexes Activating Fc Receptors in Kawasaki Disease and Multisystem Inflammatory Syndromes. Life 2024, 14, 353. [Google Scholar] [CrossRef]
- Zhang, H.-Y.; Xiao, M.; Zhou, D.; Yan, F.; Zhang, Y. Platelet and ferritin as early predictive factors for the development of macrophage activation syndrome in children with Kawasaki disease: A retrospective case-control study. Front. Pediatr. 2023, 11, 1088525. [Google Scholar] [CrossRef]
- VAERS. Vaccine Adverse Event Reporting System. U.S. Department of Health & Human Services, 2025. [Google Scholar]
- Ricke, DO. VAERS-Tools 2025.
- Gupta, K.; Subramanian, H.; Ali, H. Modulation of host defense peptide-mediated human mast cell activation by LPS. J. Endotoxin Res. 2015, 22, 21–30. [Google Scholar] [CrossRef]
- Schwarz, H.; Gornicec, J.; Neuper, T.; Parigiani, M.A.; Wallner, M.; Duschl, A.; Horejs-Hoeck, J. Biological Activity of Masked Endotoxin. Sci. Rep. 2017, 7, srep44750. [Google Scholar] [CrossRef]
- Petruk, G.; Puthia, M.; Petrlova, J.; Samsudin, F.; Strömdahl, A.-C.; Cerps, S.; Uller, L.; Kjellström, S.; Bond, P.J.; Schmidtchen, A. SARS-CoV-2 Spike protein binds to bacterial lipopolysaccharide and boosts proinflammatory activity. J. Mol. Cell Biol. 2020, 12, 916–932. [Google Scholar] [CrossRef]
- Samsudin, F.; Raghuvamsi, P.; Petruk, G.; Puthia, M.; Petrlova, J.; MacAry, P.; Anand, G.S.; Bond, P.J.; Schmidtchen, A. SARS-CoV-2 spike protein as a bacterial lipopolysaccharide delivery system in an overzealous inflammatory cascade. J. Mol. Cell Biol. 2022, 14, 1–14. [Google Scholar] [CrossRef]
- Ciesielska, A.; Matyjek, M.; Kwiatkowska, K. TLR4 and CD14 trafficking and its influence on LPS-induced pro-inflammatory signaling. Cell. Mol. Life Sci. 2021, 78, 1233–1261. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, A.; Ikeda, K.; Hamaoka, K. Aetiological Significance of Infectious Stimuli in Kawasaki Disease. Front. Pediatr. 2019, 7, 244. [Google Scholar] [CrossRef] [PubMed]
- Mitsui, K.; Yusa, T.; Miyazaki, S.; Ohara, A.; Saji, T. Increased TLR2 and TLR4 Expression in Peripheral Neutrophils Isolated from Kawasaki Disease. Pediatr. Allergy, Immunol. Pulmonol. 2014, 27, 24–29. [Google Scholar] [CrossRef]
- Takeshita, S.; Nakatani, K.; Tsujimoto, H.; Kawamura, Y.; Kawase, H.; Sekine, I. Increased levels of circulating soluble CD14 in Kawasaki disease. Clin. Exp. Immunol. 2000, 119, 376–381. [Google Scholar] [CrossRef]
- Takeshita, S.; Tsujimoto, H.; Kawase, H.; Kawamura, Y.; Sekine, I. Increased Levels of Lipopolysaccharide Binding Protein in Plasma in Children with Kawasaki Disease. Clin. Vaccine Immunol. 2002, 9, 205–6. [Google Scholar] [CrossRef]
- Rice, J.B.; Stoll, L.L.; Li, W.-G.; Denning, G.M.; Weydert, J.; Charipar, E.; Richenbacher, W.E.; Miller, F.J.; Weintraub, N.L. Low-Level Endotoxin Induces Potent Inflammatory Activation of Human Blood Vessels. Arter. Thromb. Vasc. Biol. 2003, 23, 1576–1582. [Google Scholar] [CrossRef]
- Baker, A.L.; Lu, M.; Minich, L.L.; Atz, A.M.; Klein, G.L.; Korsin, R.; Lambert, L.; Li, J.S.; Mason, W.; Radojewski, E.; et al. Associated Symptoms in the Ten Days Before Diagnosis of Kawasaki Disease. J. Pediatr. 2009, 154, 592–595.e2. [Google Scholar] [CrossRef]
- Colomba, C.; La Placa, S.; Saporito, L.; Corsello, G.; Ciccia, F.; Medaglia, A.; Romanin, B.; Serra, N.; Di Carlo, P.; Cascio, A. Intestinal Involvement in Kawasaki Disease. J. Pediatr. 2018, 202, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Tao, E.; Lang, D. Unraveling the gut: the pivotal role of intestinal mechanisms in Kawasaki disease pathogenesis. Front. Immunol. 2024, 15, 1496293. [Google Scholar] [CrossRef]
- Curtis, N.; Zheng, R.; Lamb, J.R.; Levin, M. Evidence for a superantigen mediated process in Kawasaki disease. Arch. Dis. Child. 1995, 72, 308–311. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, K.; Akagawa, S.; Akagawa, Y.; Kimata, T.; Tsuji, S. Our Evolving Understanding of Kawasaki Disease Pathogenesis: Role of the Gut Microbiota. Front. Immunol. 2020, 11, 1616. [Google Scholar] [CrossRef]
- Fabi, M.; Corinaldesi, E.; Pierantoni, L.; Mazzoni, E.; Landini, C.; Bigucci, B.; Ancora, G.; Malaigia, L.; Bodnar, T.; Di Fazzio, G.; et al. Gastrointestinal presentation of Kawasaki disease: A red flag for severe disease? PLOS ONE 2018, 13, e0202658. [Google Scholar] [CrossRef]
- Ricke, D.O. Two Different Antibody-Dependent Enhancement (ADE) Risks for SARS-CoV-2 Antibodies. Front. Immunol. 2021, 12, 640093. [Google Scholar] [CrossRef]
- Ricke, D.; Gherlone, N.; Fremont-Smith, P.; Tisdall, P.; Fremont-Smith, M. Kawasaki Disease, Multisystem Inflammatory Syndrome in Children: Antibody-Induced Mast Cell Activation Hypothesis. J. Pediatr. Pediatr. Med. 2020, 4, 1–7. [Google Scholar] [CrossRef]
- Riollano-Cruz, M.; Akkoyun, E.; Briceno-Brito, E.; Kowalsky, S.; Reed, J.; Posada, R.; Sordillo, E.M.; Tosi, M.; Trachtman, R.; Paniz-Mondolfi, A. Multisystem inflammatory syndrome in children related to COVID-19: A New York City experience. J. Med Virol. 2020, 93, 424–433. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.F.B.; Litvinov, N.; Farhat, S.C.L.; Eisencraft, A.P.; Gibelli, M.A.B.C.; de Carvalho, W.B.; Fernandes, V.R.; Fink, T.d.T.; Framil, J.V.d.S.; Galleti, K.V.; et al. Severe clinical spectrum with high mortality in pediatric patients with COVID-19 and multisystem inflammatory syndrome. Clinics 2020, 75, e2209. [Google Scholar] [CrossRef]
- Torres, J.P.; Izquierdo, G.; Acuña, M.; Pavez, D.; Reyes, F.; Fritis, A.; González, R.; Rivacoba, C.; Contardo, V.; Tapia, L.I. Multisystem inflammatory syndrome in children (MIS-C): Report of the clinical and epidemiological characteristics of cases in Santiago de Chile during the SARS-CoV-2 pandemic. Int. J. Infect. Dis. 2020, 100, 75–81. [Google Scholar] [CrossRef]
- Sahn, B.; Eze, O.P.; Edelman, M.C.; Chougar, C.E.; Thomas, R.M.; Schleien, C.L.M.; Weinstein, T. Features of Intestinal Disease Associated With COVID-Related Multisystem Inflammatory Syndrome in Children. J. Pediatr. Gastroenterol. Nutr. 2020, 72, 384–387. [Google Scholar] [CrossRef]
- Miller, J.; Cantor, A.; Zachariah, P.; Ahn, D.; Martinez, M.; Margolis, K.G. Gastrointestinal Symptoms as a Major Presentation Component of a Novel Multisystem Inflammatory Syndrome in Children That Is Related to Coronavirus Disease 2019: A Single Center Experience of 44 Cases. Gastroenterology 2020, 159, 1571–1574.e2. [Google Scholar] [CrossRef] [PubMed]
- Kofman, A.D.; Sizemore, E.K.; Detelich, J.F.; Albrecht, B.; Piantadosi, A.L. A young adult with COVID-19 and multisystem inflammatory syndrome in children (MIS-C)-like illness: a case report. BMC Infect. Dis. 2020, 20, 716. [Google Scholar] [CrossRef]
- Tsilioni, I.; Theoharides, T.C. Recombinant SARS-CoV-2 Spike Protein Stimulates Secretion of Chymase, Tryptase, and IL-1β from Human Mast Cells, Augmented by IL-33. Int. J. Mol. Sci. 2023, 24, 9487. [Google Scholar] [CrossRef]
- Chang, L.-S.; Guo, M.M.-H.; Lo, M.-H.; Kuo, H.-C. Identification of increased expression of activating Fc receptors and novel findings regarding distinct IgE and IgM receptors in Kawasaki disease. Pediatr. Res. 2019, 89, 191–197. [Google Scholar] [CrossRef]
- Hrubisko, M.; Danis, R.; Huorka, M.; Wawruch, M. Histamine Intolerance—The More We Know the Less We Know. A Review. Nutrients 2021, 13, 2228. [Google Scholar] [CrossRef] [PubMed]
- Kovacova-Hanuskova, E.; Buday, T.; Gavliakova, S.; Plevkova, J. Histamine, histamine intoxication and intolerance. Allergol. et Immunopathol. 2015, 43, 498–506. [Google Scholar] [CrossRef]
- Ricke, DO. Etiology Scenarios for Multisystem Inflammatory Syndrome in Children and Adults Associated with SARS-CoV-2. J Integr Pediatr Heal n.d., 3, 1–5. [Google Scholar]
- Vikenes, K.; Farstad, M.; Nordrehaug, J.E. Serotonin Is Associated with Coronary Artery Disease and Cardiac Events. Circulation 1999, 100, 483–489. [Google Scholar] [CrossRef]
- Golino, P.; Piscione, F.; Willerson, J.T.; Cappelli-Bigazzi, M.; Focaccio, A.; Villari, B.; Indolfi, C.; Russolillo, E.; Condorelli, M.; Chiariello, M. Divergent Effects of Serotonin on Coronary-Artery Dimensions and Blood Flow in Patients with Coronary Atherosclerosis and Control Patients. New Engl. J. Med. 1991, 324, 641–648. [Google Scholar] [CrossRef]
- Fremont-Smith, M.; Gherlone, N.; Smith, N.; Tisdall, P.; Ricke, D.O. Models for COVID-19 Early Cardiac Pathology Following SARS-CoV-2 Infection. Int. J. Infect. Dis. 2021, 113, 331–335. [Google Scholar] [CrossRef]
- Liu, X.; Chen, Y.; Yang, Y.; Su, Z.; Wang, F.; Zhanghuang, C.; Wu, Y.; Zhang, X. Association between FGA gene polymorphisms and coronary artery lesion in Kawasaki disease. Front. Med. 2023, 10, 1193303. [Google Scholar] [CrossRef]
- Levick, S.P.; Meléndez, G.C.; Plante, E.; McLarty, J.L.; Brower, G.L.; Janicki, J.S. Cardiac mast cells: the centrepiece in adverse myocardial remodelling. Cardiovasc. Res. 2010, 89, 12–19. [Google Scholar] [CrossRef]
- Yonker, L.M.; Swank, Z.; Bartsch, Y.C.; Burns, M.D.; Kane, A.; Boribong, B.P.; Davis, J.P.; Loiselle, M.; Novak, T.; Senussi, Y.; et al. Circulating Spike Protein Detected in Post–COVID-19 mRNA Vaccine Myocarditis. Circulation 2023, 147, 867–876. [Google Scholar] [CrossRef] [PubMed]
- Bozkurt, B.; Kamat, I.; Hotez, P.J. Myocarditis With COVID-19 mRNA Vaccines. Circulation 2021, 144, 471–484. [Google Scholar] [CrossRef]
- Avolio, E.; Carrabba, M.; Milligan, R.; Williamson, M.K.; Beltrami, A.P.; Gupta, K.; Elvers, K.T.; Gamez, M.; Foster, R.R.; Gillespie, K.; et al. The SARS-CoV-2 Spike protein disrupts human cardiac pericytes function through CD147 receptor-mediated signalling: a potential non-infective mechanism of COVID-19 microvascular disease. Clin. Sci. 2021, 135, 2667–2689. [Google Scholar] [CrossRef]
- Ricke, D.O. Cardiac adverse events post-vaccination. Brain Hear. 2025, 3, 5747. [Google Scholar] [CrossRef]
- Shulman, S.T. Pediatric Coronavirus Disease-2019–Associated Multisystem Inflammatory Syndrome. J. Pediatr. Infect. Dis. Soc. 2020, 9, 285–286. [Google Scholar] [CrossRef]
- Vella, L.A.; Rowley, A.H. Current Insights Into the Pathophysiology of Multisystem Inflammatory Syndrome in Children. Curr. Pediatr. Rep. 2021, 9, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Rostad, C.A.; Chahroudi, A.; Mantus, G.; Lapp, S.A.; Teherani, M.; Macoy, L.; Tarquinio, K.M.; Basu, R.K.; Kao, C.; Linam, W.M.; et al. Quantitative SARS-CoV-2 Serology in Children With Multisystem Inflammatory Syndrome (MIS-C). Pediatrics 2020, 146. [Google Scholar] [CrossRef] [PubMed]
- Anderson, E.M.; Diorio, C.; Goodwin, E.C.; O McNerney, K.; E Weirick, M.; Gouma, S.; Bolton, M.J.; Arevalo, C.P.; Chase, J.; Hicks, P.; et al. Severe Acute Respiratory Syndrome-Coronavirus-2 (SARS-CoV-2) Antibody Responses in Children With Multisystem Inflammatory Syndrome in Children (MIS-C) and Mild and Severe Coronavirus Disease 2019 (COVID-19). J. Pediatr. Infect. Dis. Soc. 2020, 10, 669–673. [Google Scholar] [CrossRef]
- Gruber, C.N.; Patel, R.S.; Trachtman, R.; Lepow, L.; Amanat, F.; Krammer, F.; Wilson, K.M.; Onel, K.; Geanon, D.; Tuballes, K.; et al. Mapping Systemic Inflammation and Antibody Responses in Multisystem Inflammatory Syndrome in Children (MIS-C). Cell 2020, 183, 982–995.e14. [Google Scholar] [CrossRef]
- Moraleda, C.; Serna-Pascual, M.; Soriano-Arandes, A.; Simó, S.; Epalza, C.; Santos, M.; Grasa, C.; Rodríguez, M.; Soto, B.; Gallego, N.; et al. Multi-inflammatory Syndrome in Children Related to Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) in Spain. Clin. Infect. Dis. 2020, 72, e397–e401. [Google Scholar] [CrossRef]
- Grazioli, S.; Tavaglione, F.; Torriani, G.; Wagner, N.; Rohr, M.; L’hUillier, A.G.; Leclercq, C.; Perrin, A.; Bordessoule, A.; Beghetti, M.; et al. Immunological Assessment of Pediatric Multisystem Inflammatory Syndrome Related to Coronavirus Disease 2019. J. Pediatr. Infect. Dis. Soc. 2020, 10, 706–713. [Google Scholar] [CrossRef]
- Cirks, B.T.; Rowe, S.J.; Jiang, S.Y.; Brooks, R.M.; Mulreany, M.P.; Hoffner, W.; Jones, O.Y.; Hickey, P.W. Sixteen Weeks Later: Expanding the Risk Period for Multisystem Inflammatory Syndrome in Children. J. Pediatr. Infect. Dis. Soc. 2021, 10, 686–690. [Google Scholar] [CrossRef]
- Al Ameer, H.H.; AlKadhem, S.M.; Busaleh, F.; AlKhwaitm, S.; Llaguno, M.B.B. Multisystem Inflammatory Syndrome in Children Temporally Related to COVID-19: A Case Report From Saudi Arabia. Cureus 2020, 12, e10589. [Google Scholar] [CrossRef]
- Abbas, M.; Törnhage, C.-J. Family Transmission of COVID-19 Including a Child with MIS-C and Acute Pancreatitis. Int. Med Case Rep. J. 2021, ume 14, 55–65. [Google Scholar] [CrossRef]
- Bartsch, Y.C.; Wang, C.; Zohar, T.; Fischinger, S.; Atyeo, C.; Burke, J.S.; Kang, J.; Edlow, A.G.; Fasano, A.; Baden, L.R.; et al. Humoral signatures of protective and pathological SARS-CoV-2 infection in children. Nat. Med. 2021, 27, 454–462. [Google Scholar] [CrossRef]
- Clouser, K.M.; Baer, A.; Bhavsar, S.; Gadhavi, J.M.; Li, S.M.; Schnall, J.; Weiss, J.E. MIS-C After ARDS Associated With SARS-CoV-2. Pediatr. Infect. Dis. J. 2020, 39, e363–e365. [Google Scholar] [CrossRef] [PubMed]
- Onouchi, Y.; Gunji, T.; Burns, J.C.; Shimizu, C.; Newburger, J.W.; Yashiro, M.; Nakamura, Y.; Yanagawa, H.; Wakui, K.; Fukushima, Y.; et al. ITPKC functional polymorphism associated with Kawasaki disease susceptibility and formation of coronary artery aneurysms. Nat. Genet. 2007, 40, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Kuo, H.-C.; Hsu, Y.-W.; Wu, C.-M.; Chen, S.H.-Y.; Hung, K.-S.; Chang, W.-P.; Yang, K.D.; Hsieh, K.-S.; Chen, W.-C.; Onouchi, Y. A Replication Study for Association of ITPKC and CASP3 Two-Locus Analysis in IVIG Unresponsiveness and Coronary Artery Lesion in Kawasaki Disease. PLOS ONE 2013, 8, e69685. [Google Scholar] [CrossRef] [PubMed]
- Onouchi, Y.; Ozaki, K.; Buns, J.C.; Shimizu, C.; Hamada, H.; Honda, T.; Terai, M.; Honda, A.; Takeuchi, T.; Shibuta, S.; et al. Common variants in CASP3 confer susceptibility to Kawasaki disease. Hum. Mol. Genet. 2010, 19, 2898–2906. [Google Scholar] [CrossRef]
- Kim, J.; Shimizu, C.; Kingsmore, S.F.; Veeraraghavan, N.; Levy, E.; Dos Santos, A.M.R.; Yang, H.; Flatley, J.; Hoang, L.T.; Hibberd, M.L.; et al. Whole genome sequencing of an African American family highlights toll like receptor 6 variants in Kawasaki disease susceptibility. PLOS ONE 2017, 12, e0170977. [Google Scholar] [CrossRef]
- Khor, C.C.; Davila, S.; Breunis, W.B.; Lee, Y.-C.; et al.; Hong Kong–Shanghai Kawasaki Disease Genetics Consortium; Korean Kawasaki Disease Genetics Consortium; Taiwan Kawasaki Disease Genetics Consortium; International Kawasaki Disease Genetics Consortium; US Kawasaki Disease Genetics Consortium; Blue Mountains Eye Study Genome-wide association study identifies FCGR2A as a susceptibility locus for Kawasaki disease. Nat. Genet. 2011, 43, 1241–1246. [Google Scholar] [CrossRef]
- Duan, J.; Lou, J.; Zhang, Q.; Ke, J.; Qi, Y.; Shen, N.; Zhu, B.; Zhong, R.; Wang, Z.; Liu, L.; et al. A Genetic Variant rs1801274 in FCGR2A as a Potential Risk Marker for Kawasaki Disease: A Case-Control Study and Meta-Analysis. PLOS ONE 2014, 9, e103329. [Google Scholar] [CrossRef] [PubMed]
- Sim, B.K.; Park, H.; Kim, J.-J.; Yun, S.W.; Yu, J.J.; Yoon, K.L.; Lee, K.-Y.; Kil, H.-R.; Kim, G.B.; Han, M.-K.; et al. Assessment of the Clinical Heterogeneity of Kawasaki Disease Using Genetic Variants of BLK and FCGR2A. Korean Circ. J. 2019, 49, 99–108. [Google Scholar] [CrossRef]
- Kuo, H.-C.; Hsu, Y.-W.; Wu, M.-S.; Woon, P.Y.; Wong, H.S.-C.; Tsai, L.-J.; Lin, R.-K.; Klahan, S.; Hsieh, K.-S.; Chang, W.-C. FCGR2A Promoter Methylation and Risks for Intravenous Immunoglobulin Treatment Responses in Kawasaki Disease. Mediat. Inflamm. 2015, 2015, 564625. [Google Scholar] [CrossRef]
- Cariaso, M.; Lennon, G. SNPedia: a wiki supporting personal genome annotation, interpretation and analysis. Nucleic Acids Res. 2011, 40, D1308–D1312. [Google Scholar] [CrossRef]
- Kwon, Y.-C.; Kim, J.-J.; Yun, S.W.; Yu, J.J.; Yoon, K.L.; Lee, K.-Y.; Kil, H.-R.; Kim, G.B.; Han, M.-K.; Song, M.S.; et al. Male-specific association of the FCGR2A His167Arg polymorphism with Kawasaki disease. PLOS ONE 2017, 12, e0184248. [Google Scholar] [CrossRef] [PubMed]
- Onouchi, Y. Molecular Genetics of Kawasaki Disease. Pediatr. Res. 2009, 65, 46R–54R. [Google Scholar] [CrossRef] [PubMed]
- Burns, J.C. The etiologies of Kawasaki disease. J. Clin. Investig. 2024, 134. [Google Scholar] [CrossRef]
- Lee, Y.-C.; Kuo, H.-C.; Chang, J.-S.; Chang, L.-Y.; Huang, L.-M.; Chen, M.-R.; Liang, C.-D.; Chi, H.; Huang, F.-Y.; Lee, M.-L.; et al. Two new susceptibility loci for Kawasaki disease identified through genome-wide association analysis. Nat. Genet. 2012, 44, 522–525. [Google Scholar] [CrossRef]
- Onouchi, Y.; Ozaki, K.; Burns, J.C.; Shimizu, C.; Terai, M.; Hamada, H.; Honda, T.; Suzuki, H.; et al.; Japan Kawasaki Disease Genome Consortium; US Kawasaki Disease Genetics Consortium A genome-wide association study identifies three new risk loci for Kawasaki disease. Nat. Genet. 2012, 44, 517–521. [Google Scholar] [CrossRef]
- Chang, C.-J.; Kuo, H.-C.; Chang, J.-S.; Lee, J.-K.; Tsai, F.-J.; Khor, C.C.; Chang, L.-C.; Chen, S.-P.; Ko, T.-M.; Liu, Y.-M.; et al. Replication and Meta-Analysis of GWAS Identified Susceptibility Loci in Kawasaki Disease Confirm the Importance of B Lymphoid Tyrosine Kinase (BLK) in Disease Susceptibility. PLOS ONE 2013, 8, e72037. [Google Scholar] [CrossRef]
- Shrestha, S.; Wiener, H.; Shendre, A.; Kaslow, R.A.; Wu, J.; Olson, A.; Bowles, N.E.; Patel, H.; Edberg, J.C.; Portman, M.A. Role of Activating Fc γ R Gene Polymorphisms in Kawasaki Disease Susceptibility and Intravenous Immunoglobulin Response. Circ. Cardiovasc. Genet. 2012, 5, 309–316. [Google Scholar] [CrossRef]
- Shrestha, S.; Wiener, H.W.; Olson, A.K.; Edberg, J.C.; Bowles, N.E.; Patel, H.; Portman, M.A. Functional FCGR2B gene variants influence intravenous immunoglobulin response in patients with Kawasaki disease. J. Allergy Clin. Immunol. 2011, 128, 677–680.e1. [Google Scholar] [CrossRef] [PubMed]
- Makowsky, R.; Wiener, H.W.; Ptacek, T.S.; Silva, M.; Shendre, A.; Edberg, J.C.; Portman, M.A.; Shrestha, S. FcγR gene copy number in Kawasaki disease and intravenous immunoglobulin treatment response. Pharmacogenetics Genom. 2013, 23, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Nagelkerke, S.Q.; Schmidt, D.E.; de Haas, M.; Kuijpers, T.W. Genetic Variation in Low-To-Medium-Affinity Fcγ Receptors: Functional Consequences, Disease Associations, and Opportunities for Personalized Medicine. Front. Immunol. 2019, 10, 2237. [Google Scholar] [CrossRef]
- Johnson, T.A.; Mashimo, Y.; Wu, J.-Y.; Yoon, D.; Hata, A.; Kubo, M.; Takahashi, A.; Tsunoda, T.; Ozaki, K.; Tanaka, T.; et al. Association of an IGHV3-66 gene variant with Kawasaki disease. J. Hum. Genet. 2020, 66, 475–489. [Google Scholar] [CrossRef]
- Onouchi, Y.; Fukazawa, R.; Yamamura, K.; Suzuki, H.; Kakimoto, N.; Suenaga, T.; Takeuchi, T.; Hamada, H.; Honda, T.; Yasukawa, K.; et al. Variations in ORAI1 Gene Associated with Kawasaki Disease. PLOS ONE 2016, 11, e0145486–e0145486. [Google Scholar] [CrossRef]
- Kim, J.-J.; Park, Y.-M.; Yoon, D.; Lee, K.-Y.; Song, M.S.; Lee, H.D.; Kim, K.-J.; Park, I.-S.; Nam, H.-K.; et al.; Korean Kawasaki Disease Genetics Consortium Identification of KCNN2 as a susceptibility locus for coronary artery aneurysms in Kawasaki disease using genome-wide association analysis. J. Hum. Genet. 2013, 58, 521–525. [Google Scholar] [CrossRef]
- Shimizu, C.; Jain, S.; Davila, S.; Hibberd, M.L.; Lin, K.O.; Molkara, D.; Frazer, J.R.; Sun, S.; Baker, A.L.; Newburger, J.W.; et al. Transforming Growth Factor-β Signaling Pathway in Patients With Kawasaki Disease. Circ. Cardiovasc. Genet. 2011, 4, 16–25. [Google Scholar] [CrossRef]
- Choi, Y.M.; Shim, K.S.; Yoon, K.L.; Han, M.Y.; Cha, S.H.; Kim, S.K.; Jung, J.H. Transforming growth factor beta receptor II polymorphisms are associated with Kawasaki disease. Korean J. Pediatr. 2012, 55, 18–23. [Google Scholar] [CrossRef]
- Cho, J.H.; Han, M.Y.; Cha, S.H.; Jung, J.H.; Yoon, K.L. Genetic Polymorphism of SMAD5 is Associated With Kawasaki Disease. Pediatr. Cardiol. 2013, 35, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Faroldi, G.; Melo, F.R.; Rönnberg, E.; Grujic, M.; Pejler, G. Active Caspase-3 Is Stored within Secretory Compartments of Viable Mast Cells. J. Immunol. 2013, 191, 1445–1452. [Google Scholar] [CrossRef] [PubMed]
- Palma, A.M.; Hanes, M.R.; Marshall, J.S. Mast Cell Modulation of B Cell Responses: An Under-Appreciated Partnership in Host Defence. Front. Immunol. 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Hu, W. Identifying differentially expressed genes and miRNAs in Kawasaki disease by bioinformatics analysis. Sci. Rep. 2022, 12, 1–14. [Google Scholar] [CrossRef]
- Gu, W.; Mirsaidi-Madjdabadi, S.; Ramirez, F.; Simonson, T.S.; Makino, A.; Gu, \. Transcriptome meta-analysis of Kawasaki disease in humans and mice. Front. Pediatr. 2024, 12, 1423958. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Sun, Y.; Meng, L.; Ye, C.; Han, H.; Zhang, T.; Feng, Y.; Li, J.; Duan, L.; Chen, Y. Whole-exome sequencing analysis identifies novel variants associated with Kawasaki disease susceptibility. Pediatr. Rheumatol. 2023, 21, 1–7. [Google Scholar] [CrossRef]
- Yeşiltepe, E.; Duman, D.; Kuyucu, N.; Bozdoğan, S.T.; Çıtırık, L.; Yeşil, E.; Karpuz, D. FCGR2A Gene Polymorphism Association in Children with Multisystem Inflammatory Syndrome. Indian Pediatr. 2025, 62, 372–377. [Google Scholar] [CrossRef]
- Jia, S.; Li, C.; Wang, G.; Yang, J.; Zu, Y. The T helper type 17/regulatory T cell imbalance in patients with acute Kawasaki disease. Clin. Exp. Immunol. 2010, 162, 131–137. [Google Scholar] [CrossRef]
- CDC. Weekly U.S. MIS-C Cases and COVID-19 Percent Positivity Reported to CDC 2025.
- Centers for Disease Control and Prevention. COVID data tracker - Wastewater Suveillance (2021). Available online: https://covid.cdc.gov/covid-data-tracker/#wastewdatatracker-home (accessed on 11 April 2022).
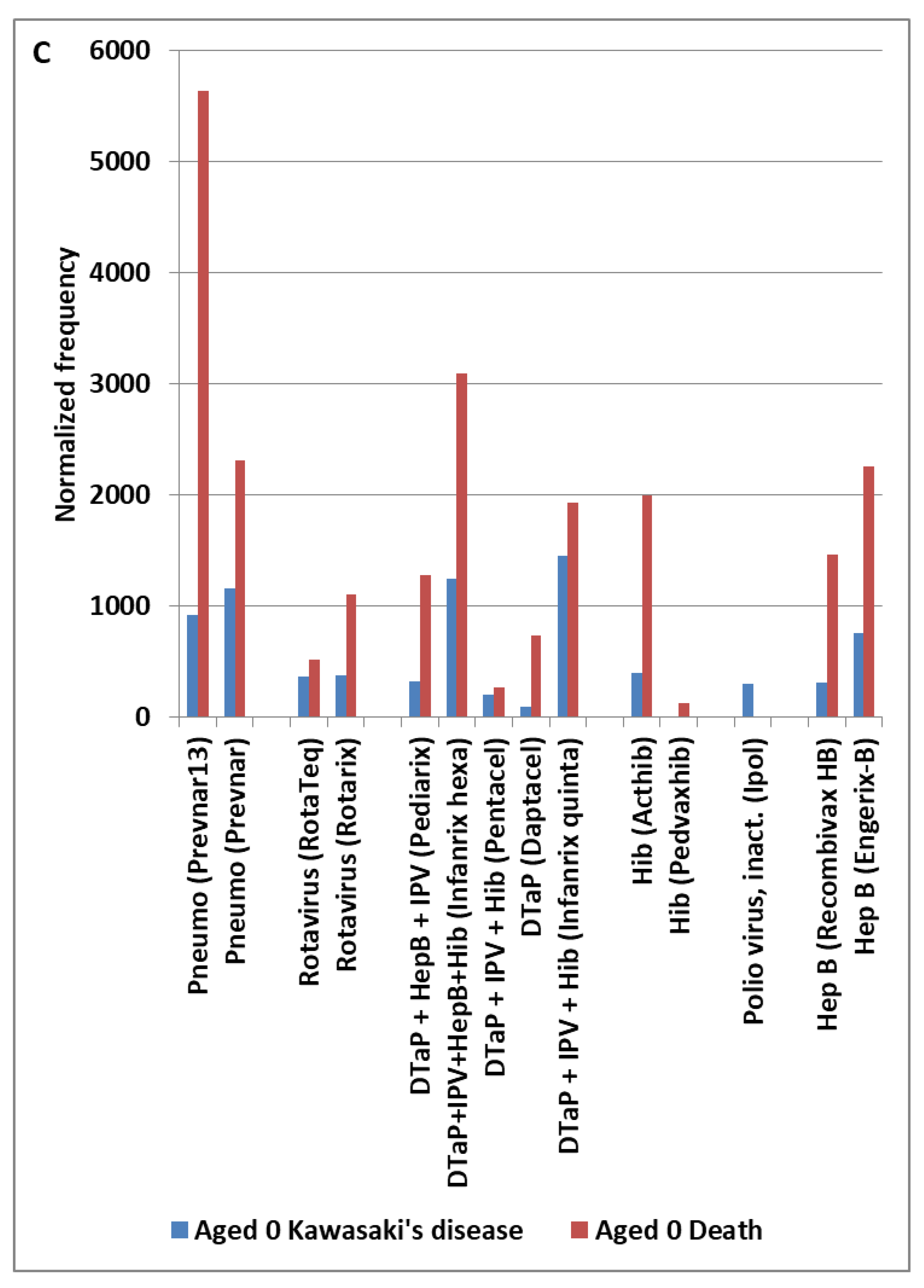
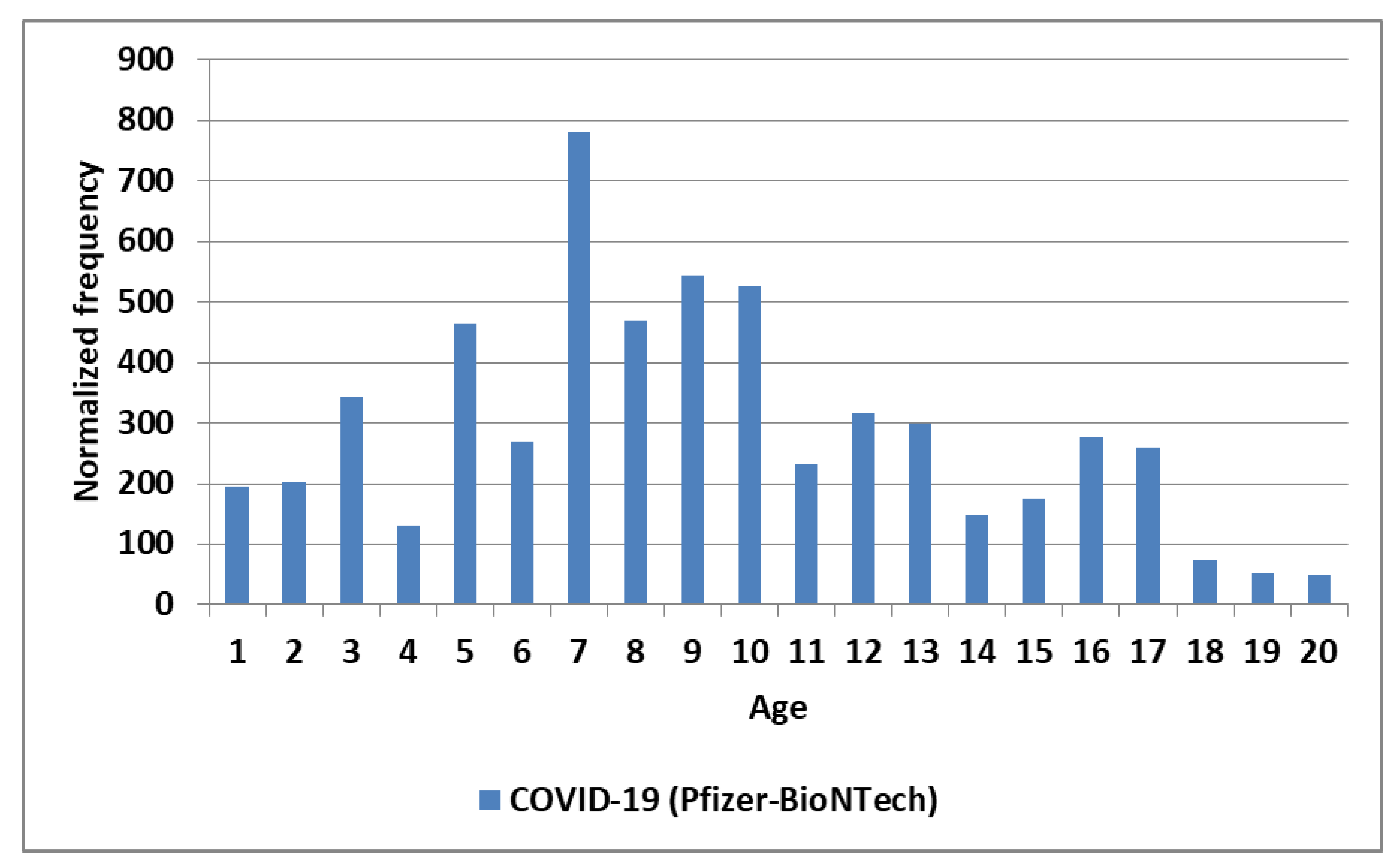
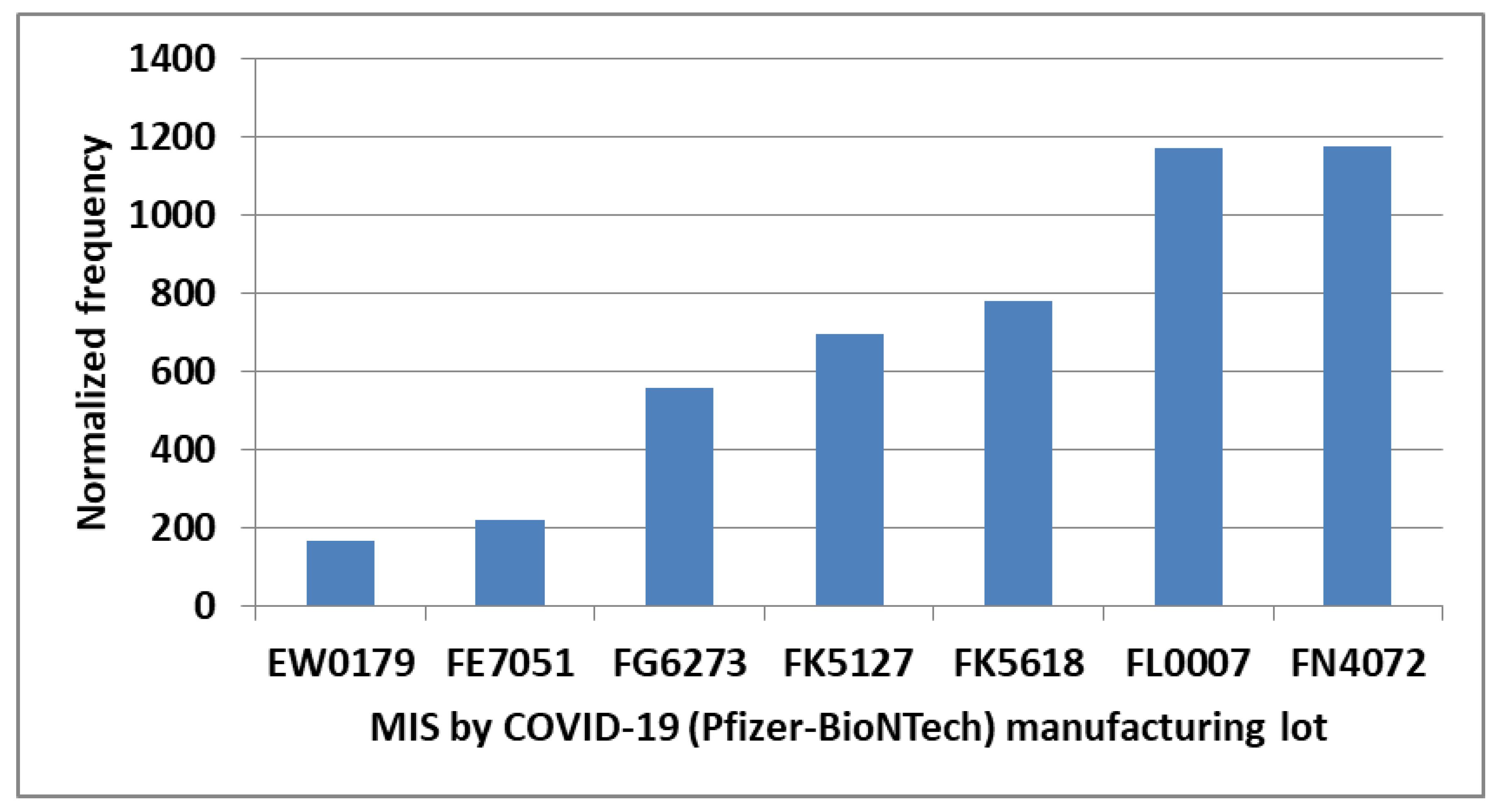
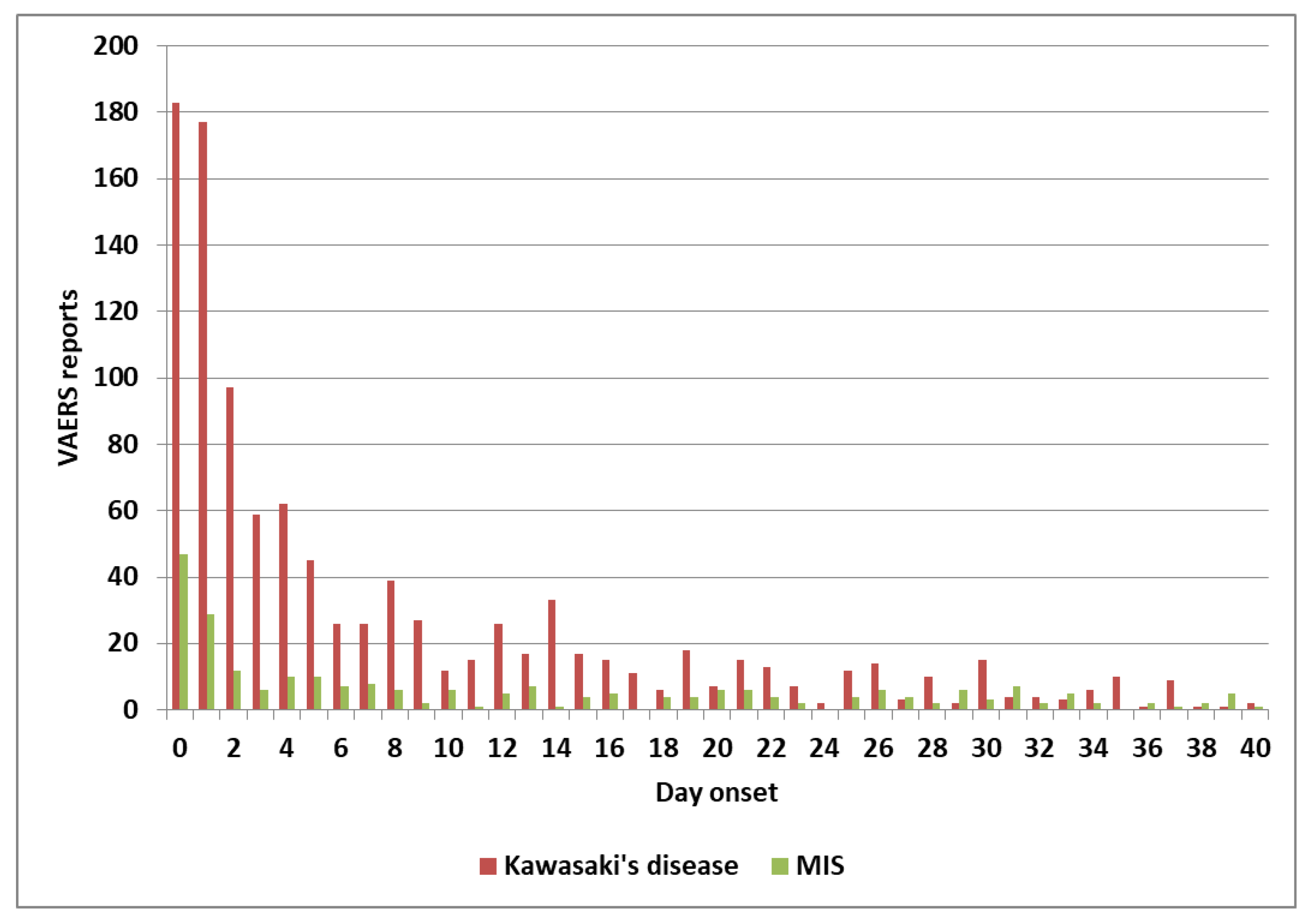
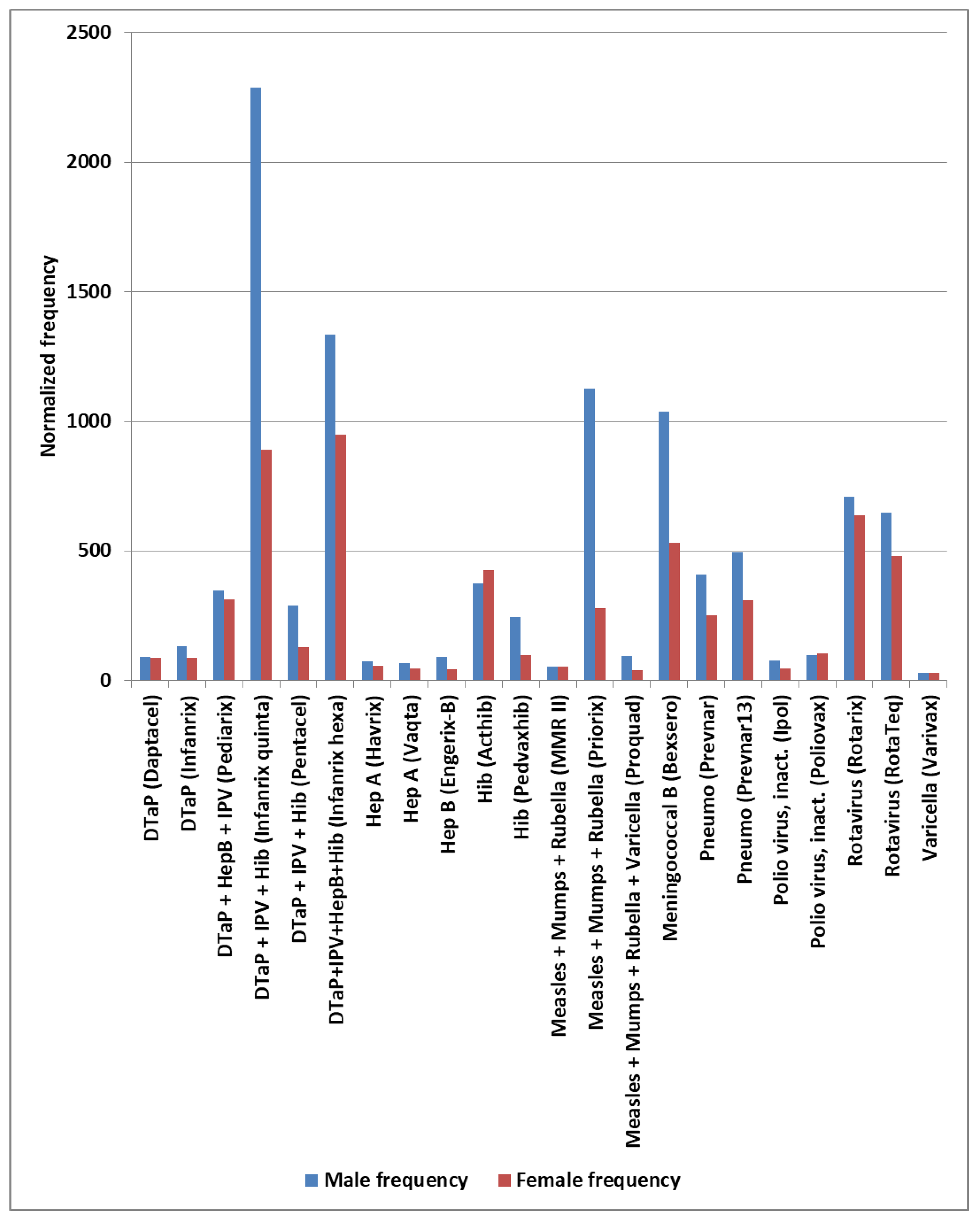
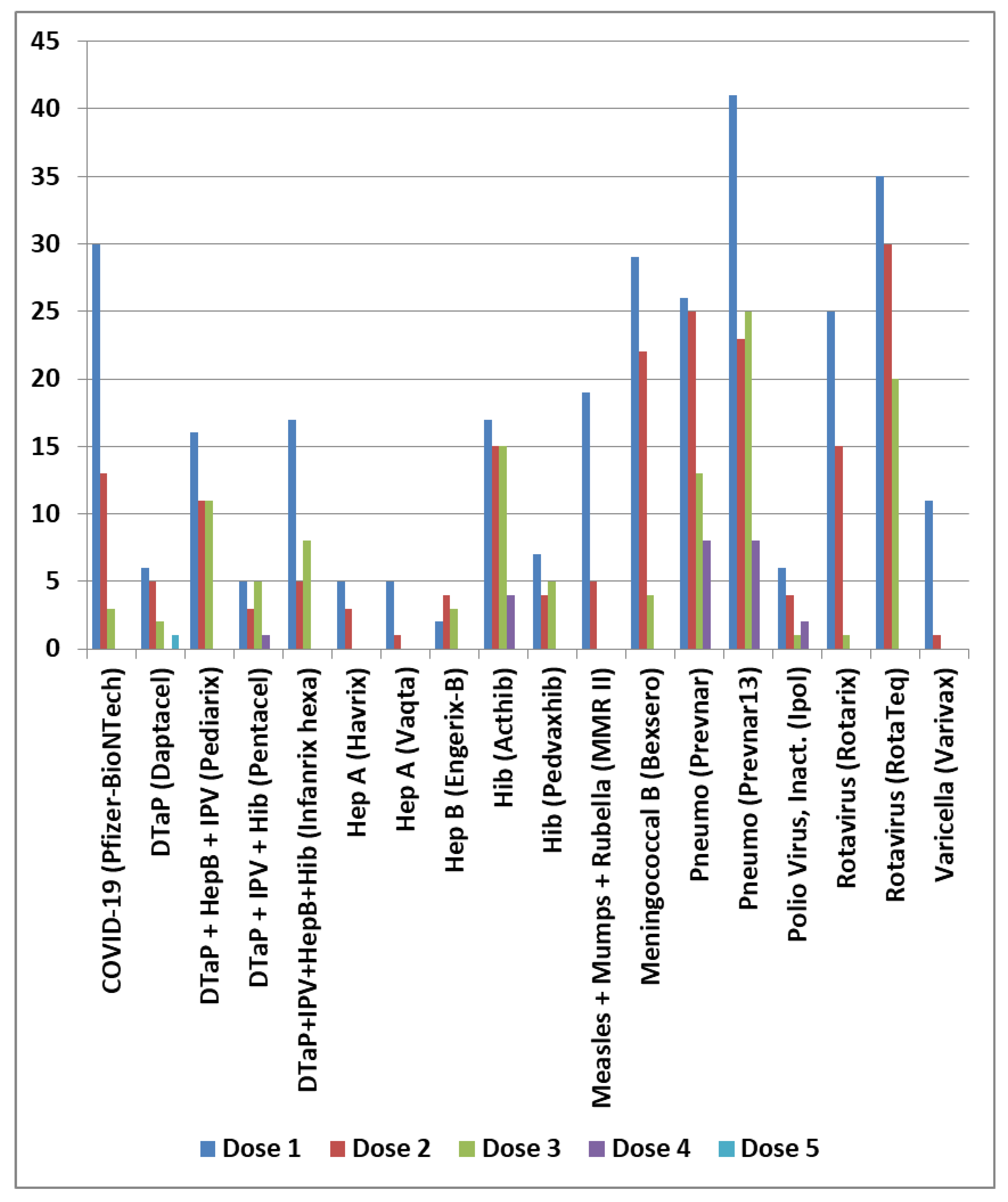
| Primary factor | Additional factor | Mast cell activator(s) | Likely IVIG resistance |
| neonate pathogen infection | maternally transferred antibodies (MatAbs) | high Ab titers | very low |
| pathogen infection | elevated Ab titers (ongoing, prior infections, …) | high Ab titers | very low |
| immunization | elevated Ab titers | high Ab titers | very low |
| immunization | manufacturing contaminant(s)—endotoxin | endotoxin or bacterial components | high |
| immunization | live attenuated vaccine GI pathogen (e.g., rotavirus)—possible disruption of gut barrier | possible high Ab titers and/or bacterial components | variable depending upon mast cell activators |
| GI infection | disruption of gut barrier [99] | endotoxin or bacterial components | high |
| environmental exposures including increased temperature | genetic risk factor | direct mast cell activation | high |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





